Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.

7.2 Dimensional quantization and low-dimensional structures 203
at the potential barrier can be either reflected or transmitted through the barrier
depending on the ratio of the potential-barrier height and the electron kinetic
energy. Structures with quantum wells as well as barriers have numerous appli-
cations in nanoelectronic devices.
The logical development of one-well and one-barrier nanostructures is the
nanostructures which contain several quantum wells and barriers, as well as
periodic nanostructures with quantum wells separated by narrow barriers. In the
case when such a periodic structure has sufficiently narrow barriers, electrons can
relatively easily tunnel from one quantum well to another and in this way can pass
through the entire structure. Such a periodic structure is called a superlattice.
Usually, the width of the barriers in a superlattice is several nanometers. In
the case of a superlattice we observe more or less three-dimensional behavior
of electrons since the motion of electrons is possible both along the layers
and perpendicular to the layers. At the same time the energy spectrum of a
superlattice is equivalent neither to the structure of energy levels of an individual
quantum well nor to the energy spectrum of the bulk materials which constitute
the quantum wells and potential barriers. As a result of the Pauli exclusion
principle the energy levels of the individual quantum wells develop into one-
dimensional energy minibands. The width and distance between the forbidden
and allowed energy minibands substantially depend on the width of the potential
wells as well as on the width and height of the potential barriers. The system
of electron energy states formed in this way can be controlled also by external
fields. The forbidden miniband is sometimes called a minigap.
In low-dimensional structures the operational range of the external electric
fields which control the energy spectrum is substantially greater than for bulk
materials. Also the temperature at which it is possible to observe most of the
quantum effects is greater. These and many other unique properties of low-
dimensional nanostructures provide the material basis for the development of
modern nanoelectronics.
In the current chapter we will discuss the peculiarities of the discrete energy
spectrum of the electron, placing emphasis on its motion along the direction of
its quantization.
Example 7.2. Using the semiclassical Bohr theory of the hydrogen atom, find
the radius of an electron orbit in the ground state of an ion with positive charge
q = Ze placed in a medium with dielectric constant equal to .
Reasoning. According to Bohr’s postulates, only the orbits which satisfy the
following condition for the angular momentum, m
e
vr
n
, can exist in the hydrogen
atom:
m
e
vr
n
= nh
-
, (7.32)
where n = 1, 2, 3,... is the orbital quantum number. The equation which
describes the rotational motion of the electron around the nucleus is the equation

204 Quantization in nanostructures
of classical mechanics and is defined by Newton’s second law:
m
e
v
2
r
n
=
k
e
Ze
2
r
2
n
, (7.33)
where m
e
is the mass of a free electron, v
2
/r
n
the electron’s centripetal acceler-
ation, v the electron velocity, r
n
the radius of the nth circular orbit, e the charge
of the electron, k
e
= 1/(4π
0
), and
0
= 8.854 × 10
−12
Fm
−1
. Excluding the
electron velocity, v, from Eq. (7.33) and taking into account Eq. (7.32), we get
the following expression for the allowed orbits’ radii, r
n
:
r
n
=
h
-
2
k
e
m
e
Ze
2
n
2
. (7.34)
The radius of the first orbit (for Z = 1), which is called the Bohr radius, is equal
to
r
1
=
h
-
2
k
e
m
e
e
2
= 5.3 × 10
−2
nm. (7.35)
If an atom is placed in a medium with dielectric constant , then the expression
for the Coulomb force on the right-hand side of Eq. (7.33) becomes
F
e
=
k
e
Ze
2
r
2
n
. (7.36)
The decrease of F
e
by a factor of increases the radius of the first orbit by a
factor of . Note that in a medium the dynamics of electron motion is defined not
by the mass of a free electron, m
e
, but by its effective mass, m
∗
. Thus, the radius
of the first orbit of an electron in the ion placed in a medium, r
∗
1
, is defined as
r
∗
1
=
m
e
Zm
∗
r
1
= 5.3 × 10
−2
m
e
Zm
∗
nm. (7.37)
For a typical semiconductor, whose dielectric constant, , is of the order of 10
and in which the effective mass of the electron is m
∗
≈ 0.1m
e
, the radius of the
first Bohr orbit, r
∗
1
, for the hydrogen-like impurity with Z = 1 is, according to
Eq. (7.37), equal to
r
∗
1
≈ 5.3nm, (7.38)
which is a hundred times larger than the radius of the first Bohr orbit in a hydrogen
atom placed in vacuum.
7.3 Quantum states of an electron in
low-dimensional structures
7.3.1 Quantum dots
If the electron motion is restricted in all three directions, i.e., Eq. (7.27)is
satisfied, then the electron energy spectrum becomes totally discrete as in an
isolated atom. Such a system is called a zero-dimensional system or quantum

7.3 Quantum states in low-dimensional structures 205
dot (we can also call it a quantum box if the shape of the nanostructure is close
to a parallelepiped). A quantum dot is analogous to an artificial atom, though
it can consist of a large number of real atoms. As in the case of a real atom,
the quantum dot may contain one or several electrons. It is necessary to keep
in mind one substantial difference between quantum dots and atoms. As we
have discussed in Section 6.4, an atom consists of positively charged protons, an
equal number of negatively charged electrons, and neutrons. The positive charge
of the nucleus, which consists of protons and neutrons, creates an attractive
potential that confines electrons. Very often we refer to such electrons as bound,
since they belong to individual atoms. A quantum dot consists of atoms and the
wavefunctions from different atoms overlap as we discussed in Section 6.6. It
is necessary to distinguish between electrons whose wavefunctions practically
do not overlap (or overlap insignificantly; these electrons can be considered as
bound to the individual atoms) and electrons with substantial overlapping of
wavefunctions. The latter belong simultaneously to a large number of atoms
and can be considered as unbound electrons. The confinement potential for the
unbound electrons is created by the barrier between the quantum dot and the
surrounding material. If a quantum dot contains only one unbound electron, then
we can consider it as an artificial hydrogen atom, if two, as an artificial helium
atom, and so on. The main characteristic of a quantum dot is its energy spectrum,
which depends on many factors, such as the geometrical form of the dot, the
surrounding material that creates potential barriers for the electron confinement,
and the number of unbound electrons.
In Chapter 4 we have already discussed the character of the energy spectrum of
an electron in three-dimensional rectangular and spherically-symmetric potential
wells with barriers of infinite height. The electron energy spectrum in a quantum
dot of cubic form (a cubic box) with the edge length equal to L according to
Eq. (4.18) contains an infinite number of levels defined by the equation
E
n
x
n
y
n
z
=
π
2
h
-
2
2m
∗
L
2
n
2
x
+ n
2
y
+ n
2
z
. (7.39)
The discrete energy levels are determined by the set of three quantum numbers
n
x
, n
y
, and n
z
. The ground state, E
(1)
, corresponds to the lowest electron energy
with n
x
= n
y
= n
z
= 1:
E
(1)
= E
min
= E
111
=
3π
2
h
-
2
2m
∗
L
2
. (7.40)
The next energy level corresponds to three sets of quantum numbers, (112, 121,
211), with the same value for the energy, E
(2)
:
E
(2)
= E
112
= E
121
= E
211
=
3π
2
h
-
2
m
∗
L
2
. (7.41)
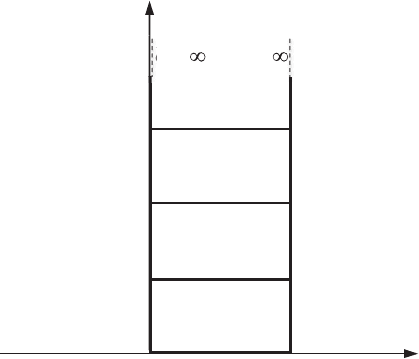
206 Quantization in nanostructures
0
L
x
U
=
E
(2)
E
(3)
E
(1)
U=
E ,U
n
x
n
y
n
z
Figure 7.5 Energy levels
in a quantum dot.
Such a level is called degenerate and has a degeneracy of three, corresponding
to three sets of quantum numbers, whereas the ground state corresponds to a
non-degenerate level with the lowest energy E
min
.
If the size of the edge of a quantum box is equal to L = 5 nm, then the
ground state has energy equal to E
(1)
≈ 0.04 eV, and the first excited state has
energy equal to E
(2)
≈ 0.08 eV. Let us note that in the case of semiconductor
nanostructures in the above-mentioned expressions we have to use the elec-
tron effective mass, m
∗
, which in general is smaller than the mass of a free
electron, m
e
, and leads to a noticeable increase in E
(1)
and E
(2)
. The distance
between lower energy levels for real quantum dots may reach several hundreds
of meV.
The energy levels in a quantum dot are shown schematically in Fig. 7.5,
where for the sake of simplicity we use only one index to denote the energy level
instead of three, i.e., E
(1)
is the lowest allowed energy in the quantum dot and
the index n increases with increasing energy E
(n)
. If the height of the potential
barrier is infinite we have an infinite number of levels in the dot. In reality only
a finite number of energy levels can exist, because the height of the potential
barriers is finite. Note that E
(1)
has a non-zero finite value, i.e., in a quantum
dot the electron cannot be at the bottom of the potential well, so it cannot have
energy equal to zero. The smaller the dot size, the higher is the energy of the
ground level, E
(1)
(see Eq. (7.40)). Only for L →∞do we get E
(1)
= E
min
→ 0.
The distance between levels E
(n)
and E
(n+1)
is finite and it increases as L
decreases (see Eq. (7.39)). For the example given above, the spacing between
the energy levels E
(1)
and E
(2)
is equal to 0.04 eV. Thus, an electron can absorb
finite portions of energy or “quanta” of energy in order for it to be transferred
from lower to higher energy levels. The wavefunction of an electron in a cubic

7.3 Quantum states in low-dimensional structures 207
quantum box corresponds to a standing wavefunction (see Eq. (4.17) with L
x
=
L
y
= L
z
= L):
ψ
n
x
n
y
n
z
(x , y, z) =
8
L
3
sin
πn
x
x
L
sin
πn
y
y
L
sin
πn
z
z
L
, (7.42)
i.e., in this case there is no classical motion of the electron in any direction. The
electron momentum cannot change continuously and the electron kinetic energy
cannot be a continuous function of momentum. Instead we have discrete values
of momentum and discrete (quantized) values of energy.
7.3.2 Quantum wires
If along one of the directions of a nanostructure (for example, along the z-axis)
the size L
z
→∞, then the electron motion along this direction becomes free.
Along the two other directions the electron motion stays quantized. As a result we
get the type of nanostructure called a one-dimensional system or a quantum wire.
We can find the energy spectrum and wavefunction of an electron moving in such
a quantum wire from Eqs. (4.17) and (4.18) if we substitute the wavefunction and
the energy of quantized motion along the z-axis with the term that describes the
free motion of the electron. Suppose that in the x- and y-directions the electron
motion is restricted due to the small size of the structure, i.e., inequalities (7.26)
are satisfied and the rectangular potential barriers are of infinite height, i.e.,
U
0
=∞. For an electron in such a quantum wire we can write
ψ
n
x
n
y
(x , y, z) =
4
L
x
L
y
sin
πn
x
x
L
x
sin
πn
y
y
L
y
e
ik
z
z
, (7.43)
E
n
x
n
y
(k
z
) =
h
-
2
2m
∗
π
2
n
2
x
L
2
x
+
π
2
n
2
y
L
2
y
+
h
-
2
k
2
z
2m
∗
, (7.44)
where h
-
k
z
= p
z
is the corresponding component of the momentum, p, along the
direction of free motion of the electron.
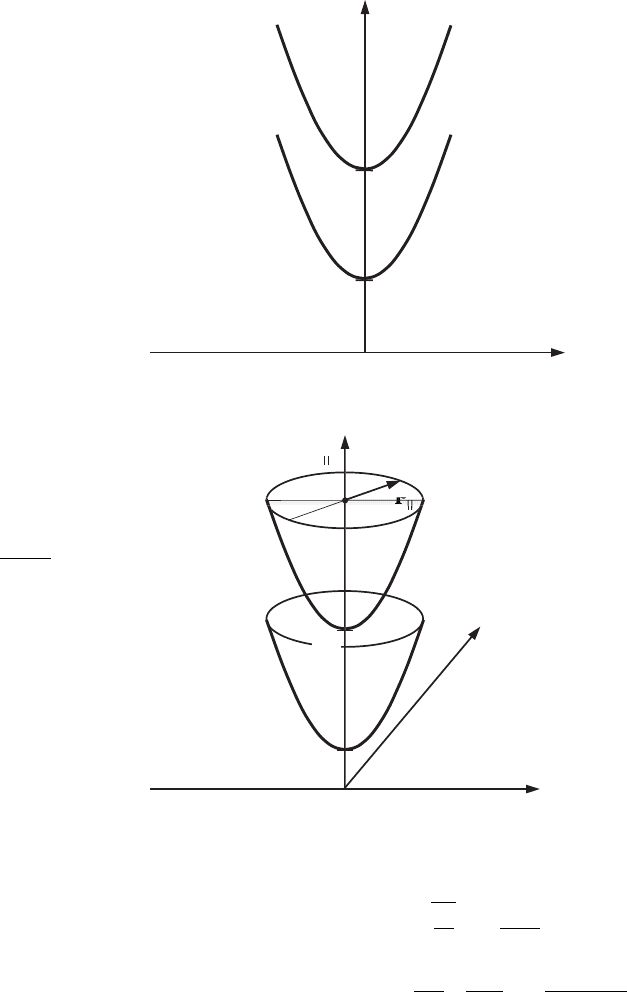
Figure 7.6 shows the energy dependences E
11
(p
z
) and E
12
(p
z
) on the momen-
tum p
z
= h
-
k
z
as well as the quantized levels E
11
(p
z
= 0) and E
12
(p
z
= 0). Note
that, in order to keep the notations less cumbersome, we will henceforth write
E
n
(p) for the energy that depends on momentum, p, while for the energy of
the nth level we will write E
n
without an argument, e.g., E
n
x
,n
y
(p
z
) and E
n
x
,n
y
,
correspondingly, in the case of a quantum wire (see Fig. 7.6).
7.3.3 Quantum wells
If we remove the restriction on electron motion also in the y-direction, i.e., con-
sider L
y
and L
z
infinitely large, then we will obtain the type of nanostructure
called a two-dimensional system or a quantum well. The forms of the wave-
function and energy spectrum of an electron in a rectangular quantum well with
208 Quantization in nanostructures
0
p
z
E
11
E
12
E ( p
z
)
n
x
n
y
Figure 7.6 Quantum wire:
the quantized energy
levels, E
11
and E
12
,as
well as the dependences
of the electron energy
E
n
x
n
y
on the momentum
p
z
defined by Eq. (7.44).
p
p
y
0
p
z
E
1
E
2
E ( )
n
x
p
Figure 7.7 Quantum well:
quantized energy levels,
E
1
and E
2
,andthe
dependences of the
electron energy, E
n
x
,on
the momentum
|p
|=h
--
k
2
y
+ k
2
z
defined
by Eq. (7.46).
barriers of infinite height are defined by the expressions
ψ
n
x
(x , y, z) =
2
L
x
sin
πn
x
x
L
x
e
i(k
y
y+k
z
z)
, (7.45)
E
n
x
(k
y
, k
z
) =
h
-
2
2m
∗
π
2
n
2
x
L
2
x
+
h
-
2
(k
2
y
+ k
2
z
)
2m
∗
. (7.46)
Figure 7.7 shows the dependences of the electron energy, E
n
x
(p
)onthe
lateral momentum |p
| as well as the quantized levels E
1
= [h
-
2
/(2m
∗
)](π
2
/L
2
x
)
and E
2
= 4E
1
.

7.3 Quantum states in low-dimensional structures 209
Equations (7.44) and (7.46) show that the energy spectra of the electron in
a quantum wire and in a quantum well consist of branches called subbands.
The total energy in these subbands is the sum of the energy of dimensional
quantization along two or one of the axes and the kinetic energy along the other
one or two directions.
Example 7.3. For an electron in the ground state in a quantum well with infinite
barriers, find the probabilities of finding the electron in the central region with
coordinates L
x
/4 ≤ x ≤ 3L
x
/4 and in the peripheral regions with coordinates
0 ≤ x ≤ L
x
/4 and 3L
x
/4 ≤ x ≤ L
x
if the width of the quantum well is equal
to L
x
. The central and peripheral regions are of the same width, which is equal
to L
x
/2.
Reasoning. According to Eq. (2.139) the probability of finding an electron in
the ground state in the peripheral regions, P
1
, can be found as
P
1
=
L
x
/4
0
|ψ
1
(x , y, z)|
2
dx +
L
x
3L
x
/4
|ψ
1
(x , y, z)|
2
dx, (7.47)
where the wavefunction ψ
1
has the form (7.45) and
|ψ
1
(x , y, z)|
2
=
2
L
x
sin
2
π x
L
x
.
Owing to the symmetry of the wavefunction ψ
1
(x, y, z) the integrals in Eq. (7.47)
are equal and the expression for the probability, P
1
, takes the form
P
1
= 2
2
L
x
L
x
/4
0
sin
2
π x
L
x
dx = 2
2
L
x
1
2
L
x
/4
0
1 −cos
2π x
L
x
dx
=
1
2
1 −
2
π
. (7.48)
In the central region the probability of finding the electron will be defined by the
same integral with the new limits of integration L
x
/4 and 3L
x
/4. As a result we
get
P
2
=
1
2
1 +
2
π
. (7.49)
Thus, in the ground state the probability, P
2
, of finding the electron in the central
part of the quantum well is larger than the probability, P
1
, of finding it at the
well edge.
Example 7.4. Find the energies of the first five levels and the order of their
degeneracy for an electron confined in a cubic quantum dot with edge size
L = 3 nm. Assume that the potential barriers in the quantum dot are of infinite
height and that the mass of the electron is equal to the free electron mass, m
e
.
Reasoning. According to Eq. (7.39) the ground state corresponds to a single
set of quantum numbers (1, 1, 1). Thus, the degeneracy of the ground state, g
1
,
210 Quantization in nanostructures
is equal to 1. The first excited state corresponds to the three possible sets of
quantum numbers (1, 1, 2), (1, 2, 1), and (2, 1, 1), i.e., the second energy level
has the order of degeneracy, g
2
, equal to 3. The next excited state, i.e., the third
energy level, corresponds to the set of quantum numbers (1, 2, 2), (2, 1, 2), and
(2, 2, 1), and thus its order of degeneracy, g
3
, is equal to 3. The fourth energy level
corresponds to the set of quantum numbers (1, 1, 3), (1, 3, 1), and (3, 1, 1), and
the order of degeneracy is g
4
= 3. Finally, the fifth energy level corresponds to a
single set of quantum numbers, (2, 2, 2), and therefore the order of degeneracy
is g
5
= 1.
On substituting the corresponding combinations of quantum numbers into
Eq. (7.39) we get for the energies of the first five levels the following numbers:
E
(1)
= 0.123 eV; E
(2)
= 0.246 eV; E
(3)
= 0.369 eV;
E
(4)
= 0.451 eV; E
(5)
= 0.492 eV.
The energy levels, E
(n)
, correspond to the following energy levels E
n
x
n
y
n
z
:
E
(1)
= E
111
; E
(2)
= E
112
= E
121
= E
211
; E
(3)
= E
122
= E
212
= E
221
;
E
(4)
= E
113
= E
131
= E
311
; E
(5)
= E
222
.
7.4 The number of states and density of states
for nanostructures
Let us explore how the number of states, N (E), and the density of states, g(E),
change when one, two, or three dimensions of the potential well become compar-
able to the de Broglie wavelength of the electron, i.e., when the electron motion
in these directions becomes confined and the electron energy spectrum cannot
be considered quasicontinuous.
7.4.1 Quantum wells
Let us consider the case when electron motion takes place in a two-dimensional
system – a quantum well. In this case the electron motion is continuous in
two directions (for example, in the y- and z-directions) with macroscopic sizes
of the system, L
y
and L
z
, and is quantized only in the x-direction. The size
of the quantum well in the x-direction, L
x
, is much smaller than L
y
and L
z
:
L
x
L
y
, L
z
. This means that there is a one-dimensional potential, U(x), which
limits electron motion in the x-direction:
U (x ) =
0, 0 ≤ x ≤ L
x
,
∞, x < 0, x > L
x
.
(7.50)
The region of quantization is 0 ≤ x ≤ L
x
. Such electron motion can be con-
sidered quasi-two-dimensional. In this case the electron wavefunctions in the
quantum well are defined by Eq. (7.45) and the energy spectrum by Eq. (7.46),
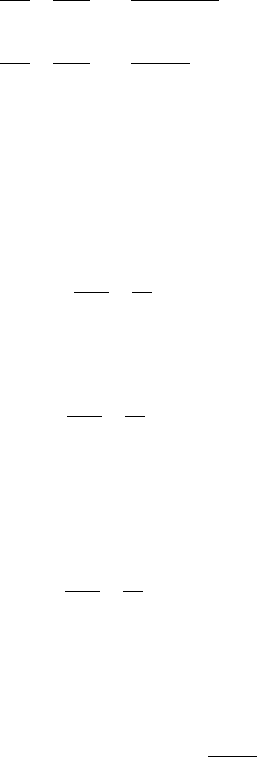
7.4 The number of states and density of states 211
which we will present in the form
E
n
x
(k
y
, k
z
) =
h
-
2
2m
∗
π
2
n
2
x
L
2
x
+
h
-
2
k
2
y
+ k
2
z
2m
∗
,
E
n
x
(p
y
, p
z
) =
h
-
2
2m
∗
π
2
n
2
x
L
2
x
+
p
2
y
+ p
2
z
2m
∗
. (7.51)
In writing this expression we took into account that the wavevector components
are defined by the expressions p
y
= h
-
k
y
and p
z
= h
-
k
z
.FromEq.(7.51) it follows
that the electron energy spectrum in the case of quasi-two-dimensional motion
consists of overlapping subbands with a parabolic dispersion relation as shown in
Fig. 7.7. The lowest energy in each of the subbands corresponds to the subbands’
bottoms with p
y
= p
z
= 0 and energy
E
n
x
= E
n
x
(0, 0) =
π
2
h
-
2
2m
∗
n
x
L
x
2
. (7.52)
The lowest energy that an electron can have in a quantum well corresponds to
n
x
= 1 and is equal to
E
1
= E
1
(0, 0) =
π
2
h
-
2
2m
∗
1
L
x
2
. (7.53)
Electron states with E < E
1
are forbidden. Therefore, for E < E
1
the number of
states is equal to zero, N (E) = 0, and the density of states is also equal to zero,
g(E) = 0. In the energy region E
1
< E < E
2
, where the lowest energy value in
the second subband is equal to
E
2
= E
2
(0, 0) =
π
2
h
-
2
2m
∗
2
L
x
2
, (7.54)
there are states that belong only to the first subband, E
1
(p
y
, p
z
).
Let us calculate the number, N , and density of states, g, in each of the
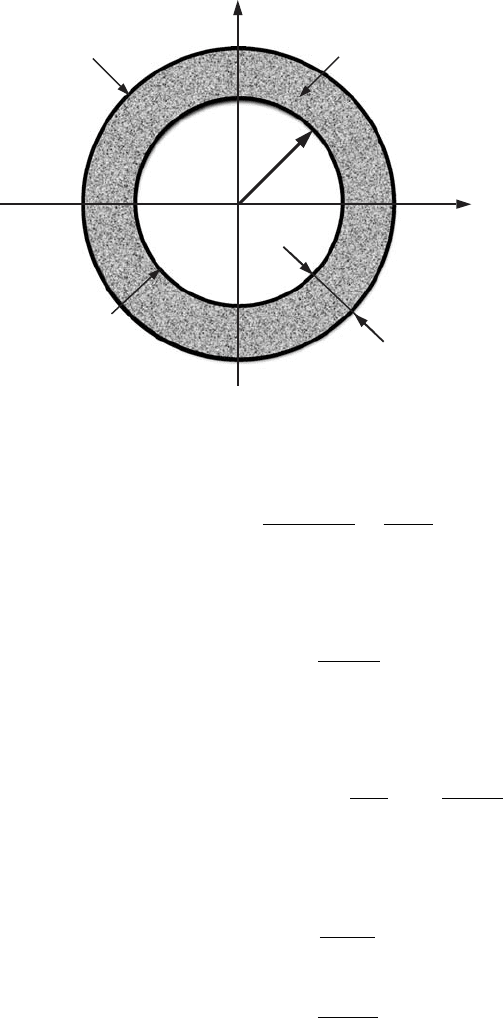
subbands. In order to do this, let us draw the surfaces of equal energy (or
isoenergetic surfaces) for this two-dimensional system in p-space. Let us take
into account that in the case of free three-dimensional electron motion the surface
of equal energy has the form of a sphere of radius p =
√
2m
∗
E. The quantum
limitation in one of the directions, for example in the x-direction, corresponds
to a constant x-component of the momentum p
x
= n
x
π h
-
/L
x
which is caused
by the quantized motion of the electron in the x-direction. Thus, the spherical
surface is divided into a set of circular cross-sections, which are perpendicular
to the axis of quantization (the x-axis) and correspond to the specific values of
the quantum number n
x
. For this kind of confinement the surface of equal energy
is a circle of radius p withagivenn
x
(see Fig. 7.8). To estimate the number of
quantum states, let us choose a circular segment with radius p and width d p,
which has an area
dS
p
= 2π p d p. (7.55)
212 Quantization in nanostructures
p
y
p
z
E
E + dE
0
p
dp
dS
p
Figure 7.8 A surface of
equal energy, E,ina
two-dimensional p-space
for a given n
x
.
The number of quantum states corresponding to this area, dS
p
, and to the area
available for an electron, S
r
= L
y
L
z
, is given by the expression
dn(p) =
S
r
× 2π p d p
(2π h
-
)
2
=
S
r
p d p
2π h
-
2
. (7.56)
Taking into account that the energy of a free electron inside the potential
well depends on the momentum quadratically, E = p
2
/(2m
∗
), we can get from
Eq. (7.56)
dn(E) =
S
r
m
∗
dE
2π h
-
2
. (7.57)
Now, we can find the total number of states in the energy interval (0, E) by inte-
gration of Eq. (7.57) within the limits of this interval, 0 and E. After integration
we arrive at the following expression:
N
0
(E) = 2
dn(E) = 2
E
0
S
r
m
∗
2π h
-
2
dE =
m
∗
L
y
L
z
π h
-
2
E. (7.58)
In Eq. (7.58) we multiplied the integral by 2 because the spin of an electron
can have two values, +1/2 and −1/2. Taking into account the definition of the
density of states,
g
0
(E) =
dN
0
(E)
dE
,
we find
g
0
(E) =
m
∗
L
y
L
z
π h
-
2
. (7.59)
From Eqs. (7.58) and (7.59) it follows that in the case of two-dimensional electron
motion the number of quantum states increases linearly with the energy and that
the density of states does not depend on energy at all.
