Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.

plant, forming nodules (knoblike growths). Root nodules help the host plant by providing
fixed nitrogen (often, the limiting nutrient for plants), while providing organic substrates
(often, the limiting nutrient for heterotrophs) produced by the plant for the bacteria. The
actinomycete Frankia also produces root nodules, most commonly in nonleguminous
woody plants such as the alder and bayberry.
Other nitrogen-fixing bacteria form associations with plants without nodule formation.
Clostridium and Desulfovibrio, for example, grow in the root zone of eelgrass, a shallow
saltwater plant, and Azotobacter and Azospirillum grow in the rhizosphere of some
grasses, including corn. Nitrogen-fixing cyanobacteria may also form symbiotic relation-
ships with fungi, as in some lichens, as well as with plants such as the water fern Azolla
(used agriculturally to fix nitrogen in rice paddies). This great diversity of organisms and
interactions further demonstrates the great adaptability of life!
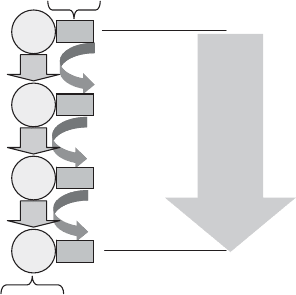
Nitrogen fixation requires an uncommonly high metabolic investment of energy and
reducing power to break molecular nitrogen’s strong trivalent bond (almost twice the
energy of oxygen’s double bond). The conversion of a single molecule of N
2
into 2
mol of reduced nitrogen requires eight protons, eight electrons, and between 16 and 24
ATP (Figure 13.19).
The highly specialized nitrogenase enzyme complex, which includes dinitrogenase,
employs cofactors containing iron and usually molybdenum or, occasionally, vanadium.
Other iron-containing enzymes and ATP are needed to transfer the required electrons to
the nitrogenase system. The reducing power (electrons) must come, in turn, from an
electron-donating substrate (usually organic, although some nitrogen fixers can use sul-
fide, hydrogen, or carbon monoxide). Although the conversion of two nitrogen atoms
from an oxidation state of 0 to 3 requires only six electrons, the process actually requires
eight, producing a molecule of hydrogen gas (H
2
).
There is some variation in the enzymes among different species, but even the nitro-
genases of aerobic bacteria are typicall y highly sensitive to irreversible inactivation by
oxygen. How, then, can these aerobes fix nitrogen? Obviously, they must have means
of preventing the contact of oxygen with the enzymes. One approach, taken by Klebsiella,
which is a facultative anaerobe, is to fix nitrogen only under anaerobic conditions. Other
Dinitrogen tetrahydride
Dinitrogen dihydride
Nitrogen
Ammonia
Dinitrogen
Oxidation
State
S
p
ecies
Nitrogen
Dinitrogenase
N
2
NH
3
N
2
H
4
N
2
H
2
-3
-2
-1
0
Nitrogen Fixation
Dinitrogenase
Dinitrogenase
4H
H
2
2H
2H
Figure 13.19 Nitrogen fixation pathway.
418
MICROBIAL TRANSFORMATIONS
strategies include producing a physical barrier, forming a protective complex with another
protein, and/or maintaining high rates of respiration to deplete oxygen near and within the
cell. Thus, another advantage of being within a root nodule is the more restricted flux of
oxygen. Klebsiella and some other free-living bacteria can also produce a thick slime
layer. Being in the rhizosphere, where root exudates produce a higher concentration of
respirable organic substrates, helps limit oxygen concentration.
Several of the filamentous cyanobacteria fix nitrogen only in a few differentiated cells
(unusual in prokaryotes) with thick walls, called heterocysts (see Section 10.5.4 and
Figure 10.20). The nitrogen is then distributed to the other cells in the filament, which
in return provide organic substrates to the nonphotosynthetic heterocyst. Many cyanobac-
teria may also prefer reduced levels of oxygen (perhaps 10% of saturation).
Most nitrogenases will also cometabolize other triple bonds, such as those of acetylene
(HC
CH) and hydrogen cyanide (HC
N). In fact, acetylene reduction to ethylene
(H
2
C
CH
2
) is often used as an indire ct means of measuring nit rogen-fixing activity.
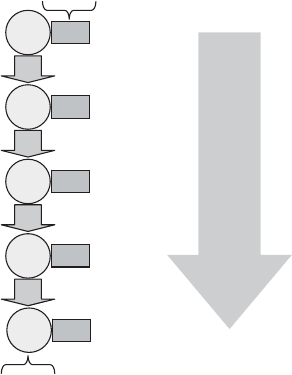
Dissimilatory Nitrate Reduction, Including Denitrification A wide variety of aerobic
prokaryotes are also able to utilize nitrate (NO
3
) as a terminal electron acceptor in the
absence of oxygen. In many case s, including with strains of Escherichia coli, Bacillus,
Staphylococcus, Spirillum , some actinomycetes, Aquifex, and some archaea, the product
of this dissimilatory nitrate reduction is nitrite ðNO
2
Þ. A few of these organisms are able
to further reduce the nitrite to ammonia, but this type of ammonification (as opposed to
the mineralization of organic nitrogen) appears to be of only minor importance in most
environments.
However, under similar conditions, anaerobic respiration by a fairly broad array of
otherwise aerobic proka ryotes, such as some Pseudomonas, Thiobacillus denitrificans,
Paracoccus, and a few archaea, results in a sequential conversion (Figure 13.20) of nitrate
Nitric oxide
Nitrogen
Nitrous oxide
Dinitrogen
Oxidation
State
Species
Nitrogen
Nitrate reductase
Nitrate
NO
3
N
2
O
NO
NO
2
+1
+2
+3
+5
Nitrite reductase
Nitric oxide
reductase
N
2
0
Nitrite
Nitrous oxide
reductase
Denitrification
-
-
Figure 13.20 Denitrification pathway.
NITROGEN 419

to nitrite to elemental nitrogen gas (N
2
). The term denitrification is applied to this pro-
cess because N
2
is unusable by most organisms, so that these reactions represent a loss of
available (fixed) nitrogen to an aquatic or soil environment. (Similarly, the release of
small amo unts of the gaseous intermediates NO and N
2
O represent losses of nitrogen
from the system.)
A general reaction for oxidation of a carbohydrate through denitrification can be writ-
ten as
5
4
CH
2
O þ NO
3
!
5
4
CO
2
þ
1
2
N
2
þ
3
4
H
2
O þ OH
ð13:10Þ
If the organic materia l oxidized is methan ol, the reaction can be written instead as
5
6
CH
3
OH þ NO
3
!
5
6
CO
2
þ
1
2
N
2
þ
7
6
H
2
O þ OH
ð13:11Þ
Example 13.5 An anaerobic groundwater contains 100 mg/L of carbohydrate with an
empirical formula of CH
2
O. How much nitrate would have to be added to meet the stoi-
chiometric requirement for complete oxidation of this contaminant?
Answer From equation (13.10), 1.0 mol of nitrate would be needed per 1.25 mol of
CH
2
O, which has an apparent molecular weight of 1ð12 Þþ2ð1Þþ1ð16Þ¼30:
required NO
3
-N ¼ 100 mg CH
2
O=L
1 mol CH
2
O
30 g CH
2
O
1 mol NO
3
1:25 mol CH
2
O
14 g NO
3
1 mol NO
3
¼ 37:3mg=LNO
3
-N
Under anaerobic conditions we often expect unpleasant odors to develop. When nitrate
is present, however, this typically does not happen. Further, the active organisms are
mainly aerobes that have the additional ability to respire using nitrate as an alternative
terminal electron acceptor. Thus, sanitary engineers, and subsequently, environmental
engineers and scientists, have traditi onally referred to conditions in which oxygen is
absent but nitrate is present as anoxic rather than anaer obic. However, please be aware
that most biologists and others outside our field do not make this distinction, using anae-
robic and anoxic as synonyms to refer to any system in which oxygen is absent.
Although most denitrifiers are organotrophs, some are lithotrophs utilizing hydrogen or
sulfide. Recently, autotrophic nitrification (Section 13.2.2) also has been linked to deni-
trification, in a process dubbed anammox (anoxic ammonia oxidation). In oxidizing
ammonia as an energy source, the organisms involved can reduce nitrate to nitrite and
then reduce the nitrite to nitrogen gas:
NH
4
þ
þ NO
2
! N
2
þ 2H
2
O ð13:12Þ
This process does not seem to play a major environmental role in nitrogen cycling. How-
ever, it may be important to the organisms involved in some circumstances, particularly
when aerobic conditions (under which the nitrate is produced) are followed by anoxic
ones. This occurs in soils that are periodically flooded, as well as in some wastewater
treatment plants.
In general, denitrification does not occur in the presence of oxygen. Since approxi-
mately 20% less energy is available to an organism when it is utilizing nitrate, there
420 MICROBIAL TRANSFORMATIONS
has been strong selective pressure for microbes to develop control mechanisms to sup-
press nitrate reduction in the presence of O
2
. However, denitrification can occur in anoxic
microenvironments (microscopically localized environments having different physical
and chemical condi tions) within heterogeneous aerobic systems, such as moist soils
and microbial colonies.
Nitrate Assimilation A wide variety of bacteria, archaea, fungi, algae, and most plants
can utilize nitrate as their source of nitrogen. However, to do this, they must reduce it to an
oxidation state of 3 (the level of ammonia and organic nitrogen). This is referred to as
assimilatory nitrate reduction. While dissimilatory reduction can be linked to energy-
yielding respiration, assimilatory reduction requires energy. As a result, the biomass yield
of organisms growing on nitrat e as their nitrogen source will be slightly lower than when
ammonia is used. Hence, an organism that is able to use both ammonia and nitrate will
utilize ammonia preferentially when it is available. On the other hand, the presence of
oxygen does not inhibit assimilatory reduction.
13.2.2 Nitrogen Oxidation
The oxidation of ammonia to nitrite, and nitrite to nitrate, is known as nitrification. There
is no known biochemical oxidation of nitrogen gas, although there are abiotic mechanisms
(electrochemical, photochemical, thermal). Similarly, there is no known pathway for
direct conversion of ammonia to nitrogen gas (th e reverse of nitrogen fixation).
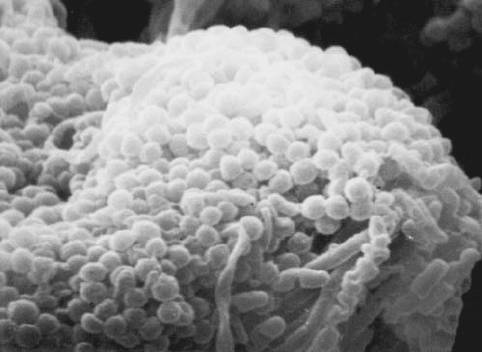
Autotrophic Nitrification Most nitrification results from the activity of aerobic, auto-
trophic chemolithotrophs, referred to as nitrifying bacteria (Figure 13.21). They utilize
reduced nitrogen (ammonia or nitrite) as their energy source, carbon dioxide as their car-
bon source, and oxygen as their electron acceptor.
The overall oxidation really consists of two separate steps, carried out by different bac-
teria (Figure 13.22). Ammonia oxidation involves the transformation of ammoni a to
nitrite, a six-electron transfer (3toþ3), and is sometimes called nitrite formatio n or
Figure 13.21 Clustered nitrifying cocci within activated sludge floc.
NITROGEN 421
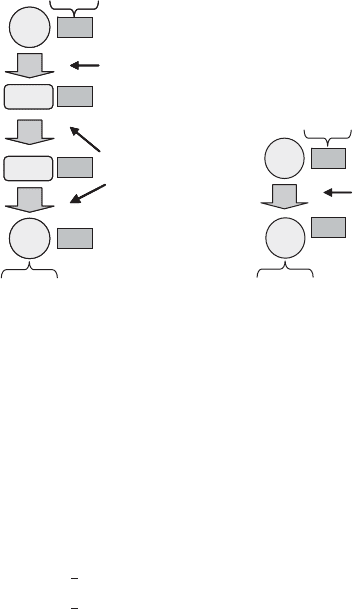
nitritification. In the second step, nitrite oxidation or nitratification, nitrite is converted
to nitrate, a two-electron change (þ3toþ5). Similarly, the bacteria can be referred to as
ammonia oxidizers (or nitritifiers) and nitrite oxidizers (nitratifiers). No known nitri-
fier can oxidize ammonia all the way to nitrate.
Balanced equations for the individual reac tions and their sum can be written as
NH
4
þ
þ
3
2
O
2
! NO
2
þ H
2
O þ 2H
þ
ð13:13Þ
NO
2
þ
1
2
O
2
! NO
3
ð13:14Þ
NH
4
þ
þ 2O
2
! NO
3
þ H
2
O þ 2H
þ
ð13:15Þ
Note that the equations are written with ammonium and nitrite as the reactants, since it
is these ionic forms that are expected to be predominant at neutral pH values. However,
there is some evidence that the forms actually used by the bacteria are nonionized
ammonia (NH
3
) and nitrous acid (HNO
2
). Also note that 2 mol of oxygen is used and
that 2 mol of strong acid is produced from 1 mol of weak acid.
Known organisms carrying out the first (nitritification) step are the proteobacteria
Nitrosomonas (b), Nitrosospira (b), and Nitrosococcus (g). The second (nitratification)
step is performed by the proteoba cteria Nitrobacter (a), Nitrococcus (g), and Nitrospina
(d), and by Nitrospira, a member of the Xenobacteria. Thus, although all of the nitrifiers
were once included in the same family because of their activities, it is now reco gnized that
they are phylogenetically diverse. Also, at one time most nitrifiers were assumed to be
either Nitrosomonas or Nitrobacter. However, it is now recognized, based on genetic tech-
niques, that although these are the most often cultured species, they are not necessarily the
most common or most active in the environment. Hence, nitrifying activity should not be
assigned to these genera unless they are actually identified.
As shown in Figure 13.22, ammonia monooxygenase (AMO) is the enzyme responsi-
ble for catalyzing the first reaction of nitrification, in which ammonia is oxidized to hydro-
xylamine. Hydroxylamine oxidoreductase then produces a transi ent intermed iate
Nitrogen
State
Species
Hydroxylamine
Oxidation
Nitroxyl
Nitrogen
Nitrite
Ammonia
NH
3
NO
2
+3
+1
-1
-3
NH
2
OH
NOH
Monooxygenase
NH
3
NO
2
+3+3
+1+1
-3
−3
−1
NH
2
OHNH
2
OH
NOHNOH
(AMO)
Ammonia
oxidoreductase
Hydroxylamine
(HAO)
Nitrogen
State
Oxidation
Species
Nitrogen
Nitrate
Nitrite
NO
2
−
NO
3
−
+5
+3
Nitrite
Oxidase
−
(a)(b)
Figure 13.22 Steps in autotrophic nitrification: ðaÞ ammonia oxidation; ðbÞ nitrite oxidation.
422
MICROBIAL TRANSFORMATIONS
(nitroxyl) while forming nitrite . Luckily, hydroxylamine itself rarely accumulates, as it is
a potential mutagen. Nitrite oxidation is catalyzed by nitrite oxidase.
AMO shows some similarity to methane monooxygenase (MMO), the enzyme used by
methanotrophs (Section 10.5.6) to oxidize methane. In fact, many ammonium oxidizers
and methanotrophs can aerobically cometabolize each other’s substrate as well as a num-
ber of other compounds, including trichloroethylene.
Compared to oxidation of organic compounds, relatively little energy is available to
nitrifiers. It takes about 35 mol of NH
4
þ
for ammonia oxidizers to fix 1 mol of CO
2
.
Nitrite oxidizers require even more substrate, about 100 mol of NO
2
per mole of CO
2
fixed. Since the nitrite typically comes from the ammonia (100 mol of ammoni a produces
100 mol of nitrite), this means that ammonia oxidizers are usually more abundant than
nitrite oxidizers. Also, cell yields based on their energy source are much lower for nitri-
fiers than for most heterotrophs, often in the range 5 to 20% rather than 50 to 60%.
The fact that nitrifiers appear to belo ng to only a few genera suggests that there may be
more limitations on their activity than would be true if they were more diverse. In fact,
compared to many heterotrophs, nitrifiers are slow growing. Under ideal conditions, mini-
mum doubling times are around 8 hours. Also, there are no known thermophilic nitrifiers,
so that autotrophic nitrification does not occur in systems with temperatures above
42
C. Furthermor e, since there are no known sporeformers, elevated temperatures actu-
ally kill the nitrifiers; this means that activity is slow to return to a system (being depen-
dent on reinvasion or reinoculation) even once elevated temperatures decrease. Optimum
temperature is usually around 28 to 30
C, and activity is usually minimal at temperatures
below 10
C.
Similarly, pH can be limiting. Optimum pH values are around 7.5 to 8, with almost no
activity below pH 6. This may in part be because of the unavailability of nonionized
ammonia at low pH values. Also, nitrite is more toxic at low pH, where it is present as
nonionized nitrous acid. At high pH, toxicity from ammonia becomes a problem. On the
other hand, although they are aerobic [with the exception of the anammox process, equa-
tion (13.12)], nitrifiers can survive for prolonged periods under anaerobic conditions and
are effective at utilizing low concentrations of oxygen. In other words, they have a low K
s
(half-saturation coefficient, Section 11.7.2) value for dissolved oxygen, typically below
0.5 mg/L. Similarly, they requi re only small amounts of their energy sources to approach
maximum activity rates (K
s
values for ammonia- or nitrite-N of 1 mg/L or less).
A close relationship between ammonia and nitrite oxidizers can be expected, since the
product of the first group is the substrate for the second. Thus, the two groups are typically
located in close physical association. Ammonia oxidizers are usually more abundant,
since about three times as much energy is available from ammonia oxidation as nitrite
oxidation. Typically, only traces of nitrite are seen in the environment. Thus, nitrification
is often treated as though it was a single step, involving one group of bacteria. However,
accumulations of nitrite can occur under transient conditions, particularly since the nitrite
oxidizers appear to be a little more sensitive to low pH and high concentrations of ammo-
nia and nitrite.
Heterotrophic Nitrification Some heterotrophic bacteria and fungi are able to oxidize
nitrite to nitrate, and/or occasionally, ammonia to nitrite. This does not appear to
provide any benefit to the organism and hence is considered a type of cometabolism.
Perhaps in some cases this represents as similatory nit rate reduct ion enzymes working
in reverse.
NITROGEN 423

It is generally believed that heterotrophic nitrification plays only a small role in nitro-
gen cycling. However, in some environments, such as acid soils, in which autotrophic
nitrification is severely inhibited, it may have a local effect.
13.2.3 Nitrogen in Environmental Engineering and Science
If nitrogen is second only to carbon in terms of the complexity of its cycling, it probably
also is second only to carbon in terms of its importance in environmental engineering and
science. The range of concerns includes wastewater and potable water treatment, surface
and groundwater contamination, agriculture, sludge and solid waste management, human
and environmental toxicity, and bioremediation. What is more, these problems may be
interdependent. For example, in agriculture, nitroge n is often the limiting nutrient for
many important crops (e.g., wheat, corn, cotton), so that loss of fixed nitrogen is a
major concern. However, overapplication of chemical fertilizers, manures, or wastewater
treatment sludges, which typically are high in nitrogen, can lead to groundwater (from
leaching) and/or surface water (from runoff) contamination. In fact, nitrate is the leading
groundwater contaminant in the United States, mostly as a result of agricultural
practices.
Oxygen Demand As noted in equation (13.15), complete stoichiometric oxidation of
ammonium requires 2 mol of O
2
per mole of ammonium-N. This translates into
64=14 ¼ 4:57 mg O
2
=mg NH
4
þ
-N oxidized to nitrat e. Because the organisms also reduce
CO
2
and assimilate some N for cell constituents, the actual ratio is a little lower, typicall y
4:33 mgO
2
=mg NH
4
þ
-N (3.22 for ammonium oxidation to nitrite and 1.11 for converting
nitrite to nitrate). This represents the N-BOD (Section 13.1.3).
Consider typical sewage (Table 13.4), with a BOD of 200 mg/L and a reduced nitrogen
content of 30 mg/L (half organic-N and half ammonium-N). During secondary treatment
(biological treatme nt required under federal law; see Chapter 16), the oxygen required to
reduce the BOD to 20 mg/L (90% removal) is 180 mg/L. For the nitrogen, typically the
organic-N is converted to ammonium, and about 5 mg/L is assimilated, leaving 25 mg/L.
If this remaining nitrogen is oxidized, it would require 25 4:33 ¼ 108 mg=L of oxygen,
an increase of 60% of the original carbonaceous demand. Some industrial wastewaters
may have much higher ammonium concentrations (even 1000s of mg/L) and thus repre-
sent even greater N-BODs.
TABLE 13.4 Typical Carbonaceous and Nitrogenous Biochemical Oxygen Demand
of Treated
a
and Untreated Sewage
C-BOD TKN N-BOD
b
Total BOD N-BOD N-BOD
(mg/L) (mg/L) (mg/L) (mg/L) (% of C-BOD) (% of total)
Sewage 200 30 130 330 65 39
Effluent nitrified 20 1 4.3 24 22 18
Effluent unnitrified 20 25 108 128 540 84
a
Assuming secondary treatment with 90% C-BOD removal and assimilation of 5 mg=LNH
þ
4
-N. The remaining
TKN is either oxidized to nitrate (nitrified) or remains unoxidized (unnitrified). Total Kjeldahl nitrogen (TKN)
consists of organic-N (usually quickly ammonified) þ ammonium-N.
b
Based on 4.33 mg O
2
demand/mg NH
4
þ
-N oxidized.
424 MICROBIAL TRANSFORMATIONS
Originally, biological sewage treatment plants produced nitrified effluents, since it was
recognized that the presence of nitrate indicated thorough stabilization of the wastewater.
However, once the BOD test became available, plants realized that they could reduce the
energy costs associated with supplying oxygen (a major portion of the overall cost of
treatment) by providing only carbonaceous removal. Thus, most U.S., plants for many
years were designed and operated so as not to nitrify.
However, this meant that ammonium was being discharged to the receiving water,
where it might also undergo nitrification and exert an N-BOD. Two classic papers in
the 1960s, one examining the Thames estuary in London and the other the Grand River
in Michigan, showed that this did in fact occur in some waters, serious ly depleting DO.
However, in some other receiving waters there was little evidence of nitrification. It now
appears that nitrification can be expected in smaller rivers and streams, where the nitrifiers
grow mainly attached to streambed surfaces (including vegetation), and in estuaries,
where plankton ic nitrifiers have time to grow. In larger rivers, where streambed surfaces
are relatively small compared to the water volume, nitrifiers appear to settle out and to
grow too slowly to oxidize most of the ammonia.
Ammonium may also enter streams from agricultural runoff, stormwater from subur-
ban and urban areas (including erosion from fertilized lawns), and from natural organic
inputs, such as falling leaves and bird droppi ngs. Of course, this nitrogen also can repre-
sent an oxygen demand.
In denitrification, nitrate is used as an alternate electron acceptor to oxygen. Thus
in systems in which carbon oxidation and nitrification take place, denitrification can
result in ‘‘recovery’’ of some of the oxygen utilized for nitrification. For example, in
equation (13.10), 1 mol of nitrate is ‘‘used’’ to oxidize 1.25 mol of carbohydrate. This
nitrate required 2 mol of oxygen when it was produced from ammonia [equation
(13.15)]. If the same amount of carbohydrate were oxidized by oxygen, 1.25 mol of O
2
would be required. Thus, using the nitrate saves using 1.25 mol of O
2
, essentially ‘‘reco-
vering’’ 62.5% (100 1.25/2) of the oxygen that was used to form the nitrate. Some
advanced wastewater treatment plants that nitrify take advantage of this to reduce aeration
costs while removing nitrogen as N
2
.
Toxicity of Nitrogen Compounds If neither the treatment plant nor the receiving water
nitrifies, the ammonia from discharges and runoff wi ll be present in the stream. This can
be a serious problem in that nonionized ammonia can be extremely toxic to aquatic organ-
isms. It is recommended that nonionized ammonia concentrations in freshwater remain
below 0.02 mg/L (20 ppb) to protect the most sensitive water uses (such as trout produc-
tion), or below 0.04 mg/L in most other cases. (These values are an exception to the gen-
eral reporting convention for nitrogen and are based on NH
3
rather than NH
3
-N.) The
amount of nonionized ammonia is strongly dependent on pH (Section 13.2), but at the
slightly alkaline pH values of some streams, these levels can be exceeded. Thus, whether
the stream nitrifies or not, there may be good arguments to require nitrification within the
treatment plant, and it is becoming more common again.
Example 13.6 In Example 13.4, a water sample had 3.0 mg/L total ammonia- þ
ammonium-N. If that sample was taken from a stream below a wastewater treatment
plant, does the stream meet an nonionized NH
3
standard of 0.04 mg/L?
Answer The nonionized NH
3
concentration calculated for that example was
0.047 mg/L NH
3
-N. Thus, the standard is not being met.
NITROGEN 425
Ammonia is also toxic to microorganisms, including nitrifiers, and at high levels can
interfere with treatment. As long as the nitrifiers are active, feed concentrations to a reac-
tor can be high, but a sudden increase in concentration or pH can lead to failure. The
increased concentration of nonionized ammonia slows down nitrifier act ivity, decreasing
ammonia oxidation rates and leading to a rapid buildup in concentration and further
increased toxicity. Typically, well-designed and well-operated reactors can handle up to
500 mg/L (or even higher) influent total ammonia concentrations, depending on pH,
because the concentration within the reactor will remain well below 100 mg/L.
Nitrite is also highly toxic to aquatic organisms. U.S. recommendations at one time
were to maintain concentrations of nitrite-N below 0.06 mg/L, and some European coun-
tries have suggested a 0.02 mg/L limit. Fortunately, nitrite rarely accumulates in the envir-
onment, since it is an intermediate for both nitrification and denitrification. Apparently for
this reason, there is no longer a U.S. nitrite criterion.
Both nitrite and nitrate may be toxic to humans, and drinking water standards have
been set at 1 mg/L for nitrite-N and 10 mg/L for nitrate-N. Upon ingestion, nitrate
may be reduced to nitrite in the digestive tract. Once absorbed into the bloodstream, nitrite
binds (preferentially over oxygen) with hemoglobin, leading to a mild to severe asphyxia
known as methemoglobinemia. This is of particular concern for bottle-fed infants, in
whom it is called ‘‘blue baby disease’’, as it can be fatal. Nitrite can also react with certain
amines released during degradation of amino acids, forming nitrosamines, some of which
are potent carcinogens.
Nutrient Enrichment Usually, either nitrogen or phosphorus is the nutrient that limits
the amount of photosynthetic growth that can occur in a water body. Typically, while
phosphorus is limiting in lakes and reservoirs, nitrogen is the limiting nutrient in streams
and coastal waters. Since a variety of organisms can utilize each form, addition of nitro-
gen as either ammonium or nitrate whe re N is limiting can lead to excessive growth of
plants, algae, and/ or cyanobacteria, speeding the process of eutrophication (Section
15.2.6). This is a major reason that some wastewater treatment plants are required to prac-
tice some form of advanced treatment to remove nitrogen, not merel y to nitrify it.
Alkalinity Consumption As can be seen from equation (13.13), each mole of ammo-
nium oxidized to nitrite produces 2 mol of acidity. This uses up 2 mol of the alkalinity
(acid-neutralizing, or pH-buffering, capacity) of the water in which it occurs. If the
water does not initially have enough alkalinity, this will result in a drop in pH. This
can, in turn, inhibit nitrification as the pH drops as low as 5.5, as well as resulting
in other harmful effects in wastewater treatment or perhaps soil. (Ammonia concentra-
tions are usually diluted enough that this level of acidification of a stream will not
occur.) To prevent this, nitrifying t reatment plants may have to a dd supplem ental
alkalinity to raise the pH to more desirable or permitted levels (usually, > 6.5).
Acidity produced by nitrification can also be one of the reasons for adding lime
(usually, it is really crushed limestone, CaCO
3
) to agricultural fields or fertilized
lawns and gardens.
Example 13.7 In a wastewater treatment plant, 20 mg/L of ammonium-nitrogen is
nitrified to nitrate. How much alkalinity is consumed?
Answer From equation (13.13), 2 mol of alkalinity is needed for every mole of
nitrogen oxidized. By convention, alkalinity is expressed as CaCO
3
equivalents, with
426 MICROBIAL TRANSFORMATIONS

2 m ol of acid neutralized per mole (100 g). Thus 1 mol of CaCO
3
is needed for every
mole of ammonium:
Alkalinity consumed
¼ 20 mg=LNH
4
þ
-N
1 mol NH
4
þ
-N
14 g NH
4
þ
-N
1 mol CaCO
3
1 mol NH
4
þ
-N
100 g CaCO
3
1 mol CaCO
3
¼ 20ð7:14 mg CaCO
3
=mg NÞ¼143 mg=LCaCO
3
alkalinity
Denitrification produces 1 mol of alkalinity for each mole of nitrite or nitrate reduced
to nitrogen gas [e.g., equations (13.10) and (13.11)]. Thus half of the alkalinity lost
through nitrification can be recovered by denitrification while removing the nitrogen
(as a gas) from the syst em. This will often eliminate the need for alkalinity addition in
wastewater treatment, but would usually be undesirable in agricul ture, where the nitrogen
loss is typically a conce rn.
Disinfection Nitrite and chlorine rapidly react to form nitrate and chloride, which has no
disinfecting power. For example, in drinking water, where hypochlorous acid is the pre-
dominant active chlorine species;
NO
2
þ HOCl ! NO
3
þ H
þ
þ Cl
ð13:16Þ
Thus, every milligram of NO
2
-N uses up 71 =14 ¼ 5:1 mg of active chlorine (which is
always expressed on the basis of Cl
2
, with molecular weight of 71).
Ammonia also reacts with chlorine, but the products depend on the relative concentra-
tions of the two. At low Cl/N molar ratios ( 1), mainly monochloramine (NH
2
Cl),
forms:
NH
4
þ
þ HOCl ! NH
2
Cl þ H
2
O þ H
þ
ð13:17Þ
As the Cl/N ratio increases, dichloramine (NHCl
2
) forms:
NH
2
Cl þ HOCl ! NHCl
2
þ H
2
O ð13:18Þ
Mono- and dichloramine are referred to as combined chlorine. Although they still repre-
sent active chlorine, they have much less disinfecting power than HOCl (1to2%as
effective). This is one reason that disinfection of was tewater (which usually contains
ammonia) is less effective than disinfection of drinking water (which typically does not).
Finally, at still higher ratios, the chlorine oxidizes the ammonia to produce nitrogen gas
or nitrate, or occasionally, trichloramine (NCl
3
), depending on pH and the specific ratio.
NHCl
2
þ HOCl ! N
2
and=or NO
3
and=or NCl
3
ð13:19Þ
This is referred to as breakpoint chlorination (Figure 13.23), and these products have
no disinfecting power. Thus, depending on the Cl/N ratio, adding more chlorine can actu-
ally result in a decrease in the remaining active chlorine, or chlorine residual. Once all of
the ammonia is converted to nitrogen gas, nitrate, or trichloramine, any additional chlor-
ine addition results in an increase in the chlorine residual.
NITROGEN 427
