Marder M.P. Condensed Matter Physics
Подождите немного. Документ загружается.


Part II
ELECTRONIC STRUCTURE
Condensed Matter
Physics,
Second Edition
by Michael P. Marder
Copyright © 2010 John Wiley & Sons, Inc.

6. The Free Fermi Gas and Single
Electron Model
6.1 Introduction
Much of condensed matter physics lies within a Hamiltonian that one easily can
write down in a single line. It is
K=Y-^+-T
«
iqi
:
. (6.1)
Z
^2M
l
2fy\R
l
-R
l
,\
The sum ranges over all electrons and nuclei in a solid; M; is the mass of
an
electron
or nucleus, and qi is its charge. The simplicity is deceptive. Equation (6.1) can be
attacked directly by computer for little more than 10 to 20 particles. Dealing with
the 10
23
particles in actual solids requires a series of approximations, few of which
are particularly well controlled. One has to replace the original equations with
simpler ones, not always possible to justify, but capturing essential features of the
system. A number of these approximations is indicated in Figure
6.1.
In the study
of electronic structure about to begin, the goal will be to start at the lower left hand
side of the sketch and work back up toward the top.
Since Eq. (6.1) so completely intractable in its original form as to be almost
useless, progress comes about by posing and solving a series of model problems.
In these model problems, one makes drastic simplifications that make it possible
to solve the resulting equations analytically or numerically. The arbiter of success
is partly comparison with experiment, and partly the ability to gain qualitative in-
sights. There is now more than 70 years' experience with approximation schemes,
and their strengths and weaknesses are fairly well understood.
The simplest model of
a
metal is the free
Fermi
gas.
In this model, the Coulomb
interactions between electrons and electrons and electrons and nuclei are all turned
off. One considers a collection of electrons moving freely around in a box, and the
only nontrivial aspect of reality to be maintained is the Pauli exclusion principle.
In applying this model to a metal, one considers just a subset of the material's
electrons, the conduction electrons. The free Fermi gas model captures the features
of some metals remarkably well, particularly the alkali metals such as sodium.
It simply cannot be employed for any solid that happens to be an insulator or a
magnet.
Some important features of the solution of the free Fermi gas do not depend
upon details of the solution. They depend upon the fact that energy of the system
of electrons can be written as the sum of energies of individual noninteracting
155
Condensed Matter
Physics,
Second Edition
by Michael P. Marder
Copyright © 2010 John Wiley & Sons, Inc.
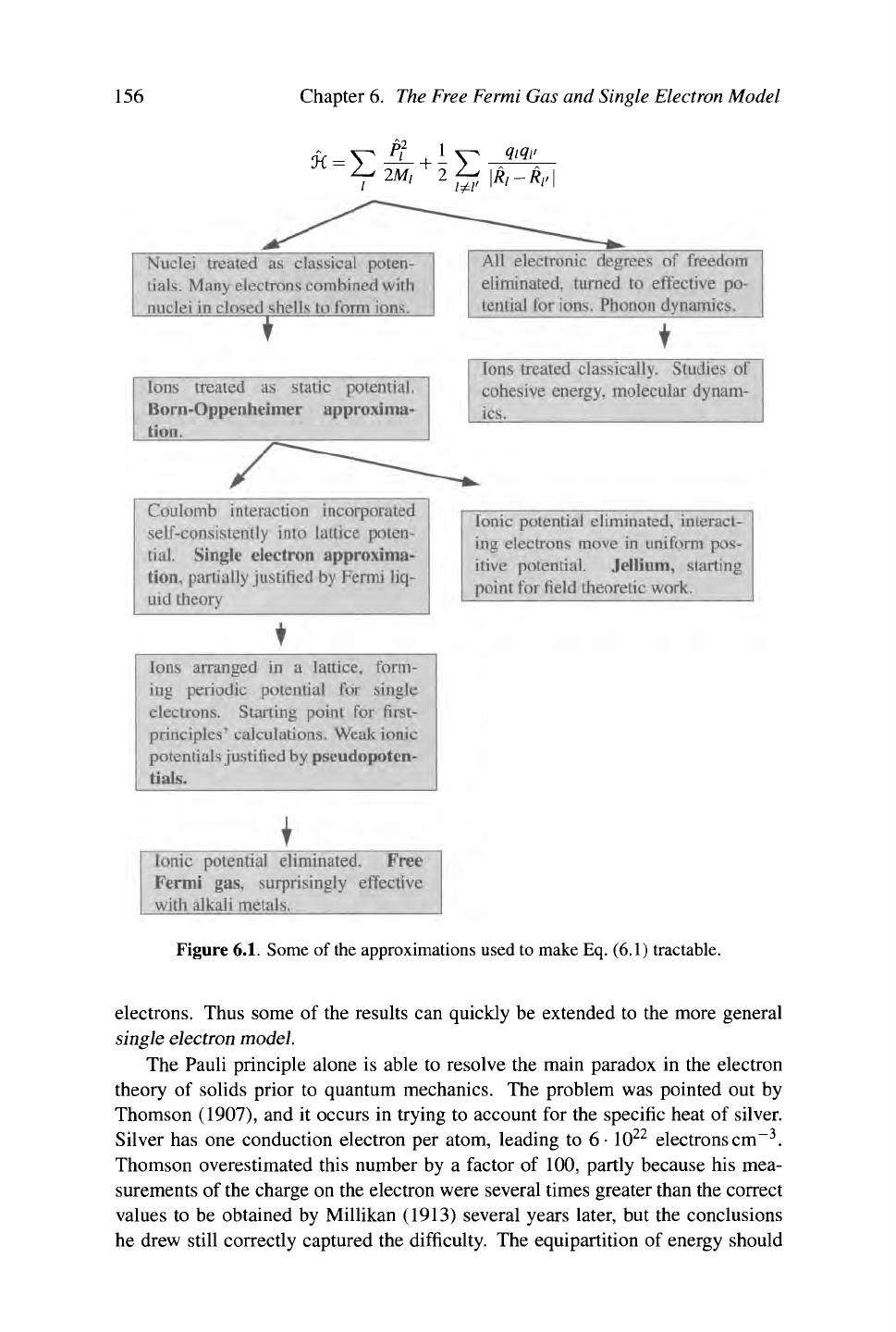
156 Chapter 6. The Free Fermi Gas and Single Electron Model
*=Y.ê,+
Pf , i v- mi
2M
'
2
Ù\
â
i-
â
''\
Figure
6.1.
Some of
the
approximations used to make
Eq.
(6.1) tractable.
electrons. Thus some of the results can quickly be extended to the more general
single electron model.
The Pauli principle alone is able to resolve the main paradox in the electron
theory of solids prior to quantum mechanics. The problem was pointed out by
Thomson (1907), and it occurs in trying to account for the specific heat of silver.
Silver has one conduction electron per atom, leading to 6 · 10
22
electrons cm
-3
.
Thomson overestimated this number by a factor of 100, partly because his mea-
surements of the charge on the electron were several times greater than the correct
values to be obtained by Millikan (1913) several years later, but the conclusions
he drew still correctly captured the difficulty. The equipartition of energy should

Starting Hamiltonian 157
contribute kgT jl to the mean energy for each of the three kinetic and potential
degrees of freedom for the free electrons plus another 3kgT for the nuclei of the
atoms, leading to a specific heat of
c\>
= 6nkß = 1.19 cal cm~
3
K '. The exper-
imental specific heat is, however, half this value, as if the electrons are somehow
immune to the demands of statistical mechanics. Thomson concluded that " We
thus get to a contradiction. The value of the specific heats of the metals shows that
the corpuscles [electrons] cannot exceed a certain number, but this number is far
too small to produce the observed conductivities...." [Thomson (1907), p. 85] The
resolution of the contradiction was not found for 20 years, and lies (as explained
below) in the fact that only a tiny fraction of the electrons is permitted by the Pauli
principle to participate in the process of absorbing energy.
The free Fermi gas and single electron models are such a crude approximations
that it may seem silly to study them at all. Two ideas eventually explained why even
the free Fermi gas can have great quantitative success for some metals. Neglect of
the periodic potential generated by the lattice of ions is justified by the pseudopo-
tential (Section 10.2.1), which shows how a redefinition of the electrons' wave
functions turns them into particles interacting with weak potentials. Neglect of the
interactions of electrons with one another
is
justified by the idea of the Fermi liquid
(Section 17.5), which shows that properly chosen linear combinations of electron
states behave like noninteracting particles. But these ideas ultimately provide only
partial justification for approximation schemes, so there is no choice but to suspend
disbelief and begin to calculate.
6.2 Starting Hamiltonian
The single-electron model is defined by the Hamiltonian
N fc2
v
2
It describes N conduction electrons, each of which interacts with an external po-
tential U but does not interact with the other conduction electrons. Equation (6.2)
is called the single-electron model because if one finds the eigenfunctions φι(7ι)
for single electrons, obeying
~
H V
+U(r)\ ψ,(η = ε
ι
ψ
ι
{?), (6.3)
y 2m
then the eigenfunctions describing many particles are simply obtained from prod-
ucts of the one-particle functions. The energy of the many-electron system is just
a sum of the energies of the one-electron functions that make it up (Problem 1).
That is, although the equation can be used to study large numbers of electrons,
their properties can be obtained one electron at a time.
Writing down Eq. (6.2) requires severe approximations outlined in Figure 6.1.
However for a general potential U it is still impossible to solve in general. To start

158 Chapter 6. The Free Fermi Gas and Single Electron Model
making progress, throw away the potential U too. The free Fermi
gas,
is described
by
-h
2
N
— T
ν
?
φ
(
?
ι
■
· ·
?
") =
εφ
(
?
ι
■
· · **)· (
6
·
4
)
2m
f^
It describes N conduction electrons, interacting neither with nuclei nor each other.
The eigenvalues and eigenfunctions of this Hamiltonian can be found exactly.
No differential equation is completely specified without naming its boundary
conditions. A natural choice would be to take Φ to vanish whenever any of its
arguments reaches the boundaries of the system, but this choice is not convenient
for calculations. Instead, one conventionally chooses a square box of side length
L,
L
3
= V and imposes periodic boundary conditions;
Φ(χι + L, y
h
z\ . . . ,
ZN)
= y{xi,y\,zi . - .
ZN)
Φ(χι,)>ι +
L,
zi ... , ΖΝ) = Ψ(*Ι,)Ί,ΖΙ -
■ ■
ZN)-
This
boundary condition is what one obtains
by
demanding that the system repeat
indef-
initely
with period L along the x, y, and z
directions.
(6.5)
This mathematical condition cannot possibly be realized experimentally. However,
almost any physical quantity one might calculate using Eq. (6.5) alters at most by
an amount proportional to
1
/L if
one
adopts more realistic boundary conditions. So
long as L is macroscopic, such corrections are negligible. It is sufficient to impose
some condition on the wave function at the system boundaries.
One Free Fermion. For the free Fermi gas, obeying Eq. (6.4), one can find the
one-electron solutions explicitly. Eigenstates of (6.3) are all of the form
Φ^ = ~j=
e
Because of the factor of l/VV, this function (6.6)
V
V is normalized.
with k of the form
- 2π
k=—{l
x
,ly,l
z
).
(6.7)
l
x
, /y, and l
z
are integers ranging from —oo to oo. Only if k is chosen in this way will a
function
of the form Eq. (6.6) satisfy the boundary condition Eq.
(6.5).
This enumeration
of
allowed values of k is more general than may now appear and it will survive unchanged
when
electrons interact with periodic potentials.
The eigenvalue corresponding to the eigenfunction (6.6) is
h
2
k
2
£9
= Simply insert Eq. (6.6) into Eq.
(6.4).
Setting (6.8)
Lm
hk = p = mv, the energy
is
just ^mv
2
.
Ground States of Many Free Fermions. The ground state of electrons obeying
Eq. (6.4) is constructed from products of the one-electron wave functions (6.6).
The Pauli exclusion principle forbids any given state from being occupied more
than once, and therefore any given state indexed by k is able to host no more than
two electrons, one for each value of spin.

Densities of States 159
The ground state of N electrons is built by first putting two electrons into the
single-particle state of lowest energy, which is \k\ = 0. Next one puts electrons
two at a time into all the states with \k\ = 2-n/L, and so on. The early stages of
this process are the subject of Problem 6.2. Because the energy Eq. (6.8) increases
with k, as one keeps adding electrons, one always wants to fill states with the lowest
available values of
£®.
Since £^ is proportional to k
2
, this means that successive
electrons get added to the ground state in successive spherical shells.
Define the occupation number f^ of a state indexed by k to be 1 if this one-
electron state is part of the ground state, and 0 otherwise. For a large number N of
electrons, the ground state corresponds to setting the occupation number _/j of all
states with k less than a certain wave vector kp, the Fermi wave vector, to 1 and
setting all others to 0. That is, the ground state is built out of one-electron wave
functions occupying a sphere in &-space, as shown in Figure 6.2. Naturally, one
wants to relate the number of electrons within the Fermi sphere to the value of kf,
and for this purpose it is necessary to recall Eq. (6.7) and analyze it more closely.
Figure 6.2. The ground state of the free electron gas is constructed by occupying vertices
in a cubic grid of states that approximate a sphere of radius k
F
in k space. Any other shape
would miss opportunities to put electrons as close as possible to the origin in k space, and
thus would not minimize the total energy.
6.3 Densities of States
The k states described by Eq. (6.7) occupy a cubic lattice in k space, also called
reciprocal space, with neighboring points separated by distances of Ιπ/L, as shown
in Figure 6.2. One can draw a Wigner-Seitz cell around each of these points and
can see as a result that the volume of the region of reciprocal space associated
with k in this way is (2TT/L)
3
. Not only is this statement true for the free Fermi

160 Chapter 6. The Free Fermi Gas and Single Electron Model
gas,
it is also true, as shown in Chapter 7, for electrons moving in general periodic
potentials, and as shown in Chapter 13 for phonons. Therefore, all the terminology
developed in this section to keep track of k states will recur constantly throughout
the remainder of this volume, and the densities of states D defined here will have
broad physical significance.
In order to calculate the total number of electrons N, or their energy £, or any
other thermodynamic quantity, one needs to carry out sums of the type
Σ
F
v
(6-9)
where F is some function of wave vector k, with the k vectors summed over values
allowed by (6.7). Mathematical convenience requests that one convert these sums
into integrals over a continuous function
F^.
Integration is defined by partitioning
space into a large number of volume elements and summing a function over these
elements, multiplied by the volume of each one. Because each k point is associated
with a volume (2π/£)
3
, one obtains
/
_
27Γ
dk
Ft = y ( ) Fr
This expression constitutes a long wavelength ap- (6.10)
*
*-^* L "·
proximation that is only accurate if
Fr
varies slowly
from one k to its neighbor, a condition that usually
obtains in practice.
^
V f -*
=>
> Fr = -^ / dk Fr.
Recall V
= Ü.
Although this formula has only (6.1
1)
TT"'
(^ττ) J
been derived for large volumes in the shape of a
k cube,
it
can be proved to hold for large volumes
of arbitrary shape.
On some occasions, the function
Fr,
may involve
a
delta function, such as
δ^.
Viewed as
a
function of
a
continuous variable, this function clearly transforms
into some multiple of ô(k
—
q). In order to remain consistent with Eq. (6.11), the
correspondence must be
δ-^—yì
'—ßß
—
uY
Only in this way do both sides of Eq. (6.11) (6.12)
kq
Ύ*
equal
1
when these delta function are inserted
6.3.1 Definition of Density of States D
The sum in Eq. (6.11) is over all wave vectors allowed for free electrons. Because
for each wave vector Pauli's exclusion principle allows two electrons, one with
spin up and the other with spin down, it is conventional to define the density of
electronic states, also sometimes called the density of levels
D^
=
2
The factor of 2 accounts for spin. (6_
\
3)
k
(2π)
3
'
which is defined to be the quantity allowing one to write
£F; =
V
j
dkD
lFl
.
(6.14)

Densities of States
161
To avoid perpetually writing Dp which does not even depend upon k, the notation
^
Ξ
|Σ=7'
dîD
i
=
jè)s
I
dl
(6
-
i5)
i
will be adopted in the equations that follow.
Energy Density
of
States. Several separate functions
D are all
referred
to as
densities
of
states. They are distinguished by their arguments. The most important
is
D(£), the
energy density
of
states which
is
useful
in
dealing with sums over
functions that depend upon
k
only through an energy function
£p
such
as
/
Electrons in periodic potentials will differ from
dED(E)F(E)
.
their free-electron counte^arts largely because
(6
16)
v
/ \ /
the energy
fcj
will differ from the tree-electron
v
'
ka value given byEq. (6.8). This definition looks
ahead to that possibility.
To find D(£), note that
J2F(E
Ï
)=V
([dk}F(E$ First use of
Eq.
(6.15).
(6
.
17
)
Ισ
/
/· Integrating
the
<5
function with respect
to E
dE
I
[dk]
δ(Ε-
Er)F(E)
J
ust
P
roduces
··
However,
if
one chooses
to
(
6
j
8)
J integrate with respect to k first, something new
comes
out.
=^D(E)
= I
[dl] 0{E
-
E-
k
)
.
Obtained by comparing Eq. (6.16) with (6.18). (6.19)
The units
of
densities
of
states are able
to
change without much warning. Often
they
are
expressed
in
units
of
l/[eV atom], which means they are related
to the
function defined by Eq. (6.19) by a factor
of
density
n.
6.3.2 Results for Free Electrons
For the free Fermi gas, one has that
D(E)
=
f[dk}S(E-E^) „„«,«,.
■
u
.
(
6
-
2
°)
v
' J
k
UseEq. (6.15), then switch
to
polar
v
'
2
roo
coordinates
in k
space to do the
=
4π-
Tj / dkk
2
S(E-E^)
integral, because
£2
depends upon the (6.21)
\
' "
magnitude but not the direction
of k.
1
f°° dE°
2mE°
1
_£(£_£°) Use Eq. (6.8)
to
express
k in
terms
of
(6.22)
π
ζ
Jo
\dE°/dk\
h
2 y
'
εΡ.
m
VïmÊ (6.23)
From Eq. (6.8) one
has
=
6.812 · 10
21
Λ/ε/eVeV"
1
cm'
3
.
àS?/dk
=
H
2
k/m; then use Eq. (6.8) (6.24)
V again
to
write
k in
terms
of £.
The number of electrons that can
fit
into
a
sphere of radius kp
is
]\j
=
\ f-
Because/j
is
1
only
if
the state
is
occupied. (6.25)
1σ

162 Chapter 6. The Free Fermi Gas and Single Electron Model
= V [ [dk] fl, Application of Eq.
(6.11 )
(6.26)
= V / \dk\ 9(kp—it) Because the occupation number vanishes for (6.27)
J k > kf.
Θ
is a Heaviside step function that
vanishes when its argument is less than 0 and
is
1
otherwise.
V 47Γ 3 _V#
= Ιζί = — Using the formula for the volume of
a
sphere, (6 28)
47Γ
3
3 3π
2
' and Eq. (6.15).
which implies that the Fermi wave vector is related to the density of electrons
n = N/V by
k
F
= (3τΛ)
1/3
= 3.09 [n-A
3
]V
3
A~\ (6.29)
The density of electrons is sometimes described by finding the volume per electron
V/N and defining the free-electron sphere to be one with the same volume, so that
4π , V
3_V
1/3
(6.30)
The energy of electrons in the most energetic occupied state is called the Fermi
energy or Fermi level and denoted by
£
F =
^
=
36.46[n-À
3
]
2
/
3
eV. (6.31)
2m
L J
The Fermi surface is the collection of k such that electrons with wave number k
have energy Ef. Finally, the Fermi velocity is defined to be
Vf = Hk
F
/m = 3.58
[n-À
3
]
1/3
· IO
8
cm s"
1
. (6.32)
The density of states D(E) has broad physical significance. Usually, it is not the
whole function that is important, but just D(Ep), the density of states at the Fermi
surface. Almost every electronic transport property of a solid will be proportional
to D(Ef-), whether it be the ability of electrons to absorb heat, or their response to
applied electric fields. Electrons lying far in energy below the Fermi surface cannot
contribute to transport properties, because all states around them are occupied, and
if they cannot change their state, they cannot respond to perturbations. States far
above the Fermi surface are not occupied at low temperatures and cannot play any
role in responding to external fields. The electron number density right at the Fermi
surface is therefore the crucial quantity to determine. For the free Fermi gas, one
gets the result
D(£
f
) = ^=4.11-10-
2
[n-Â
3
]eV-
1
À"
3
. (6.33)
2 c/r
Note that n = J
F
d£D(e) = f
Q
F
dt D(E
F
)
y/t/t
F
.
