Quinn J.J., Yi K.-S. Solid State Physics: Principles and Modern Applications
Подождите немного. Документ загружается.

Uncorrected Proof
BookID 160928 ChapID 07 Proof# 1 - 29/07/09
210 7 Semiconductors
Problems 622
7.1. Intrinsic carrier concentration can be written as 623
n
i
(T )=2.5
m
c
m
m
v
m
3/4
k
B
T
E
G
3/2
E
G
in eV
1
40
eV
3/2
e
−
E
G
2k
B
T
× 10
19
/cm
3
.
Take E
G
=1.5eV, m
h
=0.7m,andm
e
=0.06m roughly those of GaAs, and 624
plot ln n
i
vs T in the range T = 3K to 300K. 625
7.2. Plot the chemical potential ζ(T )vsT in the range T =3−−300 K for 626
values of E
G
=1.5eV, m
h
=0.7m, and m
e
=0.06 m. 627
7.3. For InSb, we have E
G
0.18eV, ε
s
17, and m
e
0.014m. 628
i) Evaluate the binding energy of a donor. 629
ii) Evaluate the orbit radius of a conduction electron in the ground state. 630
iii) Evaluate the donor concentration at which overlap effects between 631
neighboring impurities become significant. 632
iv) If N
d
=10
14
cm
3
in a sample of InSb, calculate n
c
at T =4K. 633
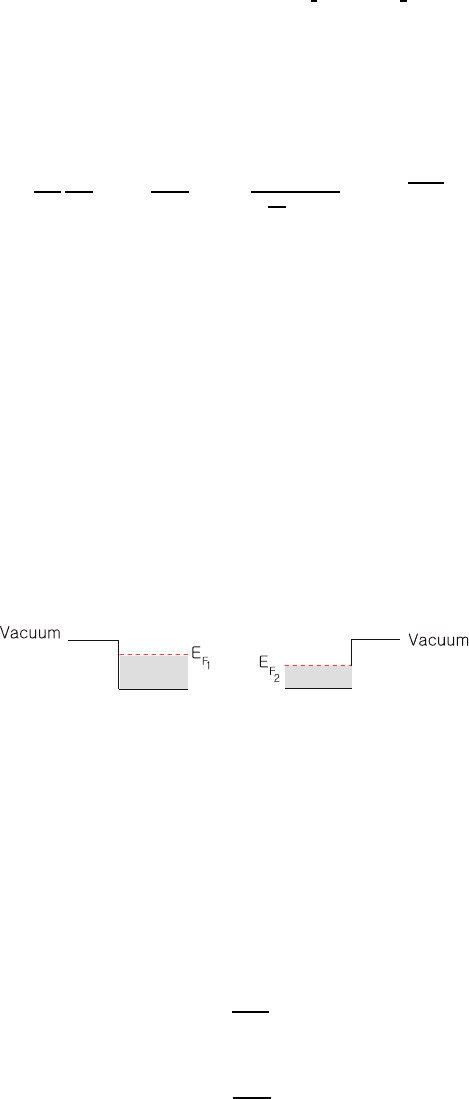
7.4. Let us consider a case that the work function of two metals differ by 2 eV; 634
E
F1
− E
F2
=2eV. 635
If the metals are brought into contact, electrons will flow from metal 1 to 636
metal 2. Assume the transferred electrons are displaced by 3 ×10
−8
cm. How 637
many electrons per square centimeter are transferred? 638
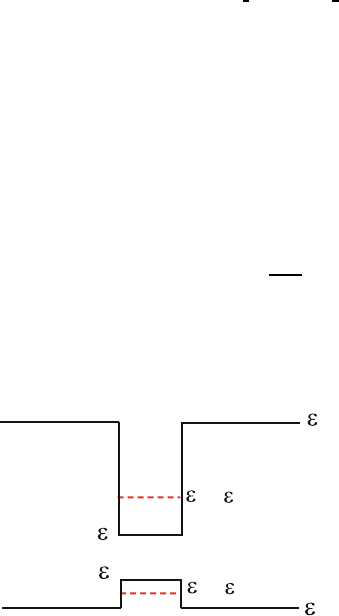
7.5. Consider a semiconductor quantum well consisting of a very thin layer 639
of narrow gap semiconductor of E
G
= ε
c
− ε
v
contained in a wide band gap 640
host material of E
G
= ε
H
c
− ε
H
v
. The conduction and valence band edges are 641
shown in the figure. The dashed lines indicate the positions of energy levels 642
associated with the quantized motion of electrons (ε
c
0
) and holes (ε
v
0
)inthis 643
quantum well. We can write the electron and hole energies, respectively, as 644
ε
c
(k)=˜ε
c
+
¯h
2
2m
c
k
2
x
+ k
2
y
and
645
ε
v
(k)=˜ε
v
−
¯h
2
2m
v
k
2
x
+ k
2
y
where ˜ε
c
= ε
c
+ ε
c
0
and ˜ε
v
= ε
v
+ ε
v
0
. 646
Uncorrected Proof
BookID 160928 ChapID 07 Proof# 1 - 29/07/09
7.8 Amorphous Semiconductors 211
(a) Calculate the two-dimensional density of states for the electrons and 647
holes assuming that other quantized levels can be ignored. Remember 648
that 649
L
2
g
c
(ε)=
k
x
,k
y
,σ
ε<ε
k
<ε+dε
1.
(b) Determine N
c
(T )andP
v
(T ) for this two-dimensional system. Remem- 650
ber that 651
N
c
(T )=
∞
˜ε
c
dεg
c
(ε)e
−
ε−˜ε
c
Θ
.
(c) Determine n
c
(T )andp
v
(T )fortheintrinsiccase. 652
(d) Determine the value of the chemical potential for this case.
c
v
v
H
c
H
c
c
0
+
v
v
0
+
653
Uncorrected Proof
BookID 160928 ChapID 07 Proof# 1 - 29/07/09
212 7 Semiconductors
Summary 654
In this chapter, we studied the physics of semiconducting material and 655
artificial structures made of semiconductors. General properties of typical 656
semiconductors are reviewed and temperature dependence of carrier concen- 657
tration is considered for both intrinsic and doped cases. Then basic physics of 658
p-n junctions is covered in equilibrium and the current–voltage characteristic 659
of the junction is described. The characteristics of two-dimensional electrons 660
are discussed for the electrons in surface space charge layers formed in metal- 661
oxide-semiconductor structures, semiconductor superlattices, and quantum 662
wells. The fundamentals of the quantum Hall effects and the effects of disorders 663
and modulation doping are also discussed. 664
The densities of states in the conduction and valence bands are given by 665
g
c
(ε)=
√
2m
3/2
c
π
2
¯h
3
(ε − ε
c
)
1/2
; g
v
(ε)=
√
2m
3/2
v
π
2
¯h
3
(ε
v
− ε)
1/2
.
In the case of nondegenerate regime, we have ε
c
−ζ Θandζ −ε
v
Θ, 666
where Θ is k
B
T . Then the carrier concentrations become 667
n
c
(T )=N
c
(T )e
−
ε
c
−ζ
Θ
; p
v
(T )=P
v
(T )e
−
ζ−ε
v
Θ
,
where
668
N
c
(T )=
∞
ε
c
dεg
c
(ε)e
−
ε−ε
c
Θ
; P
v
(T )=
ε
v
∞
dεg
v
(ε).
The product n
c
(T )p
v
(T ) is independent of ζ such that 669
n
c
(T )p
v
(T )=N
c
(T )P
v
(T )e
−E
G
/Θ
.
In the absence of impurities, n
c
(T )=p
v
(T ) and we have 670
n
i
(T )=[N
c
(T )P
v
(T )]
1/2
e
−E
G
/2Θ
.
The chemical potential now becomes
671
ζ
i
= ε
c
−
1
2
E
G
+
3
4
Θln
m
v
m
c
; ζ
i
= ε
v
+
1
2
E
G
+
3
4
Θln
m
v
m
c
.
When donors are present, the chemical potential ζ will move from its
672
intrinsic value ζ
i
to a value near the conduction band edge. If the concentration 673
of donors is sufficiently small, the average occupancy of a single donor impurity 674
state is given by 675
n
d
=
1
1
2
e
β(ε
d
−ζ)
+1
.
The numerical factor of
1
2
in n
d
comes from the fact that either spin up or 676
spin down states can be occupied but not both. 677
Uncorrected Proof
BookID 160928 ChapID 07 Proof# 1 - 29/07/09
7.8 Amorphous Semiconductors 213
At a finite temperature, we have 678
n
c
(T )=N
c
(T )e
−β(ε
c
−ζ)
,p
v
(T )=P
v
(T )e
−β(ζ−ε
v
)
,
679
n
d
(T )=
N
d
1
2
e
β(ε
d
−ζ)
+1
,p
a
(T )=
N
a
1
2
e
β(ζ−ε
a
)
+1
.
In addition, we have charge neutrality condition given by
680
n
c
+ n
d
= N
d
− N
a
+ p
v
+ p
a
.
The set of these five equations should be solved numerically in order to have
681
self consistent result for five unknowns. 682
The region of the p–n junction is a high resistance region and the electrical 683
current density becomes 684
j = e (J
gen
h
+ J
gen
e
)
e
eV /Θ
− 1
,
where J
gen
h
and J
gen
e
are hole and electron generation current densities, 685
respectively. 686
Near the interface of metal–oxide–semiconductor structure under a strong 687
enough gate voltage, the motion of the electrons is characterized by 688
ε = ε
0
+
¯h
2
2m
∗
c
k
2
x
+ k
2
y
;Ψ
n,k
x
,k
y
=
1
L
e
i(k
x
x+k
y
y)
ξ
n
(z).
Here, ξ
n
(z)isthenth eigenfunction of a differential equation given by 689
1
2m
∗
c
−i¯h
∂
∂z
2
+ V
eff
(z) − ε
n
ξ
n
(z)=0.
If a quantum well is narrow, it leads to quantized motion and subbands:
690
ε
(c)
n
(k)=ε
(c)
n
+
¯h
2
2m
∗
c
k
2
x
+ k
2
y
.
In the presence of a dc magnetic field B applied normal to the plane of
691
the 2DEG, the Hamiltonian of a single electron is written by 692
H =
1
2m
p +
e
c
A
2
.
Here, p =(p
x
,p
y
)andA(r) is the vector potential whose curl gives B = 693
(0, 0,B). The electronic states are described by 694
E
nk
=¯hω
c
n +
1
2
, Ψ
nk
(x, y, z)=e
iky
u
n
x +
¯hk
mω
c
; n =0, 1, 2,....
The density of states (per unit length) is given by g(ε) ∝
n
δ
ε−¯hω
c
(n +
1
2
)
. 695
The total number of states per Landau level is equal to the magnetic flux 696
through the sample divided by the flux quantum
hc
e
: 697
N
L
=
BL
2
hc/e
.

Uncorrected Proof
BookID 160928 ChapID 08 Proof# 1 - 29/07/09
8 1
Dielectric Properties of Solids 2
8.1 Review of Some Ideas of Electricity and Magnetism 3
When an external electromagnetic disturbance is introduced into a solid, it will 4
produce induced charge density and induced current density. These induced 5
densities produce induced electric and magnetic fields. We begin with a brief 6
review of some elementary electricity and magnetism. In this chapter, we 7
will neglect the magnetization produced by induced current density and con- 8
centrate on the electric polarization field produced by the induced charge 9
density. 10
The potential φ(r) set up by a collection of charges q
i
at positions r
i
is 11
given by 12
φ(r)=
i
q
i
|r − r
i
|
. (8.1)
The electric field E(r)isgivenbyE(r)=−∇φ(r).
13
Now, consider a dipole at position r
(see Fig. 8.1). 14
φ(r)=
q
r − r
−
d
2
−
q
r − r
+
d
2
. (8.2)
By a dipole we mean p = qd is a constant, called the dipole moment, but
15
|d| = d itself is vanishingly small. If we expand for |r − r
||d |, we find 16
φ(r)=
qd · (r − r
)
(r − r
)
3
=
p · (r − r
)
|r − r
|
3
. (8.3)
The potential produced by a collection of dipoles p
i
located at r
i
is simply 17
φ(r)=
i
p
i
·(r − r
i
)
|r − r
i
|
3
. (8.4)
Again the electric field E(r)=−∇φ(r), so
18
E(r)=
i
3(r − r
i
)[p
i
· (r − r
i
)] − (r − r
i
)
2
p
i
|r − r
i
|
5
. (8.5)
Uncorrected Proof
BookID 160928 ChapID 08 Proof# 1 - 29/07/09
216 8 Dielectric Properties of Solids
r'
r
d
r'+d/2
r'-d/2
Fig. 8.1. Electric dipole of moment p = qd located at r
8.2 Dipole Moment Per Unit Volume 19
Let us introduce the electric polarization P(r), which is the dipole moments 20
per unit volume. Consider a volume V bounded by a surface S filled with a 21
polarization P(r
) that depends on the position r
.Then 22
φ(r)=
d
3
r
P(r
) · (r − r
)
|r − r
|
3
. (8.6)
If we look at the divergence of
P(r
)
|r−r
|
with respect to r
,wenotethat 23
∇
·
P(r
)
|r − r
|
=
1
|r − r
|
∇
·P(r
)+
P(r
) · (r − r
)
|r − r
|
3
. (8.7)
We can solve for
P(r
)·(r−r
)
|r−r
|
3
and substitute into our expression for φ(r). The 24
integral of the divergence term can be expressed as a surface integral using 25
divergence theorem. This gives 26
φ(r)=
3
S
dS
P(r
) ·
ˆ
n
|r − r
|
+
V
d
3
r
[−∇
·P (r
)]
|r − r
|
. (8.8)
27
The potential φ(r) can be associated with a potential produced by a volume 28
distribution of charge density 29
ρ
P
(r)=−∇·P(r) (8.9)
30
and the potential produced by a surface charge density 31
σ
P
(r)=P(r) ·
ˆ
n. (8.10)
32
Here, of course,
ˆ
n is a unit vector outward normal to the surface S bounding 33
the volume V . 34

Uncorrected Proof
BookID 160928 ChapID 08 Proof# 1 - 29/07/09
8.4 Local Field in a Solid 217
Poisson’s equation tells us that 35
∇·E =4π (ρ
0
+ ρ
P
) , (8.11)
where ρ
0
is some external charge density and ρ
P
is the polarization charge 36
density.Sinceρ
P
= −∇ ·P,wecanwrite 37
∇·E =4πρ
0
− 4π∇·P. (8.12)
If we define D = E +4πP,then
38
∇·D =4πρ
0
. (8.13)
Thus, D is the electric field that would be produced by the external charge
39
density ρ
0
if a polarizable material were absent. E is the true electric field 40
produced by all the charge densities including both ρ
0
and ρ
P
. 41
In general, P and E need not be in the same direction. However, for 42
sufficiently small value of E, the relationship between P and E is linear. We 43
can write 44
P
i
=
j
χ
ij
E
j
, (8.14)
where χ
is called the electrical susceptibility tensor.Wecanwrite 45
D = ε · E, (8.15)
where ε
=1+4πχ is the dielectric tensor. 46
8.3 Atomic Polarizability 47
An atom in its ground state has no dipole moment. However, in the presence 48
of an electric field E, an induced dipole moment results from the relative 49
displacements of the positive and negative charges within the atom. We can 50
write 51
p
ind
= αE, (8.16)
and α is called the atomic polarizability.
52
8.4 Local Field in a Solid 53
In a dilute gas of atoms the electric field E that produces the induced dipole 54
moment on an atom is simply the applied electric field. In a solid, however, 55
all of the dipole moments produced on other atoms in the solid make a contri- 56
bution to the field acting on a given atom. The value of this microscopic field 57
at the position of the atom is called the local field. The local field E
LF
(r)is 58
different from the applied electric field E
0
and from the macroscopic electric 59
Uncorrected Proof
BookID 160928 ChapID 08 Proof# 1 - 29/07/09
218 8 Dielectric Properties of Solids
p
p
p
p
p
p
p
p
Fig. 8.2. Induced dipoles of moment p located on neighboring atoms
field E (which is the average of the microscopic field E
LF
(r) over a region 60
that is large compared to a unit cell). Clearly, the contributions to the micro- 61
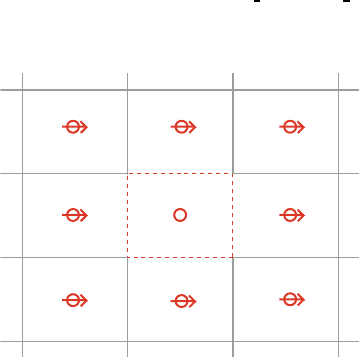
scopic field from the induced dipoles on neighboring atoms vary considerably 62
over the unit cell (see Fig. 8.2). The standard method of evaluating the local 63
field E
LF
(r) in terms of the macroscopic field E is to make use of the Lorentz 64
sphere. Before introducing the Lorentz field, let us review quickly the relation 65
between the external field E
0
and the macroscopic field E in the solid. 66
8.5 Macroscopic Field 67
Suppose the solid we are studying is shaped like an ellipsoid. It is a stan- 68
dard problem in electromagnetism to determine the electric field E inside the 69
ellipsoid in terms of the external electric field E
0
(see Fig. 8.3). 70
The applied field E
0
is the value of the electric field very far away from 71
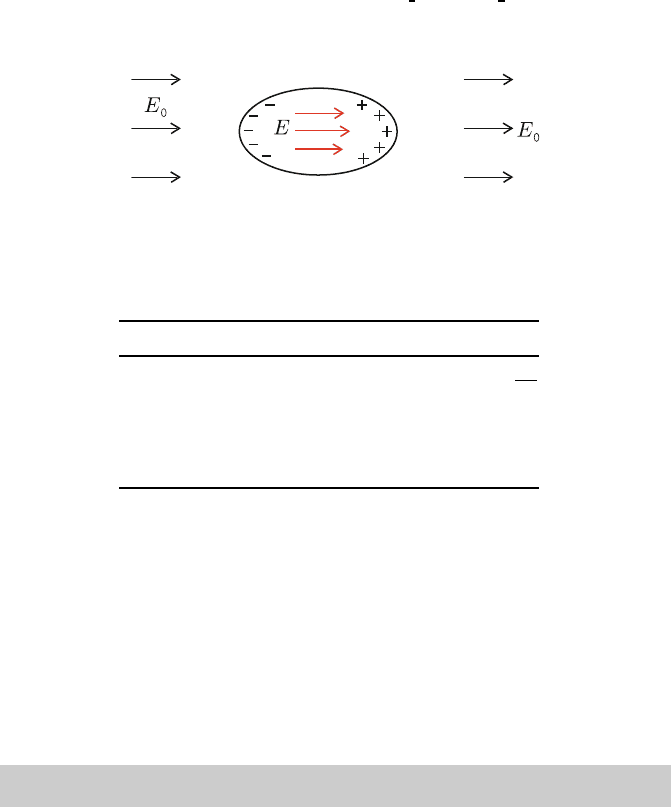
the sample. The macroscopic field inside the ellipsoid is given by 72
E = E
0
− λP = E
0
+ E
1
. (8.17)
The field E
1
= −λP is called the depolarization field, due to surface charge 73
density
ˆ
n · P on the outer surface of the specimen, and λ is called the 74
depolarization factor. 75
8.5.1 Depolarization Factor 76
The standard electromagnetic theory problem of determining λ involves 77
1. Solving Laplace’s equation ∇
2
φ(r) = 0 in cylindrical coordinates so that 78
φ(r)=
ar
l
+ br
−(l+1)
P
l
(cos θ)(C sin mφ + D cos mφ)
Uncorrected Proof
BookID 160928 ChapID 08 Proof# 1 - 29/07/09
8.6 Lorentz Field 219
Fig. 8.3. The macroscopic electric field E inside an ellipsoid located in an external
electric field E
0
is the sum of E
0
and polarization field E
1
= −λP due to the surface
charge density
ˆ
n · P
t1.1 Table 8.1. Depolarization factors λ of typical ellipsoids
t1.2 Type of ellipsoid Axis λ
t1.3 Sphere Any
4π
3
t1.4 Thin slap Normal 4π
t1.5 Thin slap Parallel 0
t1.6 Long cylinder Along axis 0
t1.7 Long circular cylinder normal to axis 2π
(a) Inside the sample (where r can approach 0) and 79
(b) Outside the sample (where r can approach ∞), 80
2. Imposing boundary conditions 81
(a) E well behaved as r → 0, 82
(b) E → E
0
as r →∞, 83
(c) D
normal
=(E +4πP)
normal
and E
trans
be continuous at the surface. 84
For an ellipsoid with the depolarization factors λ
1
, λ
2
,andλ
3
along the three 85
principal axes. 86
λ
1
+ λ
2
+ λ
3
=4π. (8.18)
87
Some examples are listed in Table 8.1. 88
8.6 Lorentz Field 89
Assume that we know E, the macroscopic field inside the solid. Now consider 90
an atom at position R. Draw a sphere of radius (named as Lorentz sphere) 91
about R where a, the interatomic spacing (see Fig. 8.4). The contribution 92
to the microscopic field at R from induced dipoles on other atoms can be 93
divided into two parts: 94
1. For atoms inside the sphere we will actually sum over the contribution 95
from their individual dipole moments p
i
. 96
2. For atoms outside the sphere we can treat the contribution macroscopically, 97
treating them as part of a continuum with polarization P. 98

Uncorrected Proof
BookID 160928 ChapID 08 Proof# 1 - 29/07/09
220 8 Dielectric Properties of Solids
0
r-R
r
R
r
r
P
Fig. 8.4. A Lorentz sphere of radius centered at R
The dipole moments outside the Lorentz sphere contribute a surface charge 99
density on the surface of the Lorentz sphere, and we can write 100
φ(R)=
Lorentzsphere
dS
P(r) ·
ˆ
n(r)
|R − r|
. (8.19)
The field E
2
caused by this surface charge on the spherical cavity (Lorentz 101
sphere) is called the Lorentz field : 102
E
2
(R)=−∇
R
φ(R)=
dS P(r) ·
ˆ
n(r)
(R − r)
|R − r|
3
. (8.20)
To evaluate this integral note, from Fig. 8.4, that
103
|r − R| = ,
P(r) ·
ˆ
n(r)=P cos θ,
dS =2π
2
sin θ dθ, and
R − r = (sin θ cos φ, sin θ sin φ, cos θ) .
Hence, we have
104
E
2
(R)=−
π
0
2π
2
d(cos θ)P cos θ
cos θ
3
.
Only the z-component of R − r survives the integration. We find that
105
E
2
(R)=2πP
1
−1
d(cos θ)cos
2
θ =
4π
3
P. (8.21)
106
E
2
=
4πP
3
is the Lorentz field. 107
The final contribution E
3
arises from the contribution of the dipoles within 108
the Lorentz sphere (L.S.). It is given by 109
E
3
=
i∈L.S.
3(p
i
· r
i
) r
i
− r
2
i
p
i
r
5
i
. (8.22)
