Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.


Appendix A lists the values of physicochemical parameters described above for several
important toxic substances.
Besides describing how substances distribute themselves within an organism, physico-
chemical parameters are important in the distribution between organisms and their envir-
onment. Of particular significance is the K
OW
value. This is a good predictor of how
substances will distribute between the lipid tissues of aquatic organisms and the water
they live in. Since K
OW
for hydrophobic organics is very large, lipid tissues of these
organisms often store concentrations of toxins far higher than is found in the environment.
This phenomenon is called bioaccumulation or bioconcentration.
Example 18.4 The aqueous solubility of Freon (CCl
2
F
2
) is 280 mg/L. If its concen-
tration in water were 0.25 mg/L, estimate the concentration in fatty tissues. Assume
that there is no elimination.
Answer The molar mass of Freon is 120.9 g/L. The solubility in molar units is
c
s
¼
0:280 g=L
120:9g=mol
¼ 0:00232 mol=L
From equation (18.7),
K
OW
¼ 10
0:7100:862 log c
s
¼ 10
0:7100:862 log ð0:00232Þ
¼ 10
2:98
¼ 959
The Freon concentration in the lipid phase can then be estimated by assuming that it parti-
tions according to the octanol–water partition coefficient. Then, using equation (18.6) [or,
equivalently, equation (18.1)], we have
C
O
¼ K
OW
C
l
¼ 959 0:25 mg=L ¼ 240 mg=L
Lipid storage prevents excretion or detoxification of a compound, and this results in
another phenomenon, called biomagnification. In biomagnification, because animals
that eat others which have already bioconcentrated the toxics will receive a higher dose
of the toxin, the concentration of toxin increases toward the top of the food pyramid.
Thus, predators at high trophic levels will be most sensitive to the presence of toxins
that bioaccumulate. The K
OW
is a good predictor of the tendency of a substance to bioac-
cumulate, bioconcentrate, or biomagnify. These concepts are discussed in more detail in
Section 18.7.
Adsorption is another type of partitioning. It is the transport of a solute from gas or
liquid to the surface of a solid, or to other interfaces, such as of membranes. Adsorbing
materials in the environment include soil, sediment, and suspended solids in air or water.
Adsorption can reduce the availability of compounds to organisms by tying them up, or it
can incr ease availability by serving as a contact mechanism. An example of the latter is
the role of atmospheric dust in carrying gaseous radon into lungs.
Two types of chemical equilibrium often need to be considered together with physico-
chemical equilibrium: acid–base reactions and complex formation. Th is is because their
equilibrium occurs fairly rapidly, so that any change in concentration of one reactant
causes rapid conversion to another form. The im portance of this is that different forms
may have different physicochemical and biological behaviors. For example, many organic
acids exist in both undis sociated (HA) and dissociated forms (H
þ
and A
). However, the
738 FATE AND T RANSPORT OF TOXINS

anion, A
, tends to concentrate in the polar aqueous phase, whereas the undissociated
form, HA, is more likely to concentrate in lipid or gas phases.
The distribution of dissociated and undissociated forms as a function of pH was de-
veloped in equation (3.3), and Figure 3.3 shows how this distribution would appear for
acetic acid. You will recall that an acid is 50% dissociated when pH equals pK
a
, whereas
it will be mostly undissociated at lower pH, and mostly in the dissociated form at higher
pH. Table 18.1 shows the pK
a
values for several pollutants.
In the case of acetic acid, for example, the dissociated form will be essentially
insoluble in lipids and thus will not pass through plasma membranes. On the other
hand, the undissociated form will have greater solubility in lipids, and therefore a greater
ability to pass through them. As a result, the effective Henry’s constant, aqueous solubi-
lity, and octanol–water partition coefficient will all vary with pH for organic acids and
bases. If the undissociated form has a phase equilibrium constant of K
u
and for the
dissociated form it is K
d
, equation (3.3) can be used to derive the effective equilibrium
constant, K
eff
:
K
eff
¼
K
u
K
d
1 þ 10
pHpK
a
þ K
d
ð18:8Þ
In each equilibrium constant of equation (18.8), the aqueous concentration is in the deno-
minator. Each equilibrium constant can represent a Henry’s constant, an octanol–water
partition coefficient, or a linear adsorption partition coefficient.
Equation (18.8) can describe the variation of toxicity of many substances with pH. An
example is hydrogen cyanide, which has a pK
a
of 9.37. Both the undissociated and dis-
sociated forms are toxic to fish, but the former is about twice as toxic. Thus, the toxicity
decreases somewhat as pH increases in the area of the pK
a
.
TABLE 18.1 Acid and Base p-Dissociation Constants
for Several Toxic Pollutants
a
Compound pK
a
Hydrogen sulfide 6.99
Hypochlorous acid 9.40
Hydrogen cyanide (1) 9.21
Phenol 9.96
p-Chlorophenol 0.80
Pentachlorophenol (2) 4.75
Trichloroacetic (1) 0.52
Analine-H
þ
(3) 10.87
Ammonium 9.26
a
Note that aniline and ammonium are given in protonated form.
These are organic bases that tend to be ionized at pH levels below
the pK
a
, as opposed to the organic acids, which are ionized
predominantly at pH levels above the pK
a
.
Source: (1) Petrucci et al. (2001); (2) Christodoulatos and
Mohiuddin (1996); (3) Austin Community College (2004).
PHYSICOCHEMICAL PROPERTIES
739

Example 18.5 Henry’s constant for undissociated hy drogen sulfide (H
2
S) is 0.0042 and
the pK
a
(for the first dissociation) is 7.1. The dissociated form is nonvolatile. What is the
effective Henry’s constant of H
2
S at pH 7.5?
Answer Use equation (18.3) with K
u
¼ 0:0042 and K
d
¼ 0:00. Then
H
c;eff
¼
H
c;H
2
S
0:0
1 þ 10
pHpK
a
þ 0:0 ¼
0:0042
1 þ 10
7:57:1
¼ 0:0012
An important example is ammonia. Being basic, it ionizes as pH goes below a pK
a
value of 9.26. In ionized form it is relatively imperme able to cell membranes and is
much less toxic to fish. However, although only a small fraction is undissociated at low
pH, that fraction increases by a factor of about 10 for each pH unit of increase. For exam-
ple, at pH 6, 0.056% of the ammonia is undissociated, but at pH 7 it is 0.55%, and at pH 8
it is 5.3%. From pH 7 to 8 the 96-h LC
50
to rainbow trout goes from 80 to 20 mg/L NH
3
-
N, a four fold increase in toxicity.
Another kind of chemical interaction with physicochemical processes is the form-
ation of soluble complexes. A compl ex is a combination of two or more species in solu-
tion held together by physicochemical or ionic forces. A notable example is the metal
ions, many of which form complexes with hydroxide and carbonate species present in
natural water. Often, a variety of different complexes can form from a particular metal,
resulting in species of varying composition and ionic charge. For example, in computing
the solubility of copper in natural waters, it is necessary to account for the formation of
the following species:
Cu
2þ
; CuOH
þ
; CuðOHÞ
2
; Cuð OHÞ
3
; Cuð OHÞ
2
4
; CuCO
3
; Cuð CO
3
Þ
2
All of these species tend to be in equilibrium with each other. At a given total copper
concentration, the relative amounts of these species will depend mostly on pH and
alkalinity. Each of these copper species may have different toxicities to an organism.
Therefore, it follows that the toxicity of copper to fish, for example, will depend on pH
and alkalinity. Many other ions complex with metals, including sulfide and phosphate.
Chloride and sulfate are significant ligands at seawater concentrations. Metals also form
complexes with organic compounds, such as to electronegative atoms; p-bonds (which
make up the C
C bond), as in unsaturated fats or aromatics; or by forming salts of
carboxylic acids.
As a result of processe s such as dissociation or complexation, a chemical may be pre-
sent in a phase in more than one form. The partition coefficient, however, only describes
the ratio of like species in the two phases. For example, in the case of acetic acid in water,
the partition coefficient is only the ratio of undissociated acid concentrations. However,
when a phase is analyzed, it is typically the total concentration of all species , including
complexes and dissociated forms, which is measured. Therefore, the mea sured ratio of
total concentrations for the two phases is given a different name, the distribution coeffi-
cient. The partition coefficient does not change with factors such as pH, but the distri-
bution coefficient does, in the same manner as equation (18.8).
Since structure has such an important influence on toxic behavior of chemicals, efforts
have been made to empirically correlate structural characteristics, as well as the physico-
chemical properties, to toxic effects. The ability to predict the toxicity of substances
is useful because given the variety of chemicals and effects, it is not feasible to perform
740 FATE AND T RANSPORT OF TOXINS
all the possible tests. Furthermore, the ability to predict effects could even be applied to
chemicals that have not yet been synthesized or purified.
Such correlations are called quantitative structure–activity relationships (QSARs).
The QSAR method uses statistical techniques, especially multiple regression or discri-
minant analysis, to find relationships between a variety of predictors and a single effect.
Regression is used to predict continuous variables, such as the dose lethal to 50% of orga-
nisms (the LD
50
value, described below). Discriminant analysis is more useful for
categorical (either/or) endpoints, such as carcinogenicity or skin irritation. Examples
of QSARs are the correlation between narcosi s and polarity, or the correlation between
toxicity and the side-chain length of organophosphate pesticides, as described in
Section 18.3.
Various types of predictors can be used. The most common are (1) physicochemical
properties, such as solubility or K
OW
; (2) functional group descriptors; and (3) molecular
indices. Prediction of narcosis from polarity is an example of the first type. Organopho-
sphate toxicity is an example of the second type.
The use of functional groups is preferable to using properties, since it can be applied
even to never-synthesized chemicals in the proposal stage of a research project. On the
other hand, since the physi cochemical properties in some cases are directly related to
the toxic effect, they can provide better predictions. The functional group descriptors
typically go well beyond the kinds of groups listed in Table 3.4 . Also included are mole-
cular descriptors such as number of double bonds, number of rings indexed by type and
number of atoms forming the ring, and length of hydrocarbon chains.
Use of the third type of data, molecular indices, can be more powerful predictors of
toxic activity than functional groups. These include the molecular connectivity indices,
electronic charge distributions, and kappa environmental descrip tors. Molecular connec-
tivity indices (MCIs) are a group of parameters that describe the topological structure of a
chemical. It contains information on molecular size, shape, branching structure, cycliza-
tion, unsaturation, and heteroatom content (presence of atoms other than carbon in the
molecule’s ‘‘backbone’’). Physicochemi cal and biological parameters are correlated to
the MCIs in the same way as they might be correlated with the K
OW
.
Despite the apparent promise of such predictors, they typically work well only when
limited to a single type of compound and a single toxic effect. For example, one might be
able to develop a model to predict the mutagenicity of polychlorinated biphenyls, but it
would be much more difficult to develop a single model to predict the mutagenicity of all
chlorinated hydrocarbons. A single effect, such as mortality, may be a consequence of
different modes of action. Thus, a model to predict LD
50
might be limited to a particular
group of compounds. Jørgensen et al. (1997) describe the use of a variety of correlation
and QSAR techniques in the prediction of a variety of physicochemical and biological
parameters, including:
Aqueous solubility
Partition coefficient (including K
OW
)
Henry’s constant
Soil adsorption and exchange coefficient
Rate constants for hydrolysis and photolysis
Enzyme inhibition
Microbial and mammalian toxicity (LC
50
,EC
50
, etc.)
PHYSICOCHEMICAL PROPERTIES 741

Uptake of organics by algae
Biosorption
Bioconcentration, bioaccumulation, and biomagnification factors
Biodegradation rate
No-effect concentration
Growth rate
Uptake rate, uptake efficiency, and excretion rate
18.2 UPTAKE MECHANISMS
Uptake is the transfer of a chemical from the environment into an organism. A toxic
substance must pass through a cell membrane to enter the organism. This can occur
naturally by several mechanisms, which were described in Section 4.1. Passive transport
occurs whenever there is a concentration gradient across the membrane. The situation is
illustrated in Figure 18.1.
The passive transport flux, F
pt
[Mt
1
L
2
], can be modeled in a simplified way as
being proportional to the concentration difference across the membrane by the diffusion
coefficient, D, and inversely proportional to the membrane thickness, h:
F
pt
¼
D
h
ðC
2
C
3
Þð18:9Þ
The diffusion coefficient depends on the prope rties of the solute and of the membrane,
but in general, it is inversely related to the square root of the molar mass of the diffusing
species. Thus, small molecules diffuse faster. The concentrations at the faces of the plasma
lipid membrane can be assumed to be in equilibrium with the aqueous phases in which
they are in contact. Thus, C
2
¼ K
M
C
1
and C
3
¼ K
M
C
4
, where K
M
is the partition coeffi-
cient between the membrane and the adjacent phases. Substituting these relations into
Membrane
h
C = 0
C
1
C
2
C
3
C
4
Flux
Donor
Compartment
Receptor
Compartment
Figure 18.1 Passive transport across a membrane by molecular diffusion.
742
FATE AND TRANSP ORT OF TOXINS

equation (18.9), we can express the flux in terms of the concentrations in the aqueous
phases:
F
pt
¼
DK
M
h
ðC
1
C
4
Þð18:10Þ
Thus, it can be seen that the partition coefficient has a direct impact on the rate of trans-
port. Since the octanol–water partition coefficient is a model for the lipid–water system,
compounds with a high K
OW
will be absorbed easily by passive transport. The constants in
equation (18.10) can be lumped into a single parameter, the mass transfer coefficient, k:
F
pt
¼ k ðC
1
C
4
Þð18:11Þ
The mass transfer coefficient is an inverse measure of the resistance posed by the
membrane to movement of the substance. A similar equation can be used to describe
the transfer between two phases in the absence of a membrane, such as from air to water
or from blood plasma to a lipid phase. This is the film theory model of interphase mass
transfer. The presence of two immiscible phases in contact with each other implies that
they will be physicochemically different and have different affinities for the solute as
described by the partition coefficient. The flux of solute can be described similar to equa-
tion (18.11), but modifying the conce ntration of one of the phases by the partition coeffi-
cient, so that the flux will be zero when the two phases are in equilibrium. For example,
the model for transfer from an organic phase with concentration C
O
to an aqueous phase
with concentration C
W
will be
F ¼ kðK
P
C
W
C
O
Þð18:12Þ
In this case the mass transfer coefficient, k, will depend on the diffusion coefficients of
the solute in the two phases, as well as other factors, such as the amount of mixing
between the interface and the bulk fluid.
Other transport mechanisms were described in Section 4.1. These include filtration,
facilitated diffusion, active transport, and endocytosis. Facilitated diffusion and active
transport, which depend on a carrier in the membrane, have a maximum flux associated
with saturation of the carrier. As a result, their dynamics can be described by a form of
Michaelis–Menten kinetics.
18.3 ABSORPTION AND ROUTES OF EXPOSURE
Absorption refers to uptake into the systemic circulation of an organism. Absorption
occurs through three main routes of exposure:
Ingestion: absorption through the lining of the gastrointestinal tract from food or
other particles
Inhalation: absorption through the lungs
Dermal contact: absorption directly through the skin
Ingestion and inhalation are forms of intake . All three are mechanisms of uptake. The
route of exposure determines how much of a toxin enters and how different organs are
ABSORPTION AND ROUTES OF EXPOSURE 743
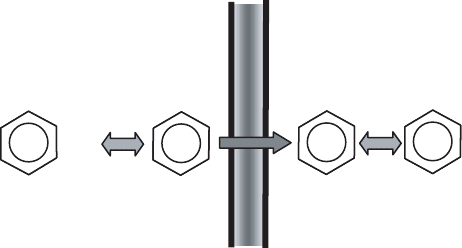
exposed. The toxicity of a substance depends on the route of exposure. For example,
arsenic is more carcinogenic by inhalation than by ingestion, whereas vinyl chloride is
the reverse. Asbestos danger from inhalation is well documented, but little evidence exists
yet for its toxicity by ingestion.
Uptake by ingestion occurs via absorption through cell membranes lining the gastro-
intestinal tract. Most of the surface area of the gastrointestinal (GI) tract is associated with
the villi of the small intestines. Thus, most neutral molecules will be absorbed there.
Organic acids and bases, on the other hand, depend on pH relationships for their absorp-
tion. The pH of the stomach ranges from 1 to 3, and the intestines from 5 to 8. Thus, weak
acids will be undissociated in the stomach and tend to be absorbed there. Conversely,
weak bases will be ionized in the stomach and therefore unable to pass membrane barriers
by passive transport. However, they can be absorbed in the intestines at the higher pH.
The pH relationships in the GI tract form a special mechanism for facilitating absorp-
tion of acids and bases. Because blood plasma has a pH close to neutral, weak acids will
be mostly dissociated in it, and only a small fraction will be in the undissociated form.
However, in the low pH environment of the stomach, weak acids will be mostly undis-
sociated. The undissociated form, being uncharged, can pass through the membranes of
the cells lining the stomach. After it crosses, it enters the bloodstream, where it become
ionized due to the high pH. This keeps the concentration gradient high, maximizing the
passive diffusion flux.
Figure 18.2 illustrates what is occurring in the case of benzoic acid. The concentration
gradient of non-ionized acid is 100 1 ¼ 99. If the driving force were based on the total
concentration instead of the non-ionized concentration, the gradient would change sign
(101 2513 ¼2412). Benzoic acid would move from the blood into the stomach
instead of the reverse. A similar situation occurs in the intestines, except in that case
the pH of the lumen is higher than that of the blood. This facilitates absorption of
weak bases by a similar mechanism.
Ingestion does not only involve food. Risk assessment for contaminated land some-
times must take into account the ingestion of soil or other solids by children. A typical
assumption is that children eat 200 mg of soil per day.
The respiratory tract is the site of absorption by inhalati on for gases, such as
carbon monoxide, vapors of mercury, and high-vapor pressure liquids such as benzene
Membrane
Stomach
pH 2
Blood
pH 7.4
COO
−
COO
−
COOH
COOH
+ H
+
+ H
+
1
100
1
2512
Figure 18.2 How pH relationships in the stomach and blood plasma facilitate absorption of weak
acids such as benzoic acid.
744
FATE AND TRANSP ORT OF TOXINS
or perchloroethylene, as well as liquid aerosols and solid particles. Gas es and vapors are
absorbed by the large surface area of the alveoli. The absorptive flux will be largely
controlled by the aqueous solubility of the solute. Very soluble gases and vapors, such
as chloroform, are quickly absorbed, and the rate of absorption is effectively controlled
by the respiration rate. Such compounds are said to be ventilation limited. The absorption
rate for less soluble gases and vapors is controlled by blood flow to the lung. These
substances are thus said to be flow limited. The absorption of flow-limited toxins is also
affected by how completely the blood is cleared of the toxin as it passes through the
body before returning to the lungs. Toxins are removed from the blood in various ways,
including the storage of lipophilic substances in fatty tissue s and complexing of the
toxin with various receptors.
Particles larger than 10 mm tend to be trapped in the nose and expelled with muc us.
Particles smaller than about 0.01 mm remain effectively suspended in the airflow and
tend to be exhaled instead of affecting lung surfaces. Particles in the range between
these values tend to be deposited in various parts of the respiratory tract. The larger
ones are deposited in the upper respiratory tract, and then may be swallowed along
with mucus, entering the GI tract. Smaller particles go deeper, first to the trachea or
bronchi. Some of these may be transported to the upper respiratory tract by the mucocilia-
ry escalator, and either be coughed up or swallowed. Others are engulfed by phagocytes
and absorbed by the lymphatic system. Particles smaller than 1 mm may reach the alveoli,
where they may be absorbed into the bloodstream. Particles of cigarette smoke range
between 0.1 and 1.0 mm, and most are between 0.2 to 0.25 mm. Thus, they tend to be
deposited in the tracheobronchial airways and alveoli.
Particles may be a source of solid toxins such as the heavy metals or polynuclear
aromatic hydrocarbons present in smoke. For example, cigarette smo ke is a source of
cadmium as well as PAHs, not to mention nicotine. Furthermore, regular smoking para-
lyzes the mucociliary escalator, which otherwise would help clear some of the particles
from the lungs. Particles may also help transport gases or vapors into the lungs by adsorb-
ing them. An example of this is radioactive radon, which otherwise is not very easily
absorbed. It has even been suggested that one reason that cigarette smoke is such a potent
cause of lung cancer is that it carries radon, naturally present in the air, into the lungs.
The human lungs contain about 70 m
2
of surface area for absorption. Adults breathe
about 8 L of air per minute at rest. For risk assessment purposes it is common to assume
a breathing rate of 20 m
3
/day (a bit less than 14 L/min). In aquatic organisms the gills
play a role similar to that of the lungs. Gills have been shown to absorb the pesticide
dieldrin, and cadmium was observed to be taken up by passive diffusion.
The stratum corneum is the main barrier to uptake of toxic substances through the skin.
Once past it, the epidermis offers little further resistance. The toxin then can enter the
dermis, from which it can be transported to other organs by the blood. Hair follicles offer
a diffusion shunt through the epidermis. Thus, the scalp and male face absorb toxins
more readily than other areas do. The palms have a thicker stratum corneum, but it is
relatively porous and thus is not a good barrier for toxins. Abrasion removes the stratum
corneum, greatly increasing absorption. Some solvents, such as dimethyl sulfoxide
(DMSO), can help carry toxins through the skin. This is taken advantage of in drug deli-
very by means of transdermal patches. Toxins that are absorbed but do not penetrate the
epidermis become washed away, since the epidermis replenishes itself about once a
month. The stratum corneum is replaced about every 14 days. It seems that all toxins
that can be absorbed by the skin do so by passive diffusion.
ABSORPTION AND ROUTES OF EXPOSURE 745
The skin can biotransfo rm substances, often increasing their toxicity. Repetitive
exposure to coal tar induces the enzyme arylhydrocarbon hydroxylase (AHH) in the
epidermis. This and other enzymes convert PAHs into more carcinogenic forms. It is inter-
esting that people with psoriasis cannot induce AHH and do not seem to develop skin
cancer upon exposure to coal tar.
Examples of toxins absorbed through the skin include hexane, carbon tetrachloride,
and some insecticides. Toxins can also be absorbed by the eyes, although in that case it
is the eye as a target organ that is usually of concern. Various invertebrates have been shown
to take up toxins through their body surface, including arthropods and crustaceans such as
Daphnia. Molting has been shown to be a way to shed heavy metals. In addition, other
routes of exposure are used experimentally, such as injection subcutaneously, intramuscu-
larly, or into bo dy cavities.
Often, the amount of uptake is reduced because some of the toxic substanc e is bonded,
complexed, or limited by diffusion in the environmental source. The fraction that is avail-
able for uptake is called the bioavailability. For example, a fraction of some pesticides
can become so strongly bound to soil particles that they are practically unavailable. It may
be necessary to take multiple routes of exposure into account for some toxins. A worker
applying pesticides by spray may absorb by both inhalation and dermal contact.
18.4 DISTRIBUTION AND STORAGE
Once a toxin enters the bloodstream, it is distributed rapidly throughout the body. How-
ever, organs do not all receive equal distributions, nor do they store them with equal
efficiency. Moreover, the location where most of a toxin is stored is not necessarily the
primary site of toxic effect. For example, 90% of the lead in adul t humans is stored in
the bones, but its effects are on the kidney, nervous system, and blood cell production.
Individual organs and tissues will then take toxins according to the blood flow through
the organ (organ perfusion), their affinity for the toxin, and the presence of any transport
barriers. Most capillaries have large pores between the cells that form their wall. In some
tissues, however, there are few or no pores. The most notable case is the blood–brain
barrier. This prevents passage of polar compounds of medium molar mass. Its behavior
is similar to that of an intact plasma membrane in that it is permeable to nonpolar com-
pounds. Thus, mercuric chloride, which is mainly in ionic form, does not penetrate,
whereas methyl mercury does. Other tissues have barriers as well, including the peripheral
nerves, the placenta, the eyes, and the testes.
The major sites of storage for toxins are (1) bound to plasma proteins, (2) the liver and
kidneys, and (3) adipose tissue. Plasma proteins form complexes with many toxicants,
serving to solubilize and transport them. The effect of protein binding depend s o n how
the proteins compete with processes that detoxify or excrete them. If they give up the
toxicants readily, they may help to transport them to the detoxification site. If, on the
other hand, protein binding is relatively strong, it may sequester the toxins away from
detoxification.
Toxins are often concentrated in the liver or kidneys, possibly due to their role in
detoxifying and excreting them. They contain their own binding proteins. An example
is metallothionein, which figures in cadmium storage and in the transfer of cadm ium
from the liver to the kidney. Toxins absorbed in the stomach and intestines must pass
through the liver before reaching other parts of the body, thus giving that organ a chance
746 FATE AND T RANSPORT OF TOXINS
to biotransform them. Toxins that have been inha led or absorbed dermally may reach
other tissues before the liver.
Adipose tissue (also called depot fat) is the tissue where energy is stored in lipid form.
The cells in these tissues contain droplets of triglycerides occupying more than 90% of the
cytoplasm. Naturally, they tend to concentrate hydrophobic toxins. Even nonhydrophobic
compounds can be stored in lipids after being conjugated with fatty acids. Toxins can be
stored in adipose tissue without causing harm there. However, lipid storage of toxins has
resulted in acute toxicity in humans who had stored subacute doses in their fatty tissues,
later liberating an acute dose to the blood by depleting their fats in a weight-loss program.
The bones also store some toxins, notably lead, radium, and fluoride.
18.5 BIOTRANSFORMATION
Once inside an organism, many toxic compounds undergo chemical changes by biochemi-
cal reactions to form metabolites. Such biochemical changes are called biotransfor-
mation. Yet another distinction in types of toxicity is between those compounds that
react directly to cause damage and those in which metabolites are responsible for the
toxicity. In the former, biotransformation produces a reduction in toxicity; in the latter
it increases it. When biotransformation increases the toxicity of a chemical, the process
is called bioac tivation. Ultimately, biotransformation serves to produce more water-
soluble compounds that are more easily eliminated by the organism, or even to eliminate
them internally by mineralization to CO
2
and water. Chemicals that are not biotrans-
formed tend to be persistent in the environment.
An example of biotransformation increasing toxicity is sodium fluoroacetate (rat
poison 1080), which is not extremely toxic itself. However, by the mechanism lethal
synthesis fluoroacetate is converted to fluorocitrate by the same Krebs cycle pathway
that converts acetate to citrate. The enzyme for the next step combines with the fluoroci-
trate, but the reaction does not occur, and the fluorocitrate does not easily desorb from the
active site of the enzyme. This amounts to reversible inhibition of the enzym e, which
slows the Krebs cycle to a crawl.
In vertebrates, much of the biotransformation is done by the liver. For example, metha-
nol is oxidized in the liver. The oxidation by-products cause the symptoms of methanol
poisoning, which include heada che, back and abdominal pain, blurry vision, and poten-
tially blindness. A latency period of 8 to 36 hours may precede the onset of symptoms as
oxidation products accumulate. Because ethanol is metabolized by the same pathway,
methanol poisoning can actually be treated with ethanol. The presence of ethanol slows
the oxidation of methanol by competing for the pathway.
Of course, many biotransformation reactions occur in other tissues; these are called
extrahepatic. Whether in the liver or not, biotransformations can be either catabolic
(breakdown reactions), such as oxidation, reduction, or hydrolysis, or they may be ana-
bolic (synthesis) reactions, which are called conjugations. The catabolic reactions are
also called phase I reactions. They usually expose or add a functional group such as
OH, NH
2
, SH, or COOH. Hydrophilicity is increased, but only slightly.
The anabolic reactions are called phase II, to indicate that they often follow phase I.
The phase II reactions involve congugating (combining) the substrate with another
molecule, such as an amino acid, or by adding sulfate, methyl, or acetyl groups. These
groups are added to the functional groups added in phase I. Phase II reactions greatly
increase hydrophilicity, speeding excretion of the compound.
BIOTRANSFORMATION 747
