Quinn J.J., Yi K.-S. Solid State Physics: Principles and Modern Applications
Подождите немного. Документ загружается.


Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
9.2 Magnetic Moment of an Atom 251
9.2.3 Total Angular Momentum and Total Magnetic Moment 70
The total angular momentum of an atom is given by 71
J = L + S. (9.25)
The total magnetic moment is given by
72
m = −μ
B
(L +2S) . (9.26)
In quantum mechanics the components of J, L,andS are operators that
73
satisfy commutation relations. As we learned in quantum mechanics, it is 74
possible to diagonalize J
2
and J
z
simultaneously. 75
J
2
|j, j
z
= j(j +1)|j, j
z
; j =0,
1
2
,
3
2
,... (9.27)
J
z
|j, j
z
= j
z
|j, j
z
; − j ≤ j
z
≤ j (9.28)
Note that j
z
=0, ±1,...,±j or j
z
= ±
1
2
, ±
3
2
,...,±j.Wecanwritethat 76
m = −ˆgμ
B
J. (9.29)
This defines the operator ˆg because we have J = L + S and
77
ˆgJ = L +2S. (9.30)
We can use these definitions to show that
78
J · J =(L + S) · (L + S)=L
2
+ S
2
+2L · S (9.31)
and
79
ˆgJ · J =(L + S) · (L +2S)=L
2
+2S
2
+3L · S. (9.32)
We can eliminate L · S and obtain
80
g
L
=
3
2
+
1
2
s(s +1)−l(l +1)
j(j +1)
(9.33)
81
This eigenvalue of ˆg is called the Land´eg-factor. 82
9.2.4 Hund’s Rules 83
The ground state of an atom or ion with an incomplete shell is determined by 84
Hund’s rules: 85
1. ThegroundstatehasthemaximumS consistent with the Pauli exclusion 86
principle. 87
2. It has the maximum L consistent with the maximum spin multiplicity 88
2S + 1 of Rule (1). 89
3. The J-value is given by L − S when a shell is less than half filled and by 90
L + S when more than half filled. 91

Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
252 9 Magnetism in Solids
t1.1 Tabl e 9. 1. Ground state electron configurations angular momentum quantum
numbers for the elements of atomic numbers 20 ≤ Z ≤ 29
t1.2 Z Element Configuration Spectroscopic notation SLJg
t1.3 20 Ca (3p)
6
(4s)
21
S
0
000–
t1.4 21 Sc (3d)
1
(4s)
22
D
3
2
1
2
2
3
2
4
5
t1.5 22 Ti (3d)
2
(4s)
23
F
2
132
2
3
t1.6 23 V (3d)
3
(4s)
24
F
3
2
3
2
3
3
2
2
5
t1.7 24 Cr (3d)
5
(4s)
17
S
3
3032
t1.8 25 Mn (3d)
5
(4s)
26
S
5
2
5
2
0
5
2
2
t1.9 26 Fe (3d)
6
(4s)
25
D
4
224
3
2
t1.10 27 Co (3d)
7
(4s)
24
F
9
2
3
2
3
9
2
4
3
t1.11 28 Ni (3d)
8
(4s)
23
F
4
134
5
4
t1.12 29 Cu (3d)
10
(4s)
12
S
1
2
1
2
0
1
2
2
Example 92
Consider an ion of Fe
2+
; it has six electrons in the 3d level. We can put five 93
of them in spin up states (since d means l =2andm
l
can be −2, −1, 0, 1, 2) 94
and to maximize S, hence, 95
↑↑↑↑↑↓ gives S =2.
The maximum value of L-value is given by
96
L = −2 − 1+0+1+2+2=2.
The J-value (since it is over half-filled) is
97
J = L + S =4.
Therefore, we have
98
g =
3
2
+
1
2
2(3) − 2(3)
4(5)
=
3
2
.
One can work out some examples listed in Table 9.1. The ground state
99
notation is
2S+1
L
J
,whereL =0, 1, 2, 3, 4,... are denoted by the letters 100
S,P,D,F,G,..., respectively. 101
9.3 Paramagnetism and Diamagnetism of an Atom 102
In the presence of a magnetic field B the Hamiltonian describing the electrons 103
in an atom can be written as 104
H = H
0
+
i
1
2m
p
i
+
e
c
A(r
i
)
2
+2μ
B
B ·
i
s
i
, (9.34)
Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
9.3 Paramagnetism and Diamagnetism of an Atom 253
where H
0
is the nonkinetic part of the atomic Hamiltonian, p
i
= −i¯h∇
i
,and 105
the sum is over all electrons in an atom. For a homogeneous magnetic field 106
B, one can choose a vector potential of 107
A = −
1
2
r × B. (9.35)
Here, we take the magnetic field B in the z-direction.
108
B =(0, 0,B
0
). (9.36)
Then, the vector potential is given by
109
A = −
1
2
B
0
y
ˆ
i − x
ˆ
j
. (9.37)
Substituting the vector potential into (9.34), we have
110
H = H
0
+
i
p
2
i
2m
e
+
eB
0
2m
e
c
i
(x
i
p
iy
− y
i
p
ix
)+
e
2
B
2
0
8m
e
c
2
i
x
2
i
+ y
2
i
+2μ
B
B
0
S
z
.
(9.38)
Here, we note that
111
x
i
p
iy
− y
i
p
ix
=(r
i
× p
i
)
iz
=¯hl
iz
.
Now, we can write the Hamiltonian as
112
H = H
0
+
i
p
2
i
2m
e
+ μ
B
(L
z
+2S
z
)B
0
+
e
2
B
2
0
8m
e
c
2
i
x
2
i
+ y
2
i
. (9.39)
But −μ
B
(L
z
+2S
z
)issimplym
z
,thez-component of the magnetic moment 113
of the atom in the absence of the applied magnetic field B. Therefore, 114
we have 115
H = H−m
z
B
0
+
e
2
B
2
0
8m
e
c
2
i
x
2
i
+ y
2
i
, (9.40)
where H = H
0
+
i
p
2
i
2m
e
. In the presence of the magnetic field B
0
, 116
v
ix
=
∂H
∂p
ix
=
1
m
p
ix
−
eB
0
2c
y
i
,
v
iy
=
∂H
∂p
iy
=
1
m
p
iy
+
eB
0
2c
x
i
, (9.41)
and the magnetic moment in the presence of B
0
is (see (9.20)) 117
μ =
i
−
e
2c
r
i
× v
i
−
e
2m
e
c
2¯hS
i
. (9.42)

Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
254 9 Magnetism in Solids
Using (9.41), the expression for v
ix
and v
iy
,oneobtains 118
μ
z
= −μ
B
L
z
− 2μ
B
S
z
−
e
2
B
0
4m
e
c
2
i
x
2
i
+ y
2
i
. (9.43)
119
Note that one can also obtain this result from (9.40) using the relation 120
μ
z
= −
∂H
∂B
0
. (9.44)
Thus, we have μ
z
, the z-component of magnetic moment of the atom in the 121
magnetic field B
0
is given by 122
μ
z
= m
z
−
e
2
B
0
4m
e
c
2
i
x
2
i
+ y
2
i
. (9.45)
It differs from m
z
,itsvaluewhenB
0
= 0 by a term that is negative and 123
proportional to B
0
. 124
If the atom is in its ground state, m
z
, the average value of m
z
is m
z
= 125
−μ
B
(L
z
+2S
z
)=−gμ
B
J
z
= −gμ
B
j
z
,wherej
z
= −J, −J +1,...,J.Fora 126
spherically symmetric atom, x
2
i
= y
2
i
= z
2
i
=
1
3
r
2
i
. Therefore, we obtain 127
μ
z
= −gμ
B
J
z
−
e
2
B
0
6m
e
c
2
i
r
2
i
. (9.46)
The second term on the right-hand side is the origin of diamagnetism.If
128
J =0(sothatJ
z
= 0), then a system containing N atoms per unit volume 129
would produce a magnetization 130
M = −N
e
2
B
0
6m
e
c
2
i
r
2
i
, (9.47)
and the diamagnetic susceptibility
131
χ
DIA
=
M
B
0
= −N
e
2
6m
e
c
2
i
r
2
i
. (9.48)
132
Here,wehaveassumedχ
Dia
1andsetχ =
M
B
instead of
M
H
. This result 133
was first derived by Langevin. 134
All substances exhibit diamagnetism. Paramagnetism occurs only in sam- 135
ples whose atoms possess permanent magnetic moments (i.e. m =0when 136
B
0
= 0). All free atoms except those having complete electronic shells are 137
paramagnetic. In solids, however, fewer substances exhibit paramagnetism 138
because the electrons form energy bands and filled bands do not contribute 139
to paramagnetism. 140

Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
9.4 Paramagnetism of Atoms 255
Examples of paramagnetism in solids are 141
1. Pauli spin paramagnetism of metals. 142
2. Paramagnetism due to incomplete shells. 143
(a) Transition elements: 144
Iron group elements with incomplete 3d shell, for example,
Ti
3+
(3d
1
) ∼ Cu
2+
(3d
9
)
.
145
Palladium group elements with incomplete 4d shell, for example,
Zr
3+
(4d
1
) ∼ Ag
2+
(4d
9
)
.
146
Platinum group elements with incomplete 5d shell, for example,
Hf
3+
(5d
1
) ∼ Au
2+
(5d
9
)
.
(b) Rare earth elements:
147
Rare earth group elements (or Lanthanides) with incomplete 4f
shell, for example,
Ce
3+
(4f
1
) ∼ Yb
3+
(4f
13
)
.
148
Transuranic group elements (or Actinides) with incomplete 5f or
6d shells, for example, elements beyond Th.
9.4 Paramagnetism of Atoms 149
We have seen that the permanent magnetic dipole moment of an atom is 150
given by 151
m = −g
L
μ
B
J. (9.49)
We will assume that the separations between atoms in the systems of interest
152
are sufficiently large that the interactions between the atoms can be neglected. 153
The energy of an atom in a magnetic field B is 154
E = −m · B = g
L
μ
B
Bm
J
, (9.50)
where m
J
= −J, −J +1,...,J − 1,J. The probability of finding an atom in 155
state |J, m
J
> at a temperature T is 156
p(m
J
)=
1
Z
e
−βE(m
J
)
, (9.51)
where β =(k
B
T )
−1
and the normalization constant Z is chosen so that 157
m
J
p(m
J
) = 1. This gives 158
Z =
J
m
j
=−J
e
−βg
L
μ
B
Bm
J
. (9.52)
Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
256 9 Magnetism in Solids
Let βg
L
μ
B
B = y. Then, Z =
J
m=−J
e
−ym
. This can be rewritten 159
Z =e
−yJ
1+e
y
+e
2y
+ ···+e
2Jy
=e
−yJ
(e
y
)
2J+1
− 1
e
y
− 1
. (9.53)
The result for Z can be rewritten
160
Z(x)=
sinh
2J+1
2J
x
sinh
x
2J
, (9.54)
where x = yJ = βg
L
μ
B
BJ. The magnetization of a system containing N 161
atoms per unit volume will be 162
M = −Ng
L
μ
B
m
J
m
J
p(m
J
)
m
J
p(m
J
)
= Ng
L
μ
B
J
∂
∂x
ln Z. (9.55)
This is usually written as
163
M = Ng
L
μ
B
JB
J
(βg
L
μ
B
BJ), (9.56)
164
where the function B
J
(x) is called the Brillouin function. It is not difficult 165
to see that 166
B
J
(x)=
2J +1
2J
coth
2J +1
2J
x −
1
2J
coth
x
2J
. (9.57)
The argument of the Brillouin function
2J+1
2J
βg
L
μ
B
BJ is small compared 167
tounity if the magnetic field B is small compared to 500 T at room tem- 168
perature. Under these conditions (use coth z
1
z
+
z
3
for z 1) one can 169
write 170
B
J
(x)
x
3
J +1
J
, (9.58)
and 171
M
Ng
2
L
μ
2
B
J(J +1)
3k
B
T
B. (9.59)
Since m · m = g
2
L
μ
2
B
J · J = g
2
L
μ
2
B
J(J +1)wecanwrite 172
χ
Para
=
M
B
=
N
m
2
3k
B
T
(9.60)
173
for the paramagnetic susceptibility of a system of atoms of magnetic moment 174
m at high temperature (g
L
μ
B
BJ k
B
T ). This is commonly known as 175
Curie’s law. Notice that when J becomes very large 176
lim
J→∞
B
J
(x) ⇒ coth x −
1
x
= L(x), (9.61)
where L is the Langevin function that we encountered in studying elec-
177
tric dipole moments. Thus, the quantum mechanical result goes over to the 178
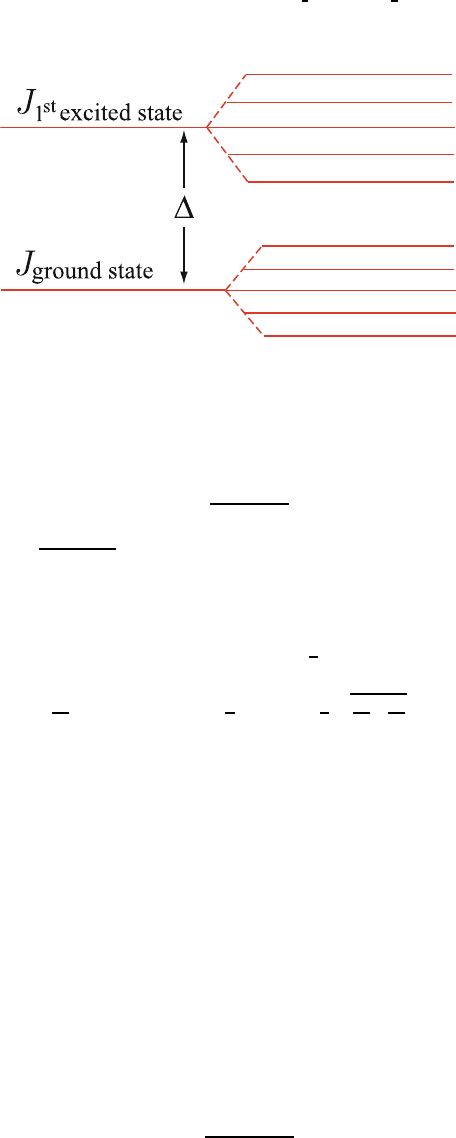
Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
9.5 Pauli Spin Paramagnetism of Metals 257
Fig. 9.2. Energy level splitting of the ground state and first excited multiplets for
an atom of the total angular momentum quantum number J
classical result as J →∞, as expected. Curie’s law is often written 179
M
Nμ
2
B
p
2
B
3k
B
T
, (9.62)
where p = g
L
J(J + 1) is called the effective number of Bohr mag- 180
netons. Knowing S, L, J and g
L
from the application of Hund’s rules 181
immediately gives us p. For example, for a Dy
3+
ion the atomic configuration is 182
(4f)
9
(5s)
2
(5p)
6
. This results from removing two 6s electrons and one 4f elec- 183
tron from the neutral atom. The S-value will be
5
2
(seven 4f-electrons in ↑ and 184
two in ↓ states), L = 5 (the two ↓ electrons have m
z
= 3 and 2 to maximize L), 185
and J = L + S =
15
2
, and hence g
L
=
4
3
and p =
4
3
2
15
2
·
17
2
10.63. 186
Observed and calculated p-values agree fairly well. There are exceptions 187
when excited state multiplets are not sufficiently high in energy (see, for 188
example, Fig. 9.2). 189
Until now we have assumed Δ k
B
T and Δ g
L
μ
B
JB.Ifthisisnot 190
true, higher multiplets can be important in evaluation of χ or p. Typically, for 191
an ion with partially filled shell with nonzero value of J, χ
Para
∼ 10
−2
−10
−3
192
at room temperature and χ
Dia
∼ 10
−5
, which is independent of temperature. 193
Therefore, we have χ
Para
∼ 500χ
DIA
at room temperature. 194
9.5 Pauli Spin Paramagnetism of Metals 195
If we used the classical theory of paramagnetism for a particle with magnetic 196
moment m, the magnetization at a temperature T (with k
B
T |m · B|) 197
would be given by Curie’s law 198
M =
N
m
2
B
3k
B
T
. (9.63)
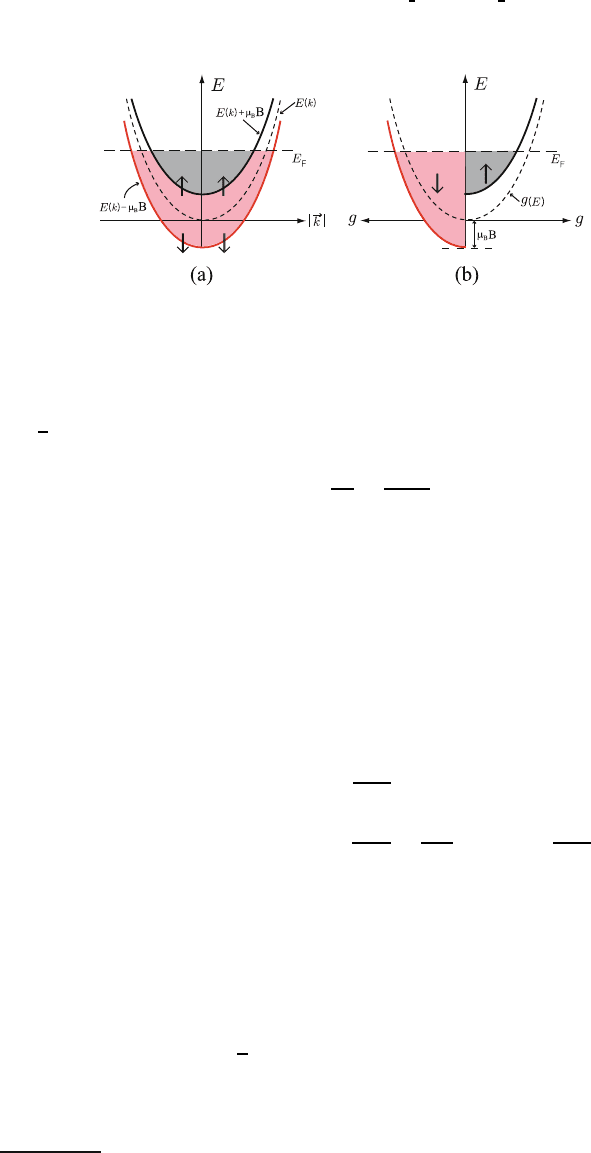
Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
258 9 Magnetism in Solids
Fig. 9.3. Energy level splitting of the electron gas in the presence of the magnetic
field B. Energy parabolas E(k)(a) and density of states g(E)(b) of electrons in
two different spin states in the presence of Zeeman splitting
For free electrons m = −2μ
B
s and
m
2
=4μ
2
s · s =4μ
2
s(s + 1). Since 199
s =
1
2
this gives
m
2
=3μ
2
and
1
200
χ
classical
=
M
B
=
n
0
μ
2
B
k
B
T
. (9.64)
As we discussed earlier, this is not what is observed experimentally. In metals
201
the observed susceptibility is approximately independent of temperature and 202
two orders of magnitude smaller than the value of χ
classical
evaluated at room 203
temperature. 204
The qualitative explanation is exactly the same as that by which the Som- 205
merfeld model explained the electronic contribution to the specific heat. At a 206
temperature T only electrons whose energy lies within a shell of width k
B
T 207
about the Fermi energy are effectively free. Other electrons are inefficient 208
because of the Pauli exclusion principle. If we replace n
0
by n
eff
,where 209
n
eff
n
0
k
B
T
ζ
. (9.65)
The spin susceptibility becomes χ
QM
n
0
μ
2
B
ζ
k
B
T
ζ
χ
classical
=
n
0
μ
2
B
ζ
. 210
To obtain χ
QM
more rigorously, we simply assume that in the presence of 211
the magnetic field B the energy of an electron is changed by an amount 212
δE = ±μ
B
B = −m · B; m = −g
L
μ
B
S
depending on whether its spin is up or down relative to the direction of B.
213
The number of particles of spin up (or down) per unit volume is 214
n
±
=
1
2
∞
0
dEf
0
(E)g (E ∓ μ
B
B) , (9.66)
where + and − in the subscript of n
±
correspond to the cases of spin up 215
(+) and spin down (−) states, respectively. (See Fig. 9.3.) We evaluated many 216
1
Here, n
0
is the number of free electrons per unit volume in a metal.

Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
9.6 Diamagnetism of Metals 259
integrals over Fermi functions in Chap. 3. Remember that the total number 217
of states per unit volume with energy less than ε is given by 218
G(ε)=
ε
0
g(ε)dε = n
0
k
k
F
3
= n
0
ε
ε
F
3/2
.
Using these results we can obtain
219
n
±
=
1
2
G(ζ ∓ μ
B
B)+
π
2
6
(k
B
T )
2
g
(ζ ∓ μ
B
B)
. (9.67)
The magnetization M is equal to μ
B
(n
−
−n
+
). Expanding for ζ μ
B
B and 220
k
B
T ζ leads to 221
M μ
2
B
B
g(ζ)+
π
2
6
(k
B
T )
2
g
(ζ)
. (9.68)
The chemical potential is determined by requiring the number of particles to
222
be n
0
= n
−
+ n
+
.Thisgives 223
n
0
= G(ζ)+
π
2
6
(k
B
T )
2
g
(ζ)+O(μ
2
B
B
2
). (9.69)
To order μ
2
B
B
2
,wenotethat 224
ζ = ζ
0
−
π
2
6
(k
B
T )
2
g
(ζ
0
)
g(ζ
0
)
. (9.70)
Using g(ζ)=
3
2
n
0
ζ
0
ε
ζ
0
1/2
gives 225
χ
QM
=
3n
0
μ
2
B
2ζ
0
1 −
π
2
12
k
B
T
ζ
0
2
+ ···
(9.71)
226
for the Pauli spin (paramagnetic) susceptibility of a metal. 227
9.6 Diamagnetism of Metals 228
According to classical mechanics there should be no diamagnetism of a free 229
electron gas. Consider the effect of a magnetic field B on the motion of an 230
electron. The force acting on the electron is 231
F = −
e
c
v × B. (9.72)
This force is always perpendicular to v, therefore F · dl = F · vdt =0.Thus,
232
no work is done on the electrons by the field B and their energy is unchanged. 233
Uncorrected Proof
BookID 160928 ChapID 09 Proof# 1 - 29/07/09
260 9 Magnetism in Solids
Further the distribution function depends only on E, T , N and will also be 234
unchanged. Thus, there can be no induced currents and no diamagnetism. 235
Quantum mechanics gives a different answer. Landau was the first to derive 236
the diamagnetic susceptibility of metals. We will not rederive his result in 237
full, but simply show how the result comes about in a quantum mechanical 238
calculation. 239
Let A =(0,xB,0) be the vector potential of a dc magnetic field B. 240
The Hamiltonian for a single electron is (here we shall neglect the intrinsic 241
magnetic moment of the electron) 242
H =
1
2m
p
2
x
+
p
y
+
e
c
Bx
2
+ p
2
z
. (9.73)
Recall that p = −i¯h∇. The Schr¨odinger equation is
243
−
¯h
2
2m
∂
2
∂x
2
+
∂
∂y
+ i
eB
¯hc
x
2
+
∂
2
∂z
2
Ψ=EΨ. (9.74)
Since the Hamiltonian is independent of y and z, let us assume a solution of
244
the form 245
Ψ(x, y, z)=e
ik
y
y+ik
z
z
φ(x). (9.75)
The equation which φ(x) must satisfy is
246
∂
2
∂x
2
−
k
y
+
eB
¯hc
x
2
− k
2
z
+
2mE
¯h
2
φ(x) = 0 (9.76)
If we let x
= x +
¯hk
y
mω
c
this equation becomes 247
−
¯h
2
2m
∂
2
∂x
2
+
1
2
mω
2
c
x
2
φ(x
)=
E −
¯h
2
k
2
z
2m
φ(x
). (9.77)
This is just the equation for a simple harmonic oscillator of mass m and
248
characteristic frequency ω
c
. The energy levels are 249
E −
¯h
2
k
2
z
2m
=¯hω
c
n +
1
2
; n =0, 1, 2,... (9.78)
Thus, the eigenfunctions and eigenvalues for an electron in the presence of a
250
magnetic field B are 251
|nk
y
k
z
= L
−1
e
ik
y
y+ik
z
z
φ
n
x +
¯hk
y
mω
c
. (9.79)
252
E
n
(k
y
,k
z
)=
¯h
2
k
2
z
2m
+¯hω
c
n +
1
2
. (9.80)
