Marder M.P. Condensed Matter Physics
Подождите немного. Документ загружается.


Part III
MECHANICAL PROPERTIES
Condensed Matter
Physics,
Second Edition
by Michael P. Marder
Copyright © 2010 John Wiley & Sons, Inc.

11.
Cohesion of Solids
11.1 Introduction
The cohesive energy of a solid is the energy needed in order to rip a sample apart
into a gas of widely separated atoms. By
itself,
this energy does not have much
significance. It is not easy to measure experimentally, and it bears no relation to
the practical strengths of
solids;
practical strength is governed by resistance to flow
and fracture, physically quite distinct from cohesive energy.
The question that the cohesive energy makes it possible to address is how crys-
tals choose their equilibrium structure. Electronic structure calculations begin by
assuming that atomic positions are known, but in studying cohesive energy, one
asks which structure leads to the lowest energy and why. In the course of this
study, crystals divide roughly into five classes: molecular, hydrogen
bonded,
ionic,
covalent, and metallic. These classes blend into each other, but still represent con-
ceptual ideal types. The molecular crystals are composed from atoms whose shells
are filled, which hold tightly onto their electrons, and which bind together only be-
cause of small induced dipole moments. Hydrogen bonded solids involve hydrogen
atoms as part of the bonding process; this might seem too specialized to warrant
a separate category except that it includes much of biology. The ionic crystals are
composed of pairs of atoms, in which one member of the pair donates an electron
to the other, and vast numbers of such pairs are held together by dipole forces.
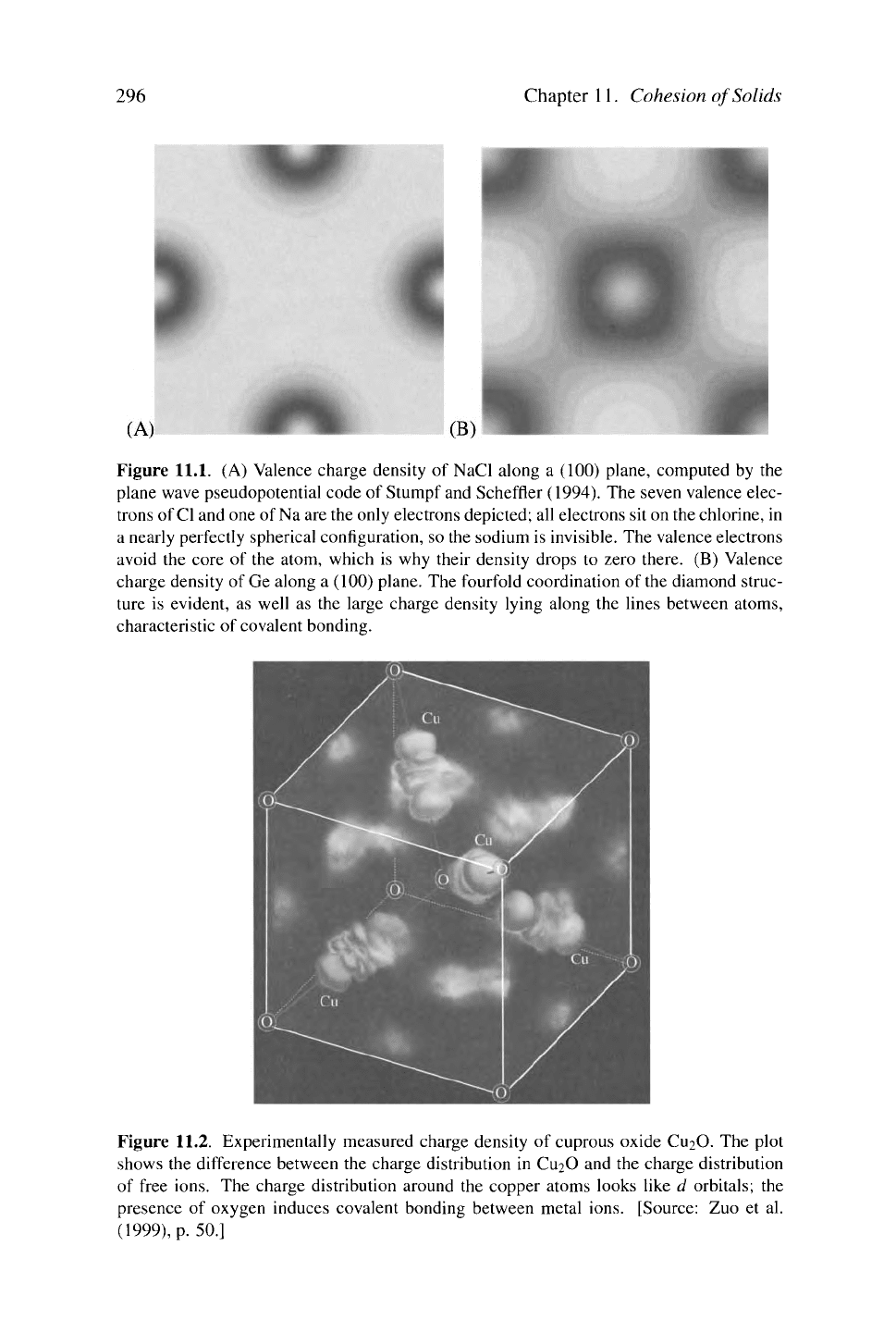
Figure 11.1(A) shows the valence charge density of NaCl; the visible charge sits
in a sphere around the chlorine, and charge density drops to very small values
between the atoms. The covalent crystals feature even more electrons wandering
away from their host atoms, and it is convenient to think of bonds between atoms,
because charge density is high along lines connecting near neighbors, as shown in
Figure
11.1
(B).
In metals, the conduction electrons are distributed quite uniformly
throughout the solid, and the net effect of the interaction between electrons and
nuclei is a hydrostatic pressure keeping the solid together. A plot of the conduction
electron density in a metal would be flat and almost featureless. As a final illustra-
tion of the connection between charge density and cohesion, Figure 11.2 presents
experimentally measured charge density for cuprous oxide. This metal oxide does
not easily fit classification. The charge density around the copper has the shape of
d orbitals, and leads to a phenomenon resembling covalent bonding between metal
ions.
295
Condensed Matter
Physics,
Second Edition
by Michael P. Marder
Copyright © 2010 John Wiley & Sons, Inc.
296
Chapter 11. Cohesion of Solids
Figure 11.1. (A) Valence charge density of NaCl along a (100) plane, computed by the
plane wave pseudopotential code of Stumpf and Scheffier (1994). The seven valence elec-
trons of
Cl
and one of Na are the only electrons depicted; all electrons sit on the chlorine, in
a nearly perfectly spherical configuration, so the sodium is invisible. The valence electrons
avoid the core of the atom, which is why their density drops to zero there. (B) Valence
charge density of Ge along a (100) plane. The fourfold coordination of the diamond struc-
ture is evident, as well as the large charge density lying along the lines between atoms,
characteristic of covalent bonding.
Figure 11.2. Experimentally measured charge density of cuprous oxide CU2O. The plot
shows the difference between the charge distribution in CU2O and the charge distribution
of free ions. The charge distribution around the copper atoms looks like d orbitals; the
presence of oxygen induces covalent bonding between metal ions. [Source: Zuo et al.
(1999),
p. 50.]

Introduction 297
Figure 11.3. Picture of the sodium chloride structure, indicating the sizes of the sodium
(small) and chlorine (large)
ions.
The chlorine ion is large because it
has
robbed
an
electron
from the sodium.
11.1.1 Radii of Atoms
In searching for ideas by which to explain how atoms assemble themselves, the
simplest is to assign each atom a radius, as if it were a hard sphere. An illustration
of NaCl, indicating the sizes of the two ions, appears in Figure 11.3. Such an
assignment would be without much consequence were it allowed to change every
time the atom formed a compound with a new neighbor, but in fact the apparent
radii of atoms remain roughly independent of their surroundings. This statement
must, unfortunately, be qualified a bit, because the apparent radii do depend upon
the class of crystal in which the atom is located.
Table 11.1 contains a summary of phenomenological rules that can be used
to estimate lattice constants if crystal structure is known; the origin of these rules
is discussed at length by Pearson (1972). Use of these rules should give lattice
constants within about 10% of experimental values, and they can serve as a quick
check on the results of either experiment, or theoretical calculation.
Example: Cristobalite. According to Wyckoff (1963-1971), quartz in the ß-
cristobalite form is cubic (a = 7.12 Â) and has a basis with eight silicon atoms and
sixteen oxygens, which in units of a/8 are at
Si:
(000) (440) (404) (044) (222) (266) (626) (662)
O: (111) (551) (515) (155) (177) (537) (573) (133)
(717) (357) (313) (753) (771) (331) (375) (735)
The nearest-neighbor distance for this structure is 1.54 Â. The silicon has four
neighboring oxygens, so Z = z = 4, while each oxygen has two neighboring sil-
icons,
and Z = 6, z = 2. According to Table 11.1, the covalent radius of silicon
is 1.17 Â, and that of oxygen is 0.74 -0.14 = 0.60 Â, which sum to 1.77 Â. The
discrepancy is more than 10%, so there are grounds for concern. Liu et al. (1993)
show that Wyckoff's structure is incorrect.

298
Chapter
11.
Cohesion of Solids
Table
11.1.
Effective radii
of the
elements
El.
Z
Ac
3+
Am
3+
Ar
0
Ag 1 +
Al
3+
As
3-
Au 1 +
Ba
2+
Be
2+
Bi
3-
B
3+
Br
1-
C
4+
4-
Ca
2+
Cd
2+
Ce
3+
Co
2-
Cl
1-
Cr
3+
Cs 1 +
Cu
1+
Dy
3+
Er
3+
Eu
2+
F
1-
Fe
2-
Ga
3+
Ge
4+
Ge
4-
Gd
3+
M
1.88
1.81
1.45
1.43
1.39
1.44
2.24
1.13
1.70
0.98
0.92
1.97
1.57
1.83
1.25
1.36
2.73
1.28
1.77
1.76
2.04
1.27
1.41
1.37
1.80
I
1.86
1.26
0.50
2.22
1.37
1.35
0.35
0.20
1.95
0.15
2.60
0.99
0.97
1.01
1.81
1.67
0.96
1.36
0.62
0.53
2.72
fli
1.53
1.25
1.21
1.52
1.98
0.89
1.52
0.80
1.14
0.77
1.74
1.49
1.65
0.99
2.35
1.35
1.59
1.57
1.85
0.72
1.27
1.22
1.61
El.
H
He
Hf
Hg
Ho
I
In
Ir
K
Kr
La
Li
Lu
Mg
Mn
Mo
N
Na
Nb
Nd
Ne
Ni
Np
O
Os
P
Pa
Pb
Pd
Po
Pr
Z
1-
0
4+
2+
3+
1-
3+
2-
1+
0
3+
1+
2+
4+
2-
3-
1+
3-
3+
0
2-
2-
2-
2-
3-
3-
4+
2-
2-
3+
M
0.78
1.58
1.57
1.77
1.66
1.36
2.38
1.88
1.56
1.60
1.30
1.40
0.88
1.91
1.47
1.83
1.25
1.56
0.89
1.35
1.28
1.63
1.75
1.38
1.76
1.83
I
2.08
1.10
2.16
0.81
1.33
2.00
1.15
0.68
0.65
1.71
0.97
1.58
1.40
2.12
0.84
Ri
1.49
1.58
1.33
1.44
2.03
1.69
1.23
1.56
1.36
0.74
1.57
1.64
0.74
1.10
1.43
1.53
1.65
El.
Pt
Pu
Rb
Re
Rh
Ru
S
Sb
Sc
Se
Si
Si
Sm
Sn
Sn
Sr
Ta
Tb
Te
Th
Ti
TI
Tm
U
V
W
Xe
Y
Yb
Zn
Zr
Z
2-
3-
1 +
2-
2-
2-
2-
3-
3+
2-
4+
4-
3+
4+
4-
2+
3-
3+
2-
4+
4+
3+
3+
2-
3-
2-
0
3+
2+
2+
4+
M
1.39
1.58
2.55
1.38
1.35
1.34
1.27
1.59
1.64
1.40
1.32
1.80
1.62
2.15
1.47
1.78
1.60
1.80
1.46
1.72
1.75
1.56
1.35
1.41
1.80
1.94
1.39
1.60
I
Ä,
1.48 2.16
1.84 1.04
2.45
1.41
0.81
1.44
1.98 1.17
0.41
1.17
2.71
1.66
0.71
1.40
2.94
1.13 1.91
1.59
2.21
1.37
0.68
0.95
1.46
1.56
2.17
0.93
1.62
1.70
0.74
1.31
0.80
El.
=
Element;
Z =
Valency;
M -
Metallic radius;
I =
Ionic radius;
R[
= Covalent radius.
The metallic radius
is
intended
for use in
close-packed structures with coordination number
12.
Ionic
and
covalent radii
may
need correction
by
additional factors.
The
ionic radius
is
intended
for
ions with coordination number near
6, and
should
be
multiplied
by the
factor
[z/6]
l
/("~
l
\
where
z
is
the
number
of
nearest neighbors,
and n is the
Born exponent
of
Table
11.2.
The
covalent radius gives best results
for
atoms with coordination number
4, and for
an atom with
Z
valence electrons
and z
nearest neighbors
is
given
by R = R\
—0.13 In[Z/z]:
for
Z < 0, use
8
-
\Z\. Source: Tosi (1964)
and
Pearson (1972),
pp.
147-153.

Noble Gases
Table 11.2. Born exponent n
Ion Type He Ne Ar Kr Xe
(inert core) Cu
+
Ag
+
Au
+
Born exponentn 5 7 9 10 12
Used in correction factor
[z/6]
1
/'"
-1
'
for ionic
radii. Source: Pearson (1972).
11.2 Noble Gases
The noble gases are characterized by a very weak attraction between the atoms.
The potential energy of a crystal conventionally takes the form of a two-body po-
tential
£
= ÌE^<;) (li.i)
'j
with
σ\ΐ2'
Ç)-0
(11.2)
Here r^ is the distance between two atoms. The potential in Eq. (11.2) is called the
Lennard-Jones 6-12 potential and arises partly from basic calculations and partly
from phenomenological considerations. The term proportional to r
-6
accurately
represents the interaction at large distances between two molecules without a per-
manent dipole moment, the van der Waals interaction. It arises in the following
way.
The interaction between two dipoles is
Φ(τ) = \?\ ■p2-?,(p
ì
-r){p2-r)]/r\ (H-3)
where r is the distance between the atoms, and r is a unit vector. Now it will be
protested that atoms without dipole moments do not have dipole moments. This
is true. However, quantum or thermal fluctuations continually induce tiny dipole
moments in each atom. The resulting electric field then polarizes the other atom,
producing a small dipole moment in it, of order r~
3
. The resulting interaction
is then of order r~
6
. The induced dipole moment is always of such a sign as to
lower the energy of the system; therefore, the interaction is always attractive. To
obtain such a result formally, see Problem 3. One places the quantum operator
for Coulomb interactions as a perturbation into the Hamiltonian for two widely
separated atoms and then finds a nonzero result in the second order of perturbation
theory. The final result is of the form
Φ—^ψ, (11.4)
where a, is the polarizability of atom i. It is only valid when the atoms are well-
separated. When they come close together, at distances comparable to the atomic

300
Chapter
11.
Cohesion of Solids
radii, the force between them becomes repulsive. For this reason, and with no
firmer foundation, one adds the term proportional to r
12
in Eq. (11.2).
In order to provide a firm check on the theory, one can measure the constants
e and σ in the gas phase and then use the results to make predictions about the
solid phase. The gas phase measurements use the following relation from the virial
expansion:
b
2
= - id? [1
—e~^
r
\
See
Landau andLifshitz(1980), pp. 231-232, (11.5)
2 J who use somewhat different notation.
The coefficient b
2
enters the virial expansion for the pressure P in the form
PV/k
B
T =
l-b
2
/V
... (11.6)
so measurements of the equation of state at low densities can be used to find b
2
,
and hence fit σ and e (Table 11.3).
Table 11.3. Parameters σ and e for the noble gases
Noble Gas
e(eV)
σ(Α)
Source:
p.
201.
He Ne
8.6· 10"
4
0.0031
2.56 2.74
Bernardes
Ar
0.0104
3.40
Kr
0.0104
3.65
(1958) and Hirschfelder et al
Xe
0.0200
3.98
. (1954),
The total energy of a crystal where atoms interact in this way is then
£/Ν
=
1*Σ
®'
2
-ψ
Taking the crystal to contain N atoms, and (\ 1.7)
adopting periodic boundary conditions so that
ϋφθ all atoms have identical neighborhoods.
Letting d be a nearest neighbor distance, one can rewrite the energy as
ε/*
=
2
^Σφ
12
φ
12
-φ
6
φ
6
di·«)
R
Ξ
26[Α
12
φ'
2
-Α
6
φνίώΑ
/Ξ
Σ (|Y. (11.9)
Written in this way, one sees that the lattice sums can be carried out once and
for all for any particular crystal structure, so that the energy depends only upon
the nearest-neighbor separation. The sums converge rapidly because the powers
involved are so high. Except for helium, all the noble gases solidify into an fee
structure, and the appropriate lattice sums in this case are shown in Table 11.4.
One can now work out the nearest-neighbor spacing in equilibrium,
4> =
*(^)
1/6
,
(ii.io)

Ionic Crystals 301
Table 11.4.
Crystal
A
6
An
Al/2An
Lattice sums,
fee
14.4539
12.1319
8.6102
A(,
and Λ n for various crystal
bec
12.2537
9.1142
8.2373
structures
hep
14.4549
12.1323
8.6111
The sums are defined in Eq. (11.9). The final row is proportional to the energy of the
crystal. While fee and hep have noticeably lower energy than bec, the energies of the
two former structures are too close for these arguments to determine which will appear
experimentally; the rare gas solids choose fee.
Table 11.5. Properties of noble gases compared with predictions of Lennard-
Jones potentials
Noble Gas Ne AT Kr Xe
Experimental do (Ä)
do from Eq. (11.10) (Â)
Experimental £/N (eV/atom)
E/N
from Eq. (11.11)
Experimental B (dyne/cm
2
)
B from Eq. (11.13)
Results show agreement at roughly the 10% level. Source: Ashcroft and Mermin
(1976),
p. 401.
3.13
2.99
-0.02
-0.027
1.1 -10
10
1.81· 10
10
3.75
3.71
-0.08
-0.089
2.7 · 10
10
3.18· 10
10
3.99
3.98
-0.11
-0.120
3.5 · 10'°
3.46· 10
10
4.33
4.34
-0.17
-0.172
3.6 · 10
10
3.81
■
10
10
the cohesive energy
P I hi — -c
'2A
12
'
A
2
E/N
= -e—4-, (11.11)
and the bulk modulus B which is given by
7
ο
2
ε
B =
V
lzri
.
(11.12)
For an fee lattice, the volume per particle V/N is related to the nearest-neighbor
separation d by V/N = d
3
/\/2· Evaluating Eq. (11.12) gives
Comparisons of this simple theory with experiment are listed in Table 11.5.
11.3 Ionic Crystals
The simplest ionic crystals are the alkali halides, built by combining an element
from the first column of the periodic table with an element from the seventh col-
umn. The distinctive feature of these crystals is that the alkali metal (Li, Na, K,

302
Chapter 11. Cohesion of Solids
Rb,
or Cs) gives up an electron relatively easily so as to obtain a closed outer shell,
while similarly the halogen (F, Cl, Br, or I) has a strong affinity for acquiring an
extra electron to fill its outer shell. In fact, the benefits of closing the electronic
shell outweigh Coulomb repulsion, and isolated halogen atoms stably bind extra
electrons. Their positive electron affinities are listed in Table 11.6, along with the
first ionization potential of the alkalis. The affinity of the halogen is typically less
than the ionization potential of the alkali, but bringing the positively and negatively
charged ions close together gives enough Coulomb energy to make the resulting
structure stable. The Coulomb energy is easily estimated by treating the two ions
as possessing a single negative or positive charge and placing them at a distance of
about 3 Â apart; this gives a Coulomb energy of around —4 eV, which is enough
to overcome the difference between electron affinity and ionization potential. The
reason that the I-VII compounds are particularly simple, however, is that the struc-
ture stabilizes in such a way that the overlap of electronic charge from neighboring
ions is negligible, as shown in Figure 11.1(A).
Four particularly common lattice structures—the sodium chloride structure, the
cesium chloride structure, the zincblende structure, and the wurtzite structure—
were sketched in Section 2.3.
11.3.1 Ewald Sums
Actually carrying out the sum over positive and negative ions to calculate the en-
ergy of the ionic solids is technically tricky. If in the first unit cell the positive ion
(cation) is at the origin, and the negative one (anion) is at d, the potential energy
associated with a particular cation is
ΪΣ[ΓΗ]-Ϊ
2
Ϊ»»-*-Ί
(1U4)
and the energy of the crystal as a whole will be N/2 times this amount. The
dif-
ficulty is that the sum (11.14) converges very slowly, and in fact it can be done so
as to have any value whatsoever. Physically, different limiting values of the sum
correspond to crystals with differing amounts of surface charge, and the Coulomb
force is so long range that the surface charges are able to make the crystal energy as
Table 11.6. Electron affinities and ionization potential for the alkalis
and the hai ides
Atom Electron affinity (eV) Atom First ionization potential (eV)
~H
Ö75
F
3.40
Cl
3.61
Br
3.36
I
3.06
Li
Na
K
Rb
Cs
5.32
5.14
4.34
4.18
3.90
Source: Hotop and Lineberger (1975) and Emsley (1998).
