Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.


B.4 Light and its wave properties 373
into four regions. In region 1 there is no absorption because
is practically equal to zero.
This region is called the optical-transparency window. In this region
> 1 and thus the
wave speed in the medium is less than the speed of light in vacuum. Region 2 is called
the optical-absorption window because in this region
has sufficiently large magnitude.
In this region the medium becomes non-transparent and waves with the corresponding
frequencies quickly fade in the medium. Note that any dielectric medium, even if it does
not have free carriers of charge, always has a frequency interval within which you can
see strong absorption. This is caused by quantum transitions in atoms. Region 3 is called
the metallic-reflection window. It begins from frequencies at which there is practically no
absorption, and it ends at frequencies for which
→ 0. In this region
< 0and
= 0.
Thus, the coefficient of wave reflection from the interface is practically equal to unity,
which is characteristic of metals. Region 4 also is a region with optical transparency, since
at these frequencies
= 0. In this region 0 <
< 1, thus the wave’s phase velocity is
greater than the speed of light in vacuum.
As we mentioned before, monochromatic waves are only an idealization and real waves
propagate in the form of wave packets transferring energy with the group velocity, v
gr
:
v
gr
=
dω
dk
. (B.195)
There is a relation between phase and group velocities:
v
gr
= v
ph
− λ
dv
ph
dλ
. (B.196)
The region where dv
ph
/dλ>0 is the region of so-called normal dispersion and in this
region v
gr
<v
ph
. In the region of so-called anomalous dispersion we have dv
ph
/dλ<0
and v
gr
>v
ph
. In the absence of dispersion, when the phase velocity does not depend on
the wavelength, v
gr
= v
ph
and the wave packet propagates without spreading. Vacuum is
a medium of this kind.
Example B.6. Estimate the real and imaginary parts of the dielectric constant, ,for
graphite at the X-ray wavelength of λ = 0.05 nm. The density of graphite is ρ = 1.6 ×
10
3
kg m
−3
.
Reasoning. Note that the carbon atom has four electrons in its outer shell (the electron
configuration of carbon is 1s
2
2s
2
2p
2
, see Table 6.7). Hard X-rays ionize these electrons
and make them free, i.e., uncouple them from their nuclei. Thus, the returning force from
the core of the atom in Eq. (B.180) is equal to zero, i.e., κ = 0, so the frequency ω
0
can
also be considered equal to zero. As a result Eqs. (B.193)and(B.194)taketheforms
(ω) = 1 −
ne
2
0
m
e
1
ω
2
+ 4β
2
= 1 −
ω
2
p
ω
2
+ 4β
2
, (B.197)
(ω) =
ne
2
0
m
e
2β
ω(ω
2
+ 4β
2
)
=
2ω
2
p
β
ω(ω
2
+ 4β
2
)
, (B.198)
where ω
p
=
ne
2
/(
0
m
e
)
1/2
is the so-called plasma frequency. For its calculation, let
us estimate the concentration of free electrons created by X-rays. The number of carbon
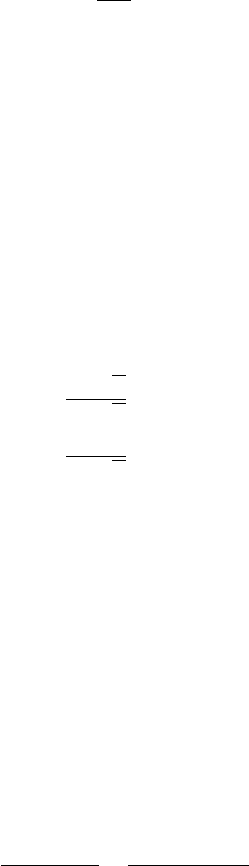
374 Appendix B. Electromagnetic fields and waves
atoms in the unit volume, n
C
,is
n
C
=
ρ N
A
A
, (B.199)
where A = 12 × 10
−3
kg mol
−1
and N
A
= 6.02 × 10
23
mol
−1
. After carrying out the
requisite calculations, we obtain
n = 4n
C
= 3.2 × 10
29
m
−3
,
and the plasma frequency is ω
p
≈ 3.2 × 10
16
s
−1
. The wavelength λ = 0.05 nm corre-
sponds to the frequency ω = 2πc/λ = 3.8 ×10
19
s
−1
, which is three orders of magnitude
higher than ω
p
. The parameter β plays the role of the scattering frequency and it has the
following order: β ∼ 10
14
−10
15
s
−1
. Therefore,
is equal to zero with high precision,
which indicates that graphite is transparent for X-rays. The magnitude of the real part of
the dielectric constant is practically equal to unity because 1 −
∼ 10
−6
. In accordance
with Eqs. (B.137)and(B.138), the coefficients of reflection, R, and transmission, T ,of
X-rays incident from vacuum on graphite are equal to
R =
1 −
√
1 +
√
≈ 0, (B.200)
T =
2
1 +
√
≈ 1. (B.201)
B.5 Summary
1. Maxwell’s equations are the main equations of classical electrodynamics, which suf-
ficiently correctly describe the processes of propagation and interaction of electro-
magnetic waves with various media up to frequencies of 10
14
−10
15
Hz. At higher
frequencies the quantum character of the interaction of electromagnetic radiation with
a substance comes to dominate.
2. Maxwell’s equations are a system of linear partial differential equations of first order,
whose unknowns are the field intensities, E and H, and flux densities D and B of
the electric and magnetic fields, respectively. For these magnitudes the superposition
principle is valid.
3. Electric and magnetic fields possess energy. The volume energy density at each point
of an electromagnetic field is equal to
w =
E · D + H · B
2
=
0
E
2
+ µµ
0
H
2
2
.
4. The vectors E, H,andk of an electromagnetic wave in an isotropic medium are
perpendicular to each other and in this order form a right-handed system of vectors.
For a monochromatic wave the oscillations of E and H are harmonic.
5. The Poynting vector, P = E ×H, defines the flux energy density which is carried by an
electromagnetic wave, which has the dimensionality [W m
−2
]. In an isotropic medium
this vector is parallel to the wavevector k.

B.6 Problems 375
6. In absorbing media the amplitude of an electromagnetic wave exponentially decreases
during its propagation. The effect of localization of electromagnetic wave energy in
the subsurface layer of an absorbing sample is called the skin effect and it can most
clearly be observed in metals.
7. At the interface of two media, which is not charged, and there is no current flowing
along this interface, the tangential components of the field intensity and normal com-
ponents of flux density of electric and magnetic fields must be continuous. To find the
amplitudes of transmitted and reflected waves we have to solve a boundary problem,
which requires sewing fields at the interface taking into account the above-mentioned
boundary conditions.
8. Interference, polarization, diffraction, and dispersion, which occur widely in nature
and are observed throughout the entire electromagnetic spectrum, are explained as
arising solely because of the wave nature of electromagnetic radiation.
B.6 Problems
Problem B.6.1. Maxwell’s equation (B.16) can be rewritten in the following form:
∇ × H = j
dis
+ j, (B.202)
where we introduced the density, j
dis
, of the displacement current, which is measured in
[A m
−2
]:
j
dis
=
∂D
∂t
. (B.203)
We see from Eq. (B.202) that it is precisely the displacement currents which are
responsible for the appearance of vortex magnetic fields in the case when there is no
current, j, of external charges. Let a monochromatic plane wave described by Eq. (B.56),
with an amplitude of the electric field of E
0
= 10
3
Vm
−1
and frequency ω = 3 × 10
15
s
−1
(red light), propagate in vacuum. Find the amplitudes of the displacement current,
j
dis,0
, and magnetic field intensity, H
0
,ofthewave.
Problem B.6.2. The point of interference, SP, of two monochromatic plane waves,
propagating in vacuum, is defined by the radius vector r, which forms angles ϕ
1
and ϕ
2
with the wavevectors, k
1
and k
2
, of the two waves (Fig. B.13). Find the frequency of the
wave at the point SP which corresponds to the intensity maximum of the wave field.
Problem B.6.3. Derive the equation for the location of maxima and minima in the
interference pattern. Assume that the setup of Young’s scheme (the two-slit experi-
ment; see Fig. B.9) is placed in water to perform the same experiment as was done in the air.
Problem B.6.4. The field intensities of electric and magnetic fields in a non-absorbing
medium oscillate in phase. Show that, for a wave in an absorbing medium, the oscillations
of the vectors E and H do not coincide in phase. Find the relation of the phase shift of
these two vectors to the conductivity of the medium, σ .
Problem B.6.5. The conductivity of sea water is σ
s.w.
≈ 5
−1
m
−1
and that of copper
is σ
Cu
≈ 6 × 10
7
−1
m
−1
. Compare the values of skin depth for these two media at
376 Appendix B. Electromagnetic fields and waves
Table B.6.1. The skin depth, δ, of sea water and copper
Frequency (Hz)
Skin depth (m) 10 10
4
10
7
10
10
Sea water 71.4 2.26 7.1 ×10
−2
2.3 ×10
−3
Copper 2.1 ×10
−2
6.6 ×10
−4
2.1 ×10
−5
6.6 ×10
−7
r
k
1
k
2
r
1
r
2
0
SP
ϕ
1
ϕ
2
Figure B.13 The
interference of two plane
waves with wavevectors
k
1
and k
2
. Here
r
1
= r cos ϕ
1
and
r
2
= r cos ϕ
2
.
frequencies f = 10, 10
4
, 10
7
, and 10
10
Hz, considering for simplicity that the dielectric
constant, , and magnetic permeability, µ, equal unity for both media. (Answer:
Table B.6.1.)
Problem B.6.6. Let us consider an optical micro-resonator that consists of two parallel-
plate mirrors applied from the opposite sides of a narrow layer of optical glass with width
L = 10 µm. Find the number, N , of possible types of electromagnetic field oscillations
in the region between the mirrors (i.e., standing waves with different wavelengths) that
can be excited in the visible range of wavelengths. Take the dielectric constant of glass
as = 1.55 and its magnetic permeability as µ = 1. (Answer: N = 27.)
Problem B.6.7. The density of charge distribution in the electron cloud of a hydrogen
atom in the ground state is defined by the following expression:
ρ(r) =−
e
πa
3
e
−2r/r
1
, (B.204)
where r
1
is the radius of the first Bohr orbit and e is the electron charge. Find the energy
of the electrostatic Coulomb interaction of the electron cloud with the nucleus of the
hydrogen atom (i.e., with the proton).
Problem B.6.8. Find the screening radius (Debye radius), r
D
, and potential, ϕ,ofa
point defect with charge q in an n-type semiconductor at room temperature, T = 300 K.
B.6 Problems 377
Assume that positively charged ions of univalent impurities are located at the points of
the lattice and that the free electrons are in thermal equilibrium. The energy distribution
of electrons is described by Boltzmann’s classical function. The density of impurities is
n
0
= 10
25
m
−3
and the dielectric constant of the crystal is = 10. (Answer: r
D
≈ 1.2 nm.)
Problem B.6.9. A plane electromagnetic wave from a medium with index of refraction
n
1
is obliquely incident on an interface with a medium with index of refraction n
2
. Find
the condition of total internal reflection, the wave field in the second medium, and the
depth of penetration into this medium.
Problem B.6.10. Find the energy, W , which is transmitted in vacuum by a plane
electromagnetic wave propagating in the x-direction through a plane contour with area
S = 1m
2
perpendicular to this direction during one second. Assume that the amplitude
of the electric field intensity is equal to E
0
= 10
−4
Vm
−1
and that the frequency is equal
to f = 10
6
Hz. (Answer: W ≈ 1.3 ×10
−15
J.)
Problem B.6.11. A linearly polarized wave is normally incident on a slab of thickness
L. The dielectric constant of the slab is , its conductivity is σ = 0, and its permeability
is µ = 1. Find the amplitudes of the waves reflected from and transmitted through the
slab and of the wave inside of the slab. Find the energy coefficients of reflection and
transmission. Find the angle of minimal wave reflection from the slab.
Appendix C
Crystals as atomic lattices
Three-dimensional quantum-dot superlattices can be considered as nanocrystals. Spherical
nanoparticles consisting of a big enough number (from 10 to 1000) of atoms or ions, which
are connected with each other and are ordered in a certain fashion, can be considered as the
structural units of such nanocrystals. Examples of nanocrystals that are of natural origin
are the crystalline modifications of boron and carbon which have as their structural units
the molecules B
12
and C
60
. The boron molecule B
12
consists of 12 boron atoms, and the
carbon molecule C
60
, which is called fullerene, consists of 60 carbon atoms. The fullerene
molecule resembles a soccer ball, i.e., it consists of 12 pentagons and 20 hexagons, with
carbon atoms at their corners. These nanoparticles form face-centered superlattices with
a period of about 1–10 nm. At these distances between molecules of C
60
weak molecular
forces, which provide the crystalline state of fullerene, act.
In addition to nanocrystals of natural origin, numerous artificial three-dimensional
superlattices consisting of various types of nanoparticles have been fabricated. The vari-
ety of nanocrystalline structures as well as of conventional crystals is defined by the
differences in the distribution of electrons over the quantum states of atoms. The most
significant role in the formation of individual nanoparticles as well as of crystals is played
by the electrons in the outer shells of atoms. First of all, the energy minimum of a nanopar-
ticle itself is predominantly defined by the interaction of valence electrons. Configurations
of nanoparticles in which electrons completely fill the energy shells have stable structures
consisting of a certain number of atoms. For this type of structure there exist so-called
magic numbers, which define the total number of atoms or ions in a nanoparticle. For
one group of metals (Ag, Au, Cu, and others) nanoparticles are formed on the basis of a
face-centered cubic elementary cell built around the central atom and have the following
series of magic numbers: N =1, 13, 55, 147, 309, 561, . . . For another group of metals
(Mg, Zn, and others) nanoparticles form on the basis of a hexagonal densely packed
elementary cell and have the following magic numbers: N =1, 13, 57, 163, 321, 581, . . .
Owing to the exchange of valence electrons or their collectivization, and due to the
spatial distribution of their wavefunctions, arrays of atoms, ions, or nanoparticles will,
under certain conditions, form regular crystalline structures of various configurations.
The stable state in such structures is almost completely provided by the forces of electro-
static attraction involving the subsystem of valence electrons in ion shells. In addition to
attraction forces at large distances, significant forces of repulsion can occur at small dis-
tances between atoms and ions because of overlap of electron shells. There are significant
differences in behavior of electrons among metals, dielectrics, and semiconductors that
378
C.1 Crystalline structures 379
are defined mainly by their different band-energy spectra. We will consider some of the
features of the formation of the above-mentioned structures and their energy spectra.
C.1 Crystalline structures
C.1.1 Spatial periodicity
All objects consist of a collection of atoms, molecules, or ions. The average distance
between them is defined by their interaction with each other and by temperature. In the
crystalline state the particles of a medium are arranged in an ordered fashion, and the type
of ordering depends on the chemical species as well as on the details of the interaction
among the particles.
In 1912 the German physicist Max von Laue discovered the diffraction of X-rays
from crystals. Since X-rays are electromagnetic waves, diffraction can take place only
in spatially-periodic structures consisting of atoms or ions with interatomic distances
comparable to the wavelength of X-rays. Modern experimental methods such as scanning
tunneling microscopy (STM) allow us to see directly the regular spatial distribution of
atoms in a crystal.
The main property of crystalline structures is their so-called translational symmetry.
The crystal can be thought of as being generated by the repetition of a basic atomic group
over a fixed vector, which is called the period of a crystalline lattice.Insuchacase
it is said that there is long-range order in a crystalline structure, i.e., the particles are
distributed in an orderly fashion throughout the entire crystal.
In addition to the translational symmetry in ideal crystals, there is so-called point
symmetry. A crystal that has point symmetry coincides with itself after an operation of
rotation and/or reflection has been carried out. The symmetry of a crystalline lattice
depends on the distribution of its structural elements in space. Symmetry defines the
physical properties of a crystal and the directions along which these properties become
apparent. For example, after a rotation of a cube around one of its three axes drawn through
the centers of the opposite cube plane, the cube will coincide with itself after each 90
◦
rotation. If we divide a cube by a plane going through the diagonals of the opposite bases
and then reflect it with respect to this plane, then the cube will coincide with itself. The
existence of such elements of symmetry can be demonstrated on the crystalline structure
of sodium chloride (NaCl). It is represented by a cubic lattice with alternating ions of
sodium (Na) and chlorine (Cl) in the sites of the lattice. Each Na
+
ion is surrounded by
six Cl
−
ions, and in its turn each Cl
−
ion is surrounded by six Na
+
ions (see Fig. C.1).
The minimal distance between ions is 2.81
˚
A. Let us note that on a macroscopic scale we
are dealing with a very large number of crystalline elements. For example, if the linear
dimensions of a crystal are about 1 cm, the crystal contains of the order of 10
22
ions.
How can one build an infinite lattice of a simple (consisting of only one type of atoms
or ions) crystal? Let us choose the origin of coordinates in any arbitrary place, and let us
put an atom (or ion) at this point. Let us draw three mutually-orthogonal vectors a
1
, a
2
,
and a
3
(in general they are not required to be mutually orthogonal), whose length is equal
to the distance between the nearest atoms (or ions). The parallelepiped whose edges are
the vectors a
1
, a
2
,anda
3
is called a unit cell. The three vectors a
1
, a
2
,anda
3
are also
called basis vectors, because, using them, one can build an infinite spatial lattice. In order
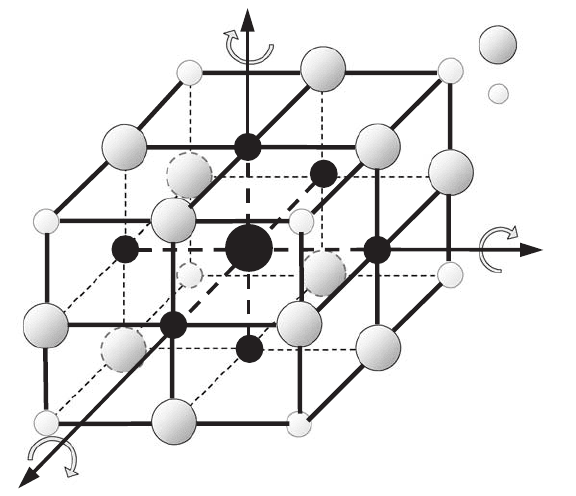
380 Appendix C. Crystals as atomic lattices
Na
+
−
Cl
x
y
z
Figure C.1 The unit cell of
the NaCl crystalline
structure.
to do this it is necessary to move all atoms (or ions) of a lattice from their initial positions
by distances equal to the translational vectors a
n
:
a
n
= n
1
a
1
+ n
2
a
2
+ n
3
a
3
, (C.1)
where n
1
, n
2
,andn
3
are arbitrary integer numbers. We can choose unit cells with other
basis vectors and, by translation of these unit cells, we may obtain the same spatial
crystalline lattice. A unit cell of minimal volume is called a primitive cell (see Fig. C.2
for examples of translational symmetry).
There are many physical phenomena in which the atomic structure of the material is
not manifested directly. While studying these phenomena the material can be considered
as a continuum, disregarding its internal structure. Such phenomena include, for example,
thermal expansion of bodies, their deformation under the influence of external forces,
dielectric permeability, and many optical phenomena. These material properties are char-
acteristic for a continuum and are called macroscopic properties. The indicated properties
significantly depend for most crystals on the direction along which the measurements are
carried out. The origin of this dependence is connected with the structure of the crystal and
its symmetry. For example, stretching a cubic crystal in the direction parallel to the edges
of cubic cells of a lattice will not be the same as stretching along the diagonals of the cells,
because the coupling energy between atoms depends on the distance between them. The
dependence of physical properties on the direction is called anisotropy. Anisotropy is the
characteristic property of crystals and in this sense they are fundamentally different from
isotropic media such as liquids and gases, whose properties are the same in all directions.
C.1 Crystalline structures 381
a
1
a
3
a
2
Figure C.2 Translational
symmetry of a
three-dimensional lattice.
Planes of
symmetry
Axes of
symmetry
Center of
symmetry
Figure C.3 Axes, planes,
and the center of
symmetry in a simple
cubic lattice.
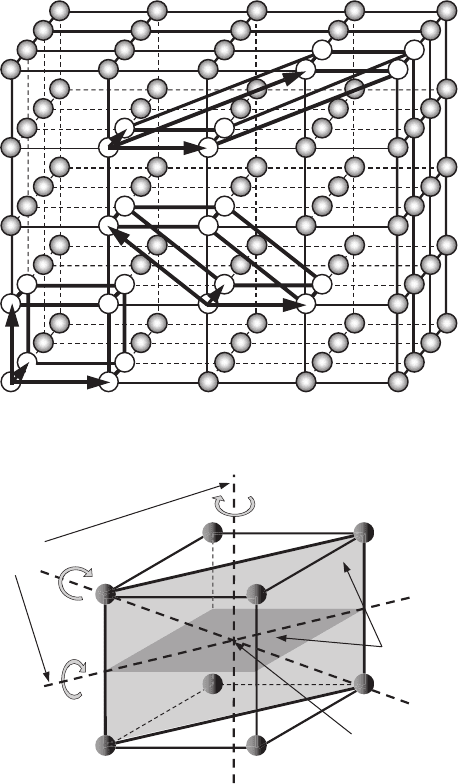
C.1.2 Symmetry elements and types of crystalline lattices
What are the main symmetry elements of an object? Let us briefly describe them.
Plane of symmetry. The presence of a plane of symmetry means that one part of
the figure coincides with the other if we move all its points equal distances to the
other side of a plane in a direction perpendicular to it. Thus the plane of symmetry
can be treated as a mirror, through which one part of the figure is reflected (see
Fig. C.3).
Axis of symmetry. An axis of symmetry is a straight line around which rotation of
the figure by a certain angle makes the figure coincide with itself. The order, n,

382 Appendix C. Crystals as atomic lattices
of the axis of symmetry is defined by the equation
n =
360
α
, (C.2)
where α is the minimal angle of rotation at which the figure coincides with itself.
Only five axes of symmetry can exist in crystals: of first, second, third, fourth,
and sixth orders (see Fig. C.3).
Center of symmetry. A crystal has a center of symmetry if any straight line drawn
through it at the opposite sides of a crystal goes through identical points. Thus,
there are equal planes, edges, and angles at the opposite sides of the crystal with
respect to the center of symmetry (see Fig. C.3).
There are 32 possible combinations of planes, axes, and centers of symmetry in
crystals. In general, a crystal does not possess only one element of symmetry. The full
set of symmetry elements is called the symmetry group. Why is the symmetry group so
important for the physics of crystals? It turns out that the symmetry group of the crystal
very often defines the physical properties of the crystal. Depending on the relation of
magnitudes and on the self-orientation of edges of the unit cell, there are 14 types of
crystalline lattices, which are called Bravais lattices (see Fig. C.4). The above-mentioned
14 types of lattices constitute 7 different systems: triclinic, monoclinic, orthorhombic,
tetragonal, rhombohedral, hexagonal,andcubic. Each of the systems is characterized
by the ratio of sides of the unit cells and the angles α, β,andγ between them. Many
important materials have simple (or primitive), base-centered, body-centered,andface-
centered Bravais lattices. If the sites of a crystalline lattice are located only at the vertices
of a parallelepiped, which represents a unit cell, then this lattice is called primitive or
simple. If there are additional sites at the center of the parallelepiped’s base, then this
lattice is called base-centered. If there is a site at the center of the intersection of the
spatial diagonals, then this lattice is called body-centered. Finally, if there are sites at the
centers of all of the lateral faces, then the lattice is called face-centered.
Almost half of the elements from the Periodic Table of the elements form crystals of
cubic or hexagonal symmetry, which we will study now in more detail.
Three lattices are possible for the crystals of cubic systems: simple, body-centered,
and face-centered (see Fig. C.5). In the cubic system all the angles of the unit cell are
equal to 90
◦
and all the edges are equal to each other. A right prism with a rhombus of
angles 60
◦
and 120
◦
as the base represents the unit cell of the hexagonal system. The two
angles between the unit cell’s axes are right angles and one of them is equal to 120
◦
(see
Fig. C.4).
In most cases we consider a crystal as a system of hard spheres that are in contact with
each other. A structure in which the spheres are as densely packed as possible corresponds
to the minimal energy of the structure. Let us compare three possible cubic structures in
such a model. In a simple cubic structure atoms are present only at the sites of a cube. In
this case one atom corresponds to one primitive cell. In a face-centered cubic lattice atoms
are present not only at the sites of a lattice, but also at the centers of six faces (NaCl has
such a structure). In a body-centered cubic lattice the atoms are at the sites of the cube and
in addition there is one at the center of the cube. The least densely packed is the simple
cubic structure, and the chemical elements prefer not to crystallize in such structures,
