Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.


7.4 The number of states and density of states 213
Now let us take into account that such motion takes place also in the other
energy subbands. The number of states, N
0
(E ), and density of states, g
0
(E ), in
the first subband are defined by Eqs. (7.58) and (7.59). Taking into account that
there are no states below energy state E
1
, they can be written as
N (E) = N
0
(E − E
1
) =
m
∗
L
y
L
z
π h
-
2
(E − E
1
), (7.60)
g(E) =
dN (E)
dE
=
m
∗
L
y
L
z
π h
-
2
. (7.61)
In the energy interval E
2
< E < E
3
the number and density of states are
equal to the sum of states of the first two subbands, i.e.,
N (E) = N
0
(E − E
1
) + N
0
(E − E
2
) =
m
∗
L
y
L
z
π h
-
2
(2E − E
1
− E
2
), (7.62)
g(E) =
2m
∗
L
y
L
z
π h
-
2
. (7.63)
For an energy in the interval E
n
< E < E
n+1
each of the n subbands will have
its input in the magnitudes of N (E) and g(E). Therefore, within this energy
interval, N (E) and g(E) can be defined as follows:
N (E) =
m
∗
L
y
L
z
π h
-
2
(nE − E
1
− E
2
−···−E
n
), (7.64)
g(E) =
nm
∗
L
y
L
z
π h
-
2
. (7.65)
Thus, the number of states, N (E), is a continuous function consisting of linear
segments with slope increasing from region to region. The angle of slope, α
n
,is
defined as
α
n
= arctan
nm
∗
L
y
L
z
π h
-
2
.
The density of states, g(E), is a step-function, which undergoes jumps,
g
n
=
m
∗
L
y
L
z
π h
-
2
,
each time the energy of the electron becomes equal to the bottom of the next
subband, i.e., when E = E
n
= n
2
E
1
. Figure 7.9 shows the dependences N (E)
and g(E) for the quantum well.
Very often these quantities are defined for the unit area of the two-dimensional
system. In this case the obtained expressions for the number and density of states
must be divided by the magnitude S
r
= L
y
× L
z
:
N (E) =
m
∗
π h
-
2
(nE − E
1
− E
2
−···−E
n
), (7.66)
g(E) =
nm
∗
π h
-
2
. (7.67)

214 Quantization in nanostructures
g
g
3D
N
(a) (b)
0 E
1
E
2
E
3
E 0 E
1
E
2
E
3
E
Figure 7.9 The
dependences of (a) the
number of states, N(E),
and (b) the density of
states, g(E), in a
rectangular quantum well
with barriers of infinite
height. Here g
3D
is the
electron density of states
in bulk
(three-dimensional)
material.
7.4.2 Quantum wires
Let us now consider electron motion in a one-dimensional system – a quantum
wire, where electron motion is continuous along only one of the directions (for
example, along the z-direction). The size of the region where the continuous
motion takes place, L
z
, is macroscopic; in the other two directions (along the
x- and y-directions) the motion is quantized. Therefore, the length of the quan-
tum wire, L
z
, satisfies the following inequality: L
x
, L
y
L
z
. This means that
the electron motion takes place in the inner-crystal periodic potential with an
additional two-dimensional potential, U(x, y), which limits electron motion in a
crystal in the x- and y-directions:
U (x , y) =
0, 0 ≤ x ≤ L
x
, 0 ≤ y ≤ L
y
,
∞, x < 0, y < 0,
∞, x > L
x
, y > L
y
.
(7.68)
Such electron motion can be considered quasi-one-dimensional. The wavefunc-
tions are defined by Eq. (7.43) and the electron energy spectrum by Eq. (7.44),
which we will present in the following form:
E
n
x
n
y
(k
z
) =
h
-
2
π
2
2m
∗
n
2
x
L
2
x
+
n
2
y
L
2
y
+
h
-
2
k
2
z
2m
∗
=
h
-
2
π
2
2m
∗
n
2
x
L
2
x
+
n
2
y
L
2
y
+
p
2
z
2m
∗
. (7.69)
The electron spectrum in a quantum wire consists of subbands with a one-
dimensional parabolic dispersion relation E
n
x
n
y
(p
z
) (see Fig. 7.6). The electron
motion along the z-direction is “free” and its dynamic properties are defined by
the effective mass, m
∗
. The minimal energy in each of the subbands corresponds
to their bottom with p
z
= 0:
E
n
x
n
y
= E
n
x
n
y
(0) =
h
-
2
π
2
2m
∗
n
2
x
L
2
x
+
n
2
y
L
2
y
. (7.70)
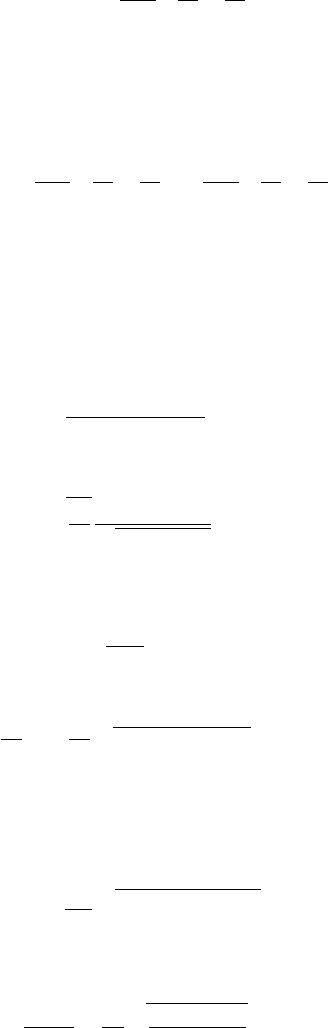
7.4 The number of states and density of states 215
The lowest energy which an electron can have in a quantum wire is equal to
E
11
= E
(1)
= E
11
(0) =
π
2
h
-
2
2m
∗
1
L
2
x
+
1
L
2
y
. (7.71)
Electron states are forbidden for energies lower than E
(1)
. Therefore, for all
energies E < E
(1)
the number and density of states in a quantum wire are equal
to zero, i.e., N (E) = 0 and g(E) = 0. In the energy interval E
(1)
< E < E
(2)
,
where the minimum energy in the second subband is equal to
E
(2)
= E
12
(0) = E
21
(0) =
π
2
h
-
2
2m
∗
1
L
2
x
+
2
2
L
2
y
=
π
2
h
-
2
2m
∗
2
2
L
2
x
+
1
L
2
y
, (7.72)
there exist states that belong only to the first subband E
11
(p
z
).
Let us find the number and density of states in each of the subbands. For quasi-
one-dimensional “free” motion in the z-direction in the considered potential well
with infinite barriers, the electron energy spectrum can be considered practically
continuous (see Example 7.5). Taking into account Eq. (7.69), the expression for
the z-component of the electron momentum in the corresponding subband can
be written as follows:
p
z
=
2m
∗
[E − E
n
x
n
y
(0)]. (7.73)
On differentiating Eq. (7.73) we get
d p
z
=
m
∗
2
dE
E − E
n
x
n
y
(0)
. (7.74)
Since the electron momentum in the potential well with infinite barriers is
defined as
p
z
=
π h
-
n
z
L
z
,
from Eq. (7.73) we find the number of energy levels:
n
z
=
L
z
π h
-
p
z
=
L
z
π h
-
2m
∗
[E − E
n
x
n
y
(0)]. (7.75)
We have already mentioned that the total number of quantum states is twice as
large as the number of energy levels, i.e., N (E) = 2n
z
. This is because of Pauli’s
principle for particles with half-integer spin, according to which on each energy
level there can be two electrons with opposite spins. Thus,
N (E) = 2n
z
=
2L
z
π h
-
2m
∗
E − E
n
x
n
y
(0)
. (7.76)
Let us find now the density of states, g(E), i.e., the number of states in the energy
interval dE:
g(E) =
dN (E)
dE
=
L
z
π h
-
2m
∗
E − E
n
x
n
y
(0)
. (7.77)
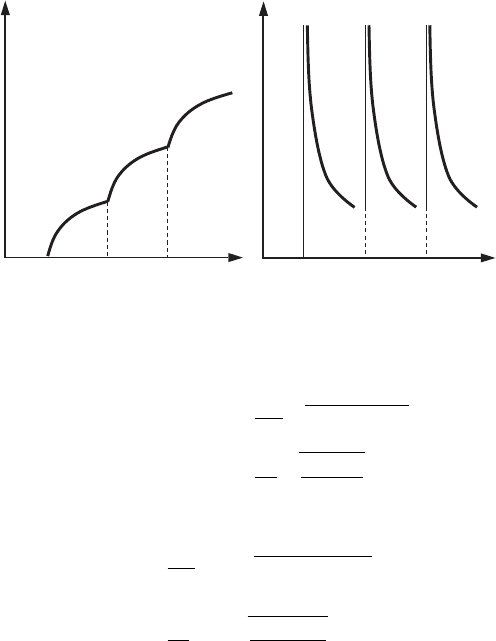
216 Quantization in nanostructures
0
g
E
0
N
E
(a) (b)
E
(1)
E
(2)
E
(3)
E
(1)
E
(2)
E
(3)
Figure 7.10 The
dependences of (a) the
number of states, N(E),
and (b) the density of
states, g(E), in the
rectangular quantum wire
with barriers of infinite
height.
Thus, in the energy interval E
(1)
< E < E
(2)
the number and density of states
are defined only by the states of the first subband and are given by the expressions
N (E) =
2L
z
π h
-
2m
∗
(E − E
11
), (7.78)
g(E) =
L
z
π h
-
2m
∗
E − E
11
. (7.79)
In the general case, for an energy E we get
N (E) =
2L
z
π h
-
n
x
,n
y
2m
∗
(E − E
n
x
n
y
) × (E − E
n
x
n
y
), (7.80)
g(E) =
L
z
π h
-
n
x
,n
y
2m
∗
E − E
n
x
n
y
× (E − E
n
x
n
y
). (7.81)
Here we introduced the Heaviside function (or the step-function):
(t) =
0, t < 0,
1, t ≥ 0.
(7.82)
The function N (E) is continuous, but at the points E = E
n
x
n
y
its derivative,
i.e., the function g(E), becomes discontinuous. The number of states within the
limited energy interval is always limited. Figure 7.10 shows the dependences
N (E) and g(E) for electron motion in a quantum wire.
7.4.3 Quantum dots
Let us consider now the electron motion in a zero-dimensional system or a
quantum dot, i.e., in a potential well whose dimensions in all three directions
are comparable to the de Broglie wavelength. In this case the electron energy
spectrum is purely discrete. For a quantum dot with L
x
= L
y
= L
z
the number
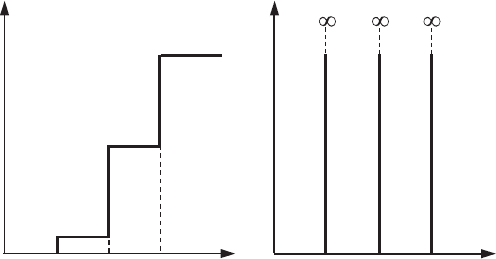
7.4 The number of states and density of states 217
g
N
E0 E
(1)
E
(2)
E
(3)
(a) (b)
E0 E
(1)
E
(2)
E
(3)
Figure 7.11 The
dependences of (a) the
number of states, N(E),
and (b) the density of
states, g (E), in a
rectangular box with
potential barriers of
infinite height.
of states doubles (taking into account the spin degeneracy) every time the energy
becomes equal to the energy level E
n
x
n
y
n
z
. If the quantum dot has the form of a
cubic box (L
x
= L
y
= L
z
) with potential barriers of infinite height, the energy
levels with different n
α
are degenerate (α = x, y, and z). In this case the jump
in the number of states, N (E), at E = E
n
x
n
y
n
z
is equal to twice the degeneracy,
g
n
x
,n
y
,n
z
, of the corresponding level.
The general expressions for the number, N (E), and density of states, g(E ),
are
N (E) = 2
n
x
,n
y
,n
z
g
n
x
,n
y
,n
z
× (E − E
n
x
n
y
n
z
), (7.83)
g(E) = 2
n
x
,n
y
,n
z
g
n
x
,n
y
,n
z
× δ(E − E
n
x
n
y
n
z
), (7.84)
where δ(E − E
n
x
n
y
n
z
)isDirac’sδ-function, which was introduced by Eq. (5.56),
(E − E
n
x
n
y
n
z
) the Heaviside function, and g
n
x
,n
y
,n
z
the degeneracy of the
(n
x
, n
y
, n
z
) state in a quantum dot. Figure 7.11 shows both dependences, N (E)
and g(E). The function N(E) for a quantum dot has the form of a step-function
and the density of states g(E) is a set of infinitely narrow and infinitely high
peaks.
Example 7.5. For a quantum well with infinite barriers and width L
x
= 10 µm,
find the number of levels that will be filled up to the energy of E = 1eVaswell
as the distance between the levels in the vicinity of the electron energy equal to
E = 1 eV. Compare the distance between energy levels with the thermal energy
at room temperature, which is of the order of k
B
T . Assume that the electron
effective mass is equal to the electron mass in vacuum: m
∗
= m
e
.
218 Quantization in nanostructures
Reasoning. According to Eq. (7.52) the electron energy spectrum in a quantum
well is defined as
E
n
x
=
π
2
h
-
2
2m
∗
n
x
L
x
2
. (7.85)
From Eq. (7.85) we get that in the vicinity of energy E
n
x
=1 eV the level number,
n
x
, is equal to
n
x
=
2m
∗
E
n
x
L
x
π h
-
≈ 10
4
. (7.86)
The distance between adjacent energy levels with quantum numbers n
x
and
n
x
+ 1 is defined by the expression
E
n
x
= E
n
x
+1
− E
n
x
=
π
2
h
-
2
2m
∗
L
2
x
(
2n
x
+ 1
)
= E
n
x
2n
x
+ 1
n
2
x
. (7.87)
For E
n
x
= 1 eV and n
x
= 10
4
we obtain
E
n
x
= 1eV×
2 ×10
4
+ 1
10
8
≈ 2 × 10
−4
eV. (7.88)
The distance between the adjacent energy levels, E
n
x
, is many orders of mag-
nitude less than the thermal energy, k
B
T , at room temperature:
E
n
x
k
B
T
=
π
2
h
-
2
(2n
x
+ 1)
2m
∗
L
2
x
k
B
T
≈ 2 × 10
−7
× (2n
x
+ 1). (7.89)
Therefore, the change in electron energy in the quantum well considered here is
practically continuous and the classical description can be used.
7.5 Double-quantum-dot structures (artificial molecules)
In the previous section we considered the behavior of an electron in an isolated
quantum dot. The main peculiarity of the energy spectrum of an electron confined
in the potential of a quantum dot is its discreteness, which is caused by the
quantization of the electron motion in all three directions.
Modern technological capabilities of epitaxial growth allow us to fabricate
types of nanostructures even with more complicated potential profiles. For exam-
ple, it is possible to fabricate nanostructures that contain two or more coupled
low-dimensional nanoobjects (a structure with size less than or about 10 nm
is called a nanoobject or a nanostructure). It becomes possible to control the
energy spectrum of electrons in such structures not only by changing the form
of an individual nanoobject, but also by changing the distance and barrier height
between neighboring nanoobjects. As we have mentioned already, if individual
nanoobjects are separated by low and narrow potential barriers, electrons can
easily tunnel from one nanoobject to another. This significantly affects the char-
acter of the electron energy spectrum in such a structure. In structures consisting
of several quantum dots, where electrons are able to tunnel between neighboring
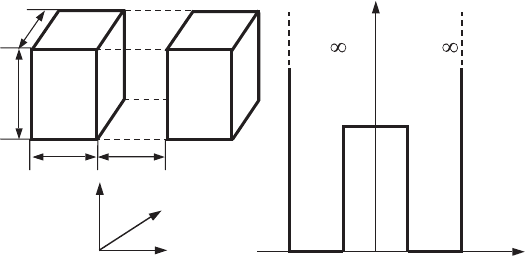
7.5 Double-quantum-dot structures (artificial molecules) 219
U
0
U
x
0
d−d−d − L
x
d+L
x
(a)
(b)
U= U=
ABA
L
x
2d
L
z
L
y
x
z
y
0
Figure 7.12 A schematic
picture of a
double-quantum-dot
structure (a) and its
potential profile U(x)(b).
A and B denote different
semiconductor materials,
which constitute quantum
dots and barriers between
them.
quantum dots, the main physical properties are similar to those of a molecule.
The energy spectrum of such an artificial molecule will be determined not only
by the parameters of the individual quantum dots but also by the parameters of
the potential barriers between them. Double-quantum-dot structures are nowa-
days the building blocks of many electronic and optoelectronic devices. Let us
consider the electron energy spectra formed by joining two nanoobjects such as
quantum dots (or quantum wires or quantum wells) into a single nanostructure.
7.5.1 Double-quantum-dot structures: qualitative analysis
Let us assume that a quantum-dot structure consists of two quantum dots sep-
arated by a rectangular potential barrier, U
0
(see Fig. 7.12).Theregionthatis
available for electrons in the y- and z-directions is given by the inequalities
0 ≤ y ≤ L
y
and 0 ≤ z ≤ L
z
. (7.90)
The rectangular distribution of the potential takes place along the x-direction.
The general dependence of the potential U (x, y, z) can be described by the
following expression:
U (x , y, z) =
∞, y < 0, z < 0,
U (x ), 0 ≤ y ≤ L
y
, 0 ≤ z ≤ L
z
,
∞, y > L
y
, z > L
z
,
(7.91)
where
U (x ) =
U
0
, |x|≤d,
0, d ≤|x|≤d + L
x
,
∞, |x | > d + L
x
.
(7.92)
220 Quantization in nanostructures
In the wavefunction of such a potential we can separate variables and write the
total wavefunction as
ψ(x, y, z) = ψ
x
(x )ψ
y
(y)ψ
z
(z), (7.93)
where the wavefunctions ψ
y
and ψ
z
describe the states of the electron in the
rectangular potential wells with infinitely high potential barriers. According to
Eq. (3.51), these wavefunctions have the following form:
ψ
y
(y) =
2
L
y
sin
n
y
π y
L
y
,ψ
z
(z) =
2
L
z
sin
n
z
π z
L
z
. (7.94)
In the x-direction the quantum dots are separated by a potential barrier of height
U
0
and width 2d.Thex-component of the wavefunction, ψ
x
(x), must satisfy the
one-dimensional Schr¨odinger equation with the potential U(x).
The total electron energy in the double-quantum-dot structure must be a sum
of the energies of quantum confinement along the y- and z-directions (defined
by Eq. (3.44)) and the energy of electron motion along the x-axis, E
x
,inthe
potential U(x):
E = E
x
+
π
2
h
-
2
2m
∗
n
2
y
L
2
y
+
n
2
z
L
2
z
. (7.95)
Thus, we have reduced the problem of finding the quantum states of an electron
in a structure consisting of two three-dimensional quantum dots to the one-
dimensional Schr¨odinger equation (3.31) with the potential U (x) defined by
Eq. (7.92).
Before finding the exact solution of the Schr¨odinger equation, let us analyze
qualitatively the dependences of the wavefunction ψ
x
and energy E
x
on the
distance between quantum dots, 2d.
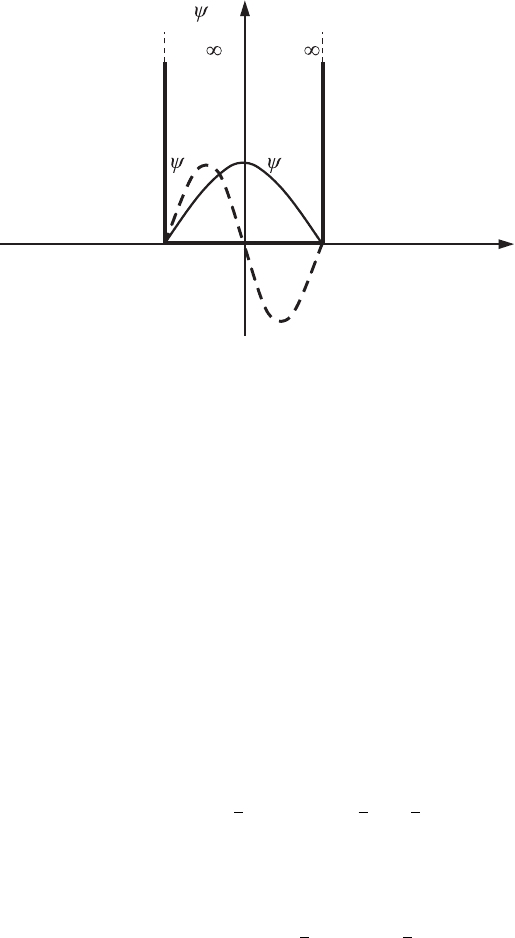
If the quantum dots are far from each other, then the wavefunction ψ
x
(x)at
the point x = 0 between them is practically equal to zero and the solution of the
Schr¨odinger equation for energies below U
0
for the double-quantum-dot structure
must practically coincide with the solution of the one-dimensional Schr¨odinger
equation (3.51) for an individual quantum dot. The only difference between these
two solutions will be that the magnitude |ψ
x
|
2
for the new solution is reduced
by a factor of 2 compared with the wavefunction of the individual quantum dot
because of the normalization condition that takes into account the possibility of
the electron being in each of the quantum dots. From those two wavefunctions
for the individual quantum dots it is possible to construct a wavefunction that
we will call a symmetric solution, ψ
s
. The wavefunction, ψ
s
, for the lowest
energy state of a double quantum dot is shown in Fig. 7.13 by a solid line. The
individual wavefunctions enter into the combined function, ψ
s
, with the same
signs. However, for the given potential profile there may exist another solution of
the Schr¨odinger equation. It differs from the symmetric solution, ψ
s
, by the signs
of the wavefunctions of the individual quantum dots which constitute the resulting

7.5 Double-quantum-dot structures (artificial molecules) 221
U =
2d
a
x
x
,U
U
0
s
U =
0− d−d − L
x
d+L
x
d
213
Figure 7.13 The
wavefunctions of a
double-quantum-dot
structure, ψ
s
and ψ
a
,
when the quantum dots
are far from each other.
wavefunction. The corresponding wavefunction, ψ
a
, is called antisymmetric and
is shown in Fig. 7.13 by the dashed line. The eigenvalues E
x
= E
s
and E
x
= E
a
of the two solutions are practically the same. This follows from the equality of the
two solutions for the average kinetic energy, which is proportional to |dψ
s,a
/dx|
2
,
and average potential energy, which is proportional to U(x)|ψ
s,a
|
2
.
Bringing the quantum dots closer to each other changes the form of the
wavefunctions. The state with the symmetric wavefunction, ψ
s
, corresponds to
a smaller energy E
s
than the energy E
a
, which corresponds to the state with the
antisymmetric wavefunction, ψ
a
. This is due to the fact that the average value of
the kinetic energy for the symmetric state is smaller than for the antisymmetric
state because
dψ
s
dx
2
<
dψ
a
dx
2
, (7.96)
where the overbar means averaging over the width of the entire structure. The
average values of the potential energy for the two states are practically the
same.
In the limiting case when the quantum dots approach each other, the barrier
between the quantum dots disappears (2d = 0). Thus, a potential well with
infinitely high barriers and width twice as large, 2L
x
, is established (see Fig. 7.14).
The symmetric wavefunction, ψ
s
, is transformed into the wavefunction of the
ground state, ψ
1
, for the quantum dot with width 2L
x
, and the antisymmetric
wavefunction, ψ
a
, is transformed into the wavefunction of the first excited state
with n
x
= 2, i.e., into ψ
2
. The energy levels in the quantum-dot structure with
the width of the potential well equal to 2L
x
are, according to Eq. (3.44),
E
x
=
π
2
h
-
2
2m
∗
(2L
x
)
2
n
2
x
= E
(0)
x
n
2
x
4
, (7.97)
222 Quantization in nanostructures
a
s
−L
x
0
x
L
x
U =U =
x
,U
Figure 7.14 The
wavefunctions ψ
s
and ψ
a
,
when the width of the
barrier between the two
quantum dots is equal to
zero.
where E
(0)
x
= π
2
h
-
2
/(2m
∗
L
2
x
) is the energy of the ground state in the individual
quantum dots. The quantum number n
x
= 1 corresponds to the state ψ
s
and
n
x
= 2tothestateψ
a
. The energy E
x
of the ground state (n
x
= 1) in the double
quantum dot formed in this way is four times smaller than the energy E
(0)
x
, which
is the initial ground state of the individual quantum dots separated by a large
distance. The energy of the quantum state with ψ
a
is exactly equal to the energy
of the initial state E
(0)
x
of the individual quantum dots. Thus, by bringing the
quantum dots closer to each other, we change the energy system of the electrons.
Let us assume that in each of the separated quantum dots in the ground state there
are only two electrons with opposite spins, and after joining of the quantum dots
these electrons fill the levels with n
x
= 1 and n
x
= 2. In the initial state, when the
quantum dots were separated, the total energy of the electrons was equal to 4E
(0)
x
.
After the joining of the quantum dots, two electrons will be in the lowest state
ψ
s
and two electrons in the state ψ
a
. Then, the energy of the electrons becomes
equal to
2E
(0)
x
×
1
4
+
2E
(0)
x
×
4
4
=
5
2
× E
(0)
x
.
As a result, after the joining of the quantum dots the energy of the system of
electrons decreases by
E
x
= 4E
(0)
x
−
5
2
× E
(0)
x
=
3
2
× E
(0)
x
.
The qualitative dependences of the energies E
(1)
x
and E
(2)
x
on the distance, d,
between the quantum dots are shown in Fig. 7.15. We see that, as in the case of the
coupled quantum dots, i.e., when the magnitude of d is small enough, the level
E
(0)
x
corresponding to the individual quantum dot splits into two levels, E
(1)
x
and
E
(2)
x
. The energy of splitting, E
x
, increases with decreasing d. If the electrons
