Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.

On the other hand, for heterotrophic organisms, it is typically the availability of
organic material that limits growth. This is particularly true for water and soil microor-
ganisms, where survival is often dependent on an ability to subsist on very low levels of
organic substrates, or to grow quickly when high concentrations of substrate suddenly
become available (e.g., through death of a plant or animal, or depositing of animal
waste products), followed by persistence until the next such event.
Thus, it is clear that the carbon cycle is of major importance in environmental engi-
neering and science, and the next two subsections deal with measurement of organics and
their biodegradation. However, it is also worth noting the potentially fragile nature of this
essential cycle. This was demonstrated on a small scale with the Biosphere II facility in
Oracle, Arizona (Section 14.2.2). That attempt to build a closed ecosystem including
humans was unable to keep the carbon cycle in balance, producing too much CO
2
and
too little O
2
. On a broader scale, global warming, largely as a result of increased atmo-
spheric CO
2
levels, represents another example of a carbon cycle that is not in balance,
with pote ntially major effects. In considering such global change, it is perhaps instructive
to keep in mind that Earth’s atmosphere was not always as we know it today. In fact, the
low CO
2
(380 ppm) and high O
2
(21%) that we consider ‘‘normal’’ is a result of a pre-
vious ‘‘imbalance’’ brought about by the advent of oxygen-producing photo synthesis
(Section 10.1).
An important lesson to be learned from these observations is the crucial balance that
comes into play with the life-sustaining biogeochemical cycling of an element such as
carbon. Compared to our planet, the scale of Biosphere II was so small that it was very
sensitive to an imbalance, and problems manifested themselves in a correspondingly short
period of time. The biogeochemical cycles of our planet, on the other hand, were estab-
lished over a far longer period (geologic time). None theless, the success of the ecosystems
that sustain us depends on a harmony in nature, one that has finite (if unknown) limits to
the levels of huma n influence that it can tolerate.
Quantification of Organic Carbon Because organic carbon is often the contaminant of
greatest concern in water pollution and waste treatment, a number of approaches to mea-
suring it have been developed. In some cases, the concentration of individual constituents
must be known, especially for toxic substances. In these instances, sophisticated instru-
mental techniques such as gas chromatography (GC) or high-performance liquid chroma-
tography (HPLC) may be used to separate and quantitate the com pounds of concern,
followed by mass spectroscopy to identify them with a high degree of certainty.
Another reason to know the amount of a compound present is to estimate the potential
oxygen demand it might exert, for example, in a stream or during wastewater treatment.
For a pure compound the theoretical oxygen demand (ThOD) can be calculated based on
the stoichiometry of its complete oxidation.
Example 13.1 What is the ThOD of glucose, C
6
H
12
O
6
?
Answer
C
6
H
12
O
6
þ 6O
2
! 6CO
2
þ 6H
2
O
Thus, 6 mol of oxygen is needed to oxidize each mole of glucose completely. Since 1 mol
of oxygen is 32 g, 6 32 ¼ 192 g of oxygen is needed for 180 g (1 mol) of gluc ose, or
1.067 g of O
2
per gram of glucose.
398 MICROBIAL TRANSFORMATIONS
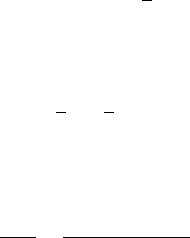
More generally, for a hydrocarbon or carbohydrate, the equation can be written as
C
a
H
b
O
c
þ nO
2
! aCO
2
þ
b
2
H
2
O ð13:7Þ
where
n ¼ a þ
1
4
b
1
2
c ð13:8Þ
Hence, with MW representing the molecular weight of the compound,
ThOD ¼
32n
MW
¼
32a þ 8b 16c
12a þ b þ16c
To calculate the ThOD of an organic compound containing other elements (besides C,
H, and O), the final form of the elements after the reaction must be known. P can be con-
sidered to be in the form of phosphate and chlorine as chloride (Cl
). However, nitrogen
might end up as ammonia or nitrate, and sulfur as sulfide or sulfate. Often, it will be
assumed that the products are in the reduced form (NH
3
and H
2
S) so that the carbonac-
eous oxygen demand is calculated; any additional oxygen demand for oxidation of the
inorganics can then be determined separately.
For mixed contaminants, such as in municipal wastewater or sludge, it is often neces-
sary or sufficient to have an estimate of the total amount of organic matter present, or
some fraction thereof, rather than trying to quantify each of the individual components
separately. One of the older of such aggregate measures, still commonly used for sludges,
is volatile solids (VS). In this test a sample is dried and its mass found to determine the
total solids (TS). It is then ignited at around 500
C, which destroys the organic material.
(Note that this is also one weakness of the method, in that some inorganics also may vola-
tilize, leading to overestimation of the organic content.) The VS is the difference in mass
between the TS and the residual ash. Rather than use the total solids, the sample also can
be filtered first and the test performed on the suspended solids (SS) trapped on the filter,
yielding the volatile suspended solids (VSS). Organisms and their associated materials
and waste products, includi ng wastewater treatment plant sludges, often have a volatile
fraction of around 85%.
This approach is also used to estimate the carbon content of soils. The volatile fraction
is determined, and then multiplied by a factor, often around 0.55, since this is the typical
fraction of the soil organic mater ial that actually consists of carbon.
Total organic carbon (TOC) is an instrumental analysis method that gives a more
selective measure of organic matter. Using combustion or chemical oxidation, the organic
matter in a sample is converted to CO
2
, which is then measured using infrared spectro-
metry. Correction must be made for the inorganic carbon present, either by acidification
(to turn it all to CO
2
) and purging (bubbling with nitrogen gas) to remove it before ana-
lysis (note that this also strips out volatile organic compounds, so that they are missed in
the analysis), or by also measuring the CO
2
present before oxidation (inorganic forms)
and subtracting it from the value obtained after oxidation (which includes both the organic
and inorganic sources).
The TOC of pure compounds can also be calculated from the chemical formula.
CARBON 399

Example 13.2 What is the TOC of glucose, C
6
H
12
O
6
?
Answer There are 6 mol of C per mole of glucose.
6 12
6 12 þ12 1 þ 6 16
¼
72 g C=mol glucose
180 g glucose=mol glucose
¼ 0:40 g TOC=g glucose
For complex mixtures of unknown composition, a ThOD cannot be calculated. How-
ever, the chemica l oxygen demand (COD) laboratory test empirically estimates this
value. It utilizes heat and strong oxidizing agents to convert most organics to CO
2
,
then estimates the amo unt of oxygen that would have been necessary to carry out the
same reaction. One limitation of the method is that despite the very vigorous conditions,
a few organic materials are not oxidized completely.
However, the most common method for estimating the strength of the oxygen-demand-
ing organic material in wastewaters—as required by many regulations—is the biochem-
ical oxygen demand (BOD). This test determines empirically, under standard conditions,
the amount of oxygen utilized during microbial oxidation of the organics present in the
sample. Thus, it measures only the biodegradable portion of the total oxygen demand.
However, this is often the fraction of interest in determining the impact of a discharge
on a receiving water, or the amount of aeration capacity needed for an aerobic treatme nt
process, since only the biodegradable organics are likely to be oxidized under such con-
ditions.
Like many of the tests described above, the BOD is operationally defined; that is, the
BOD is the number that results from conducting the test. Thus, it is important to use a
standard protocol, including a specialized bottle (Figure 13.5), so that results will be
reproducible. The sample is first oxygenated to bring the DO up to near saturation
(9.1 mg/L), the DO is measured, and the bottle is then sealed. After incubation in the
dark (to prevent photosynthetic production of oxygen) at 20
C, usually for 5 days, the
DO is measured again. The difference represents the oxygen utilized biochemically.
The BOD of domestic sewage is typically around 200 mg/L, yet the initially aerated
sample can hold no more than about 9 mg/L of DO. This means that substantial dilution is
required (w ith a specified standard buffer), and the final oxygen depletion in the bottle is
than multiplied by the dilution factor. To ensure that sufficient microorganisms are present
initially, some samples are seeded (inoculated) with a small amount of wastewater treat-
ment plant effluent or settled sewage, and the oxygen demand of this seed must be cor-
rected for in the final BOD calculation.
Figure 13.6 shows a typical plot of the BOD exerted versus time for a sewage
sample. The first surge of oxygen demand r esults from oxidation of organic materials,
while the se cond, of ten st arting at about day 7, is a result of the oxidation of ammo-
nia (nitrification; see Section 13.3.2). The standard BOD test is run for 5 days (BOD
5
),
so that usually it includes much of the carbonaceous oxygen demand (C-BOD),
but little of the nitrogenous demand (N-BOD or NOD). However, this makes the
procedure difficult to use for short-term decision-making purposes, as the results
will not be known for at least 5 days. (This choice is also unfortunate for the practical
reason that tests started on a Tuesday must be finished on a Sunday!) In some cases
the N-BOD begins to be exerted much earlier, giving a BOD
5
value that overestimates
the C-BOD. If desired, the N-BOD can be suppressed by adding to the BOD bottle
a compound that acts as a specific inhibitor of nitrific ation, preventing exertion of
the N -BOD.
400 MICROBIAL TRANSFORMATIONS

Figure 13.5 BOD bottle.
0
100
200
300
400
0 5 10 15 20
BOD Exerted (mg/L)
C-BOD
Total BOD
Time (days)
N-BOD
Figure 13.6 BOD exerted vs. time.
CARBON 401
The ultimate BOD (BOD
u
Þ is the carbonaceous BOD that results from prolonged
incubation, usually taken as 20 days. For domestic sewage, this value is often about
70% of the COD. This is because even for highly biodegradable compounds, some of
the substrate is converted to cellular material, not respired for energy. Since the BOD
5
is often about 70% of the BOD
u
, the standard BOD
5
test typically gives a value that is
about 50% of the COD. However, for various compounds and mixtures, this ratio can vary
from 0 (nonbiodegradable under the BOD test conditions) to more than 1.0 (biodegrad-
able, but not chemically oxidized in the COD test). Table 13.3 compares BOD, COD, and
TOC, showing their relative values for domestic sewage and some of the advantages and
disadvantages of each test.
Because the BOD procedure relies on oxidation by microorganisms, it is inherently
prone to variability despite efforts at standardization. Even slight variations in inoculation
or other test condi tions can lead to change s in oxidation rate and hence different oxygen
depletions at the close of the test period. Despite its weaknes ses, however, the BOD is still
highly useful and widely used.
ThOD, COD, and BOD all give a good indication of the amount of energy available
from oxidation of a compound or mixture. This is because the amount of oxygen con-
sumed in an oxidation is linked very closely to the amount of energy released during
that reaction [typically, 14,000 J/g O
2
; see the discussion of equation (16.5) in Section
16.2.3].
Biodegradation Biodegradation can be defined as the transformation of a compound
through biological activity. It was first included in some dictionaries in the 1960s. Degra-
dation is a broader term that would also include transformation as a result of other che-
mical or even physical changes. This might include photolysis (a chemical reaction in
which light provides the activation energy), for example, or abiotic catalysis at the surface
of a clay mineral.
It is interesting to note that some of the early tests of biodegradability were for plastics,
and degradation was considered undesirable. Thus, even changes in color or opacity were
considered degradation and might lead to rejection of a new polymer. On the other hand,
some plastic bags now labeled ‘‘biodegradable’’ degrade only very slowly, and thus are
TABLE 13.3 Comparison of BOD, COD, and TOC Tests
BOD
5
COD TOC
Typical value for domestic
sewage (mg/L)
200 400 150
Time for analysis 5 days 3 hours 10 minutes
Instrumentation cost Very low/low
a
Low High
Chemical cost Medium/low
a
High Low
Positive interferences
b
Ammonia, sulfide,
nitrite, ferrous iron
Chloride, sulfide,
nitrite, ferrous iron
Carbonates,
bicarbonates
Negative interferences
b
Toxics, photosynthesis Volatile organics,
pyridines
c
a
Depends on whether using chemical analysis or a probe for measuring dissolved oxygen.
b
Corrective measures are possible for most interferences. Uncorrected positive interferences lead to
overestimation; negative ones lead to underestimation.
c
Using some TOC methods, not all organic carbon is reacted and measured.
402 MICROBIAL TRANSFORMATIONS
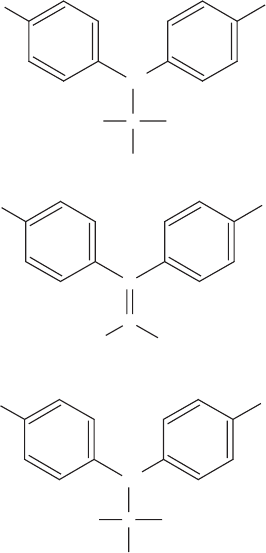
inappropriate for use in composting operations. Similarly, the pesticide DDT was found to
disappear fairly rapidly under som e conditions; unfortunately, it was simply converted to
DDD or DDE, which were at least as problematic as the parent compound (Figure 13.7).
Thus, this biotransformation (Section 18.5) did not represent substantial biodegradation of
the compound.
It is often desirable to be more specific when discussing biodegradation, and to con-
sider both the rate and the extent of the transformation. Mine ralization, or ultimate bio-
degradation, is the complete conversion of an organic molecule to inorganic products
(mainly CO
2
and H
2
O under aerobic conditions, CO
2
and CH
4
or H
2
S under anaerobic
ones). Readily biodegradable organics can be rapidly mineralized. Recalcitrant com-
pounds (also called refractory), on the other hand, are difficult to degrade, and are miner-
alized slowly if at all. However, biodegradability refers to an inherent property of the
material; whether it actually will be biodegraded depends on the environment in which
it is present. For example, readable newspapers and intact pieces of fruit buried for dec-
ades have been recovered from landfills, particularly under dry conditions. Even more
dramatically (Figure 13.8), the corpses of murder victims have been recovered from
northern bogs (with cold, acid, and anaerobic conditions) after 2000 years with much
of their clothing, skin, hair, and even stomach contents intact!
In general, conditions that promote growth are likely to speed biodegradation. Thus,
warm, moist conditions oft en lead to higher rates than cold, dry ones. Also, most—but
ClCl
Cl
CH
C
Cl
Cl
ClCl
Cl
C
C
Cl
ClCl
Cl
CH
C
H
Cl
DDT
DDE
DDD
Figure 13.7 Structures of DDT (dichlorodiphenyltrichloroethane), DDE (dichlorodiphenyldi-
chloroethene, and DDD (dichlorodiphenyldichloroethane).
CARBON 403

not all—materials are more readily biodegradable under aerobic conditions than under
anaerobic ones.
Most biodegradation occurs as a result of microbial activity in which the compound is
utilized as a carbon and energy source. Typically, complex organics are broken down
enzymatically into simpler molecules, which then enter the cell’s basic biochemical path-
ways, such as glycolysis and the Krebs cycle (Section 5.4.3). Compounds containing n itro-
gen, sulfur, or other essential elements may also be utilized as sources of these nutrients.
All naturally occurring and most human-made organic compounds are biodegradable
under appropriate conditions. However, some xenobiotic (‘‘foreign’’ to biology; i.e., syn-
thetic) compounds are extremely recalcitrant. In fact, many, such as polychlorinated
biphenyls (PCBs) and most plastics, wer e designed to resist degradation. Long hydrocar-
bon chain molecules, such as polyethylene, are slow to degrade because microbial attack
is mainly from the ends. Branching also makes biodegradation difficult, as it can hinder or
prevent the molecule from fitting into the active site of appropriate enzymes. The early
alkyl benzenesulfonate (ABS) detergents, used widely in the 1950s, for example, were
highly branched and thus recalcitrant, leading to dramat ic foaming in wastewater treat-
ment plants and receiving waters (Figure 13.9). This problem was eliminated when the
linear alkyl sulfonate (LAS) detergents were introduced as a replacement.
SO
O
−
O
CH
3
C
CH
3
CH
2
CH
CH
3
CH
2
CH
CH
3
CH
2
CH
3
SO
O
−
O
H
C
H
3
CCH
2
CH
2
CH
2
CH
3
ABS LAS
n
n
More recently, methyl tertiary butyl ether (MTBE) has become a problem in ground-
waters. The histor y of the use of this branched compound represents an interesting lesson
Figure 13.8 Tollund man: photograph of a 2000-year-old body recovered from a Danish bog.
(Photo by Lennart Larsen, National Museum of Denmark.)
404
MICROBIAL TRANSFORMATIONS

in the interrel atedness of environmental problems. For many years, lead was added to
gasoline as an antiknocking or octane-enhancing compound. However, the realization
that this lead was contributing to permanent neurological problems (including decreased
intelligence), especially in urban children, led to its reduction and eventual elimination
from gasoline in the United States as an environmental health measure (although it is
still used in many other countries, with the same unfortunate effects).
CH
3
C
CH
3
CH
3
OCH
3
MTBE
Figure 13.9 Foaming from detergents in the 1950s at (a) a municipal sewage treatment plant and
(b) the Passaic River, in New Jersey. (Photos by Joseph V. Hunter.)
CARBON 405
As a substitute for lead, MTBE was added to gasoline beginning in the mid-1970s at
about a 5% level. Almost 20 years later, when a decision was made to increase the amount
of oxygenate (oxygen-containing organic compounds) in gasoline as a means of reducing
carbon monoxide emiss ions (an air pollution problem ), MTBE was the compound of
choice in much of the country. (MTBE was favored by the oil industry, which produces
it, whereas ethanol, which can be produced from corn, was favored by agricultural inter-
ests.) Since it was already in wide use, little consideration was given to the environmental
impacts of increasing its concentration to the range 10 to 15%. However, with the wide
use of gasoline and the frequency of unintended environmental releases through leaks and
spills, MTBE quickly became a major groundwater contaminant. Compounding the pro-
blem is MTBE’s ready solubility in water and poor sorption to soil, leading to its rapid
migration. A further complication is the relative ineffectiveness of carbon adsorption and
air stripping (volatilization) as treatment technologies. Worst of all, both its branched
structure and its ether bond make MTBE resistant (although not immune) to biological
attack as well.
Molecules that are too large or otherwise not taken up by a cell are first attacked by
extracellular enzymes,orexoenzymes, released by the organisms. (Note: The term
exoenzyme is also used by biochemists to refer to enzymes that attack polymers from
the end rather than in the middle.) Many of these are hydrolases, enzymes that react
by adding or removing water from a molecule (without an oxidation or reduction).
These often are relatively specific, as is the case with many cellulases (attack cellulose),
amylases (starch), lipases (lipids), and proteases (proteins).
However, som e organisms also produc e nonspecific extracellular enzymes capable of
degrading a wide variety of compounds. One such example is the lignin-degrading basi-
diomycete fungus Phanerochaete chrysosporium. Lignin is a complex, nitrogen-contain-
ing polymer. It is produced by woody plants for structural suppor t and to protect cellulose
from degradation. Although it is very common, it is relatively recalcitrant due to its het-
erogeneous, irregularly branched structure. However, under aerobic conditions it can be
attacked by P. chrysosporium and other white rot fungi through an exocellular peroxidase
enzyme system that produces free radicals. These highly reactive substances react to non-
specifically oxidize complex organic molecules (including lignin), releasing smaller sub-
units that are more readily biodegradable.
Extracellular enzymes represent a cost to a cell in terms of energy and materials
released to the environment. The cell benefits if it is able to take up and assimilate (incor-
porate into biomass) or oxidize (for energy) some of the smaller molecules produced by
the enzyme’s activity. (Occasionally, another benefit of exocellular enzymatic activity
may be to decrease the toxicity of a compound.) However, cells that do not release extra-
cellular enzymes also benefit from the presence of these utilizable molecules (or
decreased toxicity), but witho ut paying the cost of the enzyme’s production. Such ‘‘free-
loading’’ organisms are thus in a sense parasitic on the extracellular enzyme-producing
organisms.
Oxidation of many organics involves enzymes (mainly intracellular) known as
oxygenases. Monooxygenases, which add a single oxygen atom to the molecule, are
found in both eukaryotes and prokaryotes. Dioxygenases add both atoms of an elemental
oxygen molecule to the organic and are most common in bacteria. Note that with
oxygenases, oxygen is being used as a reactant, not as an electron acceptor.
Figure 13.10 shows a typical monooxygenase oxidation of a straight-chain hydrocar-
bon, leading in several steps to production of a fatty acid. The fatty acid then can be
406 MICROBIAL TRANSFORMATIONS
mineralized via the standard b-oxidation pathway (Section 5.4.4). Note that the other
atom from the O
2
molecule is reduced to H
2
O.
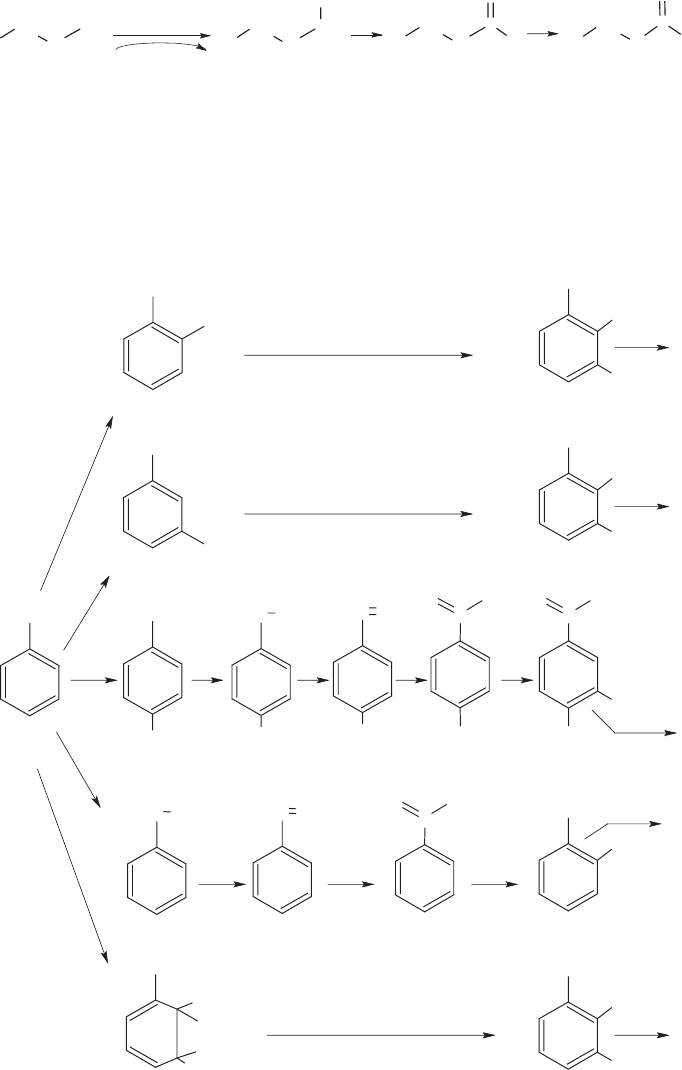
Figure 13.11 shows five bacterial pathways for the initial steps in the biodegradation
of toluene, an aromatic, naturally occurring hydrocarbon that is present in gasoline.
The first four involve a monooxygenase, the fifth a dioxygenase. The first three of the
R
CH
2
CH
2
CH
3
monooxygenase
R
CH
2
CH
2
CH
2
OH
R
CH
2
CH
2
C
O
OH
R
CH
2
CH
2
C
O
H
alkane
alcohol
aldehyde
acid
O
2
H
2
O
Figure 13.10 Monooxygenase activity on a straight-chain hydrocarbon.
C
H
3
C
H
3
O
H
C
H
3
O
H
C
H
3
O
H
C
H
2
O
H
C
H
3
O
H
O
H
H
H
C
H
3
O
H
O
H
C
H
3
O
H
O
H
C
H
3
O
H
O
H
O
H
O
H
C
O
H
O
H
O
H
O
C
H
2
O
H
O
H
C
H
O
O
H
C
H
O
C
O
H
O
H
O
C
O
H
O
toluene
o
-cresol
m
-cresol
p
-cresol
benzyl alcohol
cis
-toluene dihydrodiol
3-methyl catechol
benzaldehyde
benzoic acid catechol
p
-hydroxy
benzyl alcohol
p
-hydroxy
benzaldehyde
p
-hydroxy
benzoic acid
protocatechuate
3-methyl catechol
3-methyl catechol
cleavage
cleavage
cleavage
cleavage
cleavage
Figure 13.11 Five pathways for toluene oxidation.
CARBON 407
