Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.

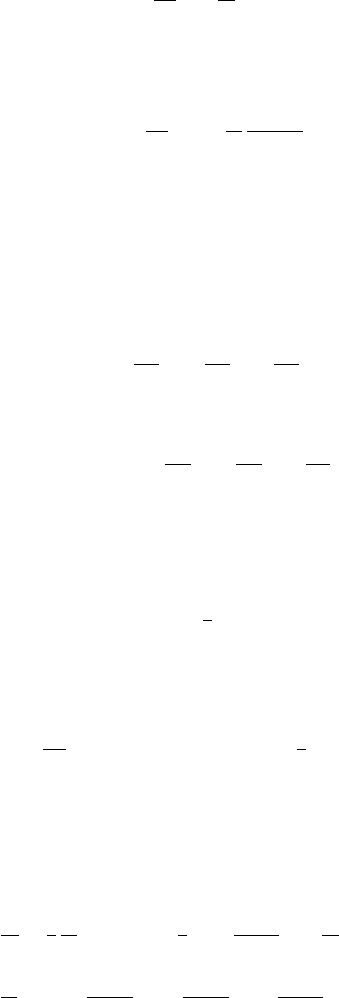
7.8 A three-dimensional superlattice of quantum dots 253
The effective mass of an electron in the cubic three-dimensional superlattice is a
scalar, i.e., it does not depend on the direction of the K-vector:
1
m
∗
j
=±
2
h
-
2
A
j
D
2
. (7.259)
In the general case of a superlattice with different periods D
α
along the directions
of the main axes the inverse effective mass becomes a tensor:
1
m
∗
αβ
=
1
h
-
2
∂
2
E
∂k
α
∂k
β
. (7.260)
Example 7.10. Show that the electron motion in the three-dimensional superlat-
tice in an external electric field, E
ext
, can be described by Newton’s second law
if the inverse mass of the electron is considered as a tensor with the components
defined by Eq. (7.260).
Reasoning. The expression for the group electron velocity in the case of three-
dimensional motion can be written as follows:
v
gr
= e
x
∂ω
∂k
x
+ e
y
∂ω
∂k
y
+ e
z
∂ω
∂k
z
= ∇
k
ω, (7.261)
where
∇
k
= e
x
∂
∂k
x
+ e
y
∂
∂k
y
+ e
z
∂
∂k
z
. (7.262)
Taking into account the relation of the electron energy to the frequency of
the electron wave, E(k) = h
-
ω, the group velocity of the wave packet which
represents the electron can be written through its energy as
v
gr
=
1
h
-
∇
k
E(k). (7.263)
If the superlattice is in an external electric field, E
ext
= e
x
E
x
+ e
y
E
y
+ e
z
E
z
,the
change in the electron energy, E, in unit time is defined as
dE
dt
= F · v = eE
ext
· v = eE
ext
·
1
h
-
∇
k
E(k)
, (7.264)
where we considered the group velocity of the electron, v
gr
, as the electron wave-
packet velocity, v. The electric force applied to the electron is equal to F = eE
ext
.
Using Eqs. (7.263) and (7.264), let us find the acceleration, a, which the electron
gains in the external field, E
ext
:
a =
dv
dt
=
1
h
-
d
dt
(
∇
k
E(k)
)
=
1
h
-
∇
k
dE (k)
dt
=
e
h
-
2
∇
k
(E
ext
·∇
k
E(k))
=
e
h
-
2
∇
k
E
x
∂ E(k)
∂k
x
+ E
y
∂ E(k)
∂k
y
+ E
z
∂ E(k)
∂k
z
. (7.265)

254 Quantization in nanostructures
Let us write the x-projection of the acceleration, a:
a
x
=
dv
x
dt
=
e
h
-
2
E
x
∂
2
E(k)
∂k
x
∂k
x
+ E
y
∂
2
E(k)
∂k
x
∂k
y
+ E
z
∂
2
E(k)
∂k
x
∂k
z
. (7.266)
Analogous expressions can be written for the other two projections of the accel-
eration, a. These expressions, according to Newton’s second law in the general
form, can be presented as
a
α
=
β
1
m
∗
αβ
F
β
, (7.267)
where the summation is done over all three projections onto the coordinate axes
β = x, y, and z, and F
β
are the projections of the force F. On comparing the
expressions obtained, we find that the magnitudes
1
m
∗
αβ
=
1
h
-
2
∂
2
E
∂k
α
∂k
β
(7.268)
are the components of the inverse effective mass tensor.
7.9 Summary
1. In a potential well of macroscopic size, the electron energy spectrum is quasicontin-
uous. With increasing energy the number of electron states in such a well increases
proportionally to E
3/2
and the density of states increases proportionally to
√
E.
These dependences hold only in the case of a quadratic dependence of energy on
momentum.
2. A structure of size less than or about 100 nm is called a nanoobject or a nanostructure.
In a nanostructure the electron energy spectrum radically changes, which leads to
changes of the physical properties of an object. For a nanostructure that is formed
from a few atoms and placed in vacuum the potential barriers are high and the spacing
between levels is about several electron-volts.
3. In quantum wells (two-dimensional structures) the electron motion in one direction is
limited and the energy that corresponds to this motion is quantized. The motion in the
two other directions stays free and is characterized by a continuous energy spectrum.
Therefore, the electron spectrum in a quantum well is a set of two-dimensional
subbands, which have the form of paraboloids.
4. In quantum wires (one-dimensional structures) electron motion is limited in two
directions, with the corresponding energy quantization. The electron motion along a
wire (the third direction) is free and the energy spectrum is continuous. The electron
spectrum in a quantum wire is a set of one-dimensional subbands, which have the
form of parabolas.
5. In quantum dots (zero-dimensional structures) the electron motion is limited in all
three directions. The energy spectrum of such motion is completely quantized.
7.10 Problems 255
6. The density of states of a quantum well in any of its subbands is constant and does not
depend on energy. Each subband has the same input into the total density of states,
which is defined by the electron effective mass and the width of the quantum well.
7. Superlattices are structures with periodic positioning of quantum-dimensional objects
along one, two, or three directions. The electron wavefunction in a superlattice
must have the form of the wavefunction of a free electron modulated by a periodic
superlattice function.
8. The periodicity of the superlattice potential leads to the conservation not of electron
momentum, but of quasimomentum. For quasimomentum Newton’s second law in
the form dp/dt = F
ext
is valid. Here F
ext
is connected with the external fields and
does not include the periodic force of a superlattice, F
SL
, which is related to the
superlattice potential by F
SL
=−
∇U , with U being the potential of the superlattice.
9. The total external force is related to the electron acceleration by F
ext
= m
∗
a,where
the effective mass of the electron, m
∗
, reflects the character of the electron energy
dispersion in a superlattice. Internal forces (potentials) define the energy dispersion,
E( p), and, through the effective mass, m
∗
, define the electron motion under the
influence of external forces.
10. The interval of quasimomenta (−π h
-
/D ≤ p ≤ π h
-
/D) that contains all physically
equivalent electron states in a one-dimensional superlattice as in the case of oscil-
lations of a chain of atoms is called the first Brillouin zone. The second Bril-
louin zone consists of two quasimomentum intervals, (−2π h
-
/D, −π h
-
/D)and
(π h
-
/D, 2π h
-
/D), and so on. The total length of each Brillouin-zone interval is equal
to 2π h
-
/D.
11. The boundaries of Brillouin zones correspond to wavevectors at which the electron
wave cannot propagate in the superlattice. At the boundaries of Brillouin zones the
electron energy in a superlattice has a discontinuity, which indicates the occurrence
in the electron energy spectrum of forbidden minibands, which alternate with allowed
minibands.
12. Each individual level of the identical quantum dots splits into the superlattice mini-
band if the quantum dots are assembled as a superlattice.
7.10 Problems
Problem 7.1. Find the expressions for the wavefunction and electron energy in a
symmetric rectangular quantum well with the following potential profile:
U (r) =
U
0
, |x| > L
x
,
0, |x|≤L
x
.
(7.269)
The electron motion along the y- and z-directions is free.
Problem 7.2. Using the solution of Problem 7.1, consider the limiting cases of
very deep and very shallow quantum wells. Show that in a shallow quantum well
there is always at least one stationary state. Derive an approximate expression

256 Quantization in nanostructures
for the energy, E
1
, of this state and estimate it for U
0
= 2.5 × 10
−3
eV and
L = 2nm.(Answer:E
1
≈ 2.375 meV.)
Problem 7.3. In an asymmetric rectangular quantum well the motion of an
electron in the yz-plane is free and motion in the x-direction is limited by the
following potential:
U (x ) =
U
1
, x < 0,
0, 0 ≤ x ≤ L
x
,
U
2
, x > L
x
,
(7.270)
where U
2
< U
1
. Find the condition for which discrete quantum states with
energy E ≤ U
2
exist. Consider the limiting cases U
1
→∞and U
1
= U
2
.
Problem 7.4. Estimate the energy of the electron ground state in a quantum well,
where electron motion in the yz-plane is free but motion along the x-direction is
limited by the potential U(x):
U (x ) =
∞, x < 0,
bx, x ≥ 0.
(7.271)
Problem 7.5. Find the energy and the wavefunctions of the stationary states of
an electron in a quantum wire. The electron motion in the z-direction is free and
motion in the plane perpendicular to the z-direction is limited by the potential
U (ρ)(ρ
2
= x
2
+ y
2
):
U (ρ) =
0,ρ≤ a,
∞,ρ>a.
(7.272)
Problem 7.6. In a quantum wire the electron motion along the z-direction is free
and that in the plane perpendicular to the z-direction is limited by the potential
U (x, y):
U (x , y) =
β(x
2
+ y
2
)
2
. (7.273)
Find the energy, the wavefunctions, and the magnitudes of various projections of
the electron angular momentum onto the z-direction and occupation probabilities
for the states with quantum numbers n
x
= 1 and n
y
= 1.
Problem 7.7. The electron motion in a spherical quantum dot happens in the
following potential:
U (r) = U
0
e
−r/b
, U
0
< 0. (7.274)
Find the average electron energy of a ground state in the given quantum well.
Problem 7.8. Electron motion takes place in an infinitely deep double-quantum-
dot structure and is limited by the potential U(x, y, z):
U (x , y, z) = U (x)U(y)U(z), (7.275)
7.10 Problems 257
where
U (x ) =
∞, x < 0, x > L
x
,
0, 0 < x < b, L
x
− b < x < L
x
,
U
0
, b ≤ x ≤ L
x
− b,
(7.276)
U (y) =
∞, y < 0, y > L
y
,
0, 0 ≤ y ≤ L
y
,
(7.277)
U (z) =
∞, z < 0, z > L
z
,
0, 0 ≤ z ≤ L
z
.
(7.278)
Find using first-order perturbation theory the expression for the electron energy
states in a double-quantum-dot structure.
Problem 7.9. Prove that in isotropic three-dimensional systems the density
of states per unit volume of momentum space (the number of states per unit
interval of momentum modulus) is proportional to the square of momentum.
Problem 7.10. The electron motion in a quantum wire is free along the z-direction
and that in the xy-plane is limited in the region 0 < x < L
x
and 0 < y < L
y
by
barriers of infinite height. Find the number and density of states in the lowest-
energy subband of the quantum wire.
Chapter 8
Nanostructures and their applications
Nanotechnology is based on the ability to manipulate individual atoms and
molecules in order to assemble them into bigger structures. Such artificial
nanoscale structures, usually fabricated using self-assembly phenomena, possess
new physical, chemical, and biological properties. The fabrication of various
types of nanostructures and study of their properties require new technological
means and new principles.
Nanotechnology has initiated a new so-called bottom-up technology. The
bottom-up technology is based on the self-assembly phenomenon, i.e., the process
of formation of complex ordered structures from simpler ones. The main idea
of this technology is in the development of the controlled self-assembly of the
atoms, molecules, and molecular chains into nanoscale objects. The bottom-up
technology allows the fabrication of nanoobjects, such as quantum dots, quantum
wires, and superlattices.
The bottom-up approach is opposite in principle to the traditional approach,
which may be called the top-down approach, which is based on the sequen-
tial decrease of the object’s size by means of mechanical or chemical pro-
cessing for the fabrication of objects of nanoscale size (nanoobjects). Thus,
for example, some of the nanoparticles can be obtained by grinding mate-
rial consisting of particles of micrometer or larger size in a special grinder.
The traditional technologies include laser methods for the processing of semi-
conductor surfaces and making masks of various configurations and sizes for
photolithography.
In this chapter we will briefly consider the main fabrication and characteriza-
tion techniques for nanostructures and will give some examples of applications
of nanostructures in modern nanoelectronics.
8.1 Methods of fabrication of nanostructures
In this section we consider the main techniques of fabrication of nanostructures.
258
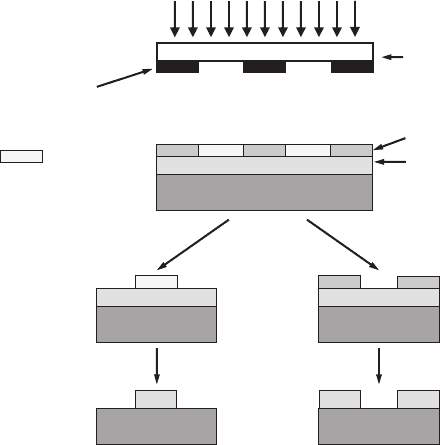
8.1 Methods of fabrication of nanostructures 259
Illuminating radiation
Mask
Photoresist
Film to be patterne
d
Substrate
Develop
Etch pattern
Imaging system
Illuminated areas
Ion-oxide layer
Quartz plate
N
egative resist:
exposed rendered
insoluble
Positive resist:
exposed is
removed
Figure 8.1 Standard steps
of a photolithographic
process.
8.1.1 Photolithography
Lithographic methods are the main representatives of the top-down techniques
in the fabrication of devices and integrated circuits (ICs). Under the term
“lithography” one usually understands a set of physicochemical processes of
layer-by-layer formation of the patterns of ICs and of elements of nanostruc-
tures. The decrease of microchip sizes can be achieved by decreasing the pattern
size which is formed with the help of lithography on the surface of solid-state
materials.
From optics it is known that the resolution of an optical system increases with
decreasing wavelength of the light. Therefore, to fabricate smaller elements by
lithographic methods it is necessary to switch to shorter wavelengths. Depending
on the wavelength and the methods of generating radiation, one can distinguish
among four types of photolithography that are in use: (1) optical lithography, (2)
X-ray lithography, (3) ion-projection lithography, and (4) electron-beam lithog-
raphy. Optical lithography, which is often also called photolithography,isaset
of methods of formation of the given relief on the sample surface with the help
of a focussed light beam.
In the simplest scenario the photolithography process consists of several steps
(see Fig. 8.1). In the first step the surface of the solid-state material (substrate)
is covered with a layer of photosensitive material. The structure of the photosen-
sitive layer changes under the influence of optical radiation. This photosensitive
260 Nanostructures and their applications
layer is called the photoresist.Aphotomask is placed on the surface of the pho-
toresist or somewhere between the photoresist and source of light. The photomask
consists of transparent and non-transparent regions. The next step of the pho-
tolithographic process is called exposure. The surface of the solid-state material
covered by the photoresist and by the photomask is subjected to radiation from
the optical or X-ray source. As a result of photochemical reactions underneath
the transparent regions of the photomask, the photoresist changes its properties.
In the case of a positive-resist process the exposed photoresist is removed during
the so-called etching process. In the case of a negative-resist process the exposed
region becomes resistant to the special chemical agents, called etchants, and
the unexposed parts of the photoresist are removed by the etching. In both cases
we will end up with a certain image on the surface of the substrate, such that some
parts of the substrate are covered by the photoresist and some parts are open. The
next step is chemical etching, which is based on the dissolving by etchants of the
part of the sample area which is not protected by the photoresistive layer. Thus,
patterns of complex configurations can be developed on the substrate.
As we have already mentioned, the radiation wavelength is one of the most
important characteristics of the source of optical radiation which is used for
the illumination of the photoresist. Because of the diffraction phenomenon the
wavelength used cannot be larger than the feature size that we would like to obtain
by photolithography. If we use a source of radiation with wavelength 1 µm, then
the minimum feature size in the pattern will be of the same order of magnitude.
To obtain a pattern with nanometer feature size we have to use so-called deep-
ultraviolet light with a wavelength of the order of tens or hundreds of nanometers.
In modern deep-ultraviolet photolithography a wavelength of λ = 200–400 nm
is used. The sources of such a radiation are powerful Hg–Xe gas-discharge
lamps and excimer lasers whose output power reaches hundreds of watts. The
radiation wavelength of the excimer lasers depends on the composition of the
active medium of the laser, for example, KrF (λ = 248 nm), ArF (λ = 193 nm),
and F
2
(λ = 157 nm) are widely used. These lasers have a pulse duration of
5–20 ns with a repetition frequency of about 4 kHz and output power up to 50 W.
Further increase of the photolithographic resolution can be achieved by using as
a source of optical radiation excimer lasers with a wavelength of λ = 13.5 nm.
In X-ray lithography soft (or low-energy) X-ray radiation with a wavelength of
0.5–5 nm is used for illumination.
In conclusion, let us note that the huge success of microelectronics in the last
quarter of the twentieth century was due to the fast advances in the development
of these processes. Many technological methods of microelectronics have the
potential to be used also in nanoelectronics, with reduction of the characteristic
feature size. In the transition from microelectronics to nanoelectronics special
attention is paid to the self-organization processes which take place during epi-
taxial growth of nanostructures, atomic-force epitaxy in colloid solutions, etc.
In particular, the rate of development of nanoelectronics in general depends
8.1 Methods of fabrication of nanostructures 261
entirely on the rate of development of industrial technologies for nanomaterials
and nanostructures.
8.1.2 Epitaxy
Epitaxy is one of the methods of nanostructure fabrication using a bottom-up
approach. Most of the fabrication methods using the assembly of nanoobjects
from individual atoms are based on the phenomenon of condensation. By con-
densation we usually understand the transition of the substance from the gaseous
state to a liquid or solid state as a result of its cooling or compression. Rain, snow,
and dew all result from natural phenomena of condensation of water vapor from
the atmosphere. The condensation of a vapor is possible only at temperatures
below a critical temperature for a given substance. In a similar fashion we can
condense atoms and molecules of other chemical elements. Condensation and
the opposite process – evaporation – are examples of phase transitions of the sub-
stance. The process of phase transition of a gas into a liquid or of a liquid into a
solid substance occurs during a certain time. During the initial stage nanoparticles
are formed, with their further transformation into macroscopic objects occurring
subsequently. We can fabricate nanoparticles by “freezing” phase transition at
an earlier stage. As a result of condensation we can obtain fullerenes, carbon
nanotubes, nanoclusters, and nanoparticles of various sizes.
Using the condensation method of fabrication of nanoparticles, we evapo-
rate from macroscopic objects atoms that are assembled into nanoobjects. The
evaporation can be done by thermal heating or by laser heating of the macro-
scopic material. The evaporated atoms have to be transferred into regions with
lower temperatures, where the condensation of atoms into nanoparticles takes
place. The controlled condensation of atoms on a crystal surface (substrate)is
the foundation of epitaxial technology.
Epitaxy (the term is derived from the Greek words epi, which means on, and
t
´
axis, which means arrangement,ororder) is an arranged growth of a substance
during the process of condensation onto a substrate. Epitaxy of atoms on a
crystalline surface can be done from the liquid phase as well as from the vapor
phase. The process of epitaxy usually begins with the nucleation on the substrate
of islands, which coalesce with each other, forming a continuous film. Modern
epitaxial techniques allow one to control the growth with resolution up to a
single atomic layer and to alternate layers with different physical and chemical
properties.
One of the modern methods of epitaxial growth of nanostructures is molecular-
beam epitaxy (MBE), which is based on the interaction of several molecular
beams with a heated monocrystalline substrate. Molecular-beam epitaxy is an
improved version of the technique of thermal sputtering under conditions of ultra-
high vacuum. The pressure of the residual gases in the vacuum chamber of MBE
is maintained at a level lower than 10
−8
Pa. The fluxes of atoms (or very rarely
262 Nanostructures and their applications
entire molecules) are formed by the evaporation of liquids or sublimation of solid-
state materials, which are located in the source – so-called Knudsen effusion cells
or simply effusion cells. The effusion cell is a crucible of cylindrical or conical
form of diameter 1–2 cm and length 5–10 cm. The outlet of the effusion cell is a
round opening called a diaphragm of diameter 5–8 mm. The crucible is usually
made of high-purity pyrolytic graphite or boron nitride (BN).
The fluxes of atoms (or molecules) of the necessary chemical elements are
directed towards and subsequently deposited onto a substrate to form a substance
of the desired composition. The number of effusion cells depends on the com-
position of the film and the dopants. To grow elementary semiconductors such
as Si or Ge, we need only one source of main material and sources of dopants
of n- and p-types. In the case of compound semiconductors (binary or ternary
alloys) we need a separate source for each component of the material which
is to be grown. The temperature of the effusion cells defines the magnitude of
the flux of the particles deposited onto the substrate and it is strictly controlled.
The control of flux is provided by so-called shutters, which shut off the various
fluxes. The homogeneity of the grown material over the surface and its crystalline
structure are defined by the homogeneity of the molecular beams. In some cases,
to increase the homogeneity of the film the substrate with the forming film is
constantly made to spin.
The epitaxial growth of semiconductor compounds involves a series of steps.
The most important steps are (1) adsorption of atoms and molecules by the
substrate, which leads to the nucleation and growth of the layer, and (2) migration
and dissociation of the adsorbed particles. The growing material establishes a
crystalline structure, which is defined by the crystalline properties of both the
substrate and the deposited material. The atoms that are deposited onto the
substrate are adsorbed by the surface. During the first stage, called physisorption,
the physical adsorption is due to weak van der Waals and (or) electrostatic
forces. During the second stage, called chemisorption (chemical adsorption), the
molecules of the substance undergo a transition to a chemisorbed state during
which electron transfer takes place, i.e., a chemical reaction between the surface
atoms and the newly arrived atoms occurs. The binding energy of chemical
adsorption is higher than of physical adsorption.
Figure 8.2 shows the main elements of the apparatus for the fabrication of
semiconductor films of Al
x
Ga
1−x
As on GaAs substrate using MBE. Each heater
has a crucible, which serves as a source of one of the components of the compound
materials. The evaporated material is deposited onto the substrate with a relatively
slow deposition rate under conditions of ultra-high vacuum. The heaters are
arranged in such a way that the maxima of the distribution intensities of the beams
are on the substrate. By selecting the temperatures of heaters and of a substrate
we can fabricate structures and films with complex chemical compositions. For
example, in the case of Al
x
Ga
1−x
As, by controlling x, i.e., by controlling the
fractions of Al and Ga, it is possible to grow a wide spectrum of materials, from
