Scheffler Immo E. Mitochondria
Подождите немного. Документ загружается.

THE ELECTRON TRANSPORT CHAIN 201
ferent organisms have been found in which some of the complex II genes are
found on the mitochondrial genome. In the red algae Chondrus crispus the
sdhB and sdhC genes are found on the mt genome (125, 126) ; in a different
red algae ( Porphyra purpurea ) and in the phylogenetically distant zoofl agel-
late Reclinomas americana, the genes for the iron – protein and the two mem-
brane anchor peptides are found in the mitochondria (127) . These data are in
agreement with the endosymbiont hypothesis for the origin of the mitochon-
drial genome, and they have been used to argue in favor of a monophyletic
origin of mitochondria (see Chapter 2 ).
Eukaryotic genes for the Ip peptides (SDH2) in complex II were fi rst
cloned in 1989 (128) , and their number has increased dramatically over the
past few years. Similarly, the gene for the Fp peptide (SDH1) is available from
numerous species. Cloning the SDH3 and SDH4 genes was initially more chal-
lenging, because the sequence conservation across species is poor, in contrast
to the Ip and Fp peptides, and probes form one organism generally do not
hybridize with genes/cDNA from another distantly related organism (102) . An
extensive review of the molecular genetics of complex II with an emphasis on
gene cloning and regulation of gene expression has been published (102) .
Further updates with the progress from the Lemire group were published
susequently (129, 130) . The major conclusions can be found in the chapter on
nuclear genes for mitochondrial proteins (Chapter 4 ).
Gene disruption experiments in yeast have provided yeast mutants with
either the Fp, Ip, or anchor protein genes deleted, and such mutants can be
used as recipients for novel gene constructs with specifi c alterations to inves-
tigate structure - function relationships (131 – 133) . For example, a series of chi-
meric Ip proteins containing yeast and human sequences were investigated
to delineate a sequence between the fi rst and second cysteine cluster which
appears to be species - specifi c and which, when replaced by a heterologous
sequence, makes it impossible to assemble complex II (134) .
An interesting mammalian cell mutant defective in SDH activity and assem-
bly of complex II has been described for Chinese hamster fi broblasts in tissue
culture (135) . The mutant cells are respiration - defi cient and unable to grow in
medium in which glucose is limiting. The mutation has been established to
result from a single nucleotide change converting a tryptophan codon to a stop
codon, thus truncating the C
II - 3
peptide before the third transmembrane
segment (136) . The fate of the truncated peptide has not been determined, but
the result is a complete failure of the assembly of complex II; not even an
active SDH enzyme complex is formed, although the Fp and Ip peptides are
made and imported normally into the mitochondria. This result can be con-
trasted with the fi ndings with the nuo21 Neurospora mutant, where the periph-
eral complex is made in an active form but not attached to the membrane arm.
The implications of this fi nding are that the Fp and Ip are not assembled sepa-
rately before their attachment to the membrane. Instead, the matrix domains
of the integral membrane proteins appear to participate in the folding and
formation of the Fe – S centers in the Ip subunit and the simultaneous associa-
202 MITOCHONDRIAL ELECTRON TRANSFER
tion with the Fp subunit. In this context it is worth noting that the Ip and Fp
peptides cannot be separated without denaturation, and they have never been
successfully reassembled in vitro into an active SDH enzyme.
In the past decade, human patients have been identifi ed with partial complex
II defi ciencies due to mutations in the SDH genes (137) . They will be discussed
later in the context of mitochondrial diseases (Chapter 7 ). While some of these
mutations lead to predictable pathologies from what we now understand
about mitochondrial diseases, there are also mutations in the SDHB, SDHC,
and SDHD genes that give rise to a very specifi c type of tumor: paraganglio-
mas (138 – 140) .
5.2.6 Complex III
Complex III is ubiquinone – cytochrome c oxidoreductase and is often named
the bc
1
complex after the two cytochromes found within it; one also fi nds the
simpler name cytochrome c reductase in the literature. The overall reaction
catalyzed by this complex is
QH cyt c 2 H Q cyt c H
2
3+ 2+
++⇒++
++
224
io
Similar to the reaction taking place with complex I, the oxidation of one of
the substrates (QH
2
) and the transfer of electrons to the mobile carrier (cyt
c) is coupled to the transfer of protons across the inner mitochondrial mem-
brane. The fate of the protons will be the subject of the discussion to follow,
and a more detailed mechanism for proton translocation across the membrane
is also deferred for the moment.
Complex III appears to be quite similar in different species including the
yeast Saccharomyces cerevisiae, Neurospora crassa , animals and plants. A
website devoted to complex III can be consulted for much recent information
( http://www.life.uiuc.edu/crofts/bc - complex_site/ ). One peptide of the complex
is encoded by the mitochondrial genome and incorporated as cytochrome b.
The other peptides (8 in yeast, ∼ 8 in Neurospora , 10 in mammals) are encoded
by the nuclear genome, synthesized in the cytosol, and imported for assembly
in the inner mitochondrial membrane. Both yeast and Neurospora have
become model systems for the study of this complex because genetic manipu-
lations allow detailed questions to be asked about (a) individual peptides and
amino acids within each peptide and (b) their role in assembly, function, and
activity. As described elsewhere in this volume, in yeast even the mitochondrial
gene is now subject to deliberate mutational modifi cation (Chapter 7 ).
Functionally, the most important subunits are the cytochromes b and c
1
and
the Rieske iron – sulfur protein, since they are the only ones participating in
electron transfer and in the accompanying proton translocation. This fact is
emphasized by the fi nding that the corresponding complex in the bacterial
electron transport chain has only those three proteins. The role of the other
proteins is diffi cult to deduce, because they have no prosthetic groups. However,
at least in Neurospora and potato the two large peripheral subunits appear to
THE ELECTRON TRANSPORT CHAIN 203
be processing proteases, and the largest subunit I has been shown to be
involved in the import and processing of proteins from the cytosol (141, 142) .
In the mammalian system, and to a large extent also in Neurospora, the
elegant experimental approaches to studying complex III have primarily relied
on solubilization, partial dissociation into subcomplexes, and reconstitution
studies (141, 143) . The original procedure by Hatefi for the isolation from
mammalian mitochondria has been simplifi ed and adapted to a smaller scale
for many purposes. When too much lipid is removed during the purifi cation,
the activity of the complex declines. A most useful technique for isolating OX -
PHOS complexes including complex III directly from homogenized human
tissues is blue - native polyacrylamide gel electrophoresis (BN - PAGE), after
solubilization of the membrane by neutral detergents (27, 144) . It is suitable
for the preparation of complexes in the microgram - to - milligram scale. As iso-
lated from bovine and Neurospora mitochondria, the complex III is a dimer,
a conclusion supported from electron microscopic studies of membrane crys-
tals of the complex from Neurospora . It remains unclear whether dimerization
is absolutely essential for the catalytic activity of the complex, but Covian and
Trumpower have provided strong experimental evidence for the transfer of
electrons between the two b
L
hemes of the dimer (145) .
Relatively mild detergent can dissociate two subunits from the bovine
complex: the Rieske iron – sulfur protein (ISP) and a small ISP - associated
protein. Further dissociation with 1.5 M guanidine yields a cytochrome c1
subcomplex (cytochrome c1, p9.2, p7.2, a core subcomplex, and several iso-
lated subunits). Cytochrome b appears to remain with the core subcomplex,
but a large fraction is lost in this procedure. When higher detergent concentra-
tions are used to remove neutral lipids from the bovine complex, activity is
lost, but can be restored by slow addition of phosphatidylcholine or phospha-
tidylethanolamine in Triton X - 100. Quantitative measurements have suggested
that a complex III dimer must be surrounded by a complete annulus of phos-
pholipid. Less easily rationalized in terms of a structural model is the obser-
vation that bovine complex III has eight or nine tightly bound cardiolipin
molecules per monomer. Their removal causes irreversible loss of activity.
All 11 mammalian complex III peptides have been purifi ed on SDS - PAGE
and used for partial sequencing and for the production of antibodies that can
be used in the exploration of the topology of the complex in the inner mem-
brane. Another experimental approach to topological studies has employed
EPR techniques. An emerging model places the heme b
562
near the middle of
the lipid bilayer, and the other heme (b
566
) and the Rieske iron sulfur cluster
as well as cytochrome c
1
are on the “ P - side ” of the membrane (facing the
intermembrane space).
Complex III, like complex I, has two reaction centers for ubiquinone, Q
N
and Q
P
. The signifi cance of these multiple sites will become more apparent
when the proton pumping mechanism will be discussed in greater detail.
Studies addressing problems related to the yeast complex III have been
predominantly of a genetic nature. All eight nuclear genes for this complex
have been cloned, and null mutants for each gene have been isolated. With
204 MITOCHONDRIAL ELECTRON TRANSFER
the exception of the mutant affecting the acidic subunit VI, these mutants are
respiration - defi cient. Therefore, even the subunits without prosthetic groups
are necessary for an active complex III, and one can speculate that they are
required for assembly and stabilization of the complex. Among the many pet
mutants analyzed (146) , mutants with a defective ubiquinone – cytochrome c
oxidoreductase activity can be found, although a direct selection or enrich-
ment for such a specifi c defect is not possible. Thus, pet mutants having verifi ed
nuclear mutations must be further screened by biochemical assays to eliminate
mutants affecting mitochondrial protein synthesis or the ubiquinone biosyn-
thetic pathway (147) . Among the mutants with a specifi c lesion in complex III
activity, several classes have been distinguished:
1. Mutants with alterations in the eight nuclear genes for the protein sub-
units of the complex. As discussed above, null mutants are respiration
defi cient. In many of these the cytochrome b is also absent, presumably
because it needs to be stabilized by association with other subunits.
2. A number of mutants are unable to make a cytochrome b detectable by
spectroscopy, even though all structural genes in the nucleus and the
COB gene on the mtDNA are normal. In another chapter on the expres-
sion of mitochondrial genes in yeast, it has been discussed that a variety
of imported proteins are required for cytochrome b mRNA maturation
(group I intron splicing and processing of the 5 ′ terminal) and for mRNA
stabilization. The COB gene encodes a protein that appears to function
both in 5 ′ end processing and mRNA stabilization (translational initia-
tion?). Additional nuclear gene products are necessary for effi cient trans-
lation of the COB mRNA, which in turn also affects the splicing of the
intron, because the COB intron encodes a maturase (see Chapter 4 ).
Another consequence may be a defect in cytochrome oxidase (complex
IV) as a result of defective splicing of the COX1 mRNA.
3. Nuclear gene products are also required for the addition of the pros-
thetic groups to the various subunits. Hemes have to be covalently
attached to apocytochromes, and another gene product, Bcs1p, has been
implicated in the “ maturation ” of the Rieske protein. The precise func-
tion of the BCS1 gene product is not known.
4. Another set of proteins defi ned by nuclear mutations are involved in the
“ late stages of the assembly pathway ” (147) . This is a problem that so far
has been defi ned exclusively by a genetic approach in yeast. Similar
assembly factors have been identifi ed for complexes IV and V. The
mechanisms and pathways of assembly of the complexes in the ETC
represent a major frontier in the quest for understanding mitochondrial
biogenesis.
Cytochrome c reductase (complex III) from potato mitochondria has
received scrutiny, because attention has been attracted more broadly to plant
mitochondria. Similarities to the complex in mammals and yeast may not be
THE ELECTRON TRANSPORT CHAIN 205
surprising, but when sequencing information became available for compara-
tive studies, the so - called core proteins from the potato complex showed the
highest sequence identity with the mitochondrial processing peptidase (MPP)
from mammals and yeast (142) . These enzymes are involved in the cleavage
of matrix targeting sequencing from precursors imported from the cytosol. It
was subsequently verifi ed that a bifunctional protein exists in potato mito-
chondria (all plants?) which plays a role in electron transport as well as in
protein import. The complex is therefore referred to as the cytochrome c
reductase/processing peptidase complex (see Section 4.6 ). It overall composi-
tion (at least 10 subunits) resembles the mammalian and fungal complexes.
A crowning achievement in the study of complex III was the completion
of the complete crystal structure of the complex at 2.9 - Å resolution by the
groups of Yu and Deisenhofer (148) . Following on the heels of the determina-
tion of the structure of complex IV (see below), it represents another major
landmark in the understanding of the complexes of the mitochondrial electron
transport chain. An atomic model has now become available to relate the
proposed reaction mechanisms of QH
2
oxidation and the reduction of cyto-
chrome c to the architecture and topology of the complex.
The model confi rms, refi nes, and expands the stuctural features deduced
from biochemical studies. In the crystal, two bc1 monomers interact to form a
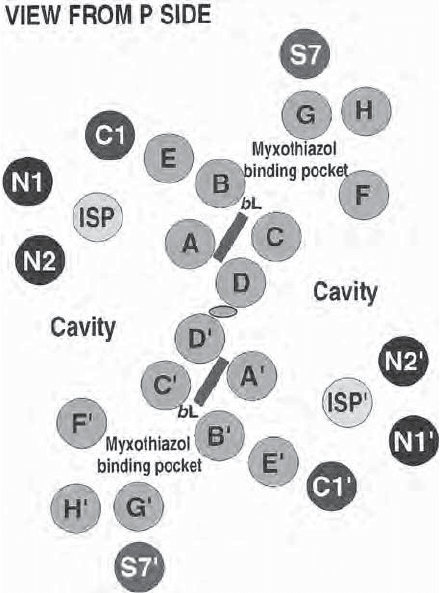
dimer with a twofold axis of symmetry perpendicular to the membrane. Each
monomer has 12 transmembrane helices: Eight are from cytochrome b, one is
from cytochrome c1, one is from the iron – sulfur protein, one from subunit 7,
and one is still unassigned (Figure 5.14 ).
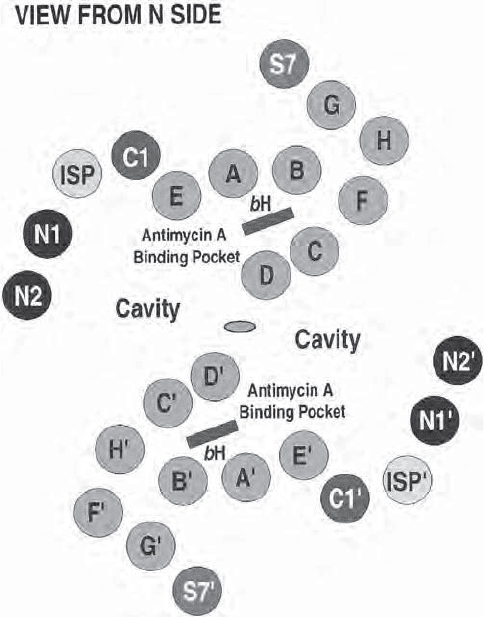
On one side of the membrane, more than half of the molecular mass of the
complex extends 75 Å into the matrix. It consists mainly of the so - called core
proteins that have a structural rather than a functional role. The peripheral
core 2 proteins contribute signifi cantly to the stabilization of the dimer, in
addition to the interaction of the cytochrome b helices within the membrane.
The two core proteins of the mammalian bc1 complex have homology to the
α and β subunits of the mitochondrial matrix protein processing peptidase
(MPP), and it is suggested that the soluble mammalian MPP has a similar
structure. As discussed above, in plants the MPP is an integral part of complex
III. A detailed side view of the complex is shown in Figure 5.15 . On the outside
of the membrane (P side), one fi nds the Rieske iron – sulfur cluster and the
domain of cytochrome c
1
containing the heme. The electron density for this
latter region is relatively poor, and the authors ascribe this problem to mobility
in the crystal, which may in fact refl ect a mobility required for interactions
with, and electron transfer to, cytochrome c. Two additional heme irons could
be located within the membrane domain, corresponding to heme b
L
and heme
b
H
, respectively. The identifi cation and distinction between the two hemes of
cyt b was also aided and confi rmed by the availability of difference density
maps comparing the native crystals with crystals obtained with the inhibitors
antimycin A or myxothiazol. The different targets of these inhibitors had been
assigned from biochemical and spectroscopic studies (see below).
206 MITOCHONDRIAL ELECTRON TRANSFER
Of special interest are the distances between the heme irons in b
H
, b
L
, and
c
1
and the [2Fe – 2S] cluster, in part to explain and confi rm previous spectro-
scopic studies, and understand the rate of electron transfer. In the dimer the
distances between two b
L
’ s and two b
H
’ s are comparable to the the b
H
– b
L
dis-
tance within a monomer, raising the possibility of electron transfer between
the monomers.
The overall model with its “ cavities ” and “ pockets ” clearly invites further
speculations about the accessibility of the complex to ubiquinone, antimycin,
and myxothiazol and ultimately about the translocation of protons and the
operation of the Q cycle.
Specifi c inhibitors of electron transport have been valuable as a means of
arresting electron fl ux at known locations for the purpose of spectroscopic
studies, for example, and to “ isolate ” a portion of the chain for detailed study.
They have also been useful and revealing probes of structure, and in some
Figure 5.14 Complex III. Schematic representation of the transmembrane helices
in the dimeric complex seen from the P and N (matrix) side (parts A and B, respec-
tively) (82) . See color plates.
A
THE ELECTRON TRANSPORT CHAIN 207
cases they have been useful to arrest electron fl ow within a complex to estab-
lish upstream and downstream portions of the complex. In the case of complex
III, the antibiotic antimycin A is the best known of the inhibitors, but a variety
of natural and synthetic inhibitors have been characterized. Initially their
utility was in the defi nition of a specifi c block in the electron transport chain
downstream from complexes I and II and upstream from cytochrome c, as well
as in the localization of the “ coupling sites ” for oxidative phosphorylation.
Later they were exploited in defi ning the distinction between center Q
N
or
center Q
P
and in elucidating the protonmotive Q cycle (Figure 5.16 ).
The formulation of the Q cycle (149 – 152) required the postulate of two
quinone reaction sites (see below). The Q
N
or quinone reduction center is
located on the matix side (N - side) of the inner membrane is associated with
the recycling of half of the electrons back into the quinone pool and the uptake
of protons from the matrix. It is inhibited by antimycin A, funiculosin, and
B
Figure 5.14 (Continued)
208 MITOCHONDRIAL ELECTRON TRANSFER
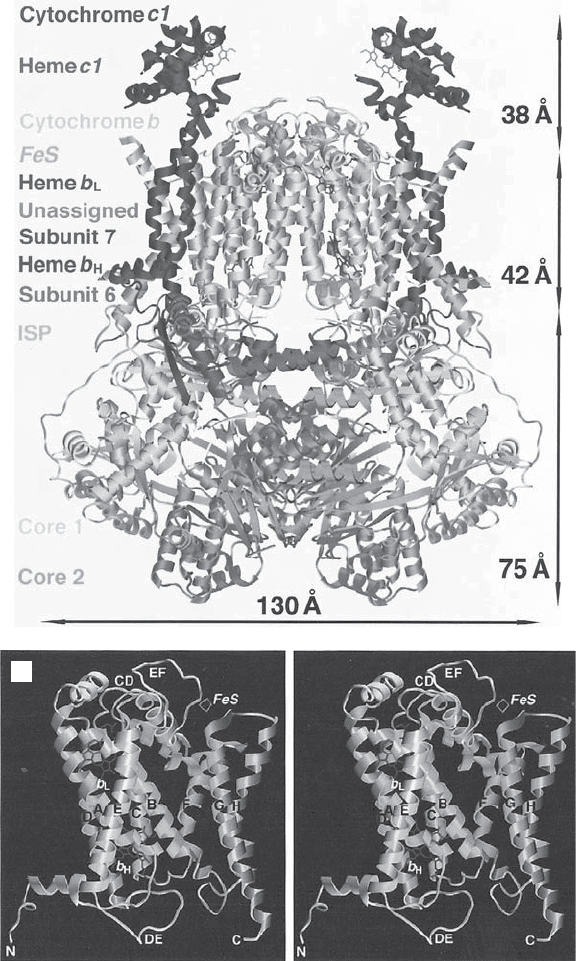
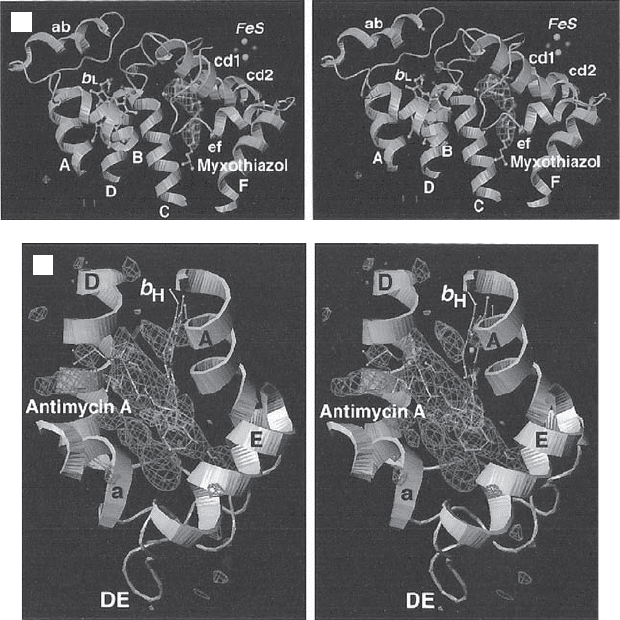
Figure 5.15 (A) Side view of complex III. The matrix side is at the bottom. Cyto-
chrome c1 extends into the intermembrane space. The membrane - spanning helices of
the various subunits are in the region indicated by the middle arrow (42 Å ). (B) Ste-
reoview of the transmembrane helices of a monomeric complex III. (C) Stereoview
from the outside showing the myxothiazol binding pocket. (D) Stereoview from the
matrix side showing the antimycin binding pocket. (From reference 147 with permis-
sion. The fi gure was generously provided by Dr. Yu.) See color plates.
A
B
THE ELECTRON TRANSPORT CHAIN 209
hydroquinoline - N - oxides, inhibitors that interfere with the electron transfer
from heme b
H
to Q or QH
·
−
. At the Q
P
center, electrons from reduced ubiqui-
none are accepted and divided into two pathways: half for recycling, and half
for transfer via the iron – sulfur center and cyt c
1
to cytochrome c. It is located
near the outer face of the inner membrane, and protons are released into the
intermembrane space. A number of different compounds can inhibit at the Q
P
center: 2 - hydroxy - 1,4 - benzoquinone drivatives, stigmatellins, and MOA inhibi-
tors containing the E - b - methoxyacrylate group (MOA - stilbene, myxothiazol).
Some further discussion of the Q cycle can be found in Section 5.4.2 .
A general review of the approaches and methodologies used with these
inhibitors (Figure 5.17 ) has been written by Link et al. (153) . Having estab-
lished the specifi city and effi cacy of each inhibitor in arresting electron trans-
port, additional studies have included light and EPR spectroscopy, the isolation
of resistant mutants of a variety of species, the identifi cation of mutated amino
acid side chains in specifi c peptides from such mutants, characterization of
D
C
Figure 5.15 (Continued)
210 MITOCHONDRIAL ELECTRON TRANSFER
mutations induced by site - directed mutagenesis (154) , and the synthesis of
additional analogues — for example, antimycin A analogues (155) .
5.2.7 Complex IV
Complex IV is generally referred to as cytochrome c oxidase. The overall reac-
tion catalyzed is the following:
44 42cyt c H 4 H O 4 cyt c H H O
2+
(s)in
+
(p)in
+
2
3+
(p)out
+
2
+++→ + +
Molecular oxygen is the terminal electron acceptor, the mobile carrier cyt c is
reoxidized, and four protons are transferred to the intermembrane space.
There are two processes involved. First, four electrons are donated on the IMS
side, while four protons are taken up on the matrix side, resulting in a transfer
of four positive charges across the membrane. Second, an average of one
proton is pumped through the enzyme for each electron transferred to oxygen
(156) .
After three decades of study and analysis, this complex had the distinction
of being the fi rst complex of the ETC to have its high - resolution crystal struc-
ture determined at ∼ 2.8 - Å resolution. Two landmark papers on the bovine
complex IV presented the largest and most complex membrane protein to be
solved at this resolution in 1996 (157, 158) . A monumental achievement, they
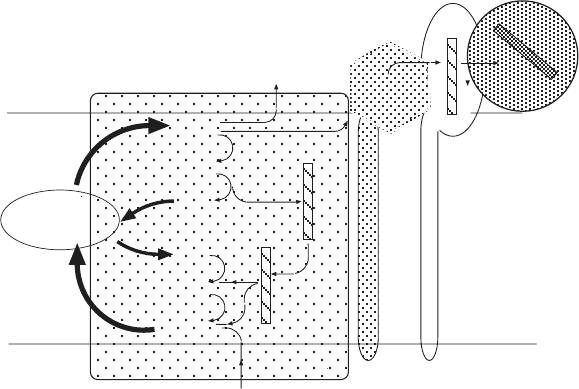
Figure 5.16 The Qcycle and electron fl ow through complex III. QH
2
is oxidized, and
electrons are transferred to the mobile carrier cytochrome c (upper right in the fi gure).
For a detailed explanation see the text. (After Trumpower (152) .)
.
Q
Q
QH
.
2QH
2Q
2Q
2
2
-
-
Q -
Pool
b
L
b
H
2H
+
4H
+
[2Fe-2S]
c
1
Q
center
P
Q
center
N
c
