Moulson A.J., Herbert J.M. Electroceramics: Materials, Properties, Applications
Подождите немного. Документ загружается.


growth process must therefore be slow and so requires a precise control over
conditions over prolonged periods.
The formation of a solid phase at the freezing point of a liquid only occurs at
the surface of existing solids, e.g. dust particles in water at 0 8C. This process is
known as heterogeneous nucleation. Liquids free from solid particles can be
cooled to some extent below their melting points. Such a supercooled liquid
crystallizes through the spontaneous formation of ordered regions closely similar
to the solid in structure, and the growth of these nuclei leads to the rapid
formation of a large number of randomly oriented crystals. Single-crystal growth
can be obtained by providing nuclei in the form of a small single crystal (a ‘seed’)
and ensuring that the conditions confine growth to the immediate vicinity of the
seed.
Where a seed crystal is not available or, by reason of the growth method,
cannot be utilized, advantage can be taken of the tendency for crystals to grow
more rapidly in one crystallographic direction than in others, as for instance in
the Bridgman–Stockbarger method outlined later. In the same way, if a thin
ceramic rod having the required stoichiometric composition is held vertically and
a small region near one end is fused by means of a laser beam and the molten
region is moved towards the other end of the rod, those crystals that grow fastest
along the axis of the rod will enlarge at the expense of those growing at angles to
it. By passing the molten zone up and down the crystal several times relatively
large single-crystal regions can be obtained. At the same time there will be a
zone-refining effect through which the impurities become concentrated at the
ends of the rod, thus leading to the growth of more perfect crystals.
Whilst every material presents its own problems in crystal growth, which must
be solved experimentally, there is a general thermodynamic principle that gives
an indication of how difficult the process is likely to be. Since crystal growth
takes place reversibly, the Gibbs energy G must be constant and so, under
isothermal conditions,
DG ¼ 0 ¼ D H TDS ð3:1Þ
where H is the enthalpy or heat content of the system, S is its entropy and T is
the thermodynamic temperature. Therefore
DS ¼
DH
T
ð3:2Þ
If we take DH ¼ L
a
, where L
a
is the latent heat per atom, Jackson’s
dimensionless parameter a is defined by
a ¼
DS
k
¼
L
a
kT
CR
ð3:3Þ
where k is Boltzmann’s constant and T
CR
is the temperature of the change of
state. a represents the decrease in entropy on going from the disordered structure
122 PROCESSING OF CERAMICS
TEAMFLY
Team-Fly
®

in the liquid state to the ordered lattice structure of the solid state. Some values
of a are given in Table 3.2.
For a 5 2 crystals grow without facets and their shape is determined by the
isotherms in the melt. Many metals, e.g. iron and lead, are in this category. For
2 5 a 5 10, which covers the bulk of materials, facetting may occur during
growth as with germanium and silicon. For a 4 10 crystals nucleate readily so
that it is difficult to avoid a polycrystalline structure. The value of a for the
growth of ice from water vapour at 0 8C is 20, which accounts for the low-density
polycrystalline character of snow. In contrast, a ¼ 2:7 for the growth of ice from
liquid water, and a dense material containing large crystals is readily formed.
An important consideration in the case of oxides containing several cations is
whether the melting point is congruent or incongruent. In the latter case crystals
cannot be grown from stoichiometric melts and a method must be found for
growth at a temperature at which the required compound is stable. A similar
difficulty occurs when the structure of the crystals formed at the melting point
differs from that required. For example, silica yields cristobalite on solidifying at
1720 8C; cristobalite transforms into tridymite below 1470 8C and tridymite
transforms to b-quartz below 867 8C, while b-quartz transforms to a-quartz
below 573 8C. a-quartz must therefore be grown below 573 8C, as explained
below.
The following methods have been used to grow crystals of oxides.
1. From an aqueous solution by cooling.
2. From solution in an oxide or fluoride flux by cooling.
3. From the liquid phase by cooling:
(a) by first freezing at the lowest point of a melt;
(b) by first freezing at the upper surface of a melt;
(c) by the flame-fusion or Verneuil method.
THE GROWTH OF SINGLE CRYSTALS 123
Table 3.2 Values of Jackson’s parameter a
Substance
Change of
state
Temperature T
CR
=K
of change
of state
Latent heat
L
m
=kJ mol
1
at T
CR
a ¼
L
m
=R
0
T
CR
Si Liquid–solid 1680 46.4 3.3
Ge Liquid–solid 1230 31.8 3.1
Fe Liquid–solid 1810 15.4 1.02
Pb Liquid–solid 601 4.77 0.96
H
2
O Liquid–solid 273 6.03 2.7
H
2
O Vapour–solid 273 45 20
4. (a) From the liquid phase kept at constant temperature by dipping a seed in
the surface and then withdrawing it into a cooler zone;
(b) from the liquid phase by deposition on a substrate of differing
composition.
5. From the vapour phase by chemical reaction close to the surface of a seed
crystal.
These methods are illustrated by the following examples.
1. Quartz is grown from an alkaline solution in water under a pressure of about
150 MPa (1500 atm). Pure mineral quartz fragments are placed at the bottom
of a tall cylindrical autoclave that is 80% filled with either a 2.2% solution of
NaOH or a 5% solution of Na
2
CO
3
. Seed crystals are held in wire frames near
the top of the autoclave. Because the base of the autoclave is kept at 400 8C
and the top is maintained at a temperature some 40 8C cooler, quartz dissolves
at the bottom of the vessel and is deposited on the seeds at the top. Crystals
weighing over a kilogram can be grown in a few weeks (Fig. 3.8). This
hydrothermal method is also of value for such materials as zinc oxide which
have high vapour pressures at their melting points.
2. Crystals of many compounds, of sufficient size for scientific purposes, can be
obtained by cooling a solution of the required compound in a suitable flux.
BaTiO
3
crystals have been prepared by using a mixture of KF with 30 wt%
BaTiO
3
and 0.2 wt% Fe
2
O
3
[15]. A temperature of 1150–1200 8Cis
maintained for 8 h, and the mixture is then cooled slowly to 900 8C when
the flux is poured off and the crystals are allowed to cool in the furnace to
room temperature. The crystals form as butterfly twins, i.e. as pairs of
triangular plates joined at one edge with an angle of about 608 between them.
The iron assists the formation of flat plates of relatively large area that are
convenient for the investigation of the ferroelectric properties of BaTiO
3
.A
small amount of iron, as Fe
3þ
, is incorporated in the BaTiO
3
lattice on Ti
4þ
sites. The charge difference is compensated by the replacement of an
equivalent number of O
2
ions by F
ions. This result is typical of flux-
growth methods which frequently result in the incorporation of a small
proportion of the solvent in the crystal lattice.
Single crystal rods of a variety of compositions, including complex garnets
(see Section 9.5.4) can be grown by the ‘floating zone’ method. For example,
two sintered Bi-substituted garnet (BIG) rods of the chosen composition are
placed end to end with a single crystal ‘seed’ of the same composition
sandwiched between them. Halogen lamps are focused onto the join area as
the rods are rotated and provide sufficient heat to form a molten zone. If the
molten zone is moved into the sintered rod in a suitably controlled way then a
124 PROCESSING OF CERAMICS

THE GROWTH OF SINGLE CRYSTALS 125
Fig. 3.8 Hydrothermally grown quartz crystals.
high quality single crystal can be grown. The whole operation is carried out in
an oxygen atmosphere to ensure the iron is in the fully oxidized state. Very
high quality crystals of size 12 mm diameter 120 mm long suited to magneto-
optical applications can be grown.
In recent years strong interest has been shown in single crystals of the
relaxor ferroelectrics for actuator applications (see Section 6.4.3). Members of
this family of electroceramics exhibit exceptionally large electric field-
induced strains. One such material has the composition approximately
0.9Pb(Zn
1/3
Nb
2/3
)O
3
-0.1PbTiO
3
abbreviated 0.9PZN–0.1PT. The starting
materials are Pb
3
O
4
, ZnO, Nb
2
O
5
and TiO
2
. The constituents, in amounts
to give the required overall composition, are well mixed and then heated with
an excess of Pb
3
O
4
which serves as the flux, in a sealed platinum crucible to
approximately 1150 8C. The melt is then slowly cooled at approximately
5 8Ch
1
when crystals with dimensions up to 20 mm grow. When the crucible
is cold the crystals are extracted with the help of hot nitric acid.
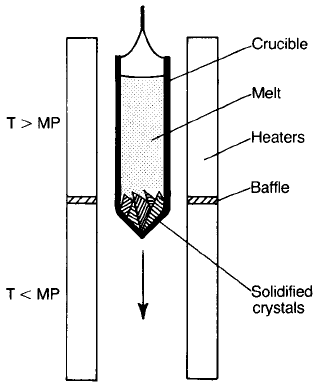
In order to form manganese zinc ferrite crystals using the Bridgman–
Stockbarger method (Fig. 3.9) a charge of the mixed oxides is melted in a Pt–Rh
crucible and kept just above its solidification temperature. The furnace is
designed such that there is a sharp drop in temperature just below the bottom tip
of the crucible in its initial position. The crucible is lowered so that the tip enters
the colder zone causing the nucleation of crystals. The crystals grow fastest in
particular crystallographic directions and those growing at angles greater than
half the cone angle terminate at the walls of the cone; only those crystals
oriented so that growth is favoured in the axial direction persist into the bulk
of the charge as the crucible is lowered into the cooler zone. As a result the
upper part of the crucible finally contains either a single crystal or a few large
crystals.
There are a number of problems associated with growing manganese zinc
ferrite single crystals by the process outlined above. Temperatures close to
1800 8C are required to melt the oxides, and then there is rapid evaporation of
zinc and loss of oxygen from the Fe
2
O
3
resulting in the formation of FeO. These
difficulties have been largely overcome through the development of a high-
pressure version of the apparatus. The crucible, now in a sealed container so that
an oxygen pressure of up to 2 MPa (20 atm) can be maintained over the melt, is
heated indirectly by a surrounding thick cylindrical Pt–Rh susceptor. The
thickness of the inductively heated susceptor is sufficient to shield the melt from
the radiofrequency field which would otherwise produce eddy current heating in
the melt, resulting in undesirable convective agitation.
A further problem arises owing to differences in composition between the
liquid and solid phases. The cation distribution can be optimized and the excess
FeO oxidized by annealing the crystal in a suitable atmosphere after cutting the
required shapes.
126 PROCESSING OF CERAMICS
Pure cubic BaTiO
3
cannot be grown from a melt of that composition because,
as shown in Fig. 5.41, the hexagonal form is in equilibrium with the liquid at the
solidification temperature (1618 8C). It can, however, be grown from a
composition containing 35 mol.% BaO, 65 mol.% TiO
2
, which solidifies below
1460 8C, the temperature below which the cubic form is stable. The melt is held
just above its solidification point, a seed crystal is dipped into its surface and the
melt is then cooled at between 0.1 and 0.5 8Ch
1
. The seed crystal is attached to a
platinum stirrer which is rotated during growth so that the liquid near the crystal
does not become depleted in barium. Only a fraction of the melt can be obtained
as single-crystal BaTiO
3
since the whole of it solidifies, forming a mixture of
cubic BaTiO
3
and Ba
6
Ti
17
O
40
, when the BaO content falls to about 32 mol.%.
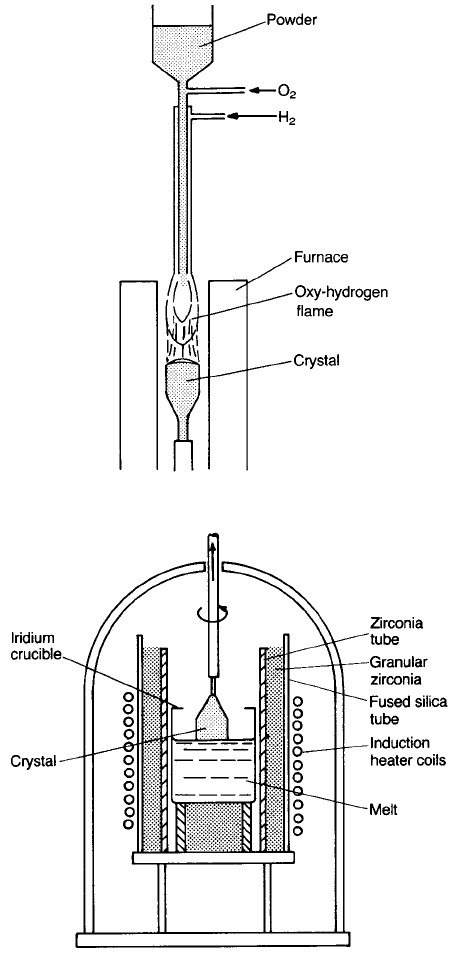
The Verneuil flame-fusion method illustrated in Fig. 3.10 is a well-established
but relatively crude method for growing refractory oxide single crystals,
particularly sapphire and ruby. In this case alumina powder is fed at a controlled
rate down a tube at the end of which it is melted by an oxyhydrogen flame; it
then drips down into a shallow pool of liquid on top of the seed crystal. As the
crystal grows, it is lowered into the annealing zone. This method suffers from the
disadvantage of poor control over the growth environment, and the quality of
crystal produced is inferior to that obtained by crystal pulling. It is adequate,
however, for jewelled bearings.
Crystal pulling is one of the most widely exploited and successful processes,
and is generally termed the Czochralski method. Silicon, gallium arsenide,
alumina, lithium niobate, lithium tantalate and gadolinium gallium garnet are
all grown on a large scale by variants of this process which is applicable to
most materials that melt congruently. The melt is kept at a temperature just
THE GROWTH OF SINGLE CRYSTALS 127
Fig. 3.9 The Bridgman–Stockbarger technique (MP, melting point).
above its freezing point and a seed crystal, firmly fixed to a rotating tube, is
lowered into the surface and then slowly withdrawn (Fig. 3.11). Air can be
blown down the tube to control the extraction of heat necessary to the growth
process. The crystal diameter can be varied by changing the rate of withdrawal.
128 PROCESSING OF CERAMICS
Fig. 3.10 The Verneuil technique.
Fig. 3.11 The Czochralski technique for pulling crystals.
Flaws and misoriented crystals can be eliminated by first enlarging and then
diminishing the crystal diameter; misoriented regions are thereby terminated at
the crystal surface in much the same way as in a Stockbarger–Bridgman
crucible. In favourable cases nearly 90% of a melt can be obtained in single-
crystal form.
Smaller crystals can be grown from solutions by a similar method; for
example, a mixture of barium and strontium titanates containing a 28% excess of
TiO
2
can be melted at 1500 8C and crystals of (Ba, Sr)TiO
3
withdrawn on a seed.
The cubic crystals are stoichiometric in their ratio of SrþBa to Ti but contain a
substantially greater proportion of strontium than the liquid phase. Provided
that the mass of the crystals constitutes less than 1% of that of the melt, they
have a highly homogeneous composition despite the inevitable change in
composition of the liquid phase as the crystal forms.
In some applications only a thin film of single-crystal material is needed on a
substrate that differs in composition. In this case advantage can be taken of the
possibility of epitaxial deposition. A seed crystal of similar crystal structure but
differing composition from the required film can be used in place of a seed of
identical composition. The method is of particular value when the deposit has a
complex composition and a suitable substrate material is available. The
magnetic garnets that were used in the now obsolete ‘bubble’ memory stores
with complex compositions such as (Y
0:9
Sm
0:5
Tm
1:1
Ca
0:5
)(Fe
2
)(Fe
2:5
Ge
0:5
)O
12
,
were grown by liquid phase epitaxy (LPE) on gadolinium gallium garnet
substrates.
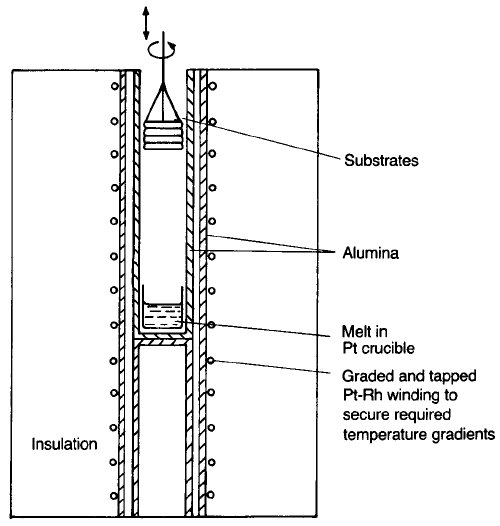
The apparatus for LPE growth of garnet film is shown schematically in
Fig. 3.12. The melt composition is typically PbO–B
2
O
3
in a 50:1 ratio by weight
to which sufficient garnet constituent oxides are added to form a saturated
solution between 950 and 1000 8C. A set of circular substrates is held in a
platinum holder above the solution which is attached to the rotatable spindle.
The melt is allowed to cool a few degrees so that it becomes supersaturated and
the substrates are then lowered into it and rotated for a sufficient time (some
minutes) for a film of a garnet of the required thickness to grow. They are then
raised from the melt and spun rapidly to remove the excess liquid. The thickness
for bubble memories was usually about 4 mm, though films of thickness up to
15 mm can be grown.
The growth of epitaxial layers from the vapour phase has been developed
extensively for III–V compounds for use in semiconductor and electro-optical
devices. The method most applicable to oxide systems is metal–organic chemical
vapour phase deposition (MOCVD). Metals form a wide range of volatile
organic compounds such as lead tetraethyl, (Pb(C
2
H
5
)
4
), silicon tetraethoxide
(Si(OC
2
H
5
)
4
) and titanium isopropoxide (Ti(C
3
H
7
O)
4
), and these are readily
converted to oxide by reaction with oxygen or water and by thermal
decomposition. Mixtures of such compounds in vapour form can be reacted in
close proximity to heated substrates to form oxide deposits that interact to form
THE GROWTH OF SINGLE CRYSTALS 129
titanates, zirconates etc. Polycrystalline layers may be adequate for some
purposes, but with suitable substrates single-crystal layers are possible.
Problems
1. Estimate the density of a compact of uniformly close-packed alumina powder,
assuming a mean particle diameter of 25 mm. What would the density be if the close-
packed particles have a mean diameter of 10 mm? Estimate the compact density if
25 wt% of the powder has a small enough particle diameter for it to occupy the
interstices in the close-packed larger fraction (crystal density of Al
2
O
3
, 4000 kg m
3
).
[Answers: 2960 kg m
3
; 2960 kg m
3
; 3730 kg m
3
].
2. A sample of alumina powder has a mean ‘particle’ diameter of 25 mm. What is the
apparent specific surface area (SSA) of the powder? If each ‘particle’ is in reality an
agglomerate of ultimate particles of diameter 0.5 mm, estimate (i) the number of
ultimate particles per agglomerate and (ii) the true SSA of the powder. [Answers:
0.06 mg
2
g
1
; 10
5
;3m
2
g
1
].
3. A colloid in suspension in an electrolyte acquires a surface charge and an ‘atmosphere’
of oppositely charged ions. An inner part of the atmosphere moves with the colloid
130 PROCESSING OF CERAMICS
Fig. 3.12 Schematic diagram of a furnace for LPE growth of magnetic garnet films.

and, together with it, constitutes an ‘electrokinetic unit’. If the net charge on the unit is
Q, and assuming that Stokes’ law applies, derive the equation
u ¼
4ez
6Z
where u is the electrophoretic mobility, e is the effective permittivity of the electrolyte
in the immediate vicinity of the colloid, z is the zeta potential and Z is the viscosity of
the liquid. Write a brief account of the significance of the zeta potential in determining
the stability of casting slips. (see [a], or similar)
4. The density of an aqueous alumina slip is 2.5 kg dm
3
. Calculate the concentration of
dry alumina in the slip on (i) a weight basis and (ii) a volume basis. [Answers: 80 wt%;
50 vol.%]
5. A pore-free polycrystalline solid contains two phases, A of density 4000 kg m
3
and B
of density 3000 kg m
3
. Calculate the density of the solid if it contains (i) 60 wt% of
phase A and (ii) 60 vol.% of phase A. [Answers: 3529 kg m
3
; 3600 kg m
3
]
6. As part of the characterization of an electrical porcelain the following weighings were
made at 25 8C using a balance which was accurate to the third decimal place:
single-piece sample of ‘body’ 15.234 g
sample saturated with water 15.408 g
saturated sample suspended in water 8.948 g
specific gravity bottle 22.432 g
bottle þ sample of finely ground dry ‘body’ 27.362 g
bottle þ powder þ water 75.379 g
bottle þ water 72.429 g
Calculate (i) the true density of the ‘body’, (ii) the bulk density of the ‘body’, (iii) the
open porosity and (iv) the total porosity (density of water at 25 8C, 997 kg m
3
).
[Answers: 2.482 Mg m
3
; 2.351 Mg m
3
; 2.7%; 5.3%]
7. Assuming a reasonable value for the surface energy of an oxide estimate the ‘negative
pressure’ tending to close spherical pores of radius (i) 1 mm and (ii) 0.01 mm during
sintering. Express your answer in megapascals and ‘atmospheres’. [Answers: 2 MPa
( 20 atm); 200 MPa (2000 atm)]
8. Compounds A and B interdiffuse a distance of approximately 1 mm in 1 h at 1000 8C. If
the activation energy for the rate-determining step is 3:2 10
19
J, calculate the
interdiffusion distance for a 10 h anneal at 1600 8C assuming the same diffusion
mechanism. [Answer: 58 mm]
9. A circular hole in an alumina plate at 20 8C is 1 cm in diameter. Calculate the diameter
at 1000 8C. If the density of alumina at 20 8C is 3980 kg m
3
, calculate the density at
1000 8C. Mean coefficient of linear expansion over the temperature range 20–1000 8C
is 8.0 MK
1
. [Answer: 1.0078 cm; 3886 kg m
3
]
PROBLEMS 131
