Quinn J.J., Yi K.-S. Solid State Physics: Principles and Modern Applications
Подождите немного. Документ загружается.

Uncorrected Proof
BookID 160928 ChapID 08 Proof# 1 - 29/07/09
8.12 Surface Waves 241
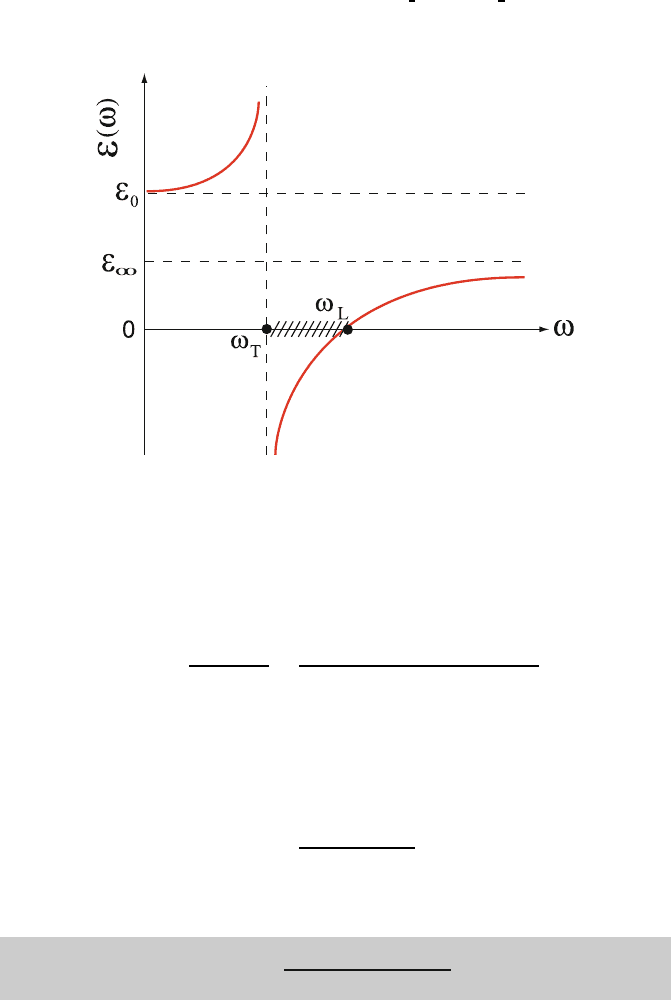
Fig. 8.21. Dielectric function ε(ω)ofapolarcrystal
Since ε
I
, α
I
,andα are all positive, this equation has a solution only in 436
the region where ε(ω) < 0. Recall that ε(ω)versusω looksasshownin437
Fig. 8.21. ε(ω) is negative if ω
T
<ω<ω
L
. The dispersion relation, (8.106), is 438
written by 439
c
2
q
2
y
=
ω
2
ε
I
ε(ω)
ε
I
+ ε(ω)
=
ε
I
ε
∞
ω
2
(ω
2
− ω
2
L
)
ε
I
(ω
2
− ω
2
T
)+ε
∞
(ω
2
− ω
2
L
)
. (8.107)
The denominator can be written as
440
D ≡ (ε
I
+ ε
∞
)ω
2
− (ε
I
ω
2
T
+ ε
∞
ω
2
L
)=(ε
I
+ ε
∞
)(ω
2
− ω
2
s
), (8.108)
where the surface phonon frequency ω
s
is given by 441
ω
2
s
=
ε
I
ω
2
T
+ ε
∞
ω
2
L
ε
I
+ ε
∞
. (8.109)
It is easy to show that ω
2
T
<ω
2
s
<ω
2
L
. The dispersion relation can be 442
written 443
c
2
q
2
y
=
ε
I
ε
∞
ω
2
(ω
2
− ω
2
L
)
(ε
I
+ ε
∞
)(ω
2
− ω
2
s
)
. (8.110)
444
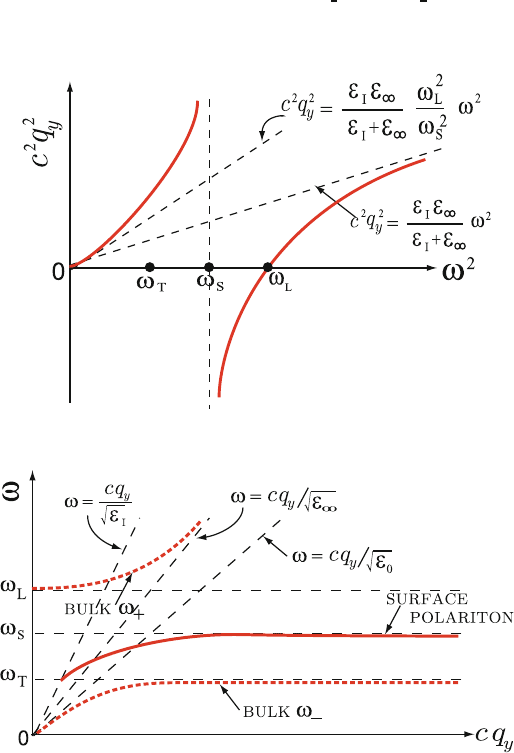
Figure 8.22 shows the right-hand side of (8.110) as a function of frequency. 445
Since surface modes can occur only where q
2
y
> 0andε(ω) < 0, we see that 446
the surface mode is restricted to the frequency region ω
T
<ω<ω
s
.Itis 447
not difficult to see that as cq
y
→∞, the surface polariton approaches the 448
frequency ω
s
. It is also apparent that at ω = ω
T
, c
2
q
2
y
= ε
I
ω
2
T
.Thisgivesthe 449
dispersion curve sketched in Fig. 8.23. 450
Uncorrected Proof
BookID 160928 ChapID 08 Proof# 1 - 29/07/09
242 8 Dielectric Properties of Solids
Fig. 8.22. Dispersion curves of phonon–polariton modes
Fig. 8.23. Dispersion relation of surface phonon–polariton modes. Also shown are
the bulk mode ω
+
and ω
−
which can occur outside the region ω
T
<ω<ω
L
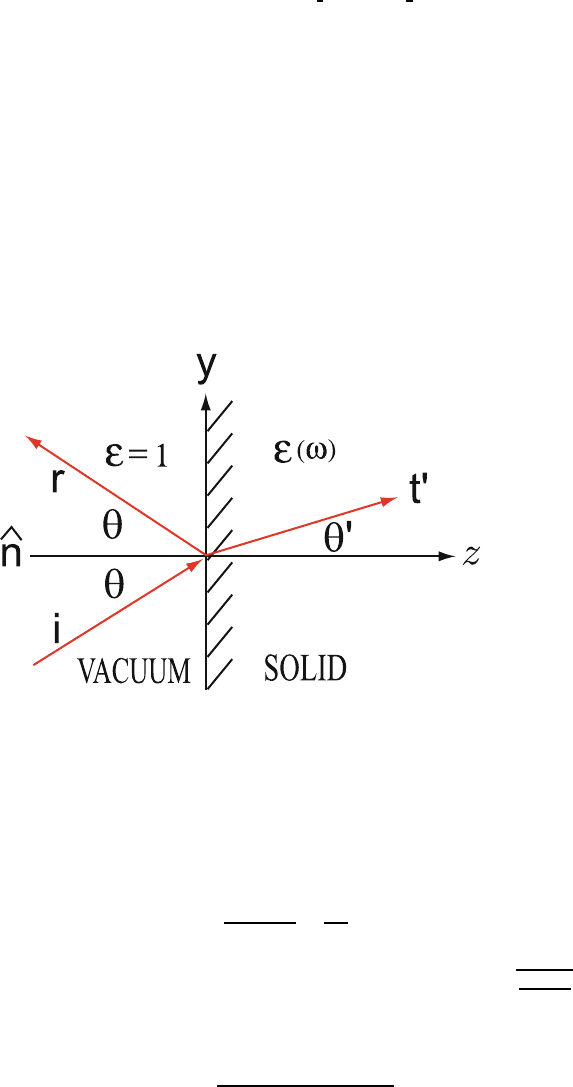
Uncorrected Proof
BookID 160928 ChapID 08 Proof# 1 - 29/07/09
8.12 Surface Waves 243
Problems 451
8.1. Use the eigenstates of the hydrogen atom to evaluate its atomic polariz- 452
ability α. 453
8.2. If atomic hydrogen formed a cubic lattice, what would its static dielectric 454
constant be? 455
8.3. Evaluate the reflectivity for an S-polarized and a P-polarized electro- 456
magnetic wave incident at an angle θ from vacuum on a material of dielectric 457
function ε(ω) as illustrated in the figure below. 458
One can take E =(E
x
, 0, 0)e
iωt−iq·r
and E =(0,E
y
,E
z
)e
iωt−iq·r
as the S- 459
and P-polarized electric fields, respectively. Remember that q · E =0and 460
q =(0,q
y
,q
z
). 461
8.4. A degenerate polar semiconductor contains n
0
free electrons per unit 462
volume in the conduction band. Its dielectric function ε(ω)isgivenby 463
ε(ω)=ε
∞
ω
2
− ω
2
L
ω
2
− ω
2
T
−
ω
2
p
ω
2
,
where ω
L
and ω
T
are the LO and TO phonon frequencies, and ω
p
=
2
4πn
0
e
2
m
. 464
1. Show that ε(ω) can be written as 465
ε(ω)=ε
∞
(ω
2
− ω
2
−
)(ω
2
− ω
2
+
)
ω
2
(ω
2
− ω
2
T
)
and determine ω
2
−
and ω
2
+
. 466

Uncorrected Proof
BookID 160928 ChapID 08 Proof# 1 - 29/07/09
244 8 Dielectric Properties of Solids
2. Make a sketch of ε(ω)versusω; be sure to indicate the locations of ω
T
, ω
L
, 467
ω
−
, ω
+
, ε
0
,andε
∞
. 468
3. Determine the dispersion relation of the longitudinal and transverse modes, 469
i.e. ω as a function of q. 470
4. In which regions of frequency are the transverse waves unable to propagate? 471
5. Consider a vacuum–degenerate polar semiconductor interface. Use the 472
results obtained in the text to determine the dispersion relations of the 473
surface modes. 474
6. Make a sketch of ω versus q
y
(q
y
is parallel to the interface) for these surface 475
modes and for the bulk modes which have q
z
=0. 476
Uncorrected Proof
BookID 160928 ChapID 08 Proof# 1 - 29/07/09
8.12 Surface Waves 245
Summary 477
In this chapter, we studied dielectric properties of solids in the presence of 478
an external electromagnetic disturbance. We first reviewed elementary elec- 479
tricity and magnetism, and introduced concept of local field inside a solid. 480
Then dispersion relations of self-sustaining collective modes and reflectivity 481
of a solid are studied for various situations. Finally, the collective modes local- 482
ized near the surface of a solid are also described and dispersion relations of 483
surface plasmon-polariton and surface phonon-polariton modes are discussed 484
explicitly. 485
When an external electromagnetic disturbance is introduced into a solid, 486
it will produce induced charge density and induced current density. These 487
induced densities produce induced electric and magnetic fields. The local field 488
E
LF
(r) at the position of an atom in a solid is given by 489
E
LF
= E
0
+ E
1
+ E
2
+ E
3
,
where E
0
, E
1
, E
2
, E
3
are, respectively, the external field, depolarization field 490
(= −λP), Lorentz field (=
4πP
3
), and the field due to the dipoles within the 491
Lorentz sphere
=
i∈L.S.
3(p
i
·r
i
)r
i
−r
2
i
p
i
r
5
i
. The local field at the center of a
492
sphere of cubic crystal is simply given by 493
E
sphere
LF
= E
0
−
4π
3
P +
4π
3
P +0=E
0
.
The induced dipole moment of an atom is given by p = αE
LF
.Thepolariza- 494
tion P is given, for a cubic crystal, by P =
Nα
1−
4πNα
3
E ≡ χE, where N is the 495
number of atoms per unit volume and χ is the electrical susceptibility. The 496
electrical susceptibility and the dielectric function (ε =1+4πχ) of the solid 497
are 498
χ =
Nα
1 −
4πNα
3
; ε =1+
4πNα
1 −
4πNα
3
.
The relation between the macroscopic dielectric function ε and the atomic
499
polarizability α is called the Clausius–Mossotti relation: 500
ε − 1
ε +2
=
4πNα
3
The total polarizability of the atoms or ions within a unit cell can usually
501
be separated into three parts: 502
1. Electronic polarizability α
e
: The displacement of the electrons relative to 503
the nucleus 504
2. Ionic polarizability α
i
: The displacement of an ion itself with respect to its 505
equilibrium position 506
3. Dipolar polarizability α
dipole
: The orientation of any permanent dipoles by 507
the electric field in the presence of thermal disorder. 508

Uncorrected Proof
BookID 160928 ChapID 08 Proof# 1 - 29/07/09
246 8 Dielectric Properties of Solids
In the presence of a field E, the average dipole moment per unit volume is 509
given by ¯p
z
= p L
pE
k
B
T
, where L(ξ) is the Langevin function. The dipolar
510
polarizability α
dipole
shows strong temperature dependence. The electronic 511
polarizability α
e
and the ionic polarizability α
ion
are almost independent of 512
temperature. 513
In a metal, the conduction electrons are free and the dielectric function 514
becomes 515
ε(ω)=1−
4πNe
2
/m
ω
2
− iω/τ
=1−
ω
2
p
ω
2
− iω/τ
.
In an ionic crystal, we have
516
ε(ω)=ε
∞
ω
2
− ω
2
L
ω
2
− ω
2
T
.
Here, ω
L
and ω
T
are the TO and LO phonon frequencies, respectively. We 517
note that ω
L
>ω
T
since ε
0
>ε
∞
in general. 518
For the propagation of light in a material characterized by ε(ω), the 519
external sources j
0
and ρ
0
vanishes. Therefore, we have 520
∇×E = −
iω
c
B; ∇×B =
iωε(ω)
c
E.
The two Maxwell equations for ∇×E and ∇×B can be combined to give a
521
wave equation: 522
ω
2
c
2
ε(ω) − q
2
E + q (q ·E)=0.
For an infinite homogeneous medium of dielectric function ε(ω), a general
523
dispersion relation of the self-sustaining waves is written as 524
ε(ω)
ω
2
c
2
ε(ω) − q
2
2
=0.
The two transverse modes and one longitudinal mode are characterized,
525
respectively, by 526
ω
2
=
c
2
q
2
ε(ω)
; ε(ω)=0.
For the interface (z = 0) of two different media of dielectric functions ε
I
527
and ε
II
, the boundary conditions give us the general dispersion of the surface 528
wave: 529
ε
I
α
I
+
ε
II
α
II
=0 or
ε
o
α
o
+
ε(ω)
α
=0.
where
530
α
o
=
8
q
2
y
−
ω
2
c
2
ε
o
and α =
8
q
2
y
−
ω
2
c
2
ε(ω).

Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
9 1
Magnetism in Solids 2
9.1 Review of Some Electromagnetism 3
9.1.1 Magnetic Moment and Torque 4
We begin with a brief review of some elementary electromagnetism. A current 5
distribution j(r) produces a magnetic dipole moment at the origin that is 6
given by 7
m =
1
2c
r × j(r)d
3
r. (9.1)
If j(r) is composed from particles of charge q
i
at positions r
i
moving with 8
velocity v
i
, j(r)=
i
q
i
v
i
δ(r − r
i
), and the magnetic moment m is 9
m =
1
2c
i
q
i
r
i
× v
i
. (9.2)
10
For a single particle of charge q moving in a circle of radius r
0
at velocity v
0
, 11
we have 12
m =
1
2c
qr
0
v
0
(9.3)
and m is perpendicular to the plane of the circle. The current i in the loop is
13
given by q divided by t =
2πr
v
0
, the time to complete one circuit. Thus 14
i =
qv
0
2πr
0
. (9.4)
We can use this in our expression for m to get
15
m =
q
2c
r
0
v
0
=
ia
c
. (9.5)
Here, a = πr
2
0
is the area of the loop. We can write m =
ia
c
if we associate 16
vector character with the a of the loop. 17
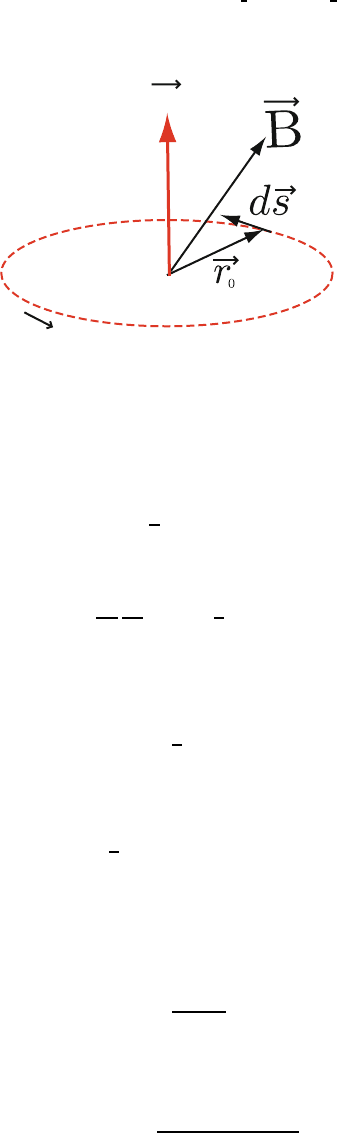
Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
248 9 Magnetism in Solids
i
m
Fig. 9.1. A magnetic moment m due to a current loop of radius r
0
located in a
magnetic field B
If a magnetic field were imposed on a magnetic moment, the magnetic 18
moment would experience a torque. To show this we begin with the Lorentz 19
force 20
F =
q
c
v
0
× B. (9.6)
For a charge dq the force dF is given by
21
dF =
dq
c
ds
dt
× B =
i
c
ds × B. (9.7)
Here, ds is an infinitesimal element of path length (see, for example, Fig. 9.1).
22
The torque τ is given by
"
r × dF. 23
τ =
r × dF =
i
c
r × ds × B. (9.8)
But
"
r × ds = a, and hence we have
24
τ =
i
c
a × B = m × B. (9.9)
9.1.2 Vector Potential of a Magnetic Dipole 25
If a magnetic dipole m is located at the origin, it produces a vector potential 26
at position r given by 27
A(r)=
m × r
r
3
. (9.10)
Of course the magnetic field B(r)=∇×A(r). If we have a magnetization
28
(magnetic dipole moment per unit volume), then A(r)isgivenby 29
A(r)=
d
3
r
M(r
) × (r − r
)
|r − r
|
3
. (9.11)

Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
9.1 Review of Some Electromagnetism 249
As we did with the electric polarization P(r), we can transform this equation 30
into two parts. 31
A(r)=
V
d
3
r
∇
r
× M(r
)
|r − r
|
+
3
S
dS
M(r
) ×
ˆ
n
|r − r
|
, (9.12)
32
where
ˆ
n is a unit vector outward normal to the surface S. The volume integra- 33
tion is carried out over the volume V of the magnetized material. The surface 34
integral is carried out over the surface bounding the magnetized object. Since 35
A(r) is related to a current density by 36
A(r)=
1
c
d
3
r
j(r
)
|r − r
|
, (9.13)
the vector potential produced by a magnetization is equivalent to volume
37
distribution of current 38
j
M
(r)=c∇×M(r) (9.14)
and a surface distribution of current
39
j
S
(r)=cM(r) × ˆn. (9.15)
Recall that if E = 0, Maxwell’s equation for ∇×B is
40
∇×B =
4π
c
(j
0
+ j
M
)=
4π
c
j
0
+4π∇×M. (9.16)
Defining H = B − 4πM gives
41
∇×H =
4π
c
j
0
(9.17)
42
which shows that H is that part of the field arising from the free current 43
density j
0
. As we stated before the two Maxwell equations 44
∇·E =4π (ρ
0
+ ρ
ind
) , (9.18)
and
45
∇×B =
1
c
˙
E +
4π
c
(j
0
+ j
ind
)+4π∇×M (9.19)
are sometimes written in terms of D and H.
46
∇·D =4πρ
0
,
(where D = E +4πP and ∇·P = −ρ
pol
with bound charge density ρ
pol
)and 47
∇×H =
1
c
˙
D +
4π
c
j
0
.
Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
250 9 Magnetism in Solids
9.2 Magnetic Moment of an Atom 48
9.2.1 Orbital Magnetic Moment 49
Let us consider the nucleus to be fixed and evaluate the orbital contribution 50
of the electron currents to the magnetic moment of an atom. 51
m =
1
2c
i
q
i
r
i
× v
i
. (9.20)
Since q
i
= − e for every electron, and every electron has mass m
e
,wecan 52
write 53
m = −
e
2m
e
c
i
r
i
×m
e
v
i
= −
e
2m
e
c
×
total angular momentum
of the electrons
. (9.21)
We know
i
r
i
× m
e
v
i
is quantized and equal to ¯hL,where|L| =0, 1, 2,... 54
and L
z
=0, ±1, ± 2,...,±L.Thus,wehave 55
m = −
e¯h
2m
e
c
L = −μ
B
L. (9.22)
Here, μ
B
=
e¯h
2m
e
c
=0.927 ×10
−20
(ergs/gauss) or 5.8 ×10
−2
(meV/T) is called 56
the Bohr magneton. The Bohr magneton corresponds to the magnetic 57
moment of a 1s electron in H. 58
9.2.2 Spin Magnetic Moment 59
In addition to orbital angular momentum ¯hL,eachelectroninanatomhasan 60
intrinsic spin angular momentum ¯hs, giving a total spin angular momentum 61
¯hS where 62
S =
i
s
i
. (9.23)
The z-component of spin is s
z
= ±
1
2
, and the spin contribution to the magnetic 63
moment is ∓μ
B
. Thus, for each electron, there is a contribution −2μ
B
s to 64
the magnetic moment of an atom. If we sum over all spins, the total spin 65
contribution to the magnetic moment is 66
m
s
= −2μ
B
i
s
i
= −2μ
B
S. (9.24)
Note that the factor of 2 appearing in this expression is not exact. It is actually
67
given by g =2(1+
α
2π
− 2.973
α
2
π
2
+ ···) 2 × 1.0011454. However, in our 68
discussion here we will take the g-factor as 2. 69
