Scheffler Immo E. Mitochondria
Подождите немного. Документ загружается.

THE ELECTRON TRANSPORT CHAIN 191
details on the structure. It provides answers to many questions, and it suggests
that the evolutionary history may be an explanation for the apparently exces-
sive number of Fe – S centers required for electrons transfer from FMN to ubi-
quinone. Alternatively, there may be an as yet undiscovered rationale linked to
the mechanism of proton translocation by this enzyme.
Understanding the structure and function of this complex has been one of
the challenges for which signifi cant progress can be reported. Two major
unsolved problems remain: (1) to understand the path of the protons pumped
by the complex and (2) to elucidate the mechanism by which electron trans-
port taking place exclusively in the peripheral subcomplex is coupled to proton
translocation that must involve the integral membrane proteins. Another chal-
lenge is to understand the assembly of this complex from 38 subunits imported
from the cytosol and seven subunits made in the mitochondrial matrix. What
regulates the expression of the widely dispersed (unlinked) nuclear genes?
What regulates the level of assembly of this complex? Is there a unique assem-
bly pathway; that is, is it possible to identify assembly intermediates? Are the
ND subunits inserted independently into the inner mitochondrial membrane,
or is their synthesis and membrane integration coupled intimately to the
assembly of subunits imported from the cytosol? Some answers to these ques-
tions have been obtained in recent years.
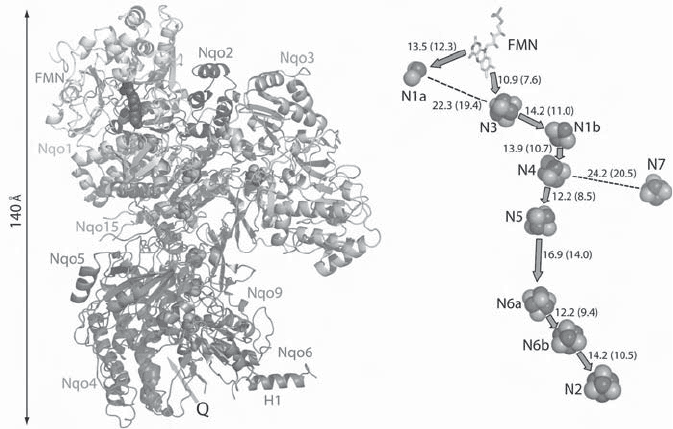
Figure 5.10 (A) Crystal structure of the peripheral subcomplex of respiratory complex
I (NADH – ubiquinone oxidoreductase) from Thermus thermophilus at 3.3 - Å resolution
(74, 76) . The subunits are labeled following the nomenclature for bacteria. (B) Location
and distances between the 9 Fe – S clusters in this subdomain. (Reproduced from refer-
ence 76 with permission.) See color plates.
AB
192 MITOCHONDRIAL ELECTRON TRANSFER
The fi rst studies to address such questions were reported for Neurospora
crassa (45) . Effectively, gene knockouts can be achieved by site - specifi c inte-
gration of a hygromycin resistance marker, and the consequences of a missing
subunit on assembly can be studied (47, 77) . The mutant nuo21 is of special
interest because in the absence of this peptide the peripheral complex is
assembled completely, while assembly of the membrane arm is only partial.
With this mutant as starting material, the peripheral subcomplex can be pre-
pared and purifi ed conveniently. It can oxidize NADH when ferricyanide is
used as an artifi cial electron acceptor, and it even has a low NADH: ubiqui-
none oxidoreductase activity. The latter is insensitive to piericidin A and thus
believed to be due to ubiquinone binding to a site on the peripheral arm which
is not normally exposed in an intact complex I. Neurospora mutants defective
in a nuclear encoded complex I peptide have a reduced growth rate, but their
mitochondria are not signifi cantly respiration defi cient. The presence of cyto-
chromes and inhibition of respiration by antimycin and cyanide suggests a
functioning electron transport chain downstream from ubiquinone, and the
absence of a piericidin - sensitive complex I is overcome by an alternative
NADH - ubiquinone oxidoreductase.
More recently the assembly of the mammalian complex has been studied
in tissue culture systems. In Scheffl er ’ s laboratory (28, 39, 40) Chinese hamster
mutants cells missing either the MWFE or the ESSS subunits in the membrane
arm were shown to lack complex I by Blue Native gel electrophoresis. The
mutants can be complemented with the respective cDNAs; moreover, the
expression of these cDNAs can be regulated from an inducible promoter.
When induced, the MWFE and ESSS subunits reach steady - state levels within
24 hours, but the appearance of the > 900 - kDa complex I on Blue Native gels
is delayed by another ∼ 24 hours, and full respiration measured by an oxygen
electrode requires even more time (28; Potluri, unpublished). Some other
subunits are missing when MWFE or ESSS are absent, presumably because
they are unstable and rapidly turned over in the absence of assembly (their
mRNAs are present at normal levels).
Other laboratories have examined complex I assembly and assembly inter-
mediates in human cells from patients with isolated complex I defi ciencies (see
Chapter 7 ) (29, 30, 78 – 80) . Such patients are alive because there is a residual
activity; and in all cases examined so far, it appears that the reduced activity
is due to a reduced steady - state level of intact complex I. The interpretation
is that assembly is slowed down due to mutations in proteins encoded by
nuclear genes (most of those are in the peripheral complex). Two precaution-
ary statements may be in order. First, the assembly intermediates in such
patient/mutant cell lines may not be exactly the same as those formed under
normal conditions. Second, the analysis requires the use of Blue - Native gels
and the solubilization of mitochondrial membranes by selected detergents.
Mutations may introduce weakened associations in the complex that lead to
dissociation into subcomplexes under the conditions of the experiment, but
not in vivo .
THE ELECTRON TRANSPORT CHAIN 193
Over the years the analysis of this complex and of its activity in mitochon-
dria has been aided by the discovery of a variety of highly specifi c, naturally
occurring inhibitors (Figure 5.11 ). The best known of these is rotenone, used
as a fi sh poison; another is the antibiotic piericidin A. Some of these are being
developed as potential insecticides and for various other pharmacological
purposes (81) . As discussed elsewhere in this volume, the “ designer drug ”
MPTP (1 - methyl - 4 - phenyl 1,2,3,6 tetrahydropyridine) can be converted to the
active metabolite 1 - methyl - 4 - phenyl prydinium (MPP
+
) by monoamine oxidase
and transported into dopaminergic neurons. At suffi ciently high concentra-
tions (mM) it is a specifi c inhibitor of complex I. This mechanism is thought
to be responsible for the death of neurons in the substantia nigra and hence
symptoms of Parkinsonism in humans and experimental primates (82, 83) . A
large and diverse series of specifi c inhibitors of complex I, some occurring
naturally and some synthetics, have been discussed and investigated systemati-
cally by Friedrich et al. (84) . On the basis of detailed kinetic analyses, these
authors were able to divide the inhibitors into two classes: class I inhibitors
(piericidin A, annonin VI, phenalamid A2, aurachins A and B, thiangazole, and
fenpyroximate (synthetic)) inhibit complex I in a partially competitive manner
with respect to ubiquinone; class II inhibitors (rotenone, phenoxan, aureothin,
and benzimidazole (synthetic)) act in a noncompetitive manner. None of the
inhibitors inhibit the NADH - ferricyanide reductase — that is, the peripheral
membrane arm of the complex extending into the matrix. Instead, all inhibitors
appear to arrest the transfer of electrons from the high - potential iron – sulfur
cluster (N - 2) to ubiquinone. Since the Fe – S clusters have not been completely
characterized in this complex, it is not possible to be more specifi c. However,
photoaffi nity labeling studies with a rotenone derivative have suggested that
this binding site is close to the peptide encoded by the mitochondrial ND1
gene. A more recent investigation of rotenone binding raising several still
controversial issues related to conformational changes within the complex has
been published by Grivennikova et al. (85) . Specifi cally, these authors propose
that one rotenone - specifi c site in complex I affects NADH oxidation by ubi-
quinone, while a second site is active in the ubiquinol – NAD
+
reductase reac-
tion. This apparent violation of the principle of microscopic reversibility clearly
deserves further clarifi cation.
Detailed studies on the mechanism of inhibition by MPP+ analogues by
Singer and colleagues (86) have led to the interpretation that there are two
MPP+ - binding sites, both of which must be occupied for complete inhibition.
A “ hydrophilic site ” is defi ned by accessibility to relatively hydrophilic
analogues, while a “ hydrophobic site ” may be less exposed to the aqueous
environment. This interpretation is consistent with fi ndings that the complex
I can contain two ubisemiquinones. Miyoshi and colleagues have reexamined
this question with the help of a new series of N - methyl - pyridinium and N -
methyl - quinolinium analogues, with the goal of fi nding a more potent inhibitor
effective at lower concentrations and, perhaps more signifi cantly, the aim of
fi nding an inhibitor which exhibits selectivity for only one of the two sites (87) .
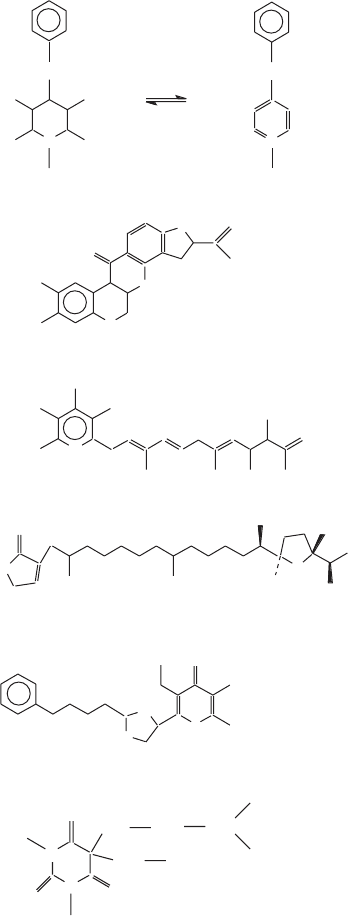
194 MITOCHONDRIAL ELECTRON TRANSFER
Figure 5.11 A and B: Structures of specifi c inhibitors of complex I frequently used in
the study of this complex (modifi ed from references 84 and 87 ). The compounds shown
in part B are N - methyl - pyridinium analogues in which various side chains can be sub-
stituted at positions 2, 3, or 4.
MTPT -------> MPP
+
Rotenone
Annonin VI
Phenoxan
Piericidin A
O
O
OCH
3
N
O
CH
2
N
CH
3
H
H
H
H
CH
2
N
CH
3
R
OH
OH
OH
O
O
OH
O
H
CH
3
O
O
CH
3
O
O
O
O
N
OH
CH
3
O
CH
3
O
OH
Amytal
N
N
O
O
O
CH
2
CH
2
CH
3
CH
2
CH
H
H
CH
3
CH
3
A
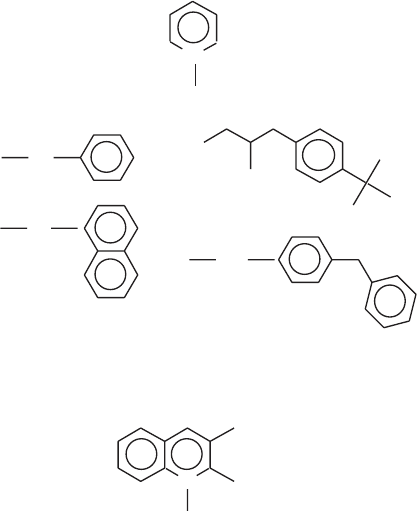
THE ELECTRON TRANSPORT CHAIN 195
The results clearly support the two - site model, and signifi cant progress toward
achieving the second aim was also made. The presence of two ubiquinone
binding sites is in good agreement with the acceptance of ubisemiquinone as
an obligatory intermediate in the two - electron transfer from NADH to ubi-
quinone (88, 89) .
5.2.5 Complex II
Complex II (succinate:ubiquinone oxidoreductase) consists of only four pep-
tides and is thus the simplest of all the complexes of the ETC. The two largest
peptides constitute the peripheral portion of the complex and function as the
enzyme succinate dehydrogenase in the Krebs cycle. They are associated with
the membrane through two integral membrane proteins, also referred to as the
“ anchor ” proteins (see Figure 5.12 ). Electrons from the oxidation of succinate
to fumarate are channeled through this complex to ubiquinone. Thus, complex
II links the Krebs cycle directly to the ETC (90 – 92) . A highly conserved and
similar complex is also found as part of the electron transport system in bacte-
rial membranes. The corresponding genes were fi rst cloned in bacteria, and the
N-Methyl-pyridinium
N-Methyl-quinolinium
O
O
O
N
1
2
3
4
(CH
2
)
11
CH
3
CH
3
4
3
2
1
N
B
Figure 5.11 (Continued)
196 MITOCHONDRIAL ELECTRON TRANSFER
bacterial systems ( Bacillus subtilis and Escherichia coli ) have been very useful
model systems in the study of the structure, function, and assembly of this
complex. Excellent authoritative reviews have been written by several authors
(93 – 95) .
Cofactors and metal ions make up the additional components of complex
II. A fl avin is linked covalently to the largest peptide (70 kDa), yielding the
fl avoprotein subunit (Fp). Singer and his colleagues established the chemical
linkage in the early 1970s, and they also identifi ed a histidine as the point of
attachment (96 – 98) . The timing and mechanism of this conjugation reaction
was an open question until recently when the Fp gene (SDH1) was cloned in
yeast. Conversion of His90 to Ser90 by site - directed mutagenesis prevented
covalent attachment of FAD, but the assembly of the complex II in mitochon-
dria was not affected (99 – 101) . It was clearly shown that fl avin attachment to
the normal Fp peptide occurred in the mitochondrial matrix, after the removal
of the targeting sequence (99) . Additional experiments were interpreted to
indicate that the enzyme responsible for this modifi cation has to recognize a
folded Fp peptide as one of its substrates, rather than a short peptide sequence
surrounding the targeted histidine side chain. Nevertheless, this region of the
peptide is highly conserved in evolution (see reference 102 for a review). When
complex II was assembled in yeast with a noncovalently bound fl avin, the
complex was active only as a fumarate reductase, not as a succinate dehydro-
genase (101) . This result supports the idea that the covalent attachment of the
fl avin raises its midpoint potential to the level required for the oxidation of
succinate (103) .
The Fp subunit is intimately associated with the iron – protein subunit (Ip),
made up of a peptide of 27 kDa containing three nonheme Fe – S centers:
[2Fe – 2S], [3Fe – 4S], [4Fe – 4S] (104) . Each has a characteristic redox potential,
and together these centers serve in the electron transport through the subunit
(93, 98, 105) . The Ip peptides from all species examined to date, and even from
prokaryotes, have three highly conserved cysteine clusters (CI, CII, CIII)
within the peptide. The fi rst cluster is used to make the [2Fe – 2S] center, and
it resembles the [2Fe – 2S] cluster also found in plant ferredoxins. The [4Fe – 4S]
center is made from the fi rst, second, and third cysteines of cluster II and the
third cysteine of cluster III, while the [3Fe – 4S] center is made from the fi rst
and second cysteines of cluster III and the fourth cysteine of cluster II. These
deductions were made from EPR studies made with the help of site - directed
mutagenesis of bacterial sdh and frd iron – proteins and were subsequently
confi rmed in the crystal structures. Thus, the assembly of the clusters depends
on the secondary and tertiary structure of the Ip subunit rather than the local
peptide sequence. It should be noted here that the Fe – S clusters are assembled
fi rst by an independent mechanism (see Section 6.8) and then delivered to
their fi nal destination in Ip and various other proteins.
The Fp – Ip complex (SDH) can be dissociated from an isolated complex II
by treatment with chaotropic ions as fi rst shown by Hatefi and colleagues (92) .
THE ELECTRON TRANSPORT CHAIN 197
It functions as a succinate dehydrogenase, when an artifi cial electron acceptor
such as ferricyanide or tetrazolium is included in the assay, but cannot interact
directly with ubiquinones. To reconstitute ubiquinone reduction, the SDH
complex must be combined with the two integral membrane proteins of
complex II, C
II - 3
and C
II - 4
, also referred to as QPs - 1 and QPs - 2 (or CybL and
CybS). These “ anchor ” proteins are small peptides of molecular weight 15 and
12 – 13 kDa, respectively (in mammals), and sequence analysis suggested that
each has three transmembrane segments, along with an N - terminal domain
extending into the matrix for interaction with the SDH enzyme (106, 107) .
Solubilization and reconstitution experiments, sequence analysis and
hydropathy plots, and some earlier radiolabeling experiments by Merli et al.
(108) suggested a domain structure for complex II that has been largely con-
fi rmed by X - ray crystallography. The fi rst structure to be solved was the struc-
ture of the E. coli fumarate reductase (109) . For good reasons (see below) the
overall structure served as an excellent model for the structure of complex II
in prokaryotes as well as eukaryotes. Since then the E. coli succinate dehydro-
genase complex has also been solved (110 – 113) , revealing in great detail the
path of electrons from succinate via the fl avin, three Fe – S cluster to ubiqui-
none (see Figure 5.13 ). Finally, a high - resolution crystal structure of a mam-
malian complex II (porcine heart) at 2.4 - Å resolution was published by Sun
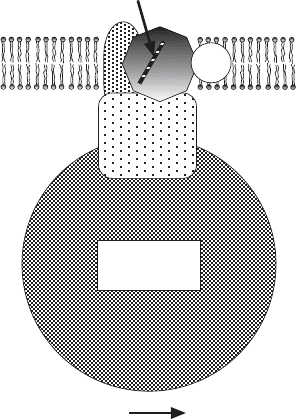
Figure 5.12 Schematic representation of complex II, succinate – ubiquinone oxidore-
ductase. Two integral membrane proteins serve to anchor the iron – protein (Ip) and the
fl avoprotein (Fp) of SDH.
FAD
Q
[2Fe-2S]
[4Fe-4S]
[3Fe-4S]
Succinate Fumarate
cytochrome
MATRIX
198 MITOCHONDRIAL ELECTRON TRANSFER
et al. (114) . This structural determination has permitted a detailed analysis of
binding sites for phospholipids, ubiquinone (two sites), and various inhibitors
of the enzyme. A comparison with the E. coli enzyme has revealed many con-
served structural components as well as informative differences.
There is some analogy with complex I, but on a much simpler scale. The
peripheral subcomplex (Fp (SdhA), Ip (SdhB)) has the substrate binding site
(succinate), the hydrogen acceptor (FAD), and 3 [Fe – S] centers for the con-
ductance of electrons to the second and relatively simple membrane subcom-
plex consisting of the two anchor proteins. This membrane complex also
contains a b - type cytochrome and the binding site(s) for ubiquinone. The
involvement of the b - type cytochrome with electron transport to ubiquinone
is still under investigation; it is attached to both anchor proteins. The C
II - 3
C
II - 4
peptides have highly conserved histidines linking the heme b to the simple
membrane complex (102) .
The reaction catalyzed by complex II is
succinate Q fumarate QH
2
+→ +
In most eukaryotic cells the reaction proceeds from left to right, not only
because of the relative concentrations of substrates and products, but also
because the enzyme has diode - like properties that prevent electron fl ow in the
opposite direction (115, 116) . Understanding the biophysical basis for this
behavior remains one of the challenges.
Specifi c inhibitors continue to play an important role in biochemical studies
of electron transport and the analysis of mitochondrial functions. For complex
II the most useful inhibitor has been the substrate analogue malonate, which
binds competitively and specifi cally at the active site on the Fp subunit.
There are examples of organisms or conditions in which the reduction of
fumarate becomes a physiologically relevant reaction. Facultative bacteria
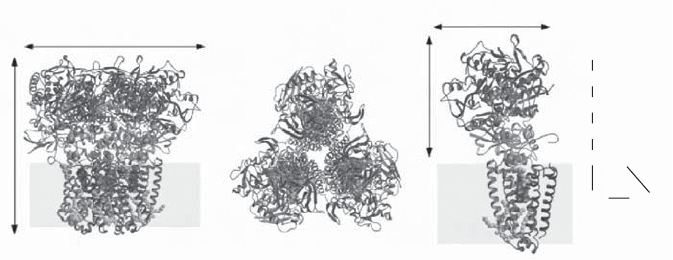
Figure 5.13 X - ray structure of succinate dehydrogenase (complex II) from E. coli .
(A) The trimer viewed parallel to the membrane. (B) Perpendicular view of the trimer
viewed from the cytoplasm. (C) View of a monomer showing the redox centers and
the distances between them. (From reference 113 with permission.)
AB
125 Å
60 Å
70 Å
cytoplasm
cytoplasm
periplasm
periplasm
SdhC
SdhA
OAA
4.6(2.5)
16.0(11.1)
12.4(9.3)
11.9(9.1)
[3Fe-4S]
[4Fe-4S]
[2Fe-2S]
18.5
(11.4)
11.0
(7.6)
Heme b
6.5(9.8)
FAD
UQ
SdhB
SdhD
CL
120
Å
C
THE ELECTRON TRANSPORT CHAIN 199
such as E. coli can proliferate under aerobic conditions and with a minimal
supply of glucose when complete combustion to carbon dioxide and the most
effi cient exploitation of available energy resources is achieved by operating
the Krebs cycle and oxidative phosphorylation. The interconversion of essen-
tial metabolites also requires the Krebs cycle. E. coli and other bacteria can
also grow under anaerobic conditions when energy metabolism has to be
altered drastically. Under such conditions the reduction of fumarate to succi-
nate becomes an essential reaction. However, the reverse reaction is not
achieved by the same SDH enzyme induced under aerobic conditions, but is
instead catalyzed by a completely distinct complex named fumarate reductase,
FRD. The two complexes are similar with respect to protein composition (Fp,
Ip, and anchor proteins), but these peptides are encoded by two distinct
operons. SdhA,
1
sdhB, sdhC, and sdhD are represented by four cistrons on a
polycistronic transcript for succinate:ubiquinone oxidoreductase, and frdA,
frdB, frdC, and frdD are encoded by four cistrons for fumarate:ubiquinone
oxidoreductase). In B. subtilis there is only one anchor protein, but it is almost
twice the size of the two E. coli peptides and may represent a gene fusion. The
regulation of the expression of these operons by oxygen and glucose has been
studied extensively in bacteria (94, 117, 118) . It remains to understand in some
detail how some changes in the primary sequence of these peptides can alter
the microenvironment of the redox couples (Fe – S, FAD) to favor reactions in
one direction or the other. Another noteworthy aspect of the SDH and FRD
activities in bacteria is that ubiquinone serves as the electron acceptor from
SDH under aerobic conditions, while reduced menaquinone serves as the
electron donor via the FRD in the reduction of fumarate to succinate under
anaerobic conditions. For a more detailed discussion of issues, the reader is
referred to the authoritative review of Ackrell et al. (93) , in which both pro-
karyotic and eukaryotic enzymes are discussed.
Another interesting situation arises for eukaryotic organisms, which spend
part of their developmental cycle under relatively anaerobic conditions and
another part of their life under aerobic conditions. For example, the sheep
nematode Haemonchus contortus switches to a fermentative, predominantly
anaerobic metabolism of the parasitic (adult) stages from the aerobic metabo-
lism found in the free - living larvae (119) . It appears that isozymes of the
iron – protein of complex II are differentially expressed in larvae and adults,
and it has been argued that a switch in isozymes is responsible for the change
in complex II from functioning predominantly as a fumarate reductase to the
conventional succinate:ubiquinone oxidoreductase under aerobic conditions.
In a related species, Ascaris suum , a similar switch and the existence of iso-
zymes for the fl avoprotein have been demonstrated (120) . The existence of
multiple genes (isozymes) for complex II may be a general observation for
1
The genes for complex II have been designated in different ways by different authors for various
organisms. The designation SDH1, SDH2, SDH3, and SDH4 is proposed for the four genes encod-
ing the subunits in the order of decreasing molecular mass of the peptides in yeast. sdhA, sdhB,
sdhC, and sdhD have been used for the bacterial genes.
200 MITOCHONDRIAL ELECTRON TRANSFER
some protozoa, parasitic helminths, and marine organisms such as annelids
and mussels, which spend their different life cycles under different conditions
(121) . It is not yet clear whether one or all of the subunits of complex II have
to be changed. The problem is not only to change the direction of electron
fl ow between succinate and fumarate, since the redox potential of all the par-
ticipants must be considered. Hence, ubiquinone (UQ/UQH
2
; E
0
′ = +100 mV)
is generally the acceptor in succinate oxidation, while menaquinone is the
donor in bacteria. Eukaryotes engaged in mitochondrial fumarate reduction
must synthesize a closely related rhodoquinone (RQ) (122) . Since these qui-
nones interact with the anchor proteins, a switch in energy metabolism during
development may require not only a different set of anchor proteins, but also
a new pathway for the synthesis of a different quinone. Since UQ and RQ
differ only in one substituent on the quinone ring (the 5 - methoxy group is
replaced by an amino group in RQ), the branchpoint can be postulated to
occur at the end of the pathway of UQ or RQ biosynthesis. In all eukaryotes
examined to date, the fi nding of RQ has been indicative of a potential for
fumarate reduction. For example, the detection of a signifi cant quantity of RQ
in the sporocysts of Schistosoma mansoni has been correlated with fumarate
reduction as an essential pathway during the parasitic stage in the intermediate
hosts (snails) where sporocysts behave as facultative anaerobes (121) . On the
other hand, in Leishmania infantum promastigotes it had also been suggested
that fumarate reduction to succinate constitutes an electron sink during anoxia.
However, the absence of RQ in these organisms suggested that this reaction
was absent, and studies have confi rmed that these organisms produce succi-
nate exclusively via the Krebs cycle under aerobic conditions and go into a
metabolic arrest during oxygen deprivation (123) .
It has been speculated that fumarate reductase was the fi rst activity in pro-
karyotes in evolution, with menaquinone as the cofactor. When oxygen con-
centrations were raised in the atmosphere, an oxidative metabolism evolved
which included the Krebs cycle and specifi cally required the conversion of an
FRD activity to an SDH activity. This was achieved by gene duplication, cova-
lent attachment of the fl avin, a raise in the standard redox potentials of the
iron – sulfur clusters, and the utilization of ubiquinone instead of menquinone
(124) . The FRD activity was subsequently lost in eukaryotic mitochondria.
However, it is conceivable that in a few facultative anaerobic eukaryotes a
“ reversed ” evolution took place. A novel activity allowed the diversion of
5 - hydroxy - 6 - methoxy - 3 - methyl - 2 - polyprenyl - 1,4 - benzoquinone to rhodoqui-
none instead of ubiquinone, while gene duplication and evolution yielded
isopeptides that could be assembled to function as fumarate reductase again.
A detailed examination of cloned genes and deduced peptide sequences will
be required to resolve and support such a hypothesis.
5.2.5.1 Nuclear Versus Mitochondrial Location of Complex II Genes For
a while it was correct to state that all complex II proteins were encoded by
nuclear genes. This generalization has to be abandoned, because several dif-
