Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.


applicable to models that require numerical solution as well, is to set all derivatives (accu-
mulation terms) to zero. The differential equations then become algebraic and are solved
as such. Do not confuse steady state with equilibrium. As long as a net reaction is occur-
ring in a compartment, it may still be balanced by flux terms. For example, an organism
could be ingesting benzene on a daily basis and biotransforming it to phenol for excretion.
As long as the rate of ingestion equals the rate of reaction, there will be no accumulation
of benzene, and the system will be at steady state.
Equilibrium formally refers to a situation when the chemical potential of reactants
and products are equal for all reactions. In practical terms, equilibrium means that instead
of using the rate laws and mass transfer flux equations to describe the reactions, one sub-
stitutes equi librium relationships such as equation (5.9) for reaction equilibrium or equa-
tion (18.1) for mass transfer equilibrium. Just as steady state does not mean ‘‘no reaction,’’
equilibrium does not mean ‘‘no reaction.’’ For example, chloroform in respired air may be
assumed to be in mass transfer equilibrium with its concentration in blood plasma, yet a
continuous transfer of the solute will continue as long as the air is changed continually.
In the next several sections some simple compartment models are developed, both to
illustrate the modeling process and because they have several important features that are
used to describe the fate and transport of toxins in biological systems.
18.7.1 Dynamic Model and the Half-Life
The most basic model, of course, is the one-compartment model, in which the compart-
ment represents a whole organism. As a hypothetical case, consider how to model a fish
that ingests zooplankton contaminated with a hydrocarbon. Having decided on the one-
compartment model, we have finished the first step of the model development. We pos-
tulate only two processes: absorption by ingestion and elimination by kidney excretion.
Let us suppose that the hydrocarbon is biotransformed completely. We treat the hydro-
carbon and its metabolite as a single compound. Thus, it is not eliminated until the meta-
bolite is excreted. Finally, let us assume that the metabolite is removed by the kidney by
glomerular filtration only and is not reabsorbed. Thus, the rate of excretion, r
e
, will be
negatively proportional to the concentration in the blood plasma:
r
e
¼k
e
C ð18:19Þ
where C is the concentration of hydrocarbon plus metabolite and k
e
is a coefficient related
to the renal clearance rate. Note that this mass transfer process can be formulated as a rate
instead of a flux. The same will be true for ingestion. The rate of absorption, r
a
, is the pro-
duct of the assimilation efficiency, a (the fraction of ingested toxicant that is absorbed),
the mas s of food ingested per unit time, W, and the average concentration of toxican t in
the food, C
f
(in units of mass of solute per unit mass of food):
r
a
¼ aWC
f
ð18:20Þ
This situation is shown schematically in Figure 18.8. Equations (18.19) and (18.20),
having units of [Mt
1
], must be multiplied by the volume before substitution into equa-
tion (18.13) along with equation (18.14). Canceling the volume yields
dC
dt
¼ aWC
f
k
e
C ð18:21Þ
758 FATE AND T RANSPORT OF TOXINS

For the initial condition that C
i
¼ 0:0att ¼ 0:0, equation (18.21) can be solved
analytically:
C ¼
aW
k
e
C
f
ð1 e
k
e
t
Þð18:22Þ
This represents a period of increasing body burden (the mass of contaminant contained
by an organism), as shown in Figure 18.8. If the contamination in the food supply were
suddenly eliminated, uptake would cease, and the only proce ss would be elimination. In
aquatic biology, this situation is called depuration. Depuration is used commercially
to eliminate contaminants from shellfish grown in polluted waters by transferring them to
clean waters for a period prior to marketing. The model of equation (18.21) applies, with
C
f
set to zero. This leaves the following:
dC
dt
¼k
e
C ð18:23Þ
This is a first-order decay model. The solution, starting from an initial concentration of
C
i
,is
C ¼ C
i
e
k
e
t
ð18:24Þ
Thus, first-order decay results in an exponential decrease in concentration. This has
the important property that the concentration decreases by equal fractions in equal time
intervals. For example, the time it takes for the concentration to drop from 100 mg/L to
50 mg/L is the same as the time to go from 2.0 mg/L to 1.0 mg/L. The time it takes for
the concentration to drop by half is called the half-life (t
1=2
). It is used as a convenient
index of how rapidly a toxin is eliminated. The rate coefficient gives the same information
but does not have such a tangible interpretation. In fact, the half-life and the rate coeffi-
cient can be computed from one another. From equation (18.24), using C=C
i
¼ 0:5 yields
t
1=2
¼
lnð0 :5Þ
k
e
¼
0:693
k
e
ð18:25Þ
0
1
0246
Time
Body Concentration
Uptake
Depuration
Exposure to
ambient pollution
Exposure
terminated
Steady-state exposure conc.
C
C
f
k
e
α
W
Figure 18.8 Schematic of a one-compartment model, and a plot of uptake and depuration of a
contaminant in the case of a fixed environmental concentration C
f
.
PHARMAKOKINETIC MODELS 759

Other characteristics times, such as t
90
, the time required for a 90% reduction, could be
computed in a similar way. Keep in mind that the half-life and similar parameters can be
used only if the elimination can be approximated as first order. (See Problem 18.3 for an
example where this is not the case.) Conveniently, this is true quite often, and half-lives
have been measured for a wide variety of compounds.
Example 18.6 Predatory animals living in a contaminated ecosystem are found to have
a tissue concentration of a toxin equal to 80 mg toxin per kilogram of tissue. Samples of
their usual prey were found to have a body burden of 10 mg/kg. Several animals are taken
into captivity and fed food without the toxin. After 100 days of this treatment, the body
burden is found to have decreased by 40%. Assuming that the animals were at a steady-
state body burden when collected and that the assimilation efficiency equals 1.0, est imate
the elimination rate coefficient, the half-life, and the mass loading rate in the wild.
Answer Rearranging equation (18.24) gives us k
e
¼lnðC=C
i
Þ=t ¼ 0:511/(40 days) ¼
0.0128 day
1
. From equation (18.25), t
1=2
¼ 0:693=k
e
¼ 0:693=ð0:013 day
1
Þ¼54:3days.
Assuming steady state and that a ¼ 1:0, equation (18.21) becomes 0 ¼ WC
f
k
e
C
ss
.
Therefore,
W ¼ k
e
C
ss
C
f
¼ð0:0128 day
1
Þ
80 mg toxin
kg body weight
kg food
10 mg toxin
¼ 0:102
kg food=day
kg body weight
The animal must eat one-tenth its body weight each day to maintain the stated
body burden.
18.7.2 Steady-State Model and Bioaccumulation
The uptake and retention of contaminants in tissues in excess of concentrations in
the source of the contaminant (such as food or water) is called bioaccumulation.In
Figure 18.8, uptake is being held constant, but elimination increases as concentration
increases. A situation is approached asymptotically where elimination balances uptake,
and the system will be at steady state. The concentration at which this will occur can
be determined in two equivalent ways, as mentioned above. One would be to take the
limit of equation (18.22) as t !1. The other is to set equation (18.21) to zero and solve
for C. Either way gives the same result for the steady-state concentration, C
ss
. The ratio of
C
ss
to C
f
is the bioaccumulation factor, K
B
(also designated BAF):
K
B
¼
C
ss
C
f
¼
aW
f
k
e
ð18:26Þ
Bioaccumulation can occur with other routes of exposure. One would develop differ-
ent models for K
B
if this were the case. In this case, since C
ss
is expressed per unit body
weight and C
f
is per weight of food, K
B
will depend on the mass of food ingested per day
per body weight. The caloric requirement of organisms is related to their surface area;
therefore, smaller animals tend to have higher bioaccumulation by ingestion (although
other routes of exposure may be more important). It can also depend on other
things that influence metabolic rate, such as stress or temperature. If elimination occurs
760 FATE AND T RANSPORT OF TOXINS

predominantly by interphase mass transfer, as is often the case with xenobiotic com-
pounds, k
e
will be inversely related to the partition coefficient, making K
B
directly related.
This accounts for the observation that bioaccumulation is strongly correlated with
K
OW
, especially if the concentration in fatty tissues is used in place of whole body
concentration.
Example 18.7 In Example 18.6, what is the bioaccumulation factor?
Answer K
B
¼ 80 mg/kg/(10 mg/kg)¼ 8; also, K
B
¼ aW
f
=k
e
¼ð1:0Þ (0.102 day
1
)/
0.0128 day
1
¼ 8.
18.7.3 Equilibrium Model and Bioconcentration
Other mechanisms can give proportional bioaccumulation relationships. For example,
consider a one-compartment model in which both uptake and elimination are by mass
transfer between the environmental concentration, C
env
, and the concentration within the
organism, C
org
. For example, this might apply to the uptake of hydrocarbons from
the air by terrestrial animals or from the water for aquatic animals. In such cases, uptake
and elimination could be described by equation (18.12), with mass transfer coefficients k
u
and k
e
for the respective rates r
u
and r
e
:
r
u
¼ k
u
ðK
P
C
env
C
org
Þ
r
e
¼k
e
ðK
P
C
env
C
org
Þ
ð18:27Þ
Assuming steady state, the mass balance leads to the relationship that the rate of uptake
equals the rate of elimination:
r
u
¼ r
e
ð18:28Þ
The process in which bioaccumulation occurs by direct uptake from the environment
is called bioconcentration. It is distinguished from bioaccumulation in that bioaccu-
mulation includes all routes of exposure, whereas bioconcentration only considers uptake
directly from the environmental medium in which the organism lives. The term biocon-
centration is usually reserved for aquatic systems.
Equations (18.27) and (18.28) can be combined and solved for the ratio of organism
concentration to environmental concentration, called the bioconcentration factor (K
C
,
also designated BCF). In this case,
K
C
¼
C
org
C
env
¼
k
u
K
P
þ k
e
K
P
k
u
þ k
e
¼ K
P
ð18:29Þ
Thus, for this model the bioc oncentration factor is equal to the partition coefficient.
Again, it must be emphasized that both the bioaccumulation factor and the bioconcentra-
tion factor can be derived from different models using different assumptions. For example,
if we assume simple first-order rate processes for uptake and elimination:
dC
org
dt
¼ k
u
C
a
k
e
C
org
ð18:30Þ
PHARMAKOKINETIC MODELS 761

where in this case k
u
and k
e
are the uptake and elimination rate coefficients, respectively,
then the bioconcentration factor would be written as
K
C
¼
k
u
k
e
ð18:31Þ
Thus, the bioconcentration and bioaccumulation factors are not defined uniquely in
terms of a particular model. More generally, they are defined as the ratio of organism
concentration to the concentration in the environment and/or the food supply.
Whatever the theoretical mechanisms are to explain the bioconcentra tion factor,
experimental measurements have shown that it correlate s with K
OW
. For example, mea-
surements with 64 organics in fish have been used to develop the following empirical
relationship:
log
10
K
C
¼ 0:76 log
10
K
OW
0:23 r
2
¼ 0:82 ð 18 :32Þ
Relationships such as this should be used cautiously, however. Although they are useful
over a wide range of K
OW
, at a particular values the upper and lower 95% confidence
levels can differ by a factor of 63. Furthermore, numerous correlations of this form
have been developed for different groups of chemicals and different species. Some studies
have correlated K
C
with aqueous solubility instead of K
OW
. Several of these physicochem-
ical parameters are given in Table 18.5.
A more complete model would separately take into account the processes of environ-
mental uptake, ingestion, biotransform ation, and excretion:
dC
org
dt
¼ aWC
f
þ k
u
C
env
k
e
C
org
ð18:33Þ
Assuming steady state, rear ranging, and substituting for K
C
and K
B
from equations
(18.26) and (18.31) shows the contribution to body burden by environmental uptake
and ingestion:
C
org
¼ K
C
C
env
þ K
B
C
f
ð18:34Þ
TABLE 18.5 Bioconcentration Factors and Physicochemical
Parameters for Some Compounds That Bioaccumulate
Compound Log K
OW
Log
10
K
C
C
S
(mg/L)
Chlordane 5.51 4.05 0.056
DDT 5.98 4.78 0.0017
Dieldrin 5.48 3.76 0.022
Lindane 4.82 2.51 0.150
Chlorpyrifos 4.99 2.65 0.3
Cycexatin 5.38 2.79 <1.0
2,4-D 1.57 1.30 900
2,4,5-T 0.6 1.63 2.38
TCDD 6.15 4.73 0.0002
Source: LaGrega et al. (2001); original source D. W. Connell (1990),
Bioaccumulation of Xenobiotic Compounds, CRC Press, Boca Raton, FL.
762 FATE AND T RANSPORT OF TOXINS
Note that K
B
in this case should be interpreted as an accumulation factor for ingestion
alone. More generally, bioaccumulation includes all sources of contaminants.
18.7.4 Food Chain Transfer and Biomagnification
A bioaccumulation mode l such as equation (18.34) can be applied separately to each
trophic level in an ecosystem, with the food at one level being the organism concentration
at the lower level:
C
i
¼ K
C;i
C
env
þ K
B;i
C
i1
ð18:35Þ
Here C
i
is the concentration of toxin in the organisms of trophic level i, C
i1
is the con-
centration of the next-lower trophic level, K
Ci
is the bioconcentration factor for trophic
level i, and K
Bi
is the bioaccumulation factor for trophic level i.
For example, suppose that the environmental concentration of a toxic substance is
1 mg/kg and K
C
¼ K
B
¼ 10 for all trophic levels. The primary producers will have a con-
centration of 10 mg/kg due to bioconcentration alone. The herbivores will have C ¼
10 1 þ10 10 ¼ 110 mg/kg. The concentration in carnivores will be C ¼ 10 1 þ
10 110 ¼ 1110 mg/kg. Secondary carnivores will have a concentration 11,110 mg/kg.
An increase of tissue concentration of a toxic substance with increasing trophic level is
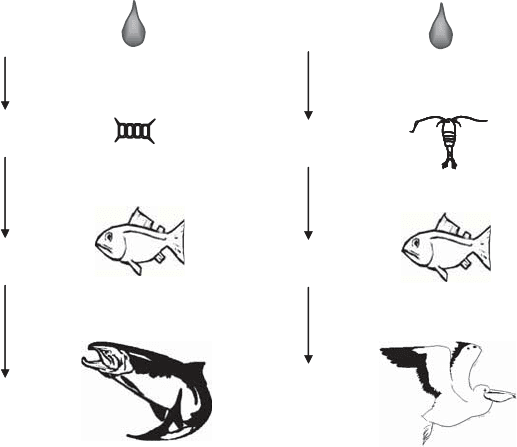
called biomagnification. Figure 18.9 illustrates the relationship for two food chains. Sub-
stances that bioaccumulate also tend to biomagn ify. Thus, the tendency to biomagnify is
usually also related to the K
OW
. However, a low rate of elimination, especially by bio-
transformation, is also necessary. These conditions are particularly true for chlorinated
organics. Biomagnification can produce extremely high body burdens in organisms at
Water
0.02 ppm
Plankton
5.3 ppm; x265
Small fish
10.0 ppm; x500
Predator fish
1,700 ppm; x85,000
Pesticide: DDD
Pesticide: Toxaphene
Water
0.2 ppm
Planktonic crustaceans
73 ppm; x365
Goldfish
200 ppm; x1,000
Pelican
1,700 ppm; x8,500
Figure 18.9 Biomagnification of two organochlorine pesticides in aquatic food chains.
PHARMAKOKINETIC MODELS 763

the top of the food chain, the secondary carnivores or predators. The bioaccumulation
factor (and therefore, biomagnification) can be related to the bioconcentration factor by
a food chain multiplier (FM):
BAF ¼ FM BCF ð18:36Þ
The U.S. EPA recommends using food chain multipliers based on K
OW
and trophic
level as shown in Table 18.6. At log K
OW
values higher than those in the table, the FM
becomes uncertain and may even decrease due to slow transport kinetics and bioavail-
ability. Thus, if log K
OW
is 5.0, the bioaccumulation factor for carnivores (trophic
level 3) will be about twice the bioconcentration factor.
Because organochlorine pesticides are not only lipophilic and persistent but also toxic
by their nature, they are the substances most capable of causing harm by biomagnifi-
cation. Furthermore, biomagnification limits the effectiveness of pesticides. Since in
agriculture pesticides are often used to control plant pests, they are applied at levels
toxic to the base of the food pyramid. If the pesticide biomagnifies, this will neces sarily
result in more harm to predatory insects, which are the ones needed to control the pests.
Thus, ironic ally, it was sometimes found that applications of these substances resulted in
increased pest problems. For these reasons, organochlorine pesticides have been replaced
in many applications by other pesticides that do not biomagnify.
18.7.5 Multicompartment Models
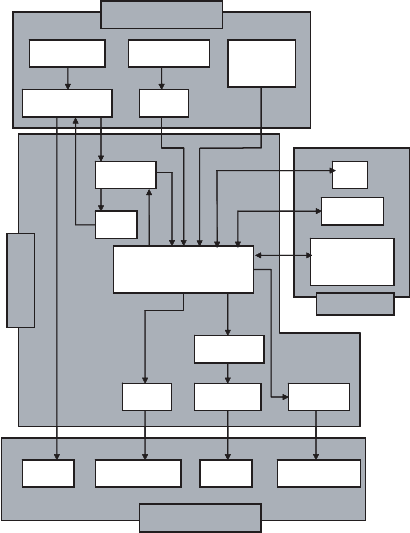
If sufficient information is available, or if contaminant behaviors that are more complex
are to be studied, multiple-compartment models can be used. For example, Figure 18.10
shows a model that could be used to predict narcosis by inhalation of a hydrocarbon. A
separate mass-balance equation must be written for each compartment. Each arr ow
represents a flux, and a term must be added to the liver compartment for elimination
by biotransformation. Although it may not be possible to define a half-life or a bioaccu-
mulation factor for some of the more complex multicomponent systems, their behavior
often may be approximated by one of those parameters.
TABLE 18.6 Thoman’s Food Chain Multipliers
Trophic Level
Log K
OW
23 4
3.5 1.0 1.0 1.0
4.0 1.1 1.0 1.0
4.5 1.2 1.2 1.2
5.0 1.6 2.1 2.6
5.5 2.8 5.9 11.0
6.0 6.8 21 67.0
6.5 19 45 100.0
Source: U.S. EPA (1994).
764 FATE AND T RANSPORT OF TOXINS
18.8 EFFECT OF EXPOSURE TIME AND MODE OF EXPOSURE
Toxic effects can be affected by the time distribution of exposure. Consider three cases:
(1) a single, instantaneous exposure; (2) a continuous, chronic exposure; and (3) periodic
or intermittant exposures. The difference between these modes will depend on the type of
effect, such as whether it is a local or a systemic toxin, whether or not there is a threshold,
and whether the damage can be repaired or accumulates. If there is a threshold, or if the
effect depends on the concentration of the toxicant or a metabolite in a compartment,
the effect would be correlated with the concentration in the compartment containing the
receptor. If the damage accumulates, an effect could be seen even at a low dose if suffi-
cient prior doses were applied. Here we might expect the effect to correlate with the time
integral of the concentration in the compartment.
For the first case, Figure 18.11 shows how concentr ation will vary for an instanta-
neous dose in a two-compartment model. The first, or systemic, compartment, peaks
immediately. This is what would be found in the bloodstream if a substance were to be
injected intravenously by hypodermic syringe, for example. The systemic compartment
then clears by some process, such as the first-order process illustrated. The second com-
partment represents one containing a receptor, such as neurons in the brain. It exchanges
toxicant only with the systemic compartment. Its concentration increases until it exceeds
that in the first compartment. Then it decreases slowly by exchange with the first compart-
ment. Here there would not be a great difference with type of effect, except that integrated
Absorption
Excretion
Storage
Bile
Liver
LungGI tract
Dermal
or eye
InhalationIngestion
ExhalationFeces
Bones and
tissues
Organs
Fat
Kidneys
BladderLung
SecretionsUrine
Blood and
Lymph
Glands
Distribution
Absorption
Excretion
Storage
Bile
Liver
LungGI tract
Dermal
or eye
InhalationIngestion
ExhalationFeces
Bones and
tissues
Organs
Fat
Kidneys
BladderLung
SecretionsUrine
Blood and
lymph
Glands
Distribution
Figure 18.10 Multicompartment model of an animal.
EFFECT OF EXPOSURE TIME AND MODE OF EXPOSURE 765

effects would take longer to appear. Biotransformation reactions are neglected in these
examples. If the system behaved more like a one-compartment model, the toxicant
would be cleared in a depuration process similar to that shown in the second part of
Figure 18.8.
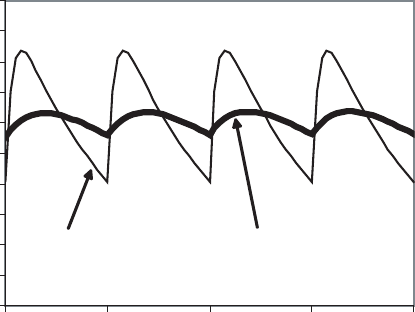
Figure 18.12 shows a similar plot for the second two cases. Intermittent exposure
results in higher peak concentrations, which may cause a greater effect either if the effect
is concentration associated or if there is a threshold. On the other hand, the periodic
decrease in concentration may enable repair mechanisms to eliminate previous damage.
This has been observed in exposing rats to ozone. A concentration that caused pulmonary
edema when applied continuously had no effect when applied intermittently. However, if
damage is cumulative, the integral of both curves will be similar, as will be their effect.
An example is the application of organophosphate pesticides, which cumulatively deplete
acetocholinesterase. There is a repair mechanism, in the form of hydrolysis of the
enzyme-OP compound and replacement of the enzyme, but it can be overwhelmed at
moderate doses.
0.0
0.2
0.4
0.6
0.8
1.0
01234
C
2
C
1
Systemic
component
C
1
Peripheral
component
C
2
k
e
K
p
Time
Concentration
dC
1
/dt = {K
p
*(C
2
-C
1
) - k
e
*C
1
} / V
1
dC
2
/dt = {-K
p
*(C
2
-C
1
)} / V
2
Figure 18.11 Typical concentration–time plot for two-compartment model. The systemic
compartment typically represents blood concentration. The peripheral compartment can represent
another tissue or organ, such as the brain or muscle. C and V are the concentration and volume,
respectively, of the compartments. In this simulation k
e
; K
p
, and V
2
¼ 1:0, and V
1
¼ 3:0.
0.0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.0 2.0 4.0 6.0
Time
Concentration
Figure 18.12 Concentration–time plot for continuous and intermittent exposure.
766
FATE AND TRANSP ORT OF TOXINS
Finally, the same dosing regimen could result in very different concentration–time
relationships, depending on the pharmacokinetics involved. Slow adsorption and elimina-
tion or the presence of a large storage compartment could smooth out changes in con-
centration. In Figure 18.13 the concentration in the peripheral compartment does not
vary greatly when the two compartments have the same volume (V
1
¼ V
2
¼ 1:0). But
when the peripheral compartment is much smaller (V
2
¼ 0:1), large concentration swings
occur.
PROBLEMS
18.1. The OSHA eight-hour inhalati on standard for phenol is approximately 19 mg/m
3
.
What is this in ppmv?
18.2. The OSHA eight-hour inhalation standard for chloroform is 50 ppmv, and its
Henry’s constant is 0.148 (conc./conc.). If wastewater flowing through a sewer was
contaminated with 0.50 mg of chloroform per liter, the air space in the sewer and
in the manholes would eventually achieve equilibrium with the chloroform in the
water. Would the air concentration of the TCE in the manhole exceed the OSHA
limit?
18.3. Use equations (18.7) and (18.32) to estimate the octanol–water partition coeffi-
cient and the bioconcentration factor of chlordane. Compare to the values given in
Table 18.5.
18.4. Ethyl alcohol is eliminated from the body by a zero-order process. That is, it
is eliminated by a constant mass per unit time. Assume that the removal rate is
0.0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1.0
01234
Time
Concentration
V
2
= 0.1 V
2
= 1.0
Figure 18.13 Effect of differing pharmacokinetics on the time course of concentration in an
organism. k
e
, K
p
, and V
1
¼ 1:0, and V
2
¼ 1:0 or 0.1 as indicated.
PROBLEMS 767
