Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.

Other In the soil, ammonium is often held at cation-exchange sites of clay minerals.
Nitrification mobilizes this nitrogen, making it easier for plants to absorb if the ammo-
nium in their rhizosphere has been depleted, but also easier for it to leach into the ground-
water. On the other hand, assimilation of ammonium or nitrate by bacteria can make this
nitrogen immobilized and unavailable to plants. Thus, if waste materials with a high car-
bon/nitrogen ratio, (such as some crop residues and some solid wastes) are applied to soil,
the microbial activity on the carbonaceous materials can lead to a depletion of available
soil N for plants. This is referred to as nitrogen robbing.
Some potable waters naturally contain ammonia, and others may add it intentionally.
This results in the formation of chloramines, which although weaker disinfectants, are less
likely to react with organic compounds in the water to produce trihalomethanes and other
potential carcinogens. However, nitrifiers have been found growing as biofilms attached to
the walls of the water distribution pipes in some of these systems, where their relative
resistance to chlorination aids in their survival. Concerns arising from these observations
include the production of nitrite (which inactivates chlorine), organic material (promoting
growth of heterotrophs), and acidity (potentially contributing to corrosion, which might
further increase concentrations of lead in the water).
During anaerobic digestion of sewage sludges, concentrations of ammonium (from
ammonification), magnesium, and phosphate will sometimes become high enough to lead
to the precipitation of magnesium ammonium phosphate, or struvite (MgNH
4
PO
4
).
Heavy deposition of this mineral can clog pipes, coat heat exchangers, and otherwise
interfere with digester operation.
Because of the limited metabolic diversity of nitrifiers, some compounds act as selec-
tive inhibitors, limiting nitrification while having little or no effect on other organisms.
Allyl thiourea was one compound used early for this purpose in both agriculture (to pre-
vent loss of nitrogen through nitrification followed by leaching or denitrification) and the
BOD test (to prevent interference in the measurement of the C-BOD, Section 13.1.3),
but it biodegrades fairly rapidly. Other compounds have also been used, most containing
sulfur and/or nitrogen. The compound now recommended for the C-BOD test is 2-chloro-
6-(trichloromethyl) pyridine, also called N-serve, nitrapyrin, and TCMP. For research
purposes, nitrite oxidation has been inhibited using potassium perchlorate, KClO
3
, but
this compound may also inhibit nitrate reduction. It is also possible that a compound
entering a wastewater treatment plant may interfere with nitrification without having a
noticeable effect on other treatment processes.
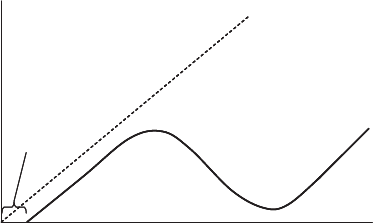
Chlorine Dose
Chlorine Residual
combined chlorine
free chlorine
chlorine
demand
no demand
Figure 13.23 Breakpoint chlorination.
428
MICROBIAL TRANSFORMATIONS
13.3 SULFUR
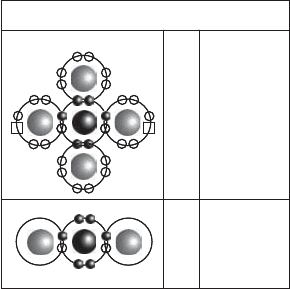
The biogeochemical cycling of sulfur involves substantial abiotic chemical as well as the
biochemical conversions. There are five sulfur oxidation states of appreciable biochemical
importance in the environment. With an atomic number of 16, sulfur’s outer orbital shell
has six electrons. By either depleting or filling this shell, therefore, sulfur may reach
extreme oxidation states of 2 (sulfide, S
2
) and þ6 (sulfate, SO
4
2
) (Figure 13.24),
as well as residing at 0 (elemental sulfur, S
0
), þ2 (thiosulfate, S
2
O
3
2
), and þ4 (sulfite,
SO
3
2
). (Note: In pyrite, FeS
2
, the sulfur has an average oxidation state of 1, although
this may result from a combination of S
2
and S
0
.)
As with reduced nitrogen in the form of ammonia, reduced sulfide can also be volatile
(as hydrogen sulfide, H
2
S). However, these two gases have opposite responses to pH.
Whereas ammonia gas is a weak base and tends to form at higher pH levels (pK
a
¼
9:3), hydrogen sulfide is a weak acid (pK
a
¼ 7:2 for first H
þ
, 11.9 for the second).
Thus, it tends to be nonionized, and hence volatile, only at neutral or lower pH. Reduced
sulfur in alkaline solutions tends to remain ionized, mostly as HS
(or S
2
at very high
pH). Sulfide also tends to react with metals to form insoluble precipitates. Ferrous sulfide
(FeS), in particular, gives many anaerobic sediments and biofilms their black color.
The organic sulfur of the amino acids methionine and cysteine (Table 3.6) is also at an
oxidation state of 2 and thus has two bonds with adjacent atoms (Section 3.2). One of
these is to a carbon atom, but the other may be to a hydrogen (sulfhydryl bond) or to the
sulfur (disulfide bond) of a second amino acid, an important factor in the three-dimen-
sional configurations of proteins.
In comparison to some of the other elements, which are considerably more limiting,
sulfur tends to be readily available in most environments. Fresh water usually contains
at least 10 mg/L of sulfate, and sometimes much more (especially near the coast), and
sewage usually has 30 mg/L more sulfate than the drinking water from which it is
derived. The concentration in seawater is 2700 mg/L. The limit for sulfate in drinkin g
water is 250 mg/L, but this is a secondary standard (protecting public welfare rather than
health) because of taste and a laxative effect.
The reactions of the sulfur cycle are depicted schematically in Figure 13.25. As with
the nitrogen cycle, some of the steps are carried out only by prokaryotes. Bacteria and
S HH
S
O
S
O
O
O
+6
−2
Sulfate
SO
4
2
−
Hydrogen
H S
2
Sulfide
- -
SULFUR
S
HH
S
S
O
S
O
O
O
O
-- --
Figure 13.24 Sulfur oxidation state extremes.
SULFUR 429
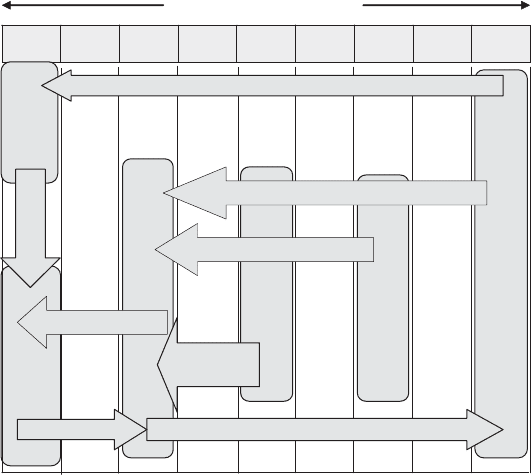
archaea associated with some of the nonassimilative sulfur reductions (use as an electron
acceptor) and oxidations (use as an energy source) are listed in Table 13.5.
Also as with nitrogen, mineralization of organic sulfur does not involve oxidation or
reduction of the sulfur, which is released as sulfide. However, assimilation of sulfide is
uncommon, largely because of its toxicity. Instead, most organisms utilize sulfate (requir-
ing a reduction) and/or organic sulfur.
13.3.1 Sulfur Reduction
Some bacteria and archaea, including chemoorganotrophs and chemolithotrophs, use sul-
fate or other oxidized forms of sulfur as an electron acceptor for anaerobic respiration,
producing further reduced int ermediates (sulfite, thiosulfate, elemental sulfur) or fully
reduced sulfide. In addition, many organisms can assimilate sulfate through a reductive
process.
Dissimilatory Sulfate and Sulfur Reduction The complete reduction of sulfate to
hydrogen sulfide, during its use as an electron acceptor for anaerobic respiration, can
be achieved by specialized but common and widespread groups of strictly anaerobic bac-
teria, referred to functionally as sulfate-reducing bacteria (SRBs). The best known of
these are Desulfovibrio, a proteobacteria, and Desulfotomaculum, a gram-positive endo-
sporeformer, although other genera are now recognized as well. Interestingly, one archaea,
Archaeoglobus, is also known that can carry out this reaction. Some other anaerobic bac-
teria and archaea can reduce elemental sulfur to sulfide.
It was initially believed that SRBs could only utilize a few fermentation products, such
as H
2
, lacta te, and pyruvate. However, it is now realized that some can utilize acetate and
S
2
O
3
2
-
Thio-
sulfate
SO
4
2
-
Sulfate
SO
3
2
-
Sulfite
-2
+5+4
+3
+2
+1
0
-1 +6
-2
+5+4
+3
+2
+1
0
-1 +6
-2
+5+4
+3
+2
+1
0
-1 +6
-2
+5+4
+3
+2
+1
0
-1 +6
Elemental sulfur oxidizersSulfide Oxidizers
Sulfur Oxidation States
Thiosulfate
reducers
Sulfide generators
S
0
H
2
S
Hydrogen
Sulfide
Sulfur
Organic
Sulfur
Sulfite reducers
Sulfate reducers
Sulfate Assimilation
mineralization
Figure 13.25 Biochemical sulfur transformations.
430
MICROBIAL TRANSFORMATIONS

other short- and long-cha in fatty acids, ethanol and other alcohols, benzoate (which has
an aromatic ring), and hexadecane (a hydrocarbon), among other compounds.
Sulfate reduction does not involve the electron transport chain utilized by aerobes and
denitrifiers. In fact, unlike those reactions, it requires an initial activation of the sulfur
with adenosine triphosphate (ATP) and thus involves an initial expenditure of energy.
The sulfate within the adenosine phosphosulfate (APS) formed is then reduced (with
the addition of two electrons) to sulfite. Sulfite reductase then catalyzes the sequential
loss of three more electron pairs, yielding H
2
S, which is released to the environment.
SO
4
2
þ ATP ! APS þ pyrophosphate (pyrophosphate is two phospha tes joined )
APS þ 2e
! AM P þ SO
3
2
SO
3
2
þ 6e
! H
2
S
Assimilatory Sulfate Reduction The assimilatory reduction of sulfate can be carried out
by most bacteria, archaea, fungi , algae, and plants, while many protozoa and animals
require organic sulfur. This reduction also starts with the production of APS, but a second
ATP is then used to attach another phosphate , forming phosphoadenosine 5
0
-phosphosul-
fate (PAPS). The sulfur is then reduced to sulfite, followed by further reduction to sulfide.
To avoid toxicity, as the sulfide is formed it is quickly combined to form an organic sulfur
compound such as cysteine.
Sulfur Disproportionation Some SRBs can simultaneously oxidize and reduce an inter-
mediate sulfur compound such as sulfite or thiosulfate to provide energy. For example,
TABLE 13.5 Examples of Prokaryotes Able to Oxidize or Reduce (Dissimilatory)
Inorganic Sulfur
Forms of Sulfur
Genus Kingdom: Class Substrate
Reduce Desulfovibrio d-Proteobacteria SO
4
2
Desulfotomaculum Firmicutes: Clostridia SO
4
2
Thermodesulfobacterium Aquaficae SO
4
2
Pyrolobus Crenarchaeota: Thermoprotei S
2
O
3
2
Pyrodictium Crenarchaeota: Thermoprotei S
Thermoplasma Euryarchaeota: Thermoplasma S
Archaeoglobus Euryarchaeota: Thermococci SO
4
2
Thermococcus, Pyrococcus Euryarchaeota: Thermococci S
Oxidize Chloroflexus Thermomicrobia: Chloroflexi H
2
S
(green nonsulfur)
Chlorobium Chlorobia (green sulfur) H
2
S, S
Rhodospirillum a-Proteobacteria (purple nonsulfur) H
2
S
Chromatium g-Proteobacteria (purple sulfur) H
2
S, S
Thiobacillus b-Proteobacteria H
2
S, S, S
2
O
3
2
Beggiatoa, Thiothrix g-Proteobacteria H
2
S; S; S
2
O
3
2
Aquafex Aquaficae S; S
2
O
3
2
Sulfolobus Crenarchaeota: Sulfolobi H
2
S; S
Ferroglobus Euryarchaeota: Thermococci H
2
S
SULFUR 431
three molecules of sulfite are oxidized to sulfate at the same time that one molecule is
reduced to hydrogen sulfide.
4H
2
SO
3
! 3H
2
SO
4
þ H
2
S
13.3.2 Sulfur Oxidation
A wide range of bacteria and archaea are able to oxidize reduced sulfur, including photo-
trophs and lithotrophs (Table 13.5). Thiobacillus, for example, is an aerobic or denitrify-
ing (one species) lithotroph that can grow on sulfides. It is of particular interest because of
the acid tolerance of several species; one has a pH optimum of 2! This is particularly use-
ful to the organism, as the product of complete sulfide oxidation (sulfate) is actually sul-
furic acid.
Beggiatoa, a filamentous proteobacteria, has a neutral pH range but is of interest
because of its gliding motility. This allows it to move so that it can stay right at the bound-
ary between an anaerobic environment (where the sulfide is formed) and the aerobic
one from which it gets oxygen. When growing on sulfide, it deposit s, internally, elemental
sulfur that is readily visible as yellow granules under the microscope (see Figure 10.22).
These may serve as a substrate reserve to be used if sulfide is depleted. However, they
may also have the benefit of minimizing sulfuric acid production.
There are also a number of phototrophic bacteria that oxidize reduced sulfur (Table 10.4).
The green and purple nonsulfur bacteria oxidize hydrogen sulfide but do not use or deposit
elemental sulfur. The green and purple sulfur bacteria, on the other hand, utilize both H
2
S
and elemental sulfur, which they also deposit (externally for the green, internally for the
purple).
13.3.3 Sulfur in Environmental Engineering and Science
Sulfur plays an important role in several areas of concern to environmental engineer s and
scientists, from corrosion to odors. Its important contribution to acid mine drainage is dis-
cussed below as part of the iron cycle (Section 13.4.3). Sulfur oxides are also a major
component of acid rain, or acid deposition (see Section 15.7), but this stems mainly
from combustion rather than microbial transformations.
Corrosion Water reacts spontaneously with iron (Fe
0
) to produce a thin layer of hydro-
gen, H
2
, and ferrous hydroxide, Fe(OH)
2
. Still, it was originally thought that iron and steel
would not corrode under anaerobic conditions, since no oxygen would be present to
oxidize the H
2
and thereby allow the reaction to continue. However, if sulfate is present,
sulfate-reducing bacteria can utilize the H
2
, perpetuating the reaction and leading to exten-
sive deterioration:
4Fe
0
þ 4H
2
O þ SO
4
2
! FeS þ 3FeðOHÞ
2
þ 2OH
ð13:20Þ
The microbial reactions of the sulfur cycle are also responsible for the crown corro-
sion of sewers, particularly those made of concrete. Unlike water distribution pipes, which
are pressurized, flow in sewers is usually by gravity, with the pipes often less than half
full. During periods of low flow, wastewater velocities may drop to the point where orga-
nic matter deposits on the bottom. This is particularly common in flat areas, such as shore
432 MICROBIAL TRANSFORMATIONS
communities built on the coastal plain. After sufficient time, these bottom deposits and the
wastewater itself may become septic, or go anaerobic, and if sufficient sulfate is present
(as is also typical in shore communities), hydrogen sulfide will be formed by sulfate-
reducing bacteria. A portion of this H
2
S volatilizes from the liquid phase and is then
absorbed in the moist biofilm at the crown (top) of the sewer. Here, under aerobic condi-
tions, it can be reoxidized by bacteria such as Thiobacillus to sulfate, in the form of sul-
furic acid (H
2
SO
4
), which then reacts with and dissolves the concrete. In fact, one of the
sulfide-oxidizing bacteria involved is called T. concretivorus (‘‘concrete eater’’). Prior to
an understanding of this process, it seemed mysterious that sewers corroded at the crown
rather than at the bottom.
Odors Decomposition of proteins can lead to the release of mercaptans, such as ethyl
mercaptan (C
2
H
5
SH), from sulfur-containing amino acids. These volatile organic sulfur
compounds are highly odorous. Both protein decomposition and sulfate reduction als o
lead to the production of H
2
S, with its penetrating rotten egg odor. Thus, very strong
and unpleasant odors can be released from a variety of ‘‘natural’’ anaerobic processes,
including decomposition of vegetative debris in salt marshes and piles of manure. Odor
problems from sulfides and mercaptans (as well as amines, organic acids, and other com-
pounds) are also a major area of complaint for a variety of waste treatment facilities,
including pumping stations on sewer lines, slud ge tanks, composting facilities, land appli-
cation sites, and landfills. In addition to aesthetic considerations, odors also appear to be
associated with health effects in some people. Thus, control of odors is a major expense at
some sites. Sulfides and mercaptans can be removed from gas streams by alkaline scrub-
bers (since at high pH the molecules ionize and are then nonvolatile), chemical oxidation
(e.g., with chlorine), biological oxidation (e.g., in biofilters), or reaction with metals (e.g.,
iron). Alternatively, the formation or release of odorous compounds can be minimized by
controlling oxygen levels, maintaining alkaline pH, and adding iron (to bind sulfides) or
nitrate (leading to denitrification instead of sulfate reduction).
Toxicity Hydrogen sulfide may be highly toxic, in both aquatic and soil systems, to both
prokaryotes and eukaryotes. Once inside the cell, it binds rapidly to metals, inactivating
cytochromes and other critical cell constituents. Low concentrations of H
2
S in air are
readily detectable by the odor. However, at higher concentrations it deadens the olfactory
cells and can no longer be smelled. The buildup of hydrogen sulfide in confined spaces is
one of the reasons (others include lack of oxygen and buildup of combustible gases) that
entry into such areas without proper precautions is so dangerous, potentially resulting in
death. Accidents involving H
2
S frequently produce multiple fatalities, as would-be res-
cuers rush into contaminated spaces, only to become victims themselves. The gas is so
toxic that even professional responders have been felled by gas leaking around their
gas masks.
Wastewater Treatment The sulfide present in wastewater can be oxidized to sulfate
under aerobic conditions. Usually, this is a minor part of the secondary treatment process.
However, some wastewaters may contain considerable amounts of sulfide, either from
industrial inputs or from extensive sulfate reduction due to anaerobic conditions in the
sewer lines or in earlier treatment steps. Under these conditions the sulfide may represent
a considerable oxygen demand and in fact would be measured as C-BOD in standard
tests. In activated sludge treatment (Section 16 .1.3) under such conditions, filamentous
SULFUR 433

sulfide-oxidizing bacteria such as Thiothrix may grow sufficiently to interfere with the
settling that is an essential part of the process.
In another secondary treatment process involving rotating biological contactors (RBCs;
Section 16.1.2), an anaerobic biofilm layer may develop that leads to H
2
S production. The
motile filamentous sulfide-oxidizing bacteria Beggiatoa may then grow at the interface
between this region and the overlying aerobic zone. Growths of Beggiatoa under these
conditions are sometimes so massive that they lead to the physical collapse of the system
(e.g., shaft failure).
Methanogenic anaerobic treatment systems are commonly used for wastewater sludges
(anaerobic digestion, Section 16.2.1) and occasionally, for high-strength wastewaters.
However, if too much H
2
S is produced (from amino acid mineralization or sulfate reduc-
tion), it can be toxic to the methanogens. One control measure is to add iron, which binds
with the sulfide as FeS.
Sulfide can also interfere with disinfection. Active chlorine is rapidly consumed by
reaction with sulfides and other reduced sulfur compounds.
Example 13.8 A wastewater contains 2 mg/L of hydrogen sulfide. How much chlorine
will this consume?
Answer Although the active form of chlorine in water is usually hypochlorous acid,
HOCl, or chloramines, NH
2
Cl (especially in wastewater), by convention (since 1 mol of
either comes from 1 mol of molecular chlorine), chlorination calculations are based on the
equivalent amount of Cl
2
(molecular weight ¼ 71):
H
2
S þ 4Cl
2
þ 4H
2
O ! SO
4
2
þ 8Cl
þ 10H
þ
chlorine consumed ¼ 2mg=LH
2
S
1 mol H
2
S
34 g H
2
S
4 mol Cl
2
1 mol H
2
S
71 g Cl
2
1 mol Cl
2
¼ 2ð8:35 mg Cl
2
=mg H
2
SÞ¼17 mg=LCl
2
Dimethyl Sulfide The biogenic production, fate, and impact of dimethyl sulfide (DMS),
CH
3
SCH
3
, in remote oceans has only recently come under investigation. This reduced
(oxidation state ¼2) volatile compound is considered to be the most abundant form of
organic sulfur in nature and the largest source of sulfur in the atmosp here. The environ-
mental significance of DMS stems from its eventual contribution to the formation of
atmospheric aerosols and cloud-forming particles that can impose a large-scale effect
on the incidence of solar radiation over remote ocean areas.
Metabolic production of DMS has been estimated to amount to 4:5 10
7
metric tons
per year. It results from the transformation of dimethylsulfoni um propionate (DMSP),
which is used by various marine algae, such as dinoflagellates, as a means of controlling
their osmotic pressure. Bacteria metabo lize released DMSP as a carbon and energy
source, forming DMS as one of the end products. DMS then can be released into the
atmosphere (Figure 13.26), where it undergoes photochemical oxidation to methane sul-
fonic acid (CH
3
SO
3
H), sulfur dioxide (SO
2
), and non-sea-salt sulfate (as opposed to aero-
solized sulfate from ocean spray).
S
+
CH
2
CH
2
C
H
3
C
H
3
C
O
O
−
DMSP
434 MICROBIAL TRANSFORMATIONS
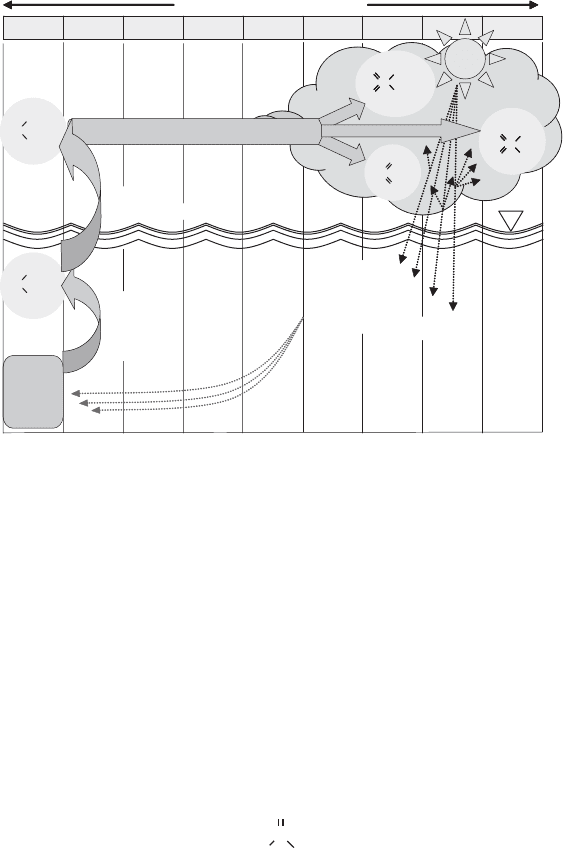
These aerosols themselves, along with the clouds that form when they serve as cloud
condensation nuclei, affect the sola r radiation reaching the ocean surface through scatter-
ing of incoming light and direct shading. In addition to the effects on the global radiation
(and heat) balance, it may be that large-scale oceanic algal blooms promote atmospheric
shading indirectly, thereby decreasing their own activity.
A variety of chemoorganotrophic and chemolithotrophic bacteria are able to oxidize
DMS remaining in the water phase, producing dimethyl sulfoxide (DMSO), which is
also frequently generated as a waste contaminant in paper manufacturing operations. Bac-
teria such as Hyphomicrobium are then capable of using DMSO aerobically as a sole
source of carbon, energy, and sulfur. Alternatively, DMSO can be reduced under anaero-
bic conditions by organisms utilizing it as an electron acceptor, produc ing DMS again.
S
H
3
C
CH
3
O
DMSO
13.4 IRON
In addition to its metallic elemental form (Fe
0
), iron has only the þ2 ferrous, Fe(II), and
þ3 ferric, Fe(III), oxidation states, separated by a one-electron difference. Most iron
cycling involves oxidations and reductions between these two states, via both abiotic
and biochemical mechanisms.
Iron is one of the most abundant elements on Earth, but it is confined primarily to the
lithosphere (soil averages about 7% Fe). It is nonvolatile, and its concentration in air is
+4
+3+2
−10
−1− 2+5
+6
S
CH
3
CH
3
S
O
O
CH
3
OH
S
O
O
S
DMS
S
CH
3
CH
3
DMS
S
CH
3
CH
3
DMS
O
S
O
O
O
-
-
S
-
S
Marine
Algae
with
DMSP
DMSP
and
cleavage
DMS release
DMS Photochemical oxidation
DMS
volatilization
and shading by
and cloud
Light reflecting
aerosol particles
condensation nuclei
Sulfur Oxidation State
Methane
sulfonic
acid
Sulfur
NSS
Sulfate
dioxide
Figure 13.26 Dimethyl sulfide reactions over remote ocean waters. (DMSP, dimethylsulfonium
propionate).
IRON 435
virtually zero except for the small amounts present in airborne soil particles. In water, the
solubility of ferric iron is low (<0.02 mg/L at pH 6), except under very acidic conditions
or in some organic complexes. Ferrous iron is more soluble (about 20 mg/L at pH 6, but
depending strongly on alkalinity). However, under the anaerobic conditions in which it is
usually found, it is likely to form a precipitate with sulfide. Although iron is an essential
element, it typically is present in organisms on ly at low concentrations (e.g., 0.2% of the
dry weight of E. coli, Table 11.1). Thus, although iron is abundant and only small amounts
are needed, it still may be available only at levels that limit growth in some environments,
especially the open ocean.
13.4.1 Iron Reduction
In the absence of oxygen and nitrate, ferric iron can be used for respiration by a variety of
aerobic bacteria. In the process, the Fe(III) is reduced to Fe(II). Both organotrophic and
lithotrophic iron reducers are known. The process seems to be carried out by many of the
same organisms that can reduce nitrate.
There are a number of natural oxygen-deficient conditions under which reduction
might take place. These include anoxic biofilm layers, lake bottoms and sediments,
water-saturated soils, groundwa ters, and bogs. Reduction is a principal mea ns of solubi-
lizing iron-bearing solids. Iron reduction and resolubilization also occur in landfills and
anaerobic digesters. Where sulfides are present, FeS will form, precipitating the iron while
helping to limit odor and toxicity problems associated with H
2
S.
13.4.2 Iron Oxidation
A diverse assortment of aerobic bacteria and archaea can oxidize ferrous iron, apparently
using it as an energy source. Because only a small amount of energy is available from this
transformation, large amounts of iron must be processed to support their growth.
At neutral pH values, ferrous iron is unstable under aerobic conditions and will be oxi-
dized rapidly and spontaneously (abiotically) to the þ3 form. Thus, microorganisms
utilizing iron at such pH values must be located at the interface between aerobic and anae-
robic zones, depending on this specialized niche so that they may capture this energy
before it is lost. On the other hand, at low pH, ferrous iron is sufficiently stable that micro-
organisms can utilize it in aerobic systems.
Thus, iron oxidizers can be subdivided into two groups based on their pH preferences.
The acidophiles include several Thiobacillus, Leptospirillum (a xenobacteria), and Sulfo-
lobus (an archaea). Many of the same organisms can also oxidize reduced sulfur, an
advantage since ferrous iron and sulfides often occur together. T. ferrooxidans, an auto-
troph, is the best known; it has a pH range of 0.5 to 6, with an optimum at 2! It utilizes a
copper-containing protein, rusticyanin, for transferring the electron from iron during its
oxidation (Figure 13.27). The amount of energy available from the oxidation is too
small to use directly for ATP synthesis; instead, the proton gradient is utilized for this
purpose.
Gallionella (Fig ure 13.28) grows at neutral pH, usually at a point where anaerobic
groundwater comes in contact with oxygen (such as in wells). It is commonly heavily
coated with ferric hydroxide, Fe(OH)
3
. Sphaerotilus and Leptothrix also deposit oxidized
iron on their sheath, but it is not certain that they are able to capture the energy from iron-
oxidation. Other iron, oxidizing bacteria include Planctomyces and Hyphomicrobium;
436 MICROBIAL TRANSFORMATIONS

interestingly, these bacteria both have stalks, although they are not related phylogeneti-
cally. Ferroglobus, an archaea, is also of interest in that it oxidizes Fe(II) to Fe(III)
under anoxic conditions, using nitrate as the electron acceptor.
13.4.3 Iron in Environmental Engineering and Science
Iron cycling plays a role in a number of issues of relevance to environmental engineers
and scientists. These include problems of acidification, particularly acid mine drainage,
and drinking water.
ATP
H
2
OH
2
O
H
2
OH
2
O
Fe
+3
Fe
3+
Fe
+2
Fe
2+
Periplasm
Cytoplasm
Membrane
Rusticyanin
Rusticyanin
Cytochrome a
1
1
/
2
O
2
2H
+
Cytochrome c
O
2
O
2
H
+
H
+
+
Proton Gradient
Energy Source
Proton Gradient
Energy Source
Elevated H
+
at low pH conditions
Cytoplasmic H
+
sink
(more neutral
p
H)
e
-
e
-
e
-
e
-
ATPsynthase
Figure 13.27 Energetic proton gradient for acidophilic iron-oxidizing chemolithotrophs.
Figure 13.28 Gallionella-type bacteria causing iron precipitation in wells.
IRON 437
