Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.

Chapter 7
Quantization in nanostructures
In Chapters 3 and 4 we have discussed electron behavior in potential wells of
various profiles and dimensionalities. We have established that localization of
electrons in such potential wells, regardless of their form, leads to the discretiza-
tion of the electron energy spectrum whereby the distance between energy levels
substantially depends on the geometrical size of the potential wells. If this size
is macroscopic then the distance between the energy levels is so small that we
can consider the energy spectrum to be practically continuous (or quasicontinu-
ous). Electrons in metallic samples of macroscopic sizes have this kind of energy
spectrum. Another limiting case is that of small clusters consisting of just a few
atoms, where the distance between energy levels is of the order of electron-volts.
Gradual decrease of one or several geometrical dimensions of the potential well
from macroscopic to about 1 µm practically does not change the form of the
electron energy spectrum. Very often macroscopic materials (or macroscopic
crystals) are referred to as bulk materials or bulk crystals. Changes happen
only when the size of structures is of the order of or less than 100 nm. Such
structures are called nanostructures. The change of the electron spectrum from
quasicontinuous to discrete implies changes in most of the physical properties
of nanostructures compared with those in bulk crystals. In this chapter we will
consider the main peculiarities of the electron energy spectrum in nanostructures
of various dimensionalities.
7.1 The number and density of quantum states
Electric, optical, thermal, and other properties of macroscopic crystalline mater-
ials significantly depend on the energy states of electrons. Let us define two
quantities, which are important for describing the features of the electron energy
spectrum in these materials. The first quantity, N (E), is the number of quantum
states corresponding to the energy interval (0, E). The second quantity, g(E),
called the density of states, defines the number of quantum states, N , correspond-
ing to the unit energy interval in the vicinity of E. In order to calculate these
quantities we consider first a three-dimensional potential well, which represents
193

194 Quantization in nanostructures
bulk crystal. Such a structure is called three-dimensional because of the num-
ber of dimensions along which the electron can freely move. In such a case the
electron momentum space is also three-dimensional. For the analysis of elec-
tron behavior in a crystal in an external field an approximate method, which is
called the effective-mass approximation, is widely used. In this approximation the
Schr¨odinger equation for an electron with free electron mass, m
e
, in the periodic
inner-crystal potential is reduced to the Schr¨odinger equation which describes
the behavior of free electrons with mass equal to the so-called effective mass, m
∗
.
We will show in Section 7.6.3 how to define the effective mass of an electron.
To find how electrons are distributed over the allowed quantum states in a
crystal with linear dimensions L
x
, L
y
, and L
z
, where they can freely move, we will
assume the boundary conditions (4.4): ψ (0, y, z) = ψ(x, 0, z) = ψ(x, y, 0) =
ψ(L
x
, y, z) = ψ (x, L
y
, z) = ψ(x, y, L
z
) = 0. The components of momentum
according to Eq. (4.15) can take the following values:
p
x
=
π h
-
L
x
n
x
, p
y
=
π h
-
L
y
n
y
, p
z
=
π h
-
L
z
n
z
, (7.1)
where n
α
=1, 2, 3, . . . and α =x, y, and z. We will consider first the case of large
L
x
, L
y
, and L
z
. Thus, the spacing between components of the momentum, p
α
=
π h
-
/L
α
, is small and we can consider the momentum, p,tobequasicontinuous.
Therefore, in order to calculate the number of states we can use integration over
p instead of summation.
Let us choose an infinitesimally small volume of momentum space, dV
p
,
around some momentum p:
dV
p
= d p
x
d p
y
d p
z
. (7.2)
It is easy to carry out calculations for a macroscopic crystal, i.e., for macroscopic
values of L
x
, L
y
, and L
z
. In this case the change of momentum projections, p
x
,
p
y
, and p
z
, is practically continuous and the electron motion can be considered
as a classical motion. Taking into account Eq. (7.1), we obtain the following
expression for the quantum numbers n
α
, which define the number of quantum
states:
n
α
=
L
α
π h
-
p
α
, (7.3)
where α = x, y, and z. On differentiating Eq. (7.3), we find the number of
quantum states dn
α
in the interval dp
α
:
dn
α
=
L
α
π h
-
d p
α
. (7.4)
The number of quantum states is equal to the number of sets of n
x
, n
y
, and
n
z
in this three-dimensional interval of numbers dn = dn
x
dn
y
dn
z
.Wehaveto
take into account the fact that each value of n
α
corresponds to the two values
of p
α
with the same absolute value, but with opposite signs. Therefore, the
total number of states in the three-dimensional interval of momentum space is

7.1 The number and density of quantum states 195
equal to
dn =
L
x
L
y
L
z
(2π h
-
)
3
d p
x
d p
y
d p
z
. (7.5)
In this expression the physical quantity V = L
x
L
y
L
z
represents the volume in
real space which is available for an electron, and the quantity dV
p
= d p
x
d p
y
d p
z
represents the differential volume element in the momentum space available for
an electron. In the case of a free electron the momentum space is isotropic
since all directions are equivalent. Therefore, for the differential volume of the
momentum space, we can use spherical coordinates and write
dV
p
= d p
x
d p
y
d p
z
= 4π p
2
d p. (7.6)
Taking into account all the above, the expression (7.5) can be rewritten as
dn(p) =
V
2π
2
h
-
3
p
2
d p. (7.7)
The total number of states in the momentum space of radius p is defined as
follows:
N ( p) = 2
dn(p) =
V
π
2
h
-
3
p
0
p
2
d p =
V
3π
2
h
-
3
p
3
. (7.8)
Note that we put a factor of 2 in front of the integral in Eq. (7.8). This is because
the set of quantum numbers n
x
, n
y
, and n
z
does not completely define the state
of an electron. Because the electron has its own internal angular momentum
(spin) (see Section 6.3), the quantum state of the electron has to be defined by an
additional quantum number m
s
:
m
s
=±
1
2
. (7.9)
According to the Pauli exclusion principle, two electrons with opposite spins can
be in a state with the same momentum p. Therefore, the number of quantum
states is twice as large as it otherwise would be.
The number of states that corresponds to the unit interval of momentum space
is given by the density of states, g(p):
g(p) =
dN ( p)
d p
=
p
2
V
π
2
h
-
3
. (7.10)
We see that with increasing momentum, p, the number of states, N (p), increases
proportionally to the momentum raised to the third power, and that the density
of states, g( p), increases proportionally to the square of the momentum.
In many cases it is more convenient to consider energy, E, rather than momen-
tum, p, as a variable. In order to do this, we have to perform a transformation
of the equations from the momentum space to the energy space. Taking into
account that the energy of a free electron has a quadratic dependence on the
momentum, the electron surface of equal energy in the momentum space, p,isa
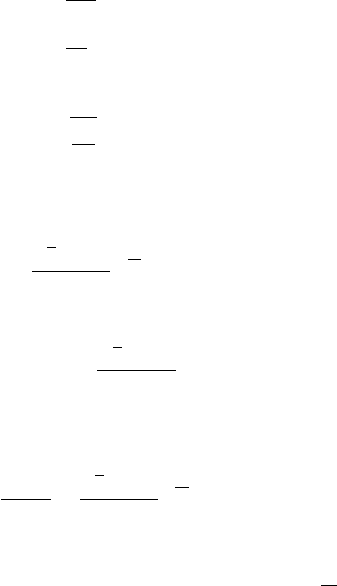
196 Quantization in nanostructures
sphere of radius p:
E =
p
2
2m
∗
, (7.11)
dE =
p
m
∗
d p, (7.12)
and
d p =
m
∗
2E
dE . (7.13)
By substituting Eqs. (7.11) and (7.13) into Eq. (7.7) we obtain the following
expression for the number of states in the energy interval dE:
dn(E) =
√
2Vm
∗
3/2
2π
2
h
-
3
√
E dE. (7.14)
The total number of quantum states in the interval (0, E) is defined by the integral
N (E) = 2
dn(E) = 2
√
2Vm
∗
3/2
3π
2
h
-
3
E
3/2
. (7.15)
For the density of states, g(E), i.e., the number of states that corresponds to the
unit energy interval, we obtain
g(E) =
dN (E)
dE
=
√
2Vm
∗
3/2
π
2
h
-
3
√
E. (7.16)
We see that the number of states, N (E), for the electron in a three-dimensional
space of macroscopic volume V increases with increasing energy proportionally
to E
3/2
, and that the density of states, g(E), increases proportionally to
√
E.Both
quantities depend linearly on the volume, V , and, therefore, they can be defined
per unit volume. In this case, the volume V in Eqs. (7.15) and (7.16) is canceled
out. Figure 7.1 shows the electron’s surface of equal energy in the momentum
space, E(p), and Fig. 7.2 shows the dependences of the number of states, N , and
the density of states, g, on energy, E, for the electron’s three-dimensional motion
in a macroscopic crystal.
Note that Eqs. (7.2)–(7.16) can be used only in the case of crystals with
macroscopic volume V . All three dimensions, L
x
, L
y
, and L
z
, are so large that
the differences between different values of electron momentum and energy are
small, and thus the magnitudes p and E can be considered as continuous (or
quasicontinuous) variables.
If there are N electrons in the volume V , then at zero temperature they occupy
the lowest energy states and on each level there are two electrons with opposite
spins defined by Eq. (4.18).
The distribution of electrons among the energy levels at an arbitrary temper-
ature, T , is described by Fermi–Dirac quantum statistics for particles with spin
m
s
=±1/2. The probability of finding an electron in the state with energy E is
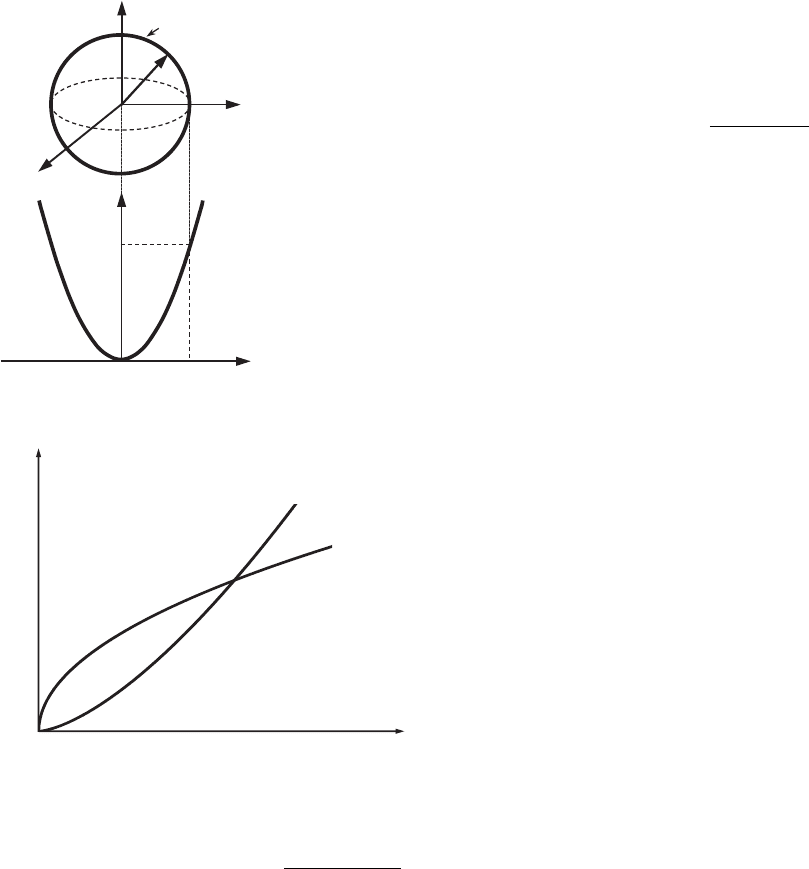
7.1 The number and density of quantum states 197
E(p)
p
y
p
x
p
z
p
0
E
||
E(p)
p
p
Figure 7.1 The surface of
equal energy in the
momentum space for the
electron’s
three-dimensional
motion. Here,
|p|=
p
2
x
+ p
2
y
+ p
2
z
.
0
N
, g
E
g(E) ~ E
1/2
N(E) ~ E
3/2
Figure 7.2 The
dependences of the
number, N(E), and the
density of states, g(E), on
energy, E, in the case of
an electron’s
three-dimensional motion
in a crystal.
given by the Fermi–Dirac distribution function, f (E):
f (E) =
1
e
(E−E
F
)/(k
B
T )
+ 1
, (7.17)
where E
F
is the Fermi energy and k
B
is Boltzmann’s constant. From Eq. (7.17)
it follows that at T = 0 K the probability of occupation of all the states with the
energy E ≤ E
F
is equal to unity, and the probability of occupation of the states
with energy E > E
F
is equal to zero (see Fig. 7.3). Thus, the Fermi energy, E
F
,
defines the energy up to which all the energy states at T = 0 K are occupied.
Energy states higher than E
F
at T = 0 K are unoccupied.
Given the importance of the notion of the Fermi energy, let us determine E
F
at T = 0 K. Let us assume that there are N electrons in the volume V . Since

198 Quantization in nanostructures
f (E)
E
0
E
F
1
T = 0 K
0.5
T
2
T
1
T
2
> T
1
Figure 7.3 The
Fermi–Dirac distribution
function, f (E), for various
temperatures. The
distribution functions at
temperatures higher than
0Kareshownbydashed
and dash–dotted lines.
Here, T
2
> T
1
.
two electrons with opposite spins occupy each available state, the number of
electrons, N , that can be placed in all states from E = 0toE = E
F
is equal to
the number of states N (E
F
). According to Eq. (7.15) this number is related to
the Fermi energy, E
F
,by
N = N (E
F
) = 2
√
2Vm
∗
3/2
3π
2
h
-
3
E
3/2
F
. (7.18)
From the above equation we obtain the following expression for the Fermi energy:
E
F
=
h
-
2
2m
∗
3π
2
N
V
2/3
. (7.19)
Let us estimate E
F
for the conduction electrons in a metal, for example in
copper. For copper the electron effective mass is close to the free electron mass:
m
∗
≈ m
e
. The electron density for copper is equal to ρ = N/V ≈ 10
29
m
−3
.
Thus, the Fermi energy is about several electron-volts and the distance between
energy levels is infinitesimally small compared with E
F
. Therefore, the energy
spectrum of the electron gas in metals can be considered to be quasicontinuous.
Example 7.1. An electron is confined in a cubic box with impenetrable walls
and with the edge lengths L = 1 µm. Find the distance between the levels E
113
and E
122
, and compare this energy with the electron thermal energy, E
th
,atroom
temperature:
E
th
=
3
2
k
B
T ≈ 38.4meV. (7.20)
Determine whether the quantum-mechanical approach is required or the clas-
sical approach with a quasicontinuous spectrum is applicable. Assume that
the mass of the electron in the box is equal to the electron mass in vacuum,
m
e
= 9.1 × 10
−31
kg.
7.2 Dimensional quantization and low-dimensional structures 199
Reasoning. According to Eq. (4.18), the energy level E
122
has the same mag-
nitude as the other two states with the sets of quantum numbers (2, 1, 2) and
(2,2,1):
E
122
= E
212
= E
221
=
π
2
h
-
2
2m
e
L
2
(1
2
+ 2
2
+ 2
2
). (7.21)
The energy level E
113
has the same magnitude as the other two states with the
sets of quantum numbers (1, 3, 1) and (3, 1, 1):
E
113
= E
131
= E
311
=
π
2
h
-
2
2m
e
L
2
(1
2
+ 1
2
+ 3
2
). (7.22)
The distance between the energy levels E
113
and E
122
is
E
113
− E
122
=
π
2
h
-
2
m
e
L
2
= 8.75 × 10
−7
eV = 8.75 × 10
−4
meV. (7.23)
According to Eq. (7.23), in the cubic box of dimensions 1 µm × 1 µm × 1 µm,
the distance between energy levels is so small in comparison with the thermal
energy at room temperature of 38.4 meV (see Eq. (7.20)) that we can consider
the electron energy spectrum to be quasicontinuous. It follows from the above
expression that in order to increase the distance between the adjacent energy
levels it is necessary to decrease the size of the region within which the electron
is confined.
7.2 Dimensional quantization and
low-dimensional structures
As we have shown previously, the quantum properties of an electron become
apparent when the electron de Broglie wavelength becomes comparable to the
size of the region within which the electron motion takes place. For estimates
of this region we have to take into account that electron motion takes place not
in vacuum but in a certain medium at a certain temperature. Therefore, we have
to replace the mass of the electron in vacuum, m
e
, by its effective mass in a
medium, m
∗
. In Section 7.6.3 we will present a detailed discussion about the
effective mass of an electron, but for now we note that the electron motion in a
crystalline material is not completely free since it takes place under the influence
of the inner-crystal potential of atoms. An electron effective mass is introduced
to take into account the crystal potential and, in many practical cases, to describe
the motion of an electron in a medium, under the influence of external fields,
using the conventional equations in which the free-electron mass, m
e
, is replaced
by the effective mass, m
∗
, of an electron in the particular medium.
We will use the effective de Broglie wavelength, λ
∗
, of an electron which is
connected with its effective mass, m
∗
, in a crystalline material, as a criterion for
200 Quantization in nanostructures
B
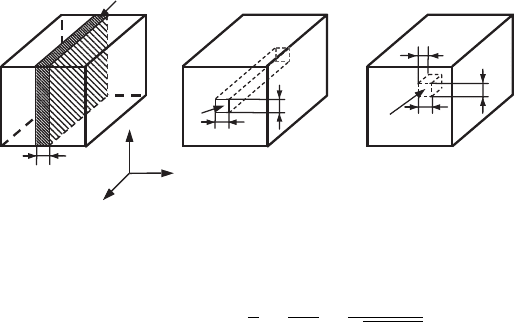
(a)
AA
(b) (c)
BB
B
A
L
x
x
y
z
L
x
L
y
L
x
L
y
L
z
Figure 7.4 Different types
of nanostructures: (a) a
quantum well, (b) a
quantum wire, and (c) a
quantum dot. A and B
denote the materials
which constitute the
nanostructures, with A
being the material of the
nanostructure itself and B
being the material of the
surrounding matrix. The
nanostructure is referred
to as free-standing if the
surrounding matrix, B, is
vacuum.
a space quantization:
λ
∗
=
h
p
=
2π h
-
m
∗
v
=
2π h
-
√
3m
∗
k
B
T
. (7.24)
In the case when electron motion along only one direction is limited (for example,
along the x-direction) (see Fig. 7.4(a)), i.e., when the condition
L
x
≤ λ
∗
(7.25)
is satisfied, the electron energy states corresponding to the motion in this direction
are quantized. At the same time the electron motion in the other two directions
(y- and z-directions) stays free, with the continuous energy spectrum. Such a
structure is called a quantum well (QW).
In the case when the electron motion is restricted along two directions (for
example, along the x- and y-directions), i.e., when the conditions
L
x
≤ λ
∗
and L
y
≤ λ
∗
(7.26)
are satisfied, the energy which corresponds to these two directions of motion is
quantized. Such a structure is called a quantum wire (QWR) (see Fig. 7.4(b)).
In the case when the electron motion is restricted along all three directions,
i.e., when the conditions
L
x
≤ λ
∗
, L
y
≤ λ
∗
, and L
z
≤ λ
∗
(7.27)
are satisfied, the energy spectrum of such a structure, which is called a quantum
dot (QD) (see Fig. 7.4(c)), is totally quantized. Quantum dots can take the form
of a cube, a short cylinder, a disk, or a sphere of nanometer size, and they have
the potential to become in the future the main building blocks of nanoelectron-
ics. For example, from a set of quantum dots we can make artificial crystals
with desired parameters. In semiconductor crystals, which are used for building
nanostructures, the ratio of the electron mass in vacuum, m
e
, to its effective
mass in a semiconductor, m
∗
, is greater than unity. For example, for GaAs,

7.2 Dimensional quantization and low-dimensional structures 201
which is one of the most widely used semiconductors in nanoelectronics, this
ratio is approximately equal to 10: m
e
/m
∗
≈ 10. Therefore, at room tempera-
ture the de Broglie wavelength of an electron in GaAs is of the order of 10 nm
according to Eq. (7.24). This defines in accordance with Eqs. (7.25)–(7.27)the
size of structures at which the quantization of the electron spectrum becomes
noticeable. Note that on lowering the temperature by a factor of 100, i.e., at
temperatures of about 3 K, λ
∗
increases by one order of magnitude. Therefore,
at 3 K the quantization is important in structures with sizes of the order of
100 nm.
The simplest nanostructure with the quantum limitation along one of the
directions, the quantum well, is a thin semiconductor film with a thickness of
about L
x
∼ λ
∗
. The electrons are mostly localized in the film and cannot escape
the film because the energy of an electron outside of the film is higher than the
energy of an electron within the film, i.e., there is a potential barrier for the
electrons outside of the film. The height of the potential barrier that forms a
quantum well for electrons in the film is equal to the work function, A
wf
, which
for most semiconductors is about 3–5 eV. This energy is two orders of magnitude
higher than the energy of thermal motion of free electrons, E
th
= 3k
B
T/2. At
room temperature, E
th
is about 4 ×10
−2
eV and E
th
decreases linearly with
decreasing temperature, T .
We can estimate the ground-state energy of an electron in a quantum well
using the uncertainty relationships. In a direction of restricted motion the electron
momentum projection is equal to p
x
∼ p
x
∼ h/L
x
. Therefore, for the energy
of the ground state we get
E
1
∼
p
2
x
2m
∗
∼
h
2
2m
∗
L
2
x
. (7.28)
In a GaAs quantum well with L
x
= 20 nm and with the effective mass of
the electron equal to m
∗
= 0.067m
e
the energy of the ground state is equal to
E
1
≈ 0.1 eV. The distances between the adjacent lower energy levels are of the
same order of magnitude. The potential well formed in the x-direction does not
affect the electron motion in the yz-plane of the semiconductor film. The electron
energy spectrum corresponding to this motion is continuous and quadratic with
respect to momentum, as it is in the case of free electron motion. The total energy
of an electron in such a thin semiconductor film has a mixed discrete–continuous
character:
E = E
n
+
p
2
y
+ p
2
z
2m
∗
, (7.29)
where E
n
is the energy of the nth level of the quantized motion in the x-direction.
Because of the continuity of the spectrum along the other two directions (the y-
and z-directions) electrons that belong to the nth quantum state may have total
energy, E, equal to any value within the interval E
n
≤ E < ∞. Such a set of

202 Quantization in nanostructures
quantum states for the given n is usually called the subband of dimensional
quantization.
In order to observe the quantization of the energy spectrum experimentally,
the distance between the adjacent energy levels has to be sufficiently large. First
of all it must be larger than the thermal energy of electrons in a quantum well,
i.e.,
E
n+1
− E
n
> k
B
T. (7.30)
This condition must be satisfied in order to exclude the possible thermal tran-
sitions of the electron between quantized energy levels which will hinder the
observation of the quantum-dimensional effects (QDEs). Second, for observa-
tion of QDEs the mean free path of the electron, l
e
, in the medium must be
substantially larger than the size of the region of quantized motion. This is
because the quantization takes place only if the electron wavefunction has the
form of a standing wave in the region of electron motion. Such a regularity
of the wavefunction is possible only in the case of weak electron scattering on
vibrations of atoms of the films as well as on defects, which are always present in
real structures and destroy the coherent character of the electron motion. To have
such a regularity, an electron in the quantum state with momentum p
n
must have
a chance to make several flights from one wall of the quantum well (quantum
wire or quantum dot) to another during its time of free flight, i.e., during the time
between two collisions:
τ
e
=
l
e
v
n
, (7.31)
where v
n
is the characteristic velocity that corresponds to momentum p
n
,
given by
v
n
=
p
n
m
∗
.
This may be possible only in the case of l
e
L, where L is the characteristic
size of a QW, QWR, or QD. Let us note that criterion (7.30) can be applied to
semiconductors in which the electron concentration is low and a small number
of levels E
n
is filled. Metallic nanostructures are not suitable for the observation
of QDEs because, due to the large number of electrons, the energy of conduction
electrons in typical metals is several electron-volts, which is much larger than
the energy distance between the levels of dimensional quantization. Therefore,
nowadays semiconductor thin-film structures are used for the observation of
QDEs. It is worth noting that the first experiments in which the quantization of
the electron spectrum was observed were carried out with semimetallic films of
bismuth (first of all the effective mass of electrons in bismuth, Bi, is very small
and second, it is easy to grow thin films of bismuth by laser ablation).
Alongside structures containing quantum wells, structures containing a poten-
tial barrier with a width of about λ
∗
are also of great interest. An electron incident
