Moulson A.J., Herbert J.M. Electroceramics: Materials, Properties, Applications
Подождите немного. Документ загружается.

10 000 h. LaCrO
3
was considered a prime candidate for this application since it
combines a melting point of 2500 8C with high electronic conductivity (about
100 S m
1
at 1400 8C) and resistance to corrosion. There is now little interest in
MHD systems but LaCrO
3
is established as a specialized heating element.
LaCrO
3
is one of the family of lanthanide perovskites RTO
3
, where R is a
lanthanide and T is a period 4 transition element. In the cubic unit cell R
occupies the cube corners, T the cube centre and O the face-centre positions. The
coordination numbers of T and R are 6 and 8 respectively. LaCrO
3
loses
chromium at high temperatures, leaving an excess of O
2
ions. The excess charge
is neutralized by the formation of Cr
4þ
which results in p-type semiconductivity
with ‘hole hopping’ via the localized 3d states of the Cr
3þ
and Cr
4þ
ions. The
concentration of Cr
4þ
can be enhanced by the substitution of strontium for
lanthanum. A 1 mol.% addition of SrO causes the conductivity to increase by a
factor of approximately 10 (see Section 2.6.2).
Heating elements are formed by normal ceramic processing. Strontium acts as
a sintering aid as well as promoting conductivity, and cobalt can be added to
limit grain growth. Sintering takes place in a reducing atmosphere
(p
O
2
10
12
atm or 10
7
Pa) at temperatures close to 1700 8C and is followed
by an anneal in oxygen that establishes the high conductivity.
Satisfactory conductivity is maintained up to 1800 8C in air but falls off at low
oxygen pressures so that the upper temperature limit is reduced to 1400 8C when
the pressure is reduced to 0.1 Pa. A further limitation arises from the volatility of
Cr
2
O
3
which may contaminate the furnace charge. The combination of high
melting point, high electronic conductivity and resistance to corrosion has led to
the adoption of lanthanum chromite for the interconnect in high temperature
solid oxide fuel cells (see Section 4.5.3).
4.1.4 Tin oxide
Tin oxide (SnO
4
) has found applications in high-temperature conductors, ohmic
resistors, transparent thin-film electrodes and gas sensors.
It crystallizes in the tetragonal rutile structure (see Fig. 5.27) with cell
dimensions a ¼ 474 pm and c ¼ 319 pm; in the single-crystal form it is known by
its mineralogical name, cassiterite. It is a wide band gap semiconductor, with the
full valence band derived from the O 2p level and the empty conduction band
from the Sn 5s level. The band gap at 0 K is approximately 3.7 eV, and therefore
pure stoichiometric SnO
2
is a good insulator at room temperature when its
resistivity is probably of the order of 10
6
O m.
In practice both natural and synthetic crystals are oxygen deficient, leading to
donor levels approximately 0.1 eV below the bottom of the conduction band and
consequently to n-type semiconductivity. Doping the crystal with group V
elements also induces n-type semiconductivity; the usual dopant is antimony.
The ground state electronic configuration of the Sb atom is 5s
2
p
3
, and when it
142 CERAMIC CONDUCTORS
TEAMFLY
Team-Fly
®
replaces Sn
4þ
(r
6
¼ 69 pm) in the SnO
2
lattice it does so as Sb
5þ
(r
6
¼ 61 pm). The
n-type conductivity is due to electrons ionized from the 5s
1
state.
Antimony-doped SnO
2
is a complex system and is far from completely
understood. The interpretation of data is complicated by the fact that both the
stoichiometry of the host lattice and the oxidation state of the dopant are
dependent upon ambient oxygen partial pressure and temperature. The
successful exploitation of semiconducting SnO
2
has been achieved largely
through development work guided by a general understanding of underlying
principles, as indeed is the case for much of electroceramics technology.
An important application of SnO
2
in ceramic form is in conducting electrodes
for melting special glasses, such as those used for optical components and lead
‘crystal’ tableware. The ideal glass-melting electrode should have a high electrical
conductivity at room temperature and, of course, at glass-melting temperatures
and a high resistance to corrosion by the glass. In addition, it must not discolour
the glass. SnO
2
is the only material, apart from platinum, which fulfils these
requirements for glasses containing lead oxide.
SnO
2
itself does not readily sinter to a dense ceramic and so sintering aids such
as ZnO (r
6
(Zn
2þ
) ¼ 75 pm) and CuO (r
6
(Cu
2þ
) ¼ 73 pm) are added, together with
group V elements such as antimony and arsenic to induce semiconductivity. Some
of the sintering aids enter the lattice, tending to negate the effect of the dopants
added to induce semiconductivity, and successful compositions, typically
containing more than 98 wt% SnO
2
, have been arrived at by trial and error.
The oxide powder, together with binders, is commonly isostatically pressed
into the forms shown in Fig. 4.3 and fired in oxidizing conditions at
approximately 1400 8C. Under these conditions sintered densities in the range
6500–6700 kg m
3
(theoretical density for SnO
2
is 7002 kg m
3
) are achieved. The
largest electrodes made in this way are in the form of cylinders about 600 mm
long and 150 mm in diameter weighing approximately 60 kg. Cooling from the
sintering temperature is carried out, in part, in a nitrogen atmosphere with the
object of creating electron-compensated oxygen vacancies and so enhancing
room temperature conductivity which is typically of the order of 10
3
Sm
1
. The
high conductivity minimizes Joule heating in the electrode region outside the
molten glass.
The form of the glass-melting furnace is shown in Fig. 4.4 although in practice
the electrodes may well be a substantial part of the furnace walls. The charge
(‘cullet’) is preheated, using gas or oil, to about 1000 8C when it has sufficient
conductivity to be directly heated to the required temperature (1300–1600 8C) by
power dissipated internally. By supplying the heat from within the body of the
glass melt rather than from the outside, the free surface temperature is kept
relatively low and thus the excessive loss of volatile elements, particularly lead, is
avoided. The process is economic since the heat is generated in the glass, where it
is required. The elements are resistant to attack by glass and so have long service
lives, typically 2 years.
HIGH-TEMPERATURE HEATING ELEMENTS AND ELECTRODES 143

144 CERAMIC CONDUCTORS
Fig. 4.3 Isostatically pressed tin oxide electrodes for producing high quality glasses; the
length of the longest electrode in the illustration is approximately 500 mm. (The contacting
stubs may be wrapped with silver foil to reduce resistance.) (Courtesy of Dyson Technical
Ceramics.)
Fig. 4.4 Heating a glass melt: the power (I
2
R) is introduced directly into the melt via tin
oxide electrodes.
4.1.5 Zirconia
The ‘Nernst filament’, once used as a light source, consisted of zirconia (ZrO
2
)
doped with thoria and ceria. Preheating the filament to about 600 8C using an
external source reduces its resistance to a value which permits direct Joule
heating to be effective when temperatures of up to 1800 8C can be attained. More
recently the development of stabilized ZrO
2
(see Section 4.5.3) has made it
possible to use ZrO
2
-based ceramics as furnace elements, although they also
always require preheating to reduce the resistance to a level at which Joule
heating is effective. Its high negative temperature coefficient of resistance makes
it necessary to have a resistor, preferably with a high positive temperature
coefficient, in series with it to limit current.
Once the element has reached temperatures exceeding about 700 8C it can also
be used as a susceptor and heated by eddy currents generated by an induction
coil and a high frequency (0.1–10 MHz) power source.
ZrO
2
was one of the earliest ceramics in which conduction by oxygen ions was
observed and now this characteristic is exploited in fuel cell and sensor
technologies (see Sections 4.5.3 and 4.6.1).
4.2 Ohmic Resistors
Most resistors for electrical and electronic applications are required to be ohmic
and to have small temperature coefficients of resistance. The major requirement in
electronics is for resistors in the range 10
3
–10
8
O, while materials with suitable
electrical properties usually have resistivities less than 10
6
O m. Fabrication of a
10
5
O resistor of length 110 mm from a material with a resistivity of 10
6
O m
requires a cross-sectional area of 10
12
m
2
, i.e. a strip 1 mm thick and 1 mm wide, for
example. This is not a technical impossibility, but other more economic routes to
resistor manufacture have been established based on the following two principles.
1. Very thin conductive layers are deposited on an insulating substrate and large
length-to-width ratios are obtained by etching a suitable pattern.
2. The conductive material is diluted with an insulating phase.
These methods are often combined.
4.2.1 Thin ¢lms
Thin films of thickness typically 10 nm are readily formed in a vacuum chamber
by evaporation, ‘sputtering’, or chemical vapour deposition (CVD). Many
metals and metal alloys, e.g. aluminium, silver, gold and Ni–Cr, can be
OHMIC RESISTORS 145
evaporated from the molten state and condensed onto suitable substrates. Ni–Cr
alloys with resistivity values of about 10
6
O m are deposited in thin-film form
and provide a basis for the manufacturer of high-value resistors.
Thin-film oxides are usually formed by ‘sputtering’. The chamber is filled with
argon at a pressure of typically about 1 Pa to which a small amount of oxygen is
added. The target is usually a disc of the oxide to be sputtered fixed to a metal
plate. The substrate on which the film is to be deposited rests on a metal plate. A
high-frequency (about 1 MHz) high-voltage (about 5 kV) is applied between the
two plates and a plasma is developed. Gaseous ions bombard the source and
detach clusters of ions or molecules from its surface which pass through the
plasma and are deposited on the substrate. SnO
2
,In
2
O
3
and mixtures of these
oxides, e.g. 90In
2
O
3
–10SnO
2
, are deposited as transparent conductive films by
this method. They are essential to the functioning of many electro-optical devices.
Indium tin oxide (ITO) films are also commonly deposited by CVD in the
manufacture of film resistors. Glass, or sometimes steatite, rods are heated to
about 700 8C in air of controlled humidity and a mixture of tin tetrachloride and
antimony pentachloride is then introduced for a few seconds. Reaction with water
occurs on the surface of the rods, resulting in the formation of a thin film of mixed
oxides which is firmly attached to the substrate. The antimony lowers the
resistivity of the SnO
2
and also enables the temperature coefficient of resistance to
be controlled near to zero; the SnO
2
alone gives a high negative coefficient. A
small fraction of the O
2
ions in the deposit are replaced by Cl
ions, and these
also lower the resistivity because the lower negative charge of Cl
compared with
O
2
is compensated by an electron in the conduction band. The resulting units
require a protective layer of impervious lacquer to inhibit the effects of moisture
but are very stable and reliable in use; because of the strong adhesion of the film
open circuits do not occur through detachment of small pieces, which
occasionally happens with sputtered carbon films. The presence of moisture
and soluble ionic material on the film can set up electrolytic action which results in
a portion of the film being converted to metallic tin with a resultant diminution in
resistance, but care in production makes this a very rare occurrence.
Enhanced length-to-width ratios can be achieved by cutting a spiral groove
through the resistive film and into the cylindrical rod substrate. Resistance values
can be kept within close tolerances by stopping the groove cutting automatically
when a preset value is reached. Patterned deposits on flat surfaces can be formed
by a number of processes, e.g. by an air jet carrying abrasive particles or by
volatilization by an infrared laser beam or an electron beam. Frequently
photolithography is used when a layer of photosensitive material, called a ‘resist’,
is formed on the deposited film. The resist is exposed to a pattern of light that
renders it insoluble and the unexposed regions are then removed with a solvent.
The deposit is then immersed in a reagent that dissolves the sputtered material
and not the resist. Finally, the remaining resist is removed. Examples of thin-film
resistors are shown in Fig. 4.5.
146 CERAMIC CONDUCTORS

4.2.2 Thick ¢lms
Rather thicker films with thicknesses typically in the range 10–15 mm are made by
what is termed the ‘thick-film’ or ‘silk-screen’ technique. Silk screening is a well-
established method for printing artwork. The screens were formerly silk but are
now meshes of either nylon or stainless steel. Patterns are formed by coating
them with a resist, exposing them to a pattern of light and washing out the
unexposed parts. The screen is held taut in a frame that is fixed 1–3 mm above
the surface to be printed. A paint of a stiff creamy consistency is swept across the
screen by a hard rubber squeegee with sufficient pressure to force the screen, now
loaded with paint, into contact with the underlying surface. The consistency of
the paint is such that, as the screen rises from the surface, it flows over the spaces
left by the threads or wires of the screen so that there is only a small variation in
the thickness of the deposit.
Resistors are formed by taking a conductive powder and mixing it with
powdered glazes and an organic vehicle that imparts the necessary rheological
properties. Mixing is carried out on a set of rollers running at different speeds as
described under calendering (see Section 3.6.7), and this ensures a very high
degree of dispersion of the components. A required resistor pattern is silk
screened onto a substrate, usually a 96% alumina. A carefully controlled firing
schedule ensures that the organic solvents evaporate (100–150 8C), the remaining
organic compounds evaporate or are burned out (200–450 8C) and, finally, the
inorganic glass and active resistive components mature and bond to the substrate
(850 8C for about 10 min). The complete firing takes about 1 h. Metal conductors
and contact pads can be made using the same technique. Examples of thick-film
devices and circuits are shown in Fig. 4.5.
Although it is simple to prepare pastes and use them to make resistors, the
structure of the resulting components is complex and the conduction
mechanisms remain a matter for debate.
The active components are usually highly conductive oxides (10
5
–10
6
Sm
1
)
such as PdO, RuO
2
,Bi
2
Ru
2
O
7
and Bi
2
Ir
2
O
7
. Electrically they behave as metals
and have low positive temperature coefficients of resistivity (TCR). The glaze is
usually a lead borosilicate, of composition typically (in wt%) 52PbO–35SiO
2
–
10B
2
O
3
–3Al
2
O
3
. The resistors normally give high resistivities and negative
temperature coefficients for low concentrations of the conductive component,
and low resistivities and positive temperature coefficients for high concentra-
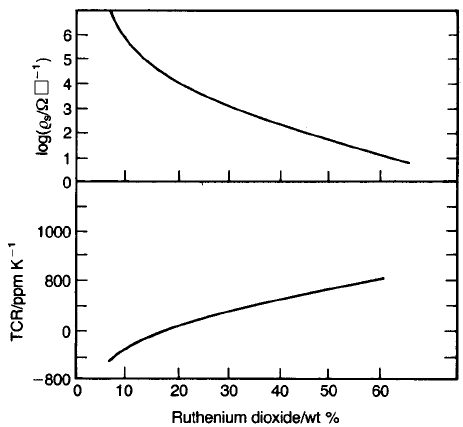
tions, as shown in Fig. 4.6
{
. This behaviour differs markedly from that to be
expected from a random distribution of conductive particles in an insulating
matrix. The conductivity of dispersions of metal particles of mean size around
OHMIC RESISTORS 147
{
In characterizing the resistance of the printed films used in microelectronics it is the practice to define a
‘surface resistivity’, r
s
¼ r=h, in which h is the film thickness. It is also common practice to express r
s
in
‘units’ of ‘ohms per square’ or simply O=
&
; r
s
is equivalent to the resistance measured between opposite
edges of a printed square.

148 CERAMIC CONDUCTORS
Fig. 4.5 Film devices and circuits: (a) thin-film resistors on glass and steatite substrates; (b)
thick-film resistor networks on ‘snapstrate’ alumina substrate; (c) various thick-film resistors;
(d) hybrid microcircuits. (Components kindly supplied by General Hybrid, C-MAC and
Welwyn Components Ltd.)
100 mm in an insulating matrix is very low when the metal concentration is small
and then increases by several orders of magnitude over a concentration range of
a few per cent around 10 vol.% (see Fig. 2.49), with only a small increase for
higher concentrations. This is understandable as a consequence of the
establishment of continuous contact between particles from electrode to
electrode at a well-defined concentration.
Transmission electron micrographs show that the dispersed conductive
particles form convoluted chains with the particles either close together or in
contact. According to this model there will be chains of conductive particles
joining the electrodes even at high dilutions. The resistance is provided by two
components, that due to the interior of the conductive particles and that due to
the areas of near contact between them. The former will provide a positive
temperature coefficient of resistance whilst the latter, if it depends on some form
of thermally activated semiconductive behaviour, will provide a negative
coefficient. So far it has not been possible to determine the properties of the
material between the surfaces of the particles. The solubility of the conductive
oxides in the glaze has been shown to be very small but, as the intergranular layer
is very thin, it may be enough to endow the glaze with a sufficient level of
conductivity.
The glaze component is resistant to moisture so that units are stable in normal
ambients. Their mode of fabrication allows whole patterns of resistors to be
made at once, although strict control of production conditions is necessary to
OHMIC RESISTORS 149
Fig. 4.6 Electrical characteristics of RuO
2
thick-film resistors.
ensure reproducibility. Generally several batches of paste are made and the
values they yield on processing are determined. The required value can then be
obtained by blending two batches whose values span that required, using
Lichtenecker’s relation (Eq. (2.131)) to determine the ratio in which the two end-
members must be combined. Values can finally be trimmed into tolerance by
volatilization using a laser beam or by jet abrasion.
4.3 Voltage-dependent Resistors (Varistors)
4.3.1 Electrical characteristics and applications
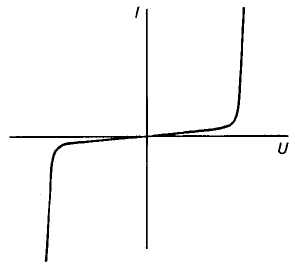
There are a number of situations in which it is valuable to have a resistor which
offers a high resistance at low voltages and a low resistance at high voltages as is
the case in the current–voltage characteristic shown in Fig. 4.7.
Such a device can be used as shown in Fig. 4.8 to protect a circuit from high-
voltage transients by providing a path across the power supply that takes only a
small current under normal conditions but takes a large current if the voltage
rises abnormally, thus preventing high-voltage pulses from reaching the circuit.
High-voltage transients are of sufficiently frequent occurrence in most power
supplies to make the protection of sensitive circuits essential. Transistors and
integrated circuits are particularly susceptible to damage by transients.
Ceramics based on SiC and ZnO are two materials in everyday use that have
the characteristic shown in Fig. 4.7. In both cases it has been established that the
resistance is controlled by the region in which the ceramic grains contact one
another.
The microstructure of a ceramic for a voltage-dependent resistor (VDR) can
be visualized as shown in Fig. 4.9(a) with IGLs of varying thickness between
grains that differ in size by about a factor of 10. An idealized structure which is
150 CERAMIC CONDUCTORS
Fig. 4.7 Typical current–voltage relation for a voltage-dependent resistor.
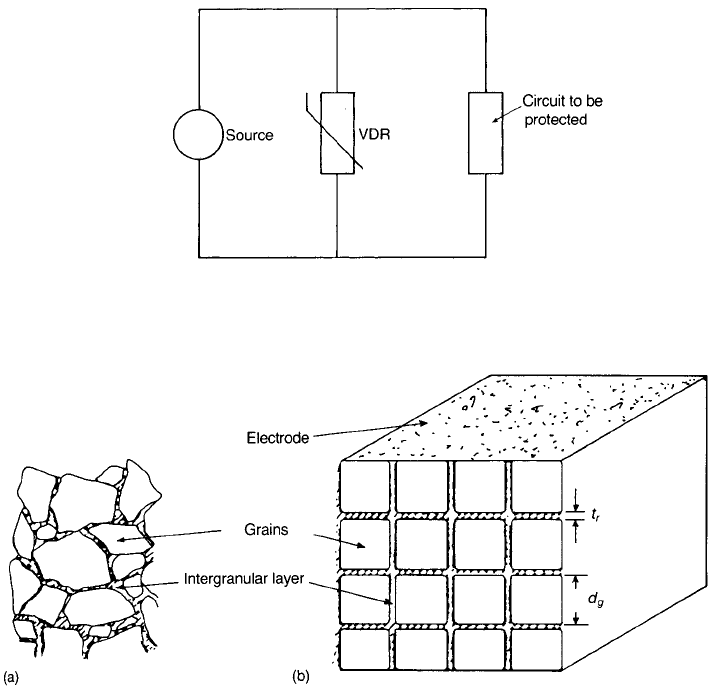
useful for calculating average properties is shown in Fig. 4.9(b), but it must be
appreciated that in practice the high-current paths are likely to be via those
particles separated only by the thinnest IGLs, so that regions of high current
density will form tortuous routes between the electrodes.
The VDR behaviour in ZnO varistors is governed by electron states that are
formed on the surfaces of crystals as a consequence of the discontinuity. These
surface states act as acceptors for electrons from the n-type semiconductor.
Electrons will be withdrawn from the region near the surface and replaced by a
positive space charge. In this way oppositely oriented Schottky barriers will be
created at the surfaces of neighbouring crystals so that a high resistance will be
offered to electron flow in either direction (Fig. 4.10(a)). The situation with an
applied field is shown in Fig. 4.10(b). With low applied fields small thermally
VOLTAGE-DEPENDENT RESISTORS (VARISTORS) 151
Fig. 4.8 Use of a voltage-dependent resistor (VDR) to protect a circuit against transients.
Fig. 4.9 Illustrations of (a) actual and (b) idealized microstructure of a varistor.
