Marder M.P. Condensed Matter Physics
Подождите немного. Документ загружается.


18.
Microscopic Theories of Conduction
18.1 Introduction
This chapter is devoted to exploring the difference between electrical conductors
and insulators. Electrical resistance arises from the scattering of electrons as elec-
tric fields drag them through a sample. To connect electrical resistivity with the
underlying physics at the atomic scale, it is necessary to grapple with the scat-
tering problem directly. The simplest version of this theory treats impurities and
thermal fluctuations as weak scattering sites, amenable to the same techniques that
solve the problem of inelastic neutron scattering.
As the strength and density of impurities that scatter electrons increases, it is
natural to expect the resistance of the solid to increase as well. What might not be
expected is that past a certain density of impurities, the solid undergoes a qualitative
change. It no longer can support traveling waves, and it turns from a conductor
to an insulator. This mechanism is a subtle quantum-mechanical effect, valid at
low temperatures. It is only one in a series of ways that qualitative shifts from
conductors to insulators can occur. The essential difference between insulators
and conductors is more than a question of filled versus unfilled bands in perfect
crystals, and the most general explanation has probably not yet been formulated.
18.2 Weak Scattering Theory of Conductivity
18.2.1 General Formula for Relaxation Time
Although the calculation of electronic states in disordered materials is an extremely
difficult problem, there is one fairly straightforward case, which is an extension of
the nearly-free electron picture to the case of disordered metals or random alloys.
Imagine that a collection of electrons sits in a weak but no longer periodic potential.
If the effects of the potential may safely be treated to low order in perturbation
theory, then its interactions with electrons may be solved in the same way that X-
ray scattering in amorphous structures can be solved. As first noted by Bhatia and
Krishnan (1948), the same correlation function governs the two cases, because the
electrons, like the X-rays, are simply waves bouncing off weak scatterers.
The starting point is Section
10.2.1,
which showed that in some cases, partic-
ularly the alkali metals, Schrodinger's equation for the conduction electrons in the
presence of a periodic potential can be recast as a problem for plane waves in weak
potentials. Formally, these weak potentials are nonlocal and energy-dependent,
because the fact that they are so weak results from the cooperative screening pro-
vided by all the different electrons. All one needs to know, however, is that there
523
Condensed Matter
Physics,
Second Edition
by Michael P. Marder
Copyright © 2010 John Wiley & Sons, Inc.

524 Chapter 18. Microscopic Theories of Conduction
are cases where regarding a metal as free electrons in weak potentials has hope
of quantitative success. Take U(R) to be an appropriate pseudopotential, centered
at R. In the case of a liquid metal or metallic glass, the locations R label all the
sites inhabited by atoms. In the case of a random alloy, or a metal with impuri-
ties,
the locations R label all the places where the second species or impurity has
been placed, and U(R) describes the difference between the pseudopotential of the
original metal and the pseudopotential of the impurity.
The Collision Term. The collision term dg/dt\
co
\\ is central to the Boltzmann
equation. It has been treated so far in the relaxation time approximation. The first
step in a microscopic theory of resistance is the attempt to calculate the relaxation
time from an underlying picture of electron collisions.
The relaxation time approximation is based upon the view that there are effec-
tively random processes causing electrons to alter their wave vectors. What form
should these processes have? The question can be answered in a purely formal
manner, using techniques to be presented in Section 20.4. The formal derivation
can obscure the physical meaning of
the
various terms that arise, so the presentation
in this section will emphasize physical arguments over formal ones.
Consider a collection of electrons described by the distribution function gj,.
There is no need for the index r, so it will not be displayed explicitly. If an electron
scatters off nonmagnetic impurities, the probability
CP
for it to make a transition
from state k and spin a to state k! with spin a' must have the form
V(k^V,t)=g
i
[l-gp]
Ôaa>W
Uf
.
(18.1)
Because the scattering potential is nonmagnetic, the jump can only occur if the
electron spin a does not change. The rate must be proportional to the number
g of electrons occupying state k, proportional to the number
1 —
g of vacancies
at destination k', and finally proportional to a rate W^, that is independent of the
occupation number g. Accounting both for electrons that jump into k by these
processes and for those that jump out gives
J[dk'}
g
%
,[\
-
gl
}
Wl
r
k
-g
%
[l-
gll
}W
rkl
.
(18.2)
dg
=
V
dt coll. 2
The leading 1/2 is due the fact that the spin state of the electron cannot change, effectively
cutting the density of states in
half.
\dk'\ defined in Eq. (6.15).
Equation (18.2) is equivalent to the relaxation time approximation when the
following conditions obtain:
1.
Scattering potentials into which electrons collide are spherically symmetrical,
and can be treated as weak.
2.
The energies of occupied electronic states £^ are isotropic, and they depend
only upon the magnitude of k.

Weak
Scattering Theory of Conductivity 525
The first assumption allows one to write the potential seen by electrons as
The functions
U
are spherically symmetrical scattering
Utnt
= / U(r
—
R).
potentials, centered
at
sites R. They are best thought
of
(18.3)
■"—^ as pseudopotentials of impurity atoms added to a perfect
R crystal, or else as atoms of the crystals out of place.
Adopting this form of scattering potential, and retracing steps starting with Fermi's
Golden Rule that led to Eq. (13.104) gives
W
M>=
E
T
HZ
{
-n\(*
f
\Û
M
\V)\
2
(18.4)
final states
= 27rS'(q,uj
e
)\U(q)\
2
^. (18.5)
Here q = k
—
Id
is the change in wave number of the electron upon scattering, and
huj
e
=
8.J.
—
£~
k
,
gives its change in energy. The inelastic structure factor
5
1
appears
just as
for
neutron scattering from phonons, now depending upon the change
in
electron energy. The number
of
scatterers N
s
may refer to
a
dilute impurity with
strong scattering properties, or to every atom in a liquid metal.
Because the potential U(r)
is
spherically symmetrical, U(q)
is
symmetric
in
k and
k'
and indeed only depends upon the angle
0
between them. The inelas-
tic structure factor is not
in
general insensitive to interchange
of k
and k', but
if
scattering is nearly elastic and uj
e
is
very small, then one sees from Eq. (13.104)
S
l
(q,
0)
=
S'(—q, 0), and assuming one can use this approximation then
W
u
,
=
W
n
.
(18.6)
Next assume that the solution of the Boltzmann equation has the form
_,
-,
Here C is a vector that either is constant, or is
gr
= fr -\-Q
•
k.
a
function
of
E-
k
;
it cannot otherwise depend
(
18.7)
upon
k
or
f.
This form is general enough to encompass spatially uniform electric fields, uniform
temperature gradients, and uniform magnetic fields. Putting Eqs. (18.6) and (18.7)
intoEq. (18.2) gives
dg
dt
coll.
-e-iv
J[dV](k-k?)W
u
„
(18.8)
and so
j„
o
—
f
Because
W-g.,
depends only on the angle between
k
and
k!, only the component
of V
parallel
to k
survives the (18.9)
dt
co
' £
integration.
If
one takes
%
=
kz, W depends only on
ki,
and
it
is easy to see that the integrals of
k'
x
and
k'
v
vanish
by symmetry. Note that C-k = g
—
f.
with
—
=
l
-yj[S!] W
Vk
,{\-k-k').
(18.10)
1
_ 1
Inserting Eq. (18.5) into Eq. (18.10), noting that
[\-k-k')
=
2(^-)\
(18.11)

526 Chapter 18. Microscopic Theories of Conduction
Table 18.1. Mean free paths of electrons in liquid metals
Metal: Li Na Cu Ag Au Zn Hg AI Ga Sn Pb Sb Bi Fe
IT (A): 45 157 34 51 27 15 5 20 17 5 6 4 4 3
Values obtained by treating the metals as free electron gases where l
T
=
VfT, and the relaxation time r is obtained from the resistivity p at the
melting point through
T
= m/e
2
np. Source: Cusack (1987) and Grigoriev
and Meilkhov (1997)
p.
555.
and averaging over initial phonon states as discussed prior to Eq. (13.113) gives
J[dq]S(q,oJ
e
)\U(q)\
2
^. (18-12)
T, ~ 2 V
In this way, the relaxation time may be related to an integral over the inelastic
structure factor and the pseudopotential U(q).
Resistivity of Liquid Metals. A liquid metal is as disordered as a metal can
be,
yet somewhat surprisingly the weak scattering limit is often appropriate, and
Eq. (18.12) allows quantitative description of resistivity. As evidence, one can use
the measured resistivity of liquid metals to deduce the characteristic mean free path
h = vpT electrons travel between scattering events. As shown in Table 18.1, this
distance is tens of angstroms in the noble metals, although it shrinks to atomic
dimensions in iron or bismuth.
For liquid metals, the static structure factor is spherically symmetric. Purely
elastic electron scattering from such a disordered array of atoms leads to nonzero
electrical resistivity. So it is enough to consider the limit
u>
e
= 0 where change in
electron energy can be neglected and write from Eqs. (3.55) and (13.104),
S{q,
Lü
e
= 0) = S(q)
H
6(E
F
- £{\k - q\)). (18.13)
In writing this expression, make use of the fact that transport formulas such as
Eq. (17.54) use k only on the Fermi surface where £^ = £/?. For elastic scattering,
£.p must lie on the Fermi surface as well.
To continue the evaluation of
Eq.
(18.12), insert Eq. (18.13). Doing the angular
integral over q
/_
6{2k
F
-q)
^
</(cos 0)5(£
F
- E(yJ4 +q
2
- 2k
F
q cos 6)) = g
£/Q
^ (18-14)
gives finally an expression due to Ziman (1961),
l l
N, r
2k
"
1 IN
Î
LKF
-, -,
— = y-: ^ dqq
3
S(q)\U(q)\
2
/V
s
is number of (18.15)
r
£
4irh
2
k
2
F
v
F
V Jo
HH KHn KHn
scatters.
>
P=
J
î-
=
^2&iW r
dqq'S{q)\U{q)\
2
.
(18.16)
ne
L
T
t
e
L
nv
F
v
V
/
Ak%
Jo

Weak
Scattering Theory of
Conductivity
527
The final expression uses free-electron relations between the electron density, the
Fermi wave vector, and the Fermi velocity vp. So in the weak scattering limit, the
resistivity is just given by an integral over the static scattering structure factor S
and the pseudopotential U.
In order to evaluate Eq. (18.16), it is necessary to know both the static structure
function S(q) and the pseudopotential U(q). The former can be measured by X-
ray scattering, and the latter can be obtained either from the density functional
calculations described in Section
10.2.1,
from de Haas-van Alphen measurements,
or else from optical data to be described in Section 23.4. An example of assembling
these various ingredients to determine the resistivity of liquid aluminum is provided
as Problem 1. The calculation outlined in the problem should not be expected to be
accurate to more than around 50%, but the more careful evaluation of Ashcroft and
Guild (1965) duplicated experimental values within a few percent. Other properties
of liquid metals are reviewed by Ashcroft and Stroud (1978) and March (1990).
Application to Phonons.
Calculation of
the
resistance of crystals proceeds in a slightly different
way.
An
absolutely perfect crystal has almost no electrical resistance, as Bloch first realized
[Section 7.1]. Formally this fact emerges without the need for any calculation
from Eq. (18.12). The leading term Eq. (13.129) in the inelastic structure function
is a product of delta functions restricting q to the reciprocal lattice, and a delta
function restricting k and k! to the Fermi surface. These two delta functions are
incompatible: when k and V are on the Fermi surface there is vanishing chance
that q = k
— K
is in the reciprocal lattice.
For metallic crystals resistivity comes from phonons, which means employing
Eq. (13.134) to take into account the creation or destruction of one phonon in each
electron scattering event.
To simplify matters, keep only longitudinal phonons corresponding to acoustic
modes, for which the polarization e^\\q. This type of interaction is called normal
scattering (N-process). The contribution of other phonon bands is not necessarily
negligible, when they enter in this context it is called called Umklapp scattering
[U-process;
Umklapp is German for "flopping over" and was introduced by Peierls
(1929)].
It is hard to see without detailed calculation whether normal or Umklapp
processes should be more important, and both should be included for accurate re-
sults.
In the interests of simplicity, the calculation that follows will keep only the
contribution from acoustic phonons, which is sufficient to give a good idea of how
the general procedure runs, but not to achieve comparison with experiment on the
order of
1 %
that is possible in the most ruthlessly correct calculations, presented
by Bass et al. (1990).
Returning to Eq. (13.134), there are two terms to inspect. In one of them, pro-
portional to
8{u)^
+
uj
e
),
the phonon of energy
%ujq
steals energy from the electron.
This requires the final electron state to have energy lower than the initial state.
Looking now at Eq. (18.1), one sees that since k is on the Fermi surface, both
terms on the right hand side of Eq. (18.1) vanish unless the final state is higher in

528 Chapter 18. Microscopic Theories of Conduction
energy than the initial state; the electron must scatter from occupied to unoccupied
states.
Terms where the phonon takes energy from the electron must be negli-
gible at temperatures low compared with the Debye temperature. In the second
process described by Eq. (13.134), proportional to
S(LO^
— u)
e
),
the phonon gives
energy to the electron. This processes is allowed because the electron moves from
occupied to unoccupied states. The main effect of keeping track of the energy as-
sociated with phonons is to decide which scattering process is allowed; otherwise
the phonon energy is negligible in comparison with the electron energies. Thus one
can write
t2 2 Keep only the phonon absorption term of
S(fl, u) « —n
q
h6(£
F
-£(\k-q\) Eq.U
3.134);
once this is done it is all
(
18#17
)
IMHLÜ
n
ght to neglect the phonon energy inside
v
'
Q the delta function. n
q
is a phonon
occupation number.
Thus
Ht/^
M^^-^-^0
(18
-
18)
3TT
/N \ 1 f
2
^
F
/j£j2
^=^(^44
X
dqq3
2lt^
U
^
USeEq(1814) (J8
-
19)
3TT
WV,N 1 H ,k
B
T^5 rWT • i ,
Il(
k
F
zTs
2
=
^(v)^2Mêy
L
dzz
F^T
|£/
(-e-)'
'
(i8
-
2o)
where c is the longitudinal wave speed, M is ion mass, 0 = hk^c/kß is a De-
bye temperature, and z =
Oq/kpT.
At temperatures T large compared with the
0, Eq. (18.20) is linear in T, while at much lower temperatures the resistivity is
predicted to vanish as T
5
, the Bloch T
5
law.
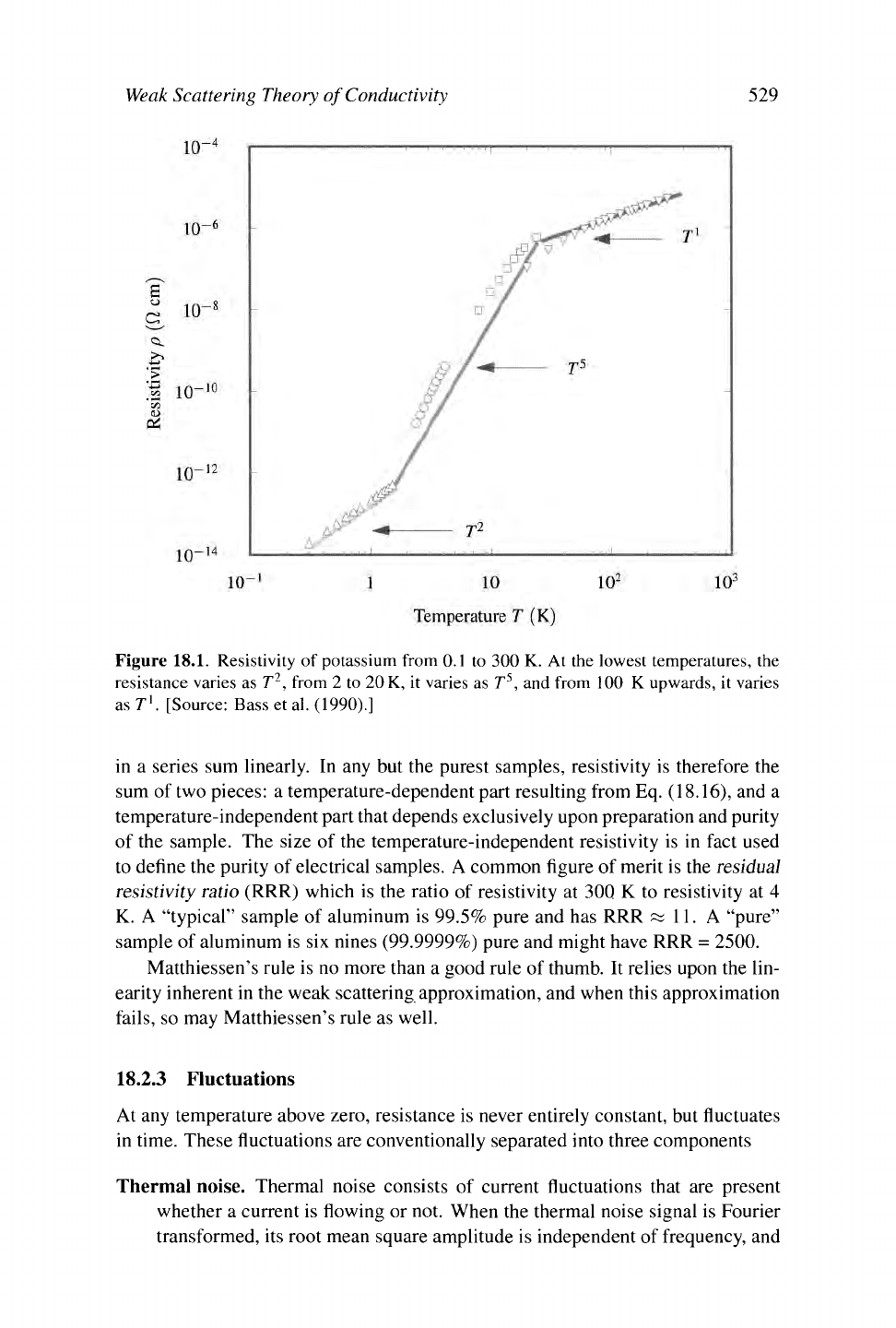
Comparison with Experiment. The most careful comparison of these predictions
with experiment has been carried out for the alkali metals. As shown in Figure
18.1,
the resistivity is linear to high accuracy at temperatures over 100 K, and it
begins crossing over to a decay as T
5
at temperatures on the order of 20 K. By a
temperature of 2 K, there is yet another crossover to a decay of resistivity as T
2
.
The origin of this behavior is electron-electron scattering, as described in Section
17.5.
Computing the coefficient of T
2
is difficult, and it is discussed by Bass et al.
(1990).
18.2.2 Matthiessen's Rule
Comparison of theory and experiment for the alkali metals relies upon extremely
pure crystalline samples. A piece of wire pulled from the shelf follows predic-
tions of Eq. (18.20) down to around 100 K, but at lower temperatures it begins
to differ. Grain boundaries, dislocations, and various trace elements scatter elec-
trons,
and therefore all contribute additively to resistivity according to Eq. (18.16).
Matthiessen
's
rule states that separate sources of resistivity, such as phonons and
impurities, sum linearly to produce the total resistivity of a sample just as resistors
Weak
Scattering Theory of
Conductivity
529
Figure 18.1. Resistivity of potassium from 0.1 to 300 K. At the lowest temperatures, the
resistance varies as T
2
, from 2 to
20
K,
it varies as T
5
, and from 100
K
upwards, it varies
as T
1
. [Source: Bass et al. (1990).]
in a series sum linearly. In any but the purest samples, resistivity is therefore the
sum of two pieces: a temperature-dependent part resulting from Eq. (18.16), and a
temperature-independent part that depends exclusively upon preparation and purity
of the sample. The size of the temperature-independent resistivity is in fact used
to define the purity of electrical samples. A common figure of merit is the residual
resistivity ratio (RRR) which is the ratio of resistivity at 30Q K to resistivity at 4
K. A "typical" sample of aluminum is 99.5% pure and has RRR « 11. A "pure"
sample of aluminum is six nines (99.9999%) pure and might have RRR = 2500.
Matthiessen's rule is no more than a good rule of
thumb.
It relies upon the lin-
earity inherent in the weak scattering approximation, and when this approximation
fails,
so may Matthiessen's rule as well.
18.2.3 Fluctuations
At any temperature above zero, resistance is never entirely constant, but fluctuates
in time. These fluctuations are conventionally separated into three components
Thermal noise. Thermal noise consists of current fluctuations that are present
whether a current is flowing or not. When the thermal noise signal is Fourier
transformed, its root mean square amplitude is independent of frequency, and

530
Chapter 18. Microscopic Theories of
Conduction
the magnitude of voltage fluctuations in frequency interval
dco
for a wire of
resistance R is
(ÔV
2
)=4k
B
TRduj. (18.21)
These noise properties were identified by Johnson (1927) and were explained
byNyquist(1928).
Shot noise. Shot noise is an additional source of noise that is proportional to mean
current. The current fluctuations 5J it produces have strength per frequency
interval duj
(SJ
2
)=2eJduj. (18.22)
1
// or flicker noise.
1
// noise is an additional noise source whose amplitude
grows with decreasing frequency / roughly as l/f. It is fairly ubiquitous,
yet the specific mechanism that produces it varies from one system to another.
Dutta and Horn (1981) provide a general explanation of how it might arise.
18.3 Metal-Insulator Transitions in Disordered Solids
18.3.1 Impurities and Disorder
No crystal produced in the laboratory is ever completely perfect. In the case of sil-
icon that has been prepared for use in electronics, the degree of perfection is very
high. Atoms such as silver and gold whose presence would disrupt electronic cir-
cuitry are excluded to better than one part in 10 '
2
, and wafers of tens of centimeters
in diameter are regularly prepared without a single dislocation or grain boundary
disrupting the crystalline order. However, silicon binds very easily to oxygen, and
silicon wafers used in electronics typically contain 10
18
atoms/cm
3
oxygen, which
is tolerated and rarely mentioned as it does not disrupt electrical properties. Other
materials such as aluminum or iron cannot be made nearly as pure, and in addition
to traces of other metals and oxygen contain many dislocations. Small mixtures
of other elements in a nearly perfect crystals are called impurities, and when one
wants to call attention to the ways a solid differs from a perfect crystal one calls it
disordered.
Disorder destroys crystalline regularity, and the conditions for Bloch's theorem
no longer apply. One must ask whether the corrections to Bloch's picture are mi-
nor, or whether they produce qualitative changes in the nature of the solid. Both
outcomes are possible, depending on how strongly electrons interact with the dis-
order. Sufficiently strong interactions can turn metals into insulators. Much of the
work in this area was instigated by Mott, and is reviewed in Mott (1990).
An important class of scattering sites in solids are point defects produced either
by
vacancies,
sites where atoms simply are missing, or else by substitutional atoms,
replacing their hosts on randomly selected lattice sites. The effects of point defects
divide into several different classes. Sometimes they have magnetic moments and
can flip the spin of passing electrons. Discussion of this case is deferred to Section
26.6.
The nonmagnetic impurities fall into two additional groups.

Metal-Insulator Transitions in Disordered Solids 531
An impurity is called compensated or isoelectronic when it comes from the
same column of the periodic table as the host crystal. Germanium or tin dissolved
in silicon, or silver dissolved in gold, provide examples. Adding an atom of this
sort amounts to a very localized perturbation on the crystal. The impurity carries a
different number of electrons with it from the host. However, the extra (or missing)
electrons are entirely confined to core states that are tightly bound to the nucleus,
localized in space, and largely inert. Section 18.4 will be dedicated to studying the
properties of electrons in such random potentials.
18.3.2 Non-Compensated Impurities and the Mott Transition
When impurities are not compensated, the electrons they carry cannot all be swept
into a static potential. The effect is as if one has scattered throughout the solid
atomic-sized spots of extra charge. All the conduction electrons in the solid inter-
act strongly with these extra charges. Simple physical considerations based upon
classical electrostatics make it possible to explain some of the main experimental
results in a simple fashion.
Consider adding an atom of phosphorus to silicon. Phosphorus lies just to
the right of silicon in the periodic table, and the perturbation it produces is best
described by adding a single proton to a silicon atom, resulting in a long-range
potential, falling off as \/r. The phosphorus comes accompanied by an extra elec-
tron as well, but this electron must be viewed as a member of the population of
conduction electrons, responding to the potential. Its wave function spreads over
hundreds of adjacent lattice sites.
Precisely because the spread is so large, the nature of the phosphorus impurity
may be captured by a simple semiclassical argument, as shown by Kohn (1957).
The static dielectric constant of silicon e, like that of other semiconductors, is large
and, according to Table 19.1, equals 11.8. The phosphorus nucleus is therefore
strongly screened. In addition, as shown in Figures 10.10 and 23.16, silicon is
technically an insulator. The valence band is filled, and any extra electron must
enter a superposition of states from the conduction band.. The lowest-lying states
behave approximately like free electrons, with the provision that the mass of the
electron be replaced by an effective mass m*, which, according to Table 19.1, is
on the order of 0.2. A proton in empty space binds an electron at radius ao
—
H
2
/me
2
with binding energy £^ = e
2
/2ao (this is a hydrogen atom). For phosphorus
in silicon, the screening of the proton replaces e
2
by e
2
/e, where e is the static
dielectric constant, and the binding energy becomes instead
eh e m* 1
o* = =■ and £* = -— = ~-13.6eV. (18.23)
m*e
z
2ea* m e
1
Because
m*
jm ~ 0.1 and e ~ 10, a* is approximately 100 times larger than the
Bohr radius, and the binding energy is approximately 1000 times smaller than the
binding energy of hydrogen. Some comparisons of
Eq.
(18.23) with actual binding
energies appear in Table 19.2, showing that it successfully captures orders of mag-
nitude despite simplifications such as assuming effective masses to be isotropic.

532 Chapter 18. Microscopic Theories of
Conduction
The large size of a* retroactively justifies using concepts such as dielectric con-
stant and effective mass that are only valid on scales much larger than a lattice
spacing. Because of its small binding energy, phosphorus is called a shallow im-
purity, and because it adds an electron to the conduction band, it is called a donor.
Elements to the left of silicon in the periodic table, such as boron or aluminum,
come with one less electron than the host and may be treated as a weakly bound
positronium atom, where a delocalized hole, built from states at the top of the va-
lence band, is attracted to a screened
—
\/r potential. These elements are called
acceptors. Other impurities, such as gold, produce much more violent changes in
the local electronic environment of silicon than do phosphorus or boron and cannot
be described as hydrogen atoms. They are called deep impurities.
Metal-Insulator Transition in
Si:P.
The subtleties of impurity potentials, as well
as the difficulty of precisely defining the difference between metals and insulators,
are both well illustrated by examining the behavior of silicon as the density of
phosphorus impurities increases. Take phosphorus-doped silicon (Si:P) down to a
temperature on the order of
1
K, so that the chance of thermally exciting a phos-
phorus electron out of
its
bound state becomes negligible. At low densities of
phos-
phorus, the material is nothing but pure silicon, an excellent insulator, and bound
phosphorus electrons, also insulating. As the density of phosphorus increases, the
bound electron wave functions begin to interact, and at a critical density the doped
semiconductor turns into a metal.
This transition cannot be explained by the view that insulators are solids with
filled Bloch bands, while metals have unfilled bands. From such a point of view,
Si:P should always be a metal. Even at low densities, the phosphorus atoms could
be arranged in a periodic way throughout the silicon. Bloch's theorem could then
be brought to bear on the rather large resulting unit cell, which would produce
bands nearly identical to those of silicon, but with the Fermi level moved upwards
to populate the bottom of the conduction band with extra electrons provided by
phosphorus.
However a dilute arrangement of phosphorus in silicon is no more likely to
produce a metal than a lattice of silver atoms, spaced one meter apart. Coulomb re-
pulsion between distant localized electrons produces an insulator that one-electron
theory cannot explain. In fact many of
the
crystalline compounds predicted by one-
electron density functional theory to be metals are in fact insulators. A prototype of
these compounds is CuO, which will be the subject of further discussion in Section
23.6.3.
Experimental realizations of Si:P do not actually array the phosphorus in a
crystalline array, but the basic physics is believed to be similar whether the ar-
rangement of the phosphorus is regular or random, with a simple semiclassical
analysis providing a simple picture of how an insulator gives way to a metal.
Polarization Argument for Mott
Transition.
View Si:P as a cubic lattice of phos-
phorus, treating the silicon as a continuous dielectric medium of dielectric constant
e. When an external electric field is applied to the sample, the phosphorus atoms
