Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.

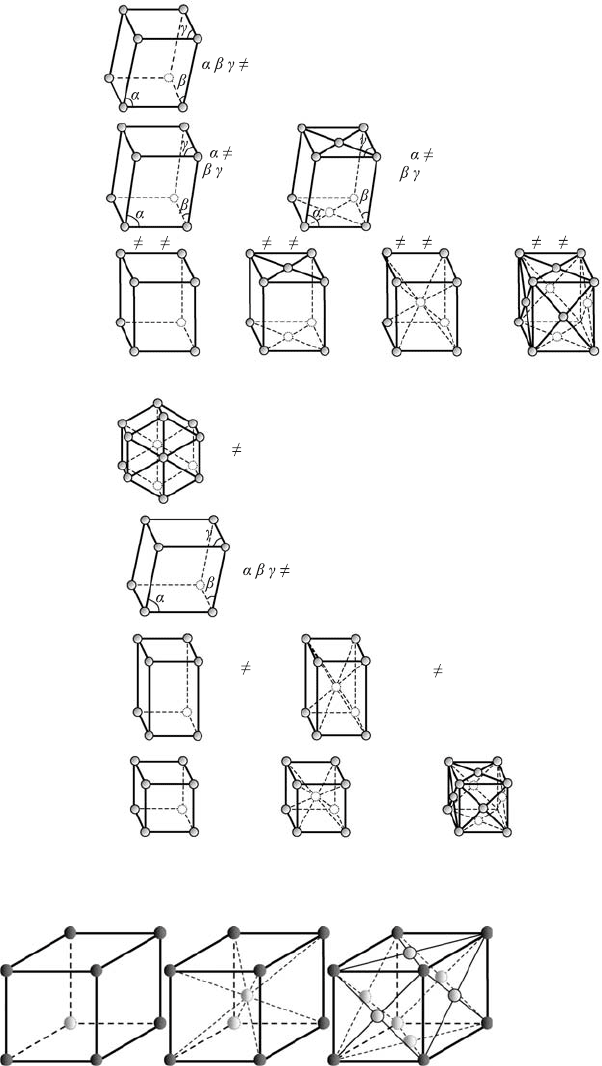
C.1 Crystalline structures 383
a b c
triclinic
, , 90°
monoclinic
90°
, = 90°
90°
, = 90°
simple
base-centered
orthorhombic
a b c
a
b ca b c
a
b
c
a
b
c
a
b
c
a
b
c
simple
base-centered body-centered
face-centere
d
hexagonal
a c
c
a
rhombohedral
a
a
a
, , 90°
a
c
a
c
aa
a
a
c
c
tetragonal
simple
body-centered
simple
body-centered
face-centered
a
a
a
a
a
a
a
a
a
cubic
Figure C.4 The 14 Bravais
lattices.
(a) (b) (c)
a
a
a
Figure C.5 Unit cells of the
cubic lattices:
(a) simple,
(b) body-centered, and
(c) face-centered.
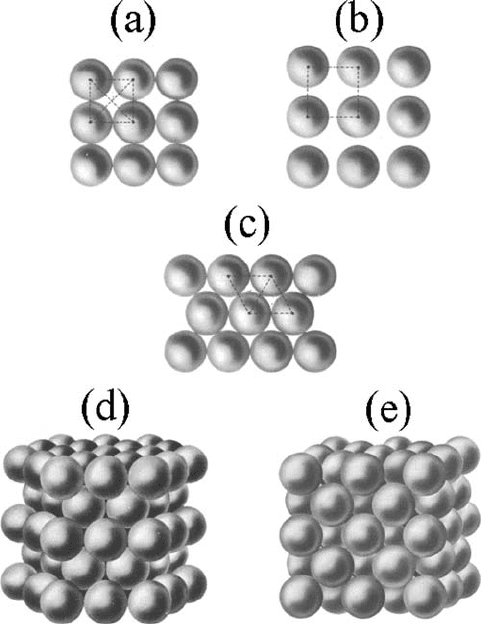
384 Appendix C. Crystals as atomic lattices
Figure C.6 Ty p es o f
packing: (a) dense
square-packed layer,
(b) loose square-packed
layer, (c) dense
hexagonally packed layer,
(d) dense-packed
hexagonal crystalline
lattice, and
(e) dense-packed
face-centered cubic
crystalline lattice.
although many substances do in their crystalline states have the structure of a simple cube
(e.g., CsCl or CuPd). The most densely packed is the face-centered structure,whichfor
this reason is also called a cubic structure with the most dense packing.However,itis
possible to have the same packing density if we were to arrange in space identical hard
spheres composing a hexagonal dense packing. In this structure the density of packing is
the same as in a face-centered cubic lattice. This is why most metals at certain temperatures
very easily change their structure from face-centered cubic to hexagonal dense packing
and vice versa. The first hexagonal plane of the skeleton of the hexagonal dense-packing
lattice corresponds to the dense packing of hard spheres (see Fig. C.6). The next atomic
plane is packed in a similar way, but it is shifted in such a manner that its atoms are in
between the atoms of the first plane; the third plane is packed in the same way, but its
atoms are exactly over the atoms of the first plane; the fourth plane has its atoms located
as in the second one, and so on.
Figure C.6 shows the two most densely packed crystalline lattices: hexagonal and
face-centered cubic lattices with dense packing.
Classification by crystal system gives us an idea about the geometrical characteristics
of a crystal, but it does not tell us about the nature of the forces that keep atoms (ions or
C.2 The nature of attraction and repulsion forces 385
molecules) at certain sites within a crystal lattice. Classification of the crystals can also be
done in terms of the physical nature of forces acting between the particles of a crystal (we
will talk about it later). In this case we will have four main types of crystalline lattices:
ionic, atomic, metallic,andmolecular.
C.2 The nature of attraction and repulsion forces
C.2.1 The potential energy of a crystal
It is well known that a large enough number of neutral atoms when brought close together
will transform into a stable crystalline state of matter. This fact indicates the existence of
attraction forces between them that at atomic distances (about 0.1 nm) are counterbalanced
by repulsion forces. In order for the atoms or ions to form a crystal, their interaction has
to be defined by a potential energy of the following form:
U (r) =
A
r
m
−
B
r
n
, (C.3)
where A, B, m,andn are positive parameters defined by a certain type of interaction.
ThefirstterminEq.(C.3) describes the repulsion forces and the second term concerns
the attraction forces. In order for repulsion forces to be greater than attraction forces at
small distances (r < r
0
), it is necessary to satisfy the condition m > n. The equilibrium
state between atoms when forces of repulsion and attraction compensate for each other is
defined by the following condition:
dU
dr
r=r
0
= 0. (C.4)
By equating the first derivative of the expression (C.3) to zero we obtain
r
0
=
nB
mA
1
m−n
. (C.5)
At this distance the potential interaction energy reaches its minimum:
U
0
=−
A
r
m
0
m
n
− 1
. (C.6)
The true theory of atomic interaction in a certain crystalline structure must be based
on quantum-mechanical consideration of the electron motion. As a rule, in such an
approach atomic nuclei are considered motionless (the so-called adiabatic approximation)
since their mass is three to four orders of magnitude larger than the mass of electrons
The main (and close to each other in strength) types of bonding in crystals are ionic,
metallic, and covalent. Other types of bonding (intermolecular interactions and hydrogen
bonding) are one to two orders weaker than the above three types. By the bonding energy,

386 Appendix C. Crystals as atomic lattices
Table C.1. Bonding energies in kJ mol
−1
for crystals
with various types of bonding
LiF 1013 V 510 C 711
NaCl 762 Fe 414 Si 448
KI 623 Cu 339 Ge 372
U
b
, we understand the energy which is required for separation of one mole of crystal to
individual neutral atoms. In Table C.1 the values of bonding energy for crystals with ionic,
metallic, and covalent bondings, which we will define, are shown. For comparison, the
characteristic energy of intermolecular bonding, which is called van der Waals bonding,
is about 1 kJ mol
−1
. It is most clearly manifested in crystals of the inert gases, which
crystallize at temperatures of about 10–100 K, and in molecular crystals formed from
individual molecules (for example, crystals of fullerene). This bonding is caused by
interaction of dipole moments of atoms, which occur as a result of fluctuations in the
distribution of charge of atoms. Even in a spherically-symmetric state the instantaneous
position of the center of the electron cloud need not coincide with the center of the
atom, which may lead to the occurrence of a non-zero dipole moment at that instant. An
instantaneous dipole moment of one atom induces instantaneous moments of neighboring
atoms of such direction that it causes attraction. For this type of bonding, in the potential
energy arising from the interaction of neighboring atoms (Eq. (C.3)) the parameter n = 6.
Let us briefly consider the main types of bonding.
C.2.2 Ionic and metallic bonding
The easiest type of bonding to interpret from the classical point of view is the bonding in
ionic crystals, which is the result of Coulomb attraction of adjacent ions. Ionic bonding
manifests itself especially clearly in such compounds as NaCl. In Example C.1 later we
will obtain the expression for the potential energy of an ion in a one-dimensional periodic
chain of ions with alternating signs:
U (r) =−α
k
e
e
2
r
, (C.7)
where r is the distance between ions with charge ±e and α is Madelung’s constant, which
is equal in the case of a one-dimensional periodic chain of atoms to α = 2ln2 = 1.386.
In the case of a three-dimensional crystal with a cubic lattice Madelung’s constant is equal
to α = 1.748. By comparing Eq. (C.7) with Eq. (C.3), we obtain B = αk
e
e
2
and n = 1.
To estimate the energy of ionic-type bonding we have to multiply Eq. (C.7)bythe
Avogadro number, N
A
:
U
ib
(r) =−α N
A
k
e
e
2
r
. (C.8)
After substituting into Eq. (C.8) the values of the physical magnitudes, we obtain for the
bonding energy U
ib
= 900 kJ mol
−1
, which is in good agreement with tabulated values
for the bonding energy of ionic crystals.

C.2 The nature of attraction and repulsion forces 387
Typical metals (Cu, Ag, Fe, and others) have relatively high conductivity, optical
reflectance, and plasticity. These properties unambiguously tell us that in metals there is
a large number of free electrons, which even under weak electric fields can move around
over large distances within crystals. The simplest model of a metal suggested by Drude
is an array of charged ions located at the points of the lattice and an ideal gas of electrons
freely moving between ions throughout the crystal. This simplified model qualitatively
explains most of the experimental data. If we take into account that an ideal electron gas
obeys quantum statistics, then the model becomes valid for the description of a wider
range of phenomena taking place in metals.
Let us consider the simplest case of a univalent metal, for which to each univalent ion
there corresponds one free electron. In the stationary state the concentration maxima of
electrons will be at positions in the middle between the ions. From the electrostatic point
of view this model can be treated as an ionic crystal with non-localized negative ions. The
potential energy of Coulomb interaction for each ion and bonding energy can be written
in the form of Eqs. (C.8)and(C.7), with substitution of α by the effective Madelung
constant, α
eff
:
U (r) =−α
eff
k
e
e
2
r
, U
ib
(r) =−α
eff
N
A
k
e
e
2
r
, (C.9)
where r = a/2 is the distance between “opposite ions,” which can be estimated as half of
the lattice constant. Thus, for the metal in Eq. (C.3), the parameters B and n are the same
as for an ionic crystal. The spreading in space of negative charge is taken into account
by the effective Madelung constant, α
eff
, which, because of this spreading of charge,
is less than the constant α and significantly depends on the distribution of the electron
wavefunction in the given metal.
C.2.3 Covalent bonding
This type of bonding is of special importance because it is the most important not
only in molecules but also in crystals. As was shown for the hydrogen molecule, this
bonding occurs between neutral atoms and is characterized by saturation and distinctive
orientation. The saturation of bonding in the case of hydrogen atoms is manifested by
the impossibility of forming a hydrogen molecule from more than two atoms. This is
because the attraction occurs only between atoms whose electrons are able to form pairs
with oppositely directed spins. The orientation of covalent bonding is defined by the
orientation (absence of spherical symmetry) of the wavefunctions of the valence electrons
of the majority of atoms that contribute to make bound states.
Thus, the nitrogen atom, N, has electron configuration 1s
2
2s
2
2p
3
. It is well known
that the spins of the three 2p-electrons are parallel, i.e., they are not spin-saturated.
Therefore, a nitrogen atom can form three covalent bonds and is a trivalent atom. For
example, in the ammonia molecule, NH
3
, the lobes of the wavefunctions of the three
2p-electrons extend along three mutually orthogonal directions. The energy gain as a
result of formation of covalent bonds is significantly defined by the overlapping of the
electron wavefunctions of the corresponding pair with anti-parallel spins. Therefore, the
hydrogen atoms in the ammonia molecule take positions along these three directions where
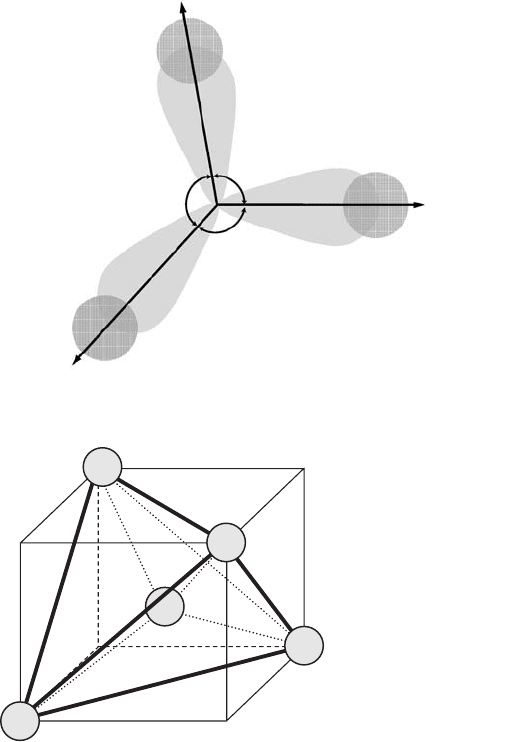
388 Appendix C. Crystals as atomic lattices
N(2p)
H(1s)
107
°
107
°
107
°
H(1s)
H(1s)
Figure C.7 The ammonia
molecule, NH
3
.
0
1
2
3
4
C
C
C
C
C
Figure C.8 The diamond
structure.
partial overlapping of the wavefunction lobes of nitrogen p-electrons and the spherically-
symmetric wavefunctions of hydrogen 1s-electrons take place (Fig. C.7). Because of the
mutual repulsion of the hydrogen atoms, the angle between bond directions is equal to
107
◦
, not 90
◦
.
The carbon atom, C, has electron configuration 1s
2
2s
2
2p
2
. Therefore, it must be
bivalent. However, carbon is quadrivalent in the majority of cases, since it participates
in chemical compounds not in its ground state but in the excited state 1s
2
2s
1
2p
3
.Inthis
case the spins of all four electrons in 2s- and 2p-states are parallel (not saturated) and
may form four covalent bonds. Thus, in diamond the directions of these bonds coincide
with directions 01, 02, 03, and 04 of the tetrahedron whose vertexes and center contain
the carbon atom (see Fig. C.8).
Crystals of the semiconductors silicon, Si, and germanium, Ge, have the same structure.
The silicon atom, Si, has four electrons in its M-shell with 3s
2
2p
2
states, and its K- and
L-shells are completely filled and are spin-saturated. Therefore, in forming crystalline

C.2 The nature of attraction and repulsion forces 389
bonds the silicon atom behaves similarly to the carbon atom. Germanium, Ge, which has
four electrons in its N-shell in 4s
2
4p
2
states, has similar properties.
Chemical compounds such as A
3
B
5
, i.e., compounds of elements from groups III
and V of the Periodic Table of the elements, share most of the properties of crystalline
structures of elements of group IV (silicon and germanium). For example, InSb and GaAs
belong to this group of compounds. Thus, indium, In, has in its O-shell three electrons
with configuration 5s
2
5p
1
and antimony, Sb, has five electrons in the states 5s
2
5p
3
. During
the formation of covalent bonds one p-electron of Sb is transferred to In and as a result the
electron configurations of the ions formed become similar to the electron configurations
of Ge and Si. The same takes place in the case of GaAs.
Covalent bonding is very strong bonding. For example, in the diamond crystal the
energy of bonding between two carbon atoms is 7.3 eV, which is comparable to the
bonding energy of ionic crystals. The magnitude of such bonding is defined mainly by
the overlapping of adjacent atoms’ wavefunctions. Therefore, it decreases exponentially
with increasing distance between atoms. If this distance is greater than several interatomic
distances, the covalent bonding becomes negligibly small.
Crystals with ionic and covalent types of bonding can be considered as extreme cases.
In between these two types of crystals there are crystals with mixed types of bonding.
Atoms with almost-filled electron shells (Li, F, Na, Cl, K, and I) have predominantly ionic
bonding. Atoms from groups III, IV, and V of the Periodic Table of the elements (Ga, In,
Si, Ge, As, and Sb) form crystals with predominantly covalent bonding. Thus, in crystals
of Si and Ge the bonding is completely covalent, whereas in crystals of InSb and GaAs
the contribution of ionic bonding to the total binding energy amounts to 32%.
C.2.4 The nature of repulsion forces
Repulsion between atoms (ions) at small distances (r ≤ 0.1 nm) has a purely quantum
origin. This is because, at uniform pressure of a crystalline specimen, electron shells
penetrate into each other and, thus, increase the specimen’s electron concentration, n.In
addition, the kinetic energy of electrons is increased, which can be ascribed to Heisenberg’s
uncertainty principle. Indeed, when the specimen’s volume decreases, the uncertainty in
coordinate position for each electron decreases, and as a result of this the uncertainty in
electron momentum increases. The increase in momentum uncertainty is possible only
with an increase of momentum and, therefore, with an increase of the average kinetic
energy of electrons
E
. To explain qualitatively the effect of repulsion, let us consider
“free” electrons in a metal. The array of these electrons constitutes a degenerate electron
gas. Taking into account the expression for the average energy,
E
, of a degenerate
electron gas (see Eqs. (C.32) and (C.38) in Section C.3.2 of this appendix), we can write
the expression for the density of kinetic energy of the electron gas, w
e
:
w
e
=
E(n)
n = γ n
5/3
, (C.10)
where
γ =
3
5/3
π
4/3
h
-
2
10m
e
. (C.11)

390 Appendix C. Crystals as atomic lattices
The internal pressure of free electrons in metals, P(n), is related to the density of electron
kinetic energy, w
e
(n), in metals by
P(n) =
2
3
w
e
(n) =
2
3
E(n)
n. (C.12)
By applying uniform pressure, we decrease the volume, and as a result we increase the
electron density in the specimen. Since
n =
N
V
≈
N
Nβr
3
=
1
βr
3
, (C.13)
where β is a factor that depends on the geometry of the lattice and is approximately equal
to unity, and r is the average distance between electrons, we have
dn =−
3
βr
4
dr =−3n
dr
r
(C.14)
(under compression dr < 0, therefore dn > 0).FromEq.(C.12) it follows that an increase
of electron density, dn, leads to an increase of the electron-gas pressure:
dP =
2
3
dw
e
dn
dn =−2n
dw
e
dn
dr
r
. (C.15)
The estimate of this quantity for copper with electron density n ≈ 8 ×10
28
m
−3
in the
case of decreasing the distance between electrons (due to uniform compression) by 10%,
i.e., at |dr|=0.1r,is
dP ≈ 4 × 10
9
Pa ≈ 4 ×10
4
atm. (C.16)
Here we used the dependence w
e
on n for a degenerate electron gas. Such overpressure is
due solely to the high forces of repulsion.
Thus, when ions in a metal approach each other, the lattice constant decreases. There-
fore, the density and pressure of the electron gas increase, which leads to the emergence
of significant repulsion forces. Let us substitute the average distance between electrons,
r ≈ n
−1/3
, (C.17)
into the expression for the average energy,
E
= γ n
2/3
=
γ
r
2
. (C.18)
This quantity can be interpreted as the potential energy of repulsion of neighboring ions
in a metal, i.e.,
U (r) =
A
r
2
, A = γ. (C.19)

C.2 The nature of attraction and repulsion forces 391
As the distance between atoms and ions decreases, the overlapping of electron wave-
functions not only of outer shells but also of inner shells increases. As a result, the
forces of repulsion sharply increase. The quantum-mechanical theory gives the following
expression for the potential energy of repulsion:
U (r) = A
e
−r/a
, (C.20)
which slightly differs from the first term in Eq. (C.3), A/r
m
, when the parameter m
takes the range of values m ≈ 9−11. Because of overlapping of electron shells, forces of
repulsion occur in dielectric crystals. Note that in dielectrics the parameter m has large
values, whereas in metals this parameter is small (m = 2). This is why most dielectric
crystals are fragile and metals are ductile.
Example C.1. Estimate the energy of van der Waals bonding of atoms located at distance
r ≈ 3 ×10
−10
m from each other.
Reasoning. Let us assume that the instantaneous dipole moment of one of the atoms,
which occurs as a result of fluctuation of charge, is equal to p
1
. At the distance r from
this atom, at the center of another atom, which is positioned on the same axis as the first
atom, an electric field, E, is created:
E =
2k
e
p
1
r
3
. (C.21)
This field induces an instantaneous dipole moment of the second atom:
p
2
= αE =
2k
e
αp
1
r
3
, (C.22)
where α is the atom’s polarizability. Now, the energy of interaction of two atoms can be
defined as the energy of interaction of two electric dipoles. The general expression for the
energy of such interaction has the form
U (r) = k
e
p
1
· p
2
r
3
−
3(p
1
· r)(p
2
· r)
r
5
. (C.23)
Since the dipole moments of atoms p
1
and p
2
are parallel to r,Eq.(C.23) takes the form
U (r) =−
2k
e
p
1
p
2
r
3
=−
4k
2
e
αp
2
1
r
6
. (C.24)
From Eq. (C.24) it follows that these two atoms are attracted to each other because of
instantaneous dipole–dipole interaction. Electron polarizability has the dimensionality
[m
3
], and for its estimation we can use the following approximate expression:
α ≈ a
3
, (C.25)

392 Appendix C. Crystals as atomic lattices
where a is the atomic radius. To estimate the dipole moment of an atom we can use the
following expression:
p
1
≈ ea. (C.26)
As a result, for the interaction energy of two atoms we obtain the following expression:
U (r) ≈−
4k
e
e
2
a
5
r
6
. (C.27)
Assuming a ≈ 10
−10
mandr ≈ 4 ×10
−10
m, we get U (r) ≈ 2.5 ×10
−21
J ≈ 0.02 eV.
Recalculating for one mole, this energy is equal to 1.5 kJ mol
−1
, which is of the same
order of magnitude as the real van der Waals binding energy.
C.3 Degenerate electron gas
C.3.1 The quantum statistics of electrons
Electrical, optical, and other properties of metals mostly are defined by the state of
conduction electrons in metals. The conduction electrons are called free electrons because
they are not bound to any particular atom of the crystalline lattice. As we have already
noted, free electrons in metals obey quantum statistics. Therefore a gas of free electrons is
called degenerate. In classical electron theory at T = 0 K the energy of all free electrons
is equal to zero, i.e., all electrons are in one lowest-energy state. Quantum statistics does
not allow such a state of the ensemble of electrons. Because of Pauli’s principle, even
at zero temperature the electrons must be in different energy states. Beginning from the
lowest state, electrons fill one after another (with two electrons with spins having opposite
directions on a single level) discrete energy levels of the allowed valence band up to an
energy level called the Fermi energy, which is denoted as E
F
. The filling of energy levels
by electrons is defined by the Fermi function, f (E):
f (E) =
1
e
(E−E
F
)/(k
B
T )
+ 1
, (C.28)
which defines the probability of filling by electrons of the corresponding level with energy
E under conditions of thermodynamic equilibrium of the electrons in crystal. Let us find
out how electrons fill the energy levels in a metal at zero and non-zero temperatures.
If the temperature of a metal tends to absolute zero, then, according to Eq. (C.28),
for electrons with E > E
F
the function f (E) → 0, and for electrons with E < E
F
we
have f (E) = 1. It follows from the above that energy levels lower than the Fermi level
are occupied by electrons, since the probability of filling these levels is equal to unity.
Energy levels higher than the Fermi level are unoccupied, since the probability of their
filling is equal to zero. Figure 7.3 shows a plot of the function f (E) at absolute zero.
Thus, the Fermi energy is the energy of the highest level occupied by electrons in a metal
at T = 0K.
In the case of non-zero temperature, i.e., at T > 0 K, the probability of occupation by
electrons of an energy level with E = E
F
is equal to 1/2. If E < E
F
, then the probability
