Quinn J.J., Yi K.-S. Solid State Physics: Principles and Modern Applications
Подождите немного. Документ загружается.

Uncorrected Proof
BookID 160928 ChapID 10 Proof# 1 - 29/07/09
304 10 Magnetic Ordering and Spin Waves
spin
s
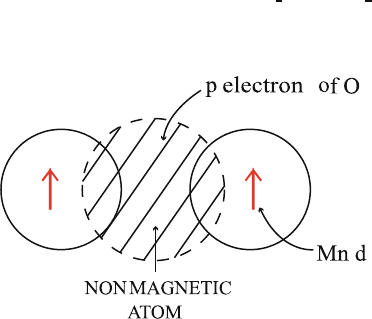
Fig. 10.15. Schematic illustration of superexchange coupling in a magnetic oxide.
Two Mn ions (each having unpaired electron in a d orbital) are separated by an
oxygen ion having two p electrons
superexchange in ionic insulators and is also called as the Ruderman- 524
Kittel-Kasuya-Yosida (RKKY) interaction. For example, the unpaired f 525
electrons in the rare earths are magnetic and they can be coupled to f 526
electrons in a neighboring rare earth ion through the exchange interaction 527
via nonmagnetic conduction electrons. 528
4. Double exchange coupling is the ferromagnetic superexchange in an ex- 529
tended system. The double exchange explains the ferromagnetic coupling 530
between magnetic ions of mixed valency. For example, La
1−x
Sr
x
MnO
3
(0 ≤ 531
x ≤ 0.175) shows ferromagnetic metallic behavior below room temperature. 532
In this material, a fraction x of the Mn ions are Mn
4+
and 1 −x are Mn
3+
, 533
becauseLaexistsasLa
3+
and Sr exists as Sr
2+
. 534
5. Itinerant ferromagnetism occurs in solids (such as Fe, Co, Ni, ···)con- 535
taining the magnetic moments associated with the delocalized electrons, 536
known as itinerant electrons, wandering through the sample. 537
10.9 Itinerant Ferromagnetism 538
Most of our discussion up to now has simply assumed a Heisenberg J
ij
S
i
·S
j
539
type interaction of localized spins. The atomic configurations of some of the 540
atoms in the 3d transition metal series are Sc (3d)
1
(4s)
2
,Ti(3d)
2
(4s)
2
,V 541
(3d)
3
(4s)
2
,Cr(3d)
5
(4s)
1
,Mn(3d)
5
(4s)
2
,Fe(3d)
6
(4s)
2
,Co(3d)
7
(4s)
2
,Ni 542
(3d)
8
(4s)
2
,Cu(3d)
10
(4s)
1
. If we simply calculate the band structure of these 543
materials, completely ignoring the possibility of magnetic order, we find that 544
the density of states of the solid has a large and relatively narrow set of 545
peaks associated with the 3d bands, and a broad but low peak associated 546
with the 4s bands as is sketched in Fig. 10.16. The position of the Fermi level 547
determines whether the d bands are partially filled or completely filled. For 548
transition metals with partially filled d bands, the electrons participating in 549
the magnetic states are itinerant. 550

Uncorrected Proof
BookID 160928 ChapID 10 Proof# 1 - 29/07/09
10.9 Itinerant Ferromagnetism 305
Fig. 10.16. Schematic illustration of the density of states of the transition metals
10.9.1 Stoner Model 551
In order to account for itinerant ferromagnetism, Stoner introduced a very 552
simple model with the following properties: 553
1. The Bloch bands obtained in a band structure calculation are maintained. 554
2. By adding an exchange energy to the Bloch bands a spin splitting, 555
described by an internal mean field, can be obtained. 556
3. States with spin antiparallel (−) to the internal field are lowered in energy 557
relative to those with parallel (+) spin. 558
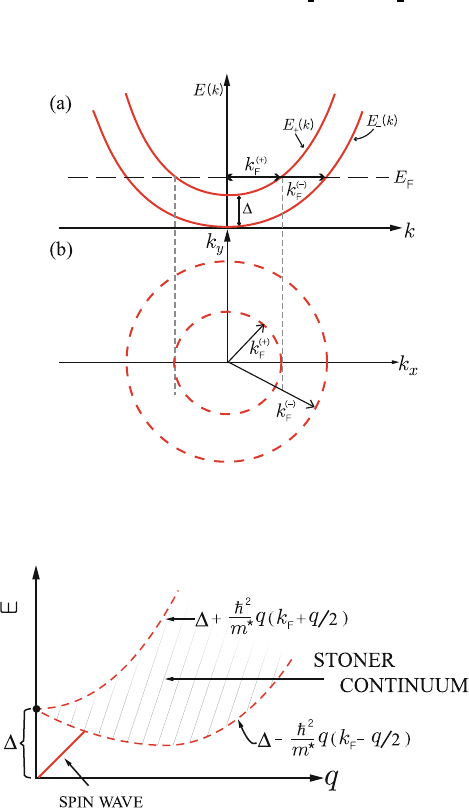
Wecanwriteforspinup(+)andspindown(−) electrons 559
E
−
(k)
¯hk
2
2m
∗
and E
+
(k)
¯hk
2
2m
∗
+Δ, (10.143)
where Δ is the spin splitting. The spin split Bloch bands and Fermi surfaces
560
for spin up and spin down electrons are illustrated in Fig. 10.17 in the presence 561
of spin splitting Δ. 562
10.9.2 Stoner Excitations 563
A single particle excitation in which an electron with wave vector k and spin 564
down (−) is excited to an empty state with wave vector k + q andspinup 565
(+) has energy 566
E = E
+
(k + q) − E
−
(k)
=
¯h
2
(k + q)
2
2m
∗
+Δ−
¯hk
2
2m
∗
=
¯h
2
m
∗
q ·
k +
q
2
+Δ.
(10.144)
These Stoner single particle excitations define the single particle continuum
567
shown in Fig. 10.18. The single particle continuum of possible values of |k| 568
for different values of |q| are hatched. Clearly when q = 0, the excitations all 569
have energy Δ. These are single particle excitations. In addition Stoner found 570
Uncorrected Proof
BookID 160928 ChapID 10 Proof# 1 - 29/07/09
306 10 Magnetic Ordering and Spin Waves
Fig. 10.17. Schematic illustration of the spin split Bloch bands in the Stoner model.
(a) Energy dispersion of the Bloch bands in the presence of spin splitting Δ (b)The
Fermi surfaces for spin up and spin down electrons
Fig. 10.18. Schematic illustration of the energy dispersion of the Stoner excitations
and spin wave modes. The hatched area shows the single particle continuum of
possible values of |k| for different values of |q|
spin waves of an itinerant ferromagnet that started at the origin (E =0at 571
q = 0) and intersected the single particle continuum at q
c
, a finite value of q. 572
The spin wave excitation is also indicated in Fig. 10.18. 573
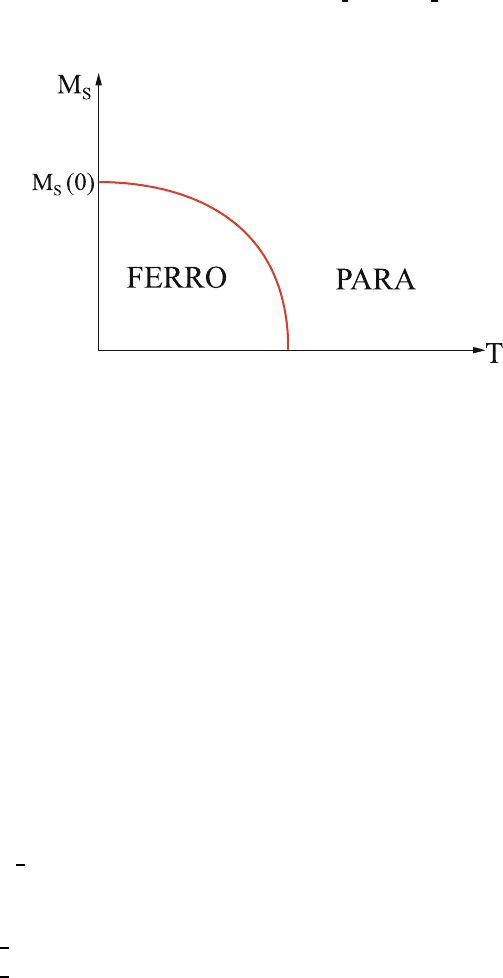
10.10 Phase Transition 574
Near T
c
, the ferromagnet is close to a phase transition. Many observable prop- 575
erties should display interesting behavior as a function of T − T
c
(see, for 576
example, Fig. 10.19). 577
Uncorrected Proof
BookID 160928 ChapID 10 Proof# 1 - 29/07/09
10.10 Phase Transition 307
Fig. 10.19. Schematic illustration of the temperature dependence of the sponta-
neous magnetization
Here, we list only a few of the interesting examples. 578
1. Magnetization:AsT increases toward T
c
the spontaneous magnetization 579
must vanish as 580
M(T ) ≈ (T
c
− T )
β
with β>0.
2. Susceptibility:AsT decreases toward T
c
in the paramagnetic state, the 581
magnetic susceptibility χ(T ) must diverge as 582
χ(T ) ≈ (T −T
c
)
−γ
with γ>0.
3. Specific heat :AsT decreases toward T
c
in the paramagnetic state, the 583
specific heat has a characteristic singularity given by 584
C(T ) ≈ (T − T
c
)
−α
with α>0.
In the mean field theory, where the interactions are replaced by their values
585
in the presence of a self-consistently determined average magnetization, we 586
find β =
1
2
and γ = 1 for all dimensions. The mean field values do not agree 587
with experiments or with several exactly solvable theoretical models for T 588
very close to T
c
. For example, 589
1. β =
1
8
in the 2 dimensional Ising model. 590
2. β
1
3
in the 3 dimensional Heisenberg model. 591
3. γ 1.25 for most 3 dimensional phase transitions instead of the mean field 592
predictions of γ =1. 593
In the early 1970’s K. G. Wilson developed the renormalization group 594
theory of phase transitions to describe the behavior of systems in the region 595
T T
c
. 596
Uncorrected Proof
BookID 160928 ChapID 10 Proof# 1 - 29/07/09
308 10 Magnetic Ordering and Spin Waves
Problems 597
10.1. Show that spin operators satisfy [
ˆ
S
2
,
ˆ
S
+
]=0and[
ˆ
S
z
,
ˆ
S
+
]=
ˆ
S
+
.Eval- 598
uate the commutator [S
+
,S
−
]and[S
±
,S
z
], and show that S
±
act as raising 599
and lowering operators. 600
10.2. If b
k
= N
−1/2
j
e
ik·x
j
a
j
and b
†
k
= N
−1/2
j
e
−ik·x
j
a
†
j
are spin wave 601
operators in terms of spin deviation operators, show that [a
j
,a
j
]=[a
†
j
,a
†
j
]=0 602
and [a
j
,a
†
j
]=δ
jj
implies [b
k
,b
k
]=
b
†
k
,b
†
k
=0and
b
k
,b
†
k
= δ
kk
. 603
10.3. In the text the Heisenberg Hamiltonian was written as 604
H = −2JS
i,j
(
8
1 −
ˆn
i
2S
a
i
a
†
j
8
1 −
ˆn
j
2S
+ a
†
i
8
1 −
ˆn
i
2S
8
1 −
ˆn
j
2S
a
j
+S
1 −
ˆn
i
S
1 −
ˆn
j
S
$
− gμ
B
B
0
S
i
1 −
ˆn
i
S
,
where ˆn
j
= a
†
j
a
j
and a
†
j
(a
j
) creates (annihilates) a spin deviation on site j. 605
Expand the square roots for small ˆn and show that the results for H
0
and H
1
606
agree with the expressions shown in (10.66) and (10.67), respectively. 607
10.4. Evaluate ω
k
, the spin wave frequency, for arbitray k within the first 608
Brillouin zone of a simple cubic lattice. Expand the result for small k and 609
compare it with the result given by (10.81). 610
10.5. An antiferromagnet can be described by H =
i,j
J
ij
S
i
· S
j
,where 611
J
ij
> 0. Show that the ground state energy E
0
of the Heisenberg antiferro- 612
magnet must satisfy 613
−S(S +1)
i,j
J
ij
≤ E
0
≤−S
2
i,j
J
ij
.
Hint: for the upper bound one can use the trial wave function
614
Φ
TRIAL
=
i ∈ A
j ∈ B
|S, S
i
|S, −S
j
,
where | S, ±S
k
is the state with S
z
= ±S on site k. 615
10.6. Prove that operators α
k
’s and β
k
’s defined in terms of spin wave 616
operators 617
α
k
= u
k
c
k
− v
k
d
†
k
and β
k
= u
k
d
k
− v
k
c
†
k
satisfy the standard commutation rules. Here, u
2
k
− v
2
k
= 1. (See (10.106)) 618
10.7. Discuss spin wave excitations in a two-dimensional square lattice. 619

Uncorrected Proof
BookID 160928 ChapID 10 Proof# 1 - 29/07/09
10.10 Phase Transition 309
Summary 620
In this chapter, we studied magnetic ordering and spin wave excitations of 621
magnetic solids. We first reviewed Heisenberg exchange interactions of atoms 622
and then discussed spontaneous magnetization and domain wall properties 623
of ferromagnets. The zero-temperature properties of Heisenberg ferromagnets 624
and antiferromagnets are described. Spin wave excitations and magnon heat 625
capacities of ferromagnets and antiferromagnets are also discussed. Finally, 626
Stoner model is introduced as an illustration of itinerant ferromagnetism. 627
The Heisenberg interaction Hamiltonian is given by 628
H = −2J
i,j
s
i
· s
j
,
where the sum is over all pairs of nearest neighbors. The exchange constant
629
J is positive (negative) for ferromagnets (antiferromagnets). For a chain of 630
magnetic spins, it is more favorable energetically to have the spin flip gradu- 631
ally. If the spin turns through an angle Φ
0
in N steps, where N is large, the 632
increase in exchange energy due to the domain wall is E
ex
= JS
2
Φ
2
0
N
. The 633
exchange energy is lower if the domain wall is very wide. 634
In the presence of an applied magnetic field B
0
oriented in the z-direction, 635
the Hamiltonian of a Heisenberg ferromagnet becomes 636
H = −
i,j
J
ij
S
iz
S
jz
−
1
2
i,j
J
ij
S
+
i
S
−
j
+ S
−
i
S
+
j
− gμ
B
B
0
i
S
iz
.
In the ground state all the spins are aligned parallel to one another and to
637
the magnetic field B
0
: |0 =
;
i
|S, S
i
. The ground state energy becomes 638
E
0
= −S
2
i,j
J
ij
− Ngμ
B
B
0
S.
For Heisenberg antiferromagnets, J is replaced by −J but a trial wave
639
function Φ
Trial
=
;
i ∈ A
j ∈ B
|S, S
i
|S, −S
j
is not an eigenfunction of H. 640
Low lying excitations of ferromagnet can be studied by introducing spin 641
deviation operator ˆn
j
defined by 642
ˆn
j
= S
j
−
ˆ
S
jz
= S −
ˆ
S
jz
≡ a
†
j
a
j
.
With a use of the Holstein–Primakoff transformation to operators a
†
j
and a
j
643
ˆ
S
+
j
=(2S
j
− ˆn
j
)
1/2
a
j
and
ˆ
S
−
j
= a
†
j
(2S
j
− ˆn
j
)
1/2
,
the Heisenberg Hamiltonian can be written, in the limit of ˆn
i
2S,as 644
H = E
0
+ H
0
+ H
1
.
Uncorrected Proof
BookID 160928 ChapID 10 Proof# 1 - 29/07/09
310 10 Magnetic Ordering and Spin Waves
Here, E
0
, H
0
,andH
1
are given, respectively, by 645
E
0
= −zJNS
2
− gμ
B
B
0
NS,
H
0
=(gμ
B
B
0
+2zJS)
i
ˆn
i
− 2JS
<i,j>
a
i
a
†
j
+ a
†
i
a
j
,
H
1
= −2J
<i,j>
ˆn
i
ˆn
j
−
1
4
ˆn
i
a
i
a
†
j
−
1
4
a
i
a
†
j
ˆn
j
−
1
4
ˆn
j
a
†
i
a
j
−
1
4
a
†
i
a
j
ˆn
i
+ higher order terms.
Introducing spin wave variables defined by
646
b
k
= N
−1/2
j
e
ik·x
j
a
j
and b
†
k
= N
−1/2
j
e
−ik·x
j
a
†
j
,
H
0
becomes H
0
=
k
¯hω
k
b
†
k
b
k
, where ¯hω
k
=2zJS(1 −γ
k
)+gμ
B
B
0
. Thus, 647
if we neglect H
1
, we have for the Hamiltonian of a state containing magnons 648
H = −
gμ
B
B
0
NS + zJNS
2
+
k
¯hω
k
b
†
k
b
k
.
We note that, at low enough temperature, the elementary excitations are
649
waves of energy ¯hω
k
. 650
At low temperature, the internal energy and magnon specific heat are 651
given by 652
U
0.45
π
2
Θ
5/2
D
3/2
and C =
∂U
∂T
=0.113k
B
Θ
D
3/2
.
The spontaneous magnetization at temperature T is given by
653
M
s
=
gμ
B
V
9
NS −
k
b
†
k
b
k
:
.
At low temperature, M
s
(T ) becomes M
s
(T )=
N
V
2μS − 0.117
μ
a
3
Θ
2SJ
3/2
. 654
In the presence of an applied field B
0
= B
0
ˆz andananisotropyfieldB
A
, 655
the Heisenberg Hamiltonian of an antiferromagnet can be written 656
H =+J
<i,j>
ˆ
S
i
·
ˆ
S
j
− gμ
B
(B
A
+ B
0
)
l∈a
ˆ
S
a
lz
+ gμ
B
(B
A
− B
0
)
p∈b
ˆ
S
b
pz
.
In the absence of magnon–magnon interaction, the ground-state energy is
657
given by 658
E
GS
= −2NzJS
2
− 2gμ
B
B
A
NS +
k
[ω
k
− (ω
A
+ ω
e
)] .
The internal energy due to antiferromagnetic magnons is given by
659
U =2
k
ω
k
e
ω
k
/Θ
− 1
.
The low temperature specific heat per particle becomes
660
C =
8π
4
5
Θ
Θ
N
3
.

Uncorrected Proof
BookID 160928 ChapID 11 Proof# 1 - 29/07/09
11 1
Many Body Interactions – Introduction 2
11.1 Second Quantization 3
The Hamiltonian of a many particle system is usually of the form 4
H =
i
H
0
(i)+
1
2
i=j
V
ij
. (11.1)
Here, H
0
(i) is the single particle Hamiltonian describing the ith particle, and 5
V
ij
is the interaction between the ith and jth particles. Suppose we know the 6
single particle eigenfunctions and eigenvalues 7
H
0
|k = ε
k
|k.
We can construct a basis set for the many particle wave functions by taking
8
products of single particle wave functions. We actually did this for bosons 9
when we discussed phonon modes of a crystalline lattice. We wrote 10
|n
1
,n
2
,...,n
k
,... =(n
1
!n
2
! ···n
k
!)
−1/2
a
†
1
n
1
a
†
2
n
2
···
a
†
k
n
k
···|0.
(11.2)
This represents a state in which the mode 1 contains n
1
excitations, ...,the 11
mode k contains n
k
excitations. Another way of saying it is that there are n
1
12
phonons of wave vector k
1
, n
2
phonons of wave vector k
2
, .... The creation 13
and annihilation operators a
†
and a satisfy 14
a
k
,a
†
k
−
= δ
kk
;[a
k
,a
k
]
−
=
a
†
k
,a
†
k
−
=0.
The commutation relations assure the symmetry of the state vector under
15
interchange of a pair of particles since 16
a
†
k
a
†
k
= a
†
k
a
†
k
.

Uncorrected Proof
BookID 160928 ChapID 11 Proof# 1 - 29/07/09
312 11 Many Body Interactions – Introduction
Thesingleparticlepartisgivenby 17
i
H
0
(i)=
k
ε
k
n
k
, (11.3)
where ε
k
= k|H
0
|k and n
k
= a
†
k
a
k
. 18
For Fermions, the single particle states can be singly occupied or empty. 19
This means that n
k
can take only two possible values, 0 or 1. It is convenient 20
to introduce operators c
†
k
and its Hermitian conjugate c
k
and to require them 21
to satisfy anticommutation relations 22
c
k
,c
†
k
+
≡ c
k
c
†
k
+ c
†
k
c
k
= δ
kk
,
[c
k
,c
k
]
+
=
c
†
k
,c
†
k
+
=0.
(11.4)
These relations assure occupancy of 0 or 1 since
c
†
k
2
=0and(c
k
)
2
=0: 23
c
†
k
,c
†
k
+
=2c
†
k
c
†
k
=0
[c
k
,c
k
]
+
=2c
k
c
k
=0
from the anticommutation relations given by (11.4). It is convenient to order
24
the possible values of the quantum number k (e.g. the smallest k’s first). Then, 25
an eigenfunction can be written 26
|0
1
, 1
2
, 0
3
, 0
4
, 1
5
, 1
6
,...,1
k
,... = ···c
†
k
···c
†
6
c
†
5
c
†
2
|0
1
, 0
2
,...0
k
,...,0
n
,....
The order is important, because interchanging c
†
6
and c
†
5
leads to 27
|0
1
, 1
2
, 0
3
, 0
4
, 1
6
, 1
5
,...,1
k
,... = −|0
1
, 1
2
, 0
3
, 0
4
, 1
5
, 1
6
,...,1
k
,....
The kinetic (or single particle) energy part is given by
28
k
occupied
k|H
0
|kc
†
k
c
k
=
k
ε
k
c
†
k
c
k
=
k
ε
k
n
k
. (11.5)
The more difficult question is “How do we represent the interaction term in
29
the second quantization or occupation number representation?”. 30
In the coordinate representation the many particle product functions must 31
be either symmetric for Bosons or antisymmetric for Fermions. Let us write 32
out the case for Fermions 33
Φ=
1
√
N!
P
(−)
P
P {φ
α
(1)φ
β
(2) ···φ
ω
(N)} (11.6)
Here,
P
means sum over all permutations and (−)
P
is −1foroddpermu- 34
tations and +1 for even permutations. For example, for a three particle state 35

Uncorrected Proof
BookID 160928 ChapID 11 Proof# 1 - 29/07/09
11.1 Second Quantization 313
the wave function Φ
αβγ
(1, 2, 3) can be written 36
Φ
αβγ
=
1
√
3!
[ φ
α
(1)φ
β
(2)φ
γ
(3) − φ
α
(1)φ
β
(3)φ
γ
(2) + φ
α
(2)φ
β
(3)φ
γ
(1)
−φ
α
(2)φ
β
(1)φ
γ
(3) + φ
α
(3)φ
β
(1)φ
γ
(2) −φ
α
(3)φ
β
(2)φ
γ
(1)] .
(11.7)
Such antisymmetrized product functions are often written as Slater determi-
37
nants 38
Φ=
1
√
N!
φ
α
(1) φ
α
(2) ···φ
α
(N)
φ
β
(1) φ
β
(2) ··· φ
β
(N)
.
.
.
.
.
.
.
.
.
.
.
.
φ
ω
(1) φ
ω
(2) ···φ
ω
(N)
(11.8)
Look at V
12
operating on a two particle wave function Φ
αβ
(1, 2). We assume 39
that V
12
= V (|r
1
− r
2
|)=V (r
12
)=V
21
.Then 40
V
12
Φ
αβ
(1, 2) =
1
√
2
V
12
[φ
α
(1)φ
β
(2) − φ
β
(1)φ
α
(2)] .
The matrix element Φ
γδ
|V
12
|Φ
αβ
becomes 41
Φ
γδ
|V
12
|Φ
αβ
=
1
2
γδ|V
12
|αβ +
1
2
δγ|V
12
|βα
−
1
2
γδ|V
12
|βα−
1
2
δγ|V
12
|αβ.
(11.9)
Since γδ|V
12
|αβ =
"
d
3
r
1
d
3
r
2
φ
∗
γ
(1)φ
∗
δ
(2)V (r
12
)φ
α
(1)φ
β
(2), we can see that 42
it must be equal to δγ|V
12
|βα by simple interchange of the dummy variables 43
r
1
and r
2
. Thus, we find, for two-particle wave function, that 44
Φ
γδ
|V
12
|Φ
αβ
= γδ|V
12
|αβ−γδ|V
12
|βα. (11.10)
45
Just as we found in discussing the Heisenberg exchange interaction, we find 46
that the antisymmetry leads to a direct term and an exchange term. Had we 47
been considering Bosons instead of Fermions, a plus sign would have appeared 48
in Φ
αβ
(1, 2) and in the expression for the matrix element. 49
Exactly the same result can be obtained by writing 50
V
12
=
λλ
μμ
λ
μ
|V
12
|λμc
†
λ
c
†
μ
c
μ
c
λ
, (11.11)
and
51
|Φ
αβ
=
1
√
2
c
†
β
c
†
α
|0, (11.12)
where |0 is the vacuum state, which contains no particles. It is clear that
52
V
12
|Φ
αβ
=
1
√
2
λλ
μμ
λ
μ
|V
12
|λμc
†
λ
c
†
μ
c
μ
c
λ
c
†
β
c
†
α
|0
