Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.

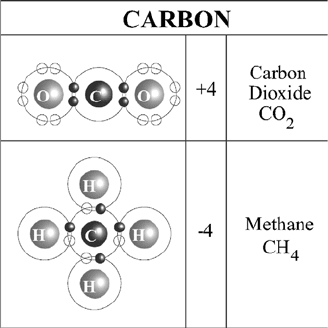
Biogeochemical cycles are not maintained by any single species; rather, they repre-
sent the collective effects of diverse, yet inherently coordinated, microbial consortia
as well as interactions of microbes with higher organisms. In the following sections
we examine the specific eleme nts carbon, nitrogen, sulfur, iron, and manganes e and
their cyclic metabolic transitions, while also discussing oxygen and hydrogen.
In each case, these cycles provide an endless exchange o f oxidation states
through which these atoms are transformed, both oxidatively and reductively (Sec-
tion 5.1.2). The oxidation state assumed by any given atom reflects the level and
mode of electrons that it shares with its neighbors. For example, carbon has a range
of oxidation states from 4toþ4, as dictated by its atomic number (AN), 6. This
means that carbon has six protons (positive charge) in the nucleus (equal to its atomic
number) and six electrons (negative charge) outside the nucleus, balancing the atom’s
charge.
The nucleus of an atom (except most hydrogen nuclei) also contains neutra l parti-
cles, called neutrons. The sum of the protons and neutrons is the atomic weight of the
atom. Carbon usually has six neutrons, so that its usual atomic weight is 12 (six pro-
tons þ six neutrons). However, some forms, or isot opes, of carbon have seven or eight
neutrons, so that they have an atomic weight of 13 or 14. These forms are relatively
rare, so that on average, carbon typically has an atomic weight of 12.011.
The electrons surround an atom’s nucleus in orbital shells. The first s hell for
any element can only have a maximum of two electrons, while the next can have
a maximum of eight. (The third shell ca n have up to 18, but is also stable with
only eight.) Therefore, after subtracting the two electrons allocated to its first
shell, carbon’s second orbital contai ns the four remaining electrons. In this config-
uration (six electrons along with the six protons in the nucle us), elemental carbon’s
net electrical charge (oxidation state) would then be zero. In fact, in elemental
form, the oxidation state of all elements is zero. This oxidation state will change,
though, as the outer shell either gains or loses electrons while bonding with adja-
cent atoms. For example (Figure 13.1), the ca rbon atom found in a methane mole-
cule, CH
4
, effectively pulls four additional negatively charged electrons into its
Figure 13.1 Carbon oxidation state extremes.
388
MICROBIAL TRANSFORMATIONS
outer shell (one from each of the four hydrogens), thereby changing its oxidation state
to 4 (10 negatively charged elect rons versus six positively charged protons). Conver-
sely, the carbon found in carbon dioxide (CO
2
) essentially lends all four of its outer
shell electrons to a pair of surrounding oxygen atoms, thereby shifting its oxidation
state to þ4.
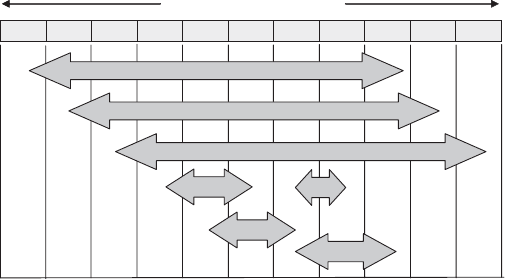
Carbon, nitrogen, and sulfur are the three main elements of biological interest
with a full eight-electron range in oxidation state. As shown in Figure 13.2, the ranges
for these three elements (C, N, and S) are each, respectively, shifted one electron
higher.
Table 13.1 shows the common oxidation states of some of the other elements
of biological importance. Oxygen’s (AN ¼ 8) outer shell has six electrons, pro-
ducing an aggressive tendency to add two additional electrons (thereby filling its
outer shell) rather than releasing any to other adjacent atoms. This characteristic
qualifies oxygen as having a high degree of electronegativity (Section 3.2). As a
result, oxygen’s metabolic range of oxidation states only includes 2(e.g.,H
2
O),
besides 0 (diatomic oxygen, O
2
). Hydrogen (AN ¼1) has p ossible sta tes of 1, 0 ,
and þ1, since its single outer shell has only one electron of the two it would take
to fill it. In living matter it typically donates its electron (thus, oxidation state ¼þ1)
to a mor e electronega tive (e.g., oxygen, carbon, sulfur, nitrogen) receptor. Metabolic
iron (AN ¼26), occurs mainly in the þ2 (ferrous) and þ3 (ferric) oxidation
states, while manganese (AN ¼ 25) is found mainly as the þ2 (manganous) and
þ4 (manganic) forms. Chlorine (AN ¼ 17) as a disinfectant in water (HOCl, hypo-
chlorous acid) has an oxidation state of þ1, but as chloride or in organic compounds
it is 1.
13.1 CARBON
Carbon can be converted from inorganic to organic form by a variety of microorganisms
and by plants. It then is transformed, by anabolic and catabolic processes (Section 5.4), to
+4-4
+3+2
+1
0
-1
-2
-3
+5 +6
Chemical Oxidation States
Carbon
Nitrogen
Sulfur
Iron
Manganese
Hydrogen
Oxygen
Figure 13.2 Redox ranges for some elements of major biochemical importance.
CARBON 389

a tremendous array of biologically important compounds in all living things. Some of
these compounds are released as waste, or eventually the organism itself dies. They are
then broken down, mainly by microorganisms, and eventually the carb on returned to inor-
ganic form.
With carbon’s central importance to all known forms of life, perhaps it is not surprising
that the redox reactions through which these atoms are cycled (Figure 13.3) have such a
high degree of breadth, complexity, and variability. In fact, its role as the dominant
element in living organisms appears to stem from the great diversity of its bonding
options. It typical ly forms fo ur bonds with adjacent at oms. This is mo re than hydrogen
(one bond) or oxygen (two), but not really different from nitrogen (three or four), and
less than phosphorus (five) or sulfur (two to six). However, its great versatility also
TABLE 13.1 Common Oxidation States of Some Biologically Important Elements
Atomic Atomic
Element Symbol Number Weight Common Oxidation States
Aluminum Al 13 26.98 0, þ3
Arsenic As 33 74.92 þ3, þ5
Bromine Br 35 79.90 1
Cadmium Cd 48 112.40 þ2
Calcium Ca 20 40.08 þ2
Carbon C 6 12.01 4, 3, 2, 1, 0, þ1 þ2, þ3 þ4
Chlorine Cl 17 35.45 1, 0, þ1
Chromium Cr 24 52.00 0, þ3, þ6
Cobalt Co 27 58.93 þ2, þ3
Copper Cu 29 63.54 0, þ1, þ2
Fluorine F 9 19.00 1
Hydrogen H 1 1.01 1, 0, þ1
Iodine I 53 126.90 0, 1
Iron Fe 26 55.85 0, þ2, þ3
Lead Pb 82 207.2 0, þ2, þ4
Magnesium Mg 12 24.31 þ2
Manganese Mn 25 54.94 þ2, þ4, þ7
Mercury Hg 80 200.59 0, þ1, þ2
Molybdenum Mo 42 95.94 þ6
Nickel Ni 28 58.69 þ2, þ3
Nitrogen N 7 14.01 3, 1, 0, þ1, þ2, þ3, þ5
Oxygen O 8 16.00 2, 0
Phosphorus P 15 30.97 þ5
Potassium K 19 39.10 þ1
Selenium Se 34 78.96 2, þ4, þ6
Silica Si 14 28.09 þ4
Sodium Na 11 22.99 þ1
Sulfur S 16 32.06 2, 0, þ2, þ4, þ6
Tin Sn 50 118.69 þ2, þ4
Vanadium V 23 50.94 þ5
Zinc Zn 30 65.38 0, þ2
390
MICROBIAL TRANSFORMATIONS
stems from the fact that it can occur in all of the oxidation states from 4toþ4
(Table 13.2).
Organic Carbon What exactly is meant by organic carbon? The term dates to the time
that such compounds were considered to be produced only by organisms. German Frei-
drich Wo
¨
hler is generally credited with the first synt hesis of an organic compound, urea,
in 1928. Now, of course, there are many synthetic organic compounds. So again, what
does organic mean?
Interestingly, most chemistry texts do not provide a real definition. A dictionary may
state that organic chemistry is the study of carbon compounds, but carbon dioxide is not
considered organic (although it is produced by organisms). Another definition that has
been used is ‘‘compounds containing a CC bond.’’ This would exclude carbon dioxide,
as desired, but would also exclude methanol, formic acid, and formaldehyde, which are
generally considered organic. (Formic acid, for example, is produced by ants for their
sting, as noted in the scientific name of the family, Formicidae.) Another definition is
‘‘compounds containing a CH bond.’’ This would exclude carbon dioxide and include
methanol, formic acid, and formaldehyde. It would also include methane, which some
people consider organic, but not carbon tetrachloride. Interestingly, neither definition
would include urea!
Perhaps this explains why most texts avoid the question—there is not a simple answer.
Once again, nature does not easily fit into human classification systems. For our purposes,
we will consider carbon-containing compounds (not the elemental forms graphite and dia-
mond) to be organics, with the exception of carbon dioxide, carbonic acid, bicarbonate,
carbonate, and carbon monoxide. Methane we consider both organic, since it is the carbon
and energy source for methanotrophs (Section 10.5.6), and inorganic, since it is a major
end product of anaerobic mineralization and also the product of carbon dioxide respiration
(Section 13.1.1).
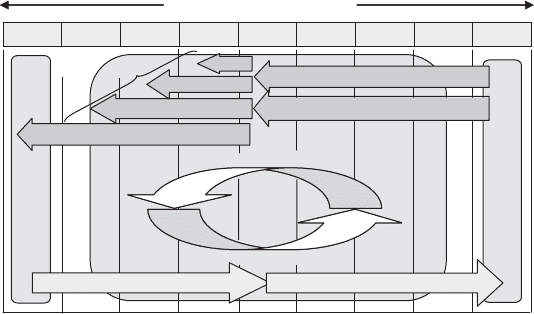
13.1.1 Carbon Reduction
The reductive side of the carbon cycle is what makes continued life on Earth possible.
Without this transformation, and specifically the biological fixation of inorganic carb on
-4
+3+2
+1
0
-1
-2
-3 +4
-4
+3+2
+1
0
-1
-2
-3 +4
-4
+3+2
+1
0
-1
-2
-3 +4
Methane
CH
4
Carbon
Dioxide
CO
2
Photosynthetic carbon fixation
Chemolithoautotrophic
Organic carbon oxidations
Organic carbon oxidations
Carbon Oxidation States
carbon fixation
Methanogenesis
Fixed
Cellular
Carbon
Fixed carbon oxidations
Fixed carbon reductions
Fixed
Cellular
Carbon
Fixed carbon oxidations
Fixed carbon reductions
Anaerobic
Fermentations
Figure 13.3 Biochemical carbon transformations.
CARBON 391
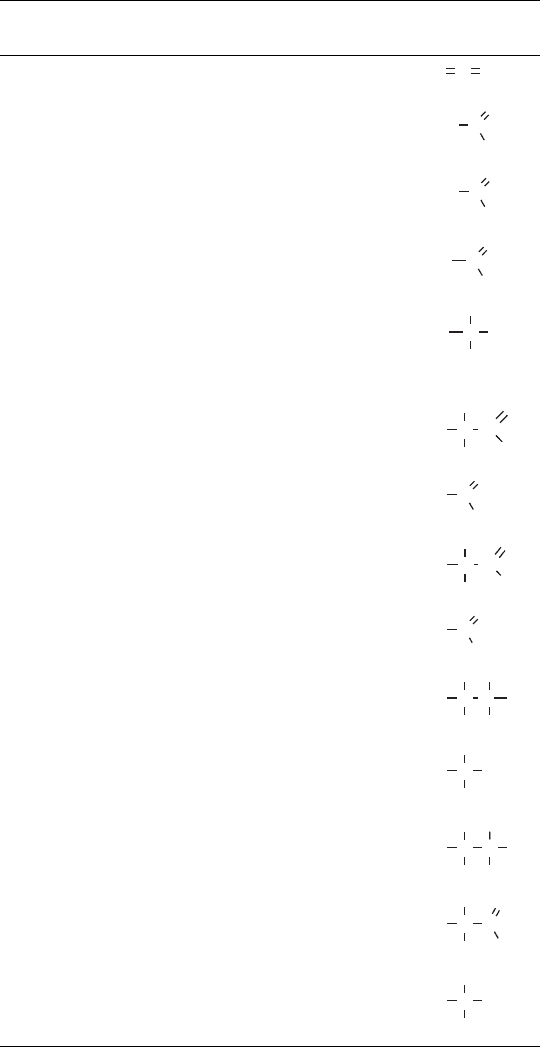
TABLE 13.2 Simple Carbon Compounds and the Oxidation State
of the Carbon
a
Oxidation State
of Carbon Chemical Name Formula Structure
þ4 Carbon dioxide CO
2
OCO
Carbonic acid H
2
CO
3
C
O
OH
HO
Bicarbonate HCO
3
C
O
O
−
HO
Carbonate CO
3
2
C
O
O
−
O
−
Carbon CCl
4
C
Cl
Cl
Cl
Cl
tetrachloride
þ3 Acetic acid
a
CH
3
C
*
OOH
C
H
C*
H
H
O
OH
þ2 Formic acid HCOOH
C
O
OH
H
þ1 Acetaldehyde
a
CH
3
C
*
HO
C
H
C*
H
H
O
H
0 Formaldehyde HCHO
C
O
H
H
1 Ethanol
a
CH
3
C
*
H
2
OH
C
H
H
HC
*
OH
H
H
2 Methanol CH
3
OH
C
H
H
HOH
3 Ethanol
a
C
*
H
3
CH
2
OH
C
H
H
HCOH
H
H
*
Acetic acid
a
C
*
H
3
COOH
C
H
H
HC
*
OH
O
4 Methane CH
4
C
H
H
HH
a
For the two-carbon compound s, the oxidation state given is for the C marked
with an asterisk.
392 MICROBIAL TRANSFORMATIONS
(carbon dioxide, carbonic acid, bicarbonate, and carbonate) to organic forms, life would
be dependent on chemically fixed organic matter, and thus extremely limited.
Organisms that can grow on inorganic carbon, thereby converting it to fixed, organic
form, are referred to as autotrophs (Section 10.3.2). Carbon reduction requires energy, a
portion of which is stored as chemical energy in the fixed carbon that is then available to
other life-forms. The primary source of energy utilized for this purpose, providing most of
the new organic carbon that is metabolically synthesized on our planet, is light. Organisms
able to utilize light as their energy source are phototrophs and are referred to as photo-
synthetic. This includes almost all plants and algae, and some bacteria. Some autotrophic
bacteria and archaea, referred to as chemolithotrophs (Section 10.3.1), are able to use
inorganic chemical energy to fix carbon.
Once carbon has been fixed, it can be further reduced by organisms to generate the
cellular constituents that they need for growth. However, this reduction also requires
energy, either directly or to regenerate intermediates.
Many organisms (including humans) also reduce carbon during a process known as
fermentation. This is an anaerobic energy-yielding process during which organic carbon
is both oxidized and reduced, either in different molecules or in different parts of the same
molecule. Because it is the oxidation that provides energy, fermentation is discussed in
Section 13.1.2.
Photosynthesis As a new day starts and light intensity builds, photosynthetic reactions
metabolically infuse new organic carbon back into the cycle of life. The net production of
glucose and oxygen generated through this process by plants and algae can be summar-
ized as
6CO
2
þ 6H
2
O þ light energy ! C
6
H
12
O
6
þ 6O
2
ð13:1Þ
Cyanobacteria carry out the same reaction, but other photosynthetic bacteria utilize hydro-
gen, hydrogen sulfide, or elemental sulfur instead of water as their electron donor, and
thus do not produce oxygen (see Table 10.4).
Almost all phototrophs use the same reaction sequence, referred to as the Calvin cycle
after its discoverer, to invest energy drawn from sunlight into fixing inorganic carbon
(Section 5.4.5). Exceptions are some green sulfur bacteria, which can run the Krebs
cycle (Section 5.4.3) in reverse to fix carbon, and some green nonsulfur bacteria, which
utilize a unique hydroxypropionate cycle to form the two carbon compound glyoxylate
(CHOCOOH) from two carbon dioxide molecules.
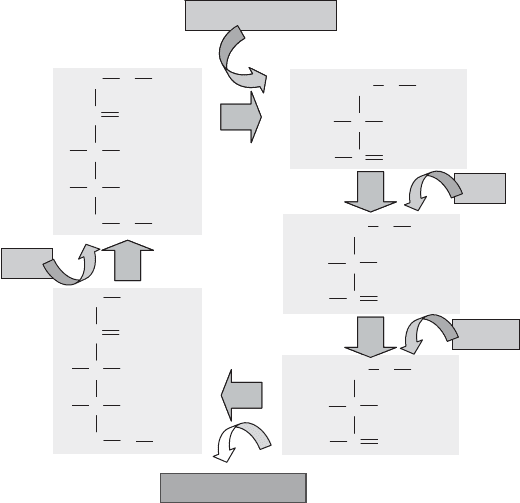
The Calvin cycle itself was summarized in Figure 5.13 and is shown in more
detail in Figure 13.4. It can be considered to consist of three parts. The first step com-
bines the carbon dioxide with a five-carbon sugar containing a phosphate on each end
[ribulose 1,5-diphosphate; also called ribulose 1,5-bisphosphate (RuBP)] . The unstable
product breaks down to produce two molecules of a phosphorylated, three-carbon com-
pound cal led 3-phosphoglycerate. This overall reaction is catalyzed by ribulose-1,5-
bisphosphate carboxylase/oxygenase (RubisCO), which may be the most abundant
enzyme on Earth.
During the secon d part of the cycle, the newly fixed carbon is reduced further,
using additional energy, to glyceraldehyde 3-phosphate. In the final part of the cycle, a
six-carbon sugar is produced, and the ribulose 1,5-diphosphate regenerated in preparation
for subsequent cycles.
CARBON 393
Photosynthetic organisms utilize part of the carbon they fix as an energy source, par-
ticularly in the dark (Section 13.1.2). However, the rest is used to produce the organic
compounds they need for growth. It is this net fixation of carbon that sustains the ecosys-
tems dependent on these primary producers, directly or indirectly, for their carbon and
energy supply.
Of course, there are considerable variations in the rates of organic carbon assimilation
around the world. Tropical rain forests, for example, have some of the highest rates of
carbon uptake on Earth, with annual net fixation of up to 1:5kg C=m
2
of land surface
area. Freshwater wetlands are almost as productive. Forests, grasslands, and cultivated
plants in the middl e latitudes typically assimilate 50% as much carbon, although intensive
agriculture can more than double the production of the rain forest. The open ocean,
deserts, and the Arctic tundra fix much less carb on (typic ally 0.05, 0.1, and 0:2kg=m
2
,
respectively, per year). Sunlight intensity, temperature, nutrient supply, and water avail-
ability are all factors (see Chapters 14 and 15).
The use of sunlight for photosynthesis is not very efficient. On average, only
about 0.1% of the energy available is utilized. Even with intensive agriculture, this
value is unlikely to exceed 1%. Each gram of carbon fixed represents about 10 kJ of
stored energy.
It is estimated that photosynthesis provides our planet with a cumulative carbon assim-
ilation rate of approximately 10
11
metric tons per year. Based on the stoichiometry in
equation (13.1), this means that at the same time, almost 3 10
11
metric tons per year
of oxygen is produced. The long-term maintenance of this essential activity, however,
6-
Carbon Sugar (1)
NADH
ATP
Carbon dioxide (6)*
3-Phospho-
glycerate (12)
1,3-
glycerate (12)
3-
-
(6)
Ribulose 1,5-
(6)
OH
H
H
H
2
C
H
2
C
OC
C
C
OH
OH
OPO
3
H
2
C
H
2
C
C
PO
3
H
2
O
H
OH
H
2
O
3
P
O
C
H
2
C
C
PO
3
H
2
O
HOH
H O
H
2
C
C OH
PO
3
H
2
H
2
C
OC
C
OH
O
O
PO
3
H
2
H
H
Carbon dioxide (6)*
3-Phospho-
glycerate (12)
1,3-
glycerate (12)
3-
1,5-
OH
H
H
H
2
C
H
2
C
OC
C
C
OH
OH
OPO
3
H
2
C
H
2
C
C
PO
3
H
2
O
H
OH
H
2
O
3
P
OC
H
2
C
C
OO
H
OHOH
H
2
O
3
P
OO
C
H
2
C
C
PO
3
H
2
O
HOH
H O
2
C
H
H
2
C
C OH
PO
3
H
2
H
2
C
OC
C
OH
O
O
PO
3
H
2
H
H
H
2
C
O
H
H
ATP
RubisCO
C
H
2
C
C
PO
3
H
2
O
HOH
OC
H
2
C
C
HOHOH
OO
-
O
phosphate
Ribulose 5-
Glyceraldehyde
phosphate (12)
Diphospho-
diphosphate
Figure 13.4 Calvin cycle carbon fixation process.
*
(No.) refers to number of molecules.
394
MICROBIAL TRANSFORMATIONS
is sensitive to disruption. Whether measured in acreage of rain forest lost to deforestation,
wetlands drainage, or accidental tanker spills and resultant oil slicks, humans certainly
have the potential to cause severe environmental stress.
Chemolithoautotrophic Carbon Fixation Although they utilize chemical energy stored
in inorganic molecules rather than light, chemolithoautotrophs also utilize the Calvin
cycle to fix inorganic carbon. Worldwide, a substantial amount of carbon is fixed
through their action. In fact, in som e environments in which sunlight is absent, they
may be the major or only primary producers. Surrounding thermal vents in the deep
ocean, for example, the release of hydrogen sulfide serves as an energy source for sul-
fide-oxidizing bacteria and archaea, which then form the base of an extensive ecosys-
tem, including giant worms and clams. It also appears that the abiotic release of
hydrogen gas in deep deposits of basalt (a volcanic rock) in the Pacific northwestern
region of the United States may support specialized autotrophic bacteria that serve as
the base of a microbial ecosystem.
Nitrifying (see more in Section 13.2.2) and sulfur-oxidizing (Section 13.3.2) bacteria
may be important in waste treatment and aquatic and soil environments, and sulfur-and
iron-oxidizing (Section 13.4.2) bacteria play a major role in acid mine drainage. However,
in these cases it is not the carbon fixation that is of greatest environmental significance. It
recently has been suggested, however, that in drinking water systems using chloramina-
tion (an ammonium/chlorine combination) for disinfection, the nitrification that some-
times occurs may actually contribute materially to the otherwise low organic carbon
content of the water.
Methanogenesis Methanogenesis, the production (genesis) of methane, is a form of car-
bon reduction carried out solely by a group of strictly anaerobic archaea referred to col-
lectively as methanogens (Section 10.6.3). One reaction commonly utilized is actually an
anaerobic respiration in which CO
2
[equation (13.2)], or occasionally, CO [equation
(13.3)], is reduced to methane during oxidation of H
2
. Many methanogens can ferment
formic acid [equation (13.4)] or methanol [equation (13.5)], with some of the molecules
being oxidized to CO
2
and others reduced to CH
4
. A few methanogens can similarly
ferment methyl amines or, less commonly, methyl sulfides. Many can also ferment acetic
acid, oxidizing part of the molecule to CO
2
while reducing the other part of same mole-
cule to CH
4
[equation (13.6)]:
4H
2
þ CO
2
! CH
4
þ 2H
2
O ð13:2Þ
3H
2
þ CO ! CH
4
þ H
2
O ð13:3Þ
4CHOOH ! CH
4
þ 3CO
2
þ 2H
2
O ð13:4Þ
4CH
3
OH ! 3CH
4
þ CO
2
þ 2H
2
O ð13:5Þ
CH
3
COOH ! CH
4
þ CO
2
ð13:6Þ
Often under methanogenic conditions, such as in landfills and anaerobic digesters, the net
gas production is almost two-thirds methane and one-third carbon dioxide, with small
amounts of other gases. Additional details about methanogenesis are given in Sections
13.1.1 and 16.2.1.
CARBON 395
13.1.2 Carbon Oxidation
The oxidation of organic carbon compounds provides energy to a cell. Even in photosyn-
thetic organisms, it is the oxidation of the fixed carbon produced (with the energy from
sunlight) that provides the cells with most of the energy they use.
The two overall types of energy-yielding metabolism are respiration (Section 5.4.3)
and fermentation (Section 5.4.2). In respiration, an inorganic molecule acts as the term-
inal electron acceptor, whereas in fermentation, an organic molecule serves this function.
Fermentation is an anaerobic process, whereas there are both aerobic and anaerobic forms
of respiration.
Respiration Respiration provides more energy to the cell then fermentation, so is
favored whenever it is possible. Aerobic respiration, in which molecular oxygen (O
2
)
is the terminal electron acceptor, provides the most energy; thus, aerobic organisms are
likely to dominate wherever oxygen is available.
Since our atmosphere is 21% oxygen, it might be expected that oxygen is readily avail-
able except in sealed systems. However, oxygen is poorly soluble in water, with only 9.1
mg/L of dissolve d oxygen (DO) present at saturation (equilibrium with the atmosphere)
at 20
C. Also, oxygen diffusion through pores in solid matrices (such as soil and compost-
ing material) occurs by a factor of 4 orders of magnitude more slowly in water than in air.
Thus, in aqueous systems (such as wastewater, liquid sludges, surface waters, river and
lake sediments , groundwater, and flooded soils) the oxidation of even small amounts of
organic material can quic kly deplete DO, leading to anaerobic conditions.
In the absence of oxygen, many bacteria can carry out respiration using nitrate as the
terminal electron acceptor (Section 5.4.3). This denitrification (Section 13.2.1) provides
about 85% of the energy available aerobic ally from oxidation of the same organic com-
pound with oxygen. The nitrate is reduced first to nitrite, then eventually to nitrogen gas.
Thus, nitrite can also be utilized, but yields still less energy. Den itrifying bacteria include
a wide diversity of aerobic bacteria that can utilize nitrate and nitrite as alternative elec-
tron acceptors when oxygen is absent. The process itself is discussed in more detail as part
of the nitrogen cycle (Section 13.2).
Ferric iron can also be utilized for respiration by some aerobic bacteria, providing a
similar amount of energy to nitrite. Some other metals may also be utilized by faculta-
tively or strictly anaerobic bacteria.
In the absence of oxygen, nitrate, nitrite, and ferri c iron, some anaerobic bacteria and
archaea can utilize sulfate as an electron acceptor, producing hydrogen sulfide. This is
particularly important in salt marshes and other systems where sulfate is abundant (Sec-
tion 13.3). Once the sulfate is also depleted, methanogens can utilize CO
2
as an electron
acceptor, producing methane [equation (13.2)].
Under anaerobic conditions, particular bacteria, especially some sulfate reducers , may
be able to utilize other ‘‘unusual’’ electron acceptors. Examples include the use of a num-
ber of chlorinated organic compounds, including important environmental contaminants
such as polychlorinated biphenyls (PCBs), trichloroethylene (TCE), tetrachloroethylene
[commonly called perchloroethylene (PCE)], and vinyl chloride, during which a hydrogen
is substituted for a chlorine atom. This represe nts both a chemical reduction and a dechlo-
rination of the organic molecule and results in the release of a chloride ion (Cl
). A simi-
lar reaction may occur with brominated and perhaps other halogenated compounds. Thus,
the more general term reductive dehalogenation is often used to describe this process.
396 MICROBIAL TRANSFORMATIONS
Fermentation Although it is usually bacteria, archaea, some fungi, and a few protozoa
that are thought of as being anaerobic, higher organisms also carry out fermentations.
During vigorous exercise, for example, not enough oxygen can be delivered to the mus-
cles, leading to buildup of the fermentation product lactic acid. This is wha t makes your
arms and legs feel heavy and tired. Gradually, as you recover, the lactic acid is transported
to the liver and converted there for other uses.
Because an organic compound serves as the electron acceptor in fermentation, some
of the organic material is reduced while oxidizing other organic molecules. In the
examples above for methanogenesis, one reaction involves four molecules of formic
acid [equation (13.4)], one of which is reduced to CH
4
while the other three are oxi-
dized to CO
2
. For the fermentation of the more reduced compound methanol [equation
(13.5)], the ratio of these two products is reversed. With acetic acid [equation (13.6)],
on the other hand, of the two carbons of a single molecule, one is oxidized while the
other is reduced.
13.1.3 Carbon in Environmental Engineering and Science
It traditionally has been the organic carbon (along with pathogens) that was of the greatest
concern in water pollution (Section 15.2.7), leading to the construction of wastewater
treatment plants (Chapter 16) that focus on its removal. Management of wastewater treat-
ment sludges often has stabilization of the organic material as a major objective. (Stabi-
lization involves conversion of readily degradable materials to those that change only
slowly; see later in this subsection). Municipal solid waste management also must stabi-
lize the organic material (e.g., by incineration or composting), or else d eal with the con-
sequences (e.g., attraction of vermin, settling, and leachate and gas production during
landfilling). Similarly, with soil and groundwater contamination, it is often organic carbon
that is the target of remediation. Undesirable tastes and odors in drinking water, and the
formation of cancer-causing compounds during disinfection, are traceable to organic com-
pounds present in the water supply. Even air pollution control may involve organics, such
as volatile organic com pounds (VOCs) and soot (which includes organic particles), as
important contaminants. Most individual toxic compounds of concern in water, soil,
and air are also organics.
Thus, much of environmental engineering and science is directed at control of organic
carbon or an understanding o f its fate and effects in the environment. In particular cases
the emphasis may be on a single compound, a particular class of compounds (such as pet-
roleum hydrocarbons or chlorinated solvents), some broad fraction (such as oil and
grease, or oxygen demanding biodegradable compounds), or the total organics. One con-
cern might be rapid biodegradation, leading to depletion of oxygen, while another mate-
rial might pose a hazard because it is very resistant to degradation. Slow-to-degrade
compounds may be bioaccumulated (Section 18.7.2) in organisms, perhaps leading to
toxic effects even if they are present only at low concentrations in the environment.
Other slowly degrading compounds, such as many plastics, may pose aesthetic problems,
or perhaps injure wildlife that eat (discarded plastic bags that are mistaken for jellyfish) or
become entangled (abandoned fishing nets or six-pack rings) in them.
In some water bodies, contamination with excess levels of nitrogen and/or phosphorus
may lead to excessive growth of aquatic plants, algae, or cyanobacteria (eutrophication;
Section 15.2.6). In this case organic carbon is not added directly, but instead, becomes
problematic after it is formed through photosynthesis.
CARBON 397
