Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.


1 Electrophoretic Techniques70
1.5.2.1 Lysine Labeling (Minimal Labeling)
It must be avoided to block all lysines of the proteins with a label,
because the proteins would become very hydrophobic and would not
stay in solution. Therefore only a small portion of the proteins is
labeled. In practice only 400 pmol/L of dye is added to 50 mg of pro-
tein. In this way only 3 to 5% of the proteins will receive a tag; the
other 95–97% proteins remain unlabeled. This measure, which is
called “minimal labeling”, prevents also the appearance of multiple
labels: the occurrence of multiple labels on proteins with several
lysines will statistically be so low that these molecules are below the
limit of detection. Overlabeling with minimal dyes could be recog-
nized very easily in the false color image (see above): Two and more
dye molecules per protein would cause vertical streaking due to
increased molecular weight of a part of the molecules of the labeled
protein.
It is, however, very important that these dyes are charge and size
matched. Therefore a basic buffering group is added to each of the
dyes, thus the isoelectric points are not changing. Also in the second
dimension the proteins migrate to the same point independently of
their label, because the molecular weights added are in the range
between 434 and 464 for the three dyes Cy2, Cy3, and Cy5. This little
difference cannot be resolved by SDS electrophoresis. The 2-D pat-
tern obtained with minimal labeling does not differ from a pattern of
post-stained 2-D gels of non-labeled proteins.
The sensitivity of minimal labeling is comparable to good quality
silver staining, however exhibiting a much wider dynamic range and
linearity. It is very rare that a protein does not pick up a label or does
not contain any lysine. To be on the safe side it is recommended to
stain a gel with a sensitive fluorescent dye to detect such proteins.
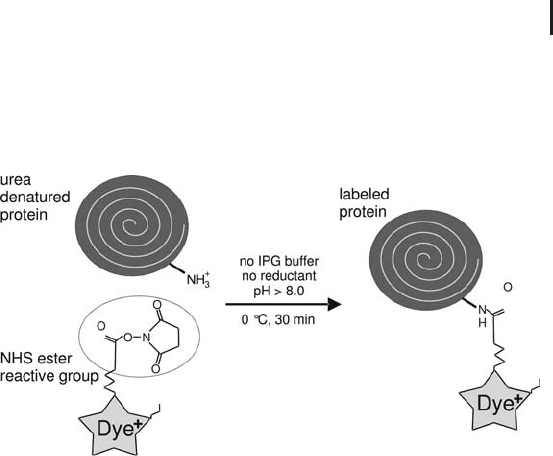
In order to make the method reliable and reproducible, the labeling
has to be performed under optimum conditions and needs to be well
controlled (see Figure 1.19). The proteins are solubilized and dena-
tured with high molar urea or thiourea/urea. The optimal concentra-
tion of the protein lysate is between 5 and 10 mg/mL. But also sam-
ples containing from 1 mg/mL to 20 mg/mL have been successfully
labeled. If the sample is too diluted, labeling will be less efficient.
Lysine labeling is performed on ice and in absence of IPG buffers or
carrier ampholytes and reductants: The primary amino groups of
amphoteric buffers would be labeled as well; the reductant would
interfere with the reaction. These compounds are added after labeling
had been completed. A sample pH value above pH 8.0 is essential,
the optimum lies at pH 8.5. Therefore the sample solution should
always contain 30 mmol/L Tris-base. Samples, which have been
In downstream analysis the
non-labeled peptides are in a
great majority versus the
labeled ones, therefore only the
non-modified peptides will be
detected with mass spectro-
metry.
In the lower molecular weight
area of the SDS gel an offset of
non-labeled from labeled
protein spots can be observed.
Therefore, when spot picking is
intended, it is recommended to
stain the gel after scanning with
a fluorescent dye and match
the spots with the labeled spots
using the spot matching algo-
rithm of the evaluation soft-
ware.
Underlabeling is not an issue: if
more protein is labeled with the
same amount of dye, the
scanned image will be the
same. However, when proteins
are overlabeled, multiple labels
can become visible, particular
in the cathodal area of the 2-D
gel.
The distribution of dyes is
uniformly and quantitative
across all the different proteins.
1.5 Two-dimensional Electrophoresis 71
extracted with TCA acetone or have been cleaned up with precipita-
tion, are often very acidic. In this case it can be necessary to adjust
the pH value by adding 50 mmol/L NaOH solution. The reaction is
carried out on ice for 30 minutes and stopped by adding a 0.1% (w/v)
lysine solution.
Fig. 1.19: Simplified diagram of lysine labeling for DIGE.
No derivatization is needed, because the e-amino group of
the lysine is readily accessible.
Subsequently the samples are combined and either submitted
directly to 2-D electrophoresis or stored in a deep freezer until use.
1.5.2.2 Cysteine Labeling (Saturation Labeling)
In contrast to lysine labeling here all available cysteines will be
tagged. Labeling the cysteines is pH neutral and does not increase
the hydrophobicity. However the resulting 2-D electrophoresis pat-
tern looks different from those originating from minimal labeled or
non-labeled proteins: proteins with many cysteines will pick up many
labels, which results in slower migration in the SDS gel and a much
stronger light emission signal. The increase of molecular weight is
650 Da per label. Because of multiple labels, here it is particularly
important that the dyes are size matched. Not all proteins contain
cysteine. It is estimated that in human, animal, and plant proteomes
about 95% of the proteins possess at least one cysteine, and can there-
fore be detected with this method.
The sensitivity of this method is very high. The application of down
to 1,000 human cells (equal to 2.5 mg protein) provides still very good
2-D gels, as shown in a paper by Sitek et al. (2005).
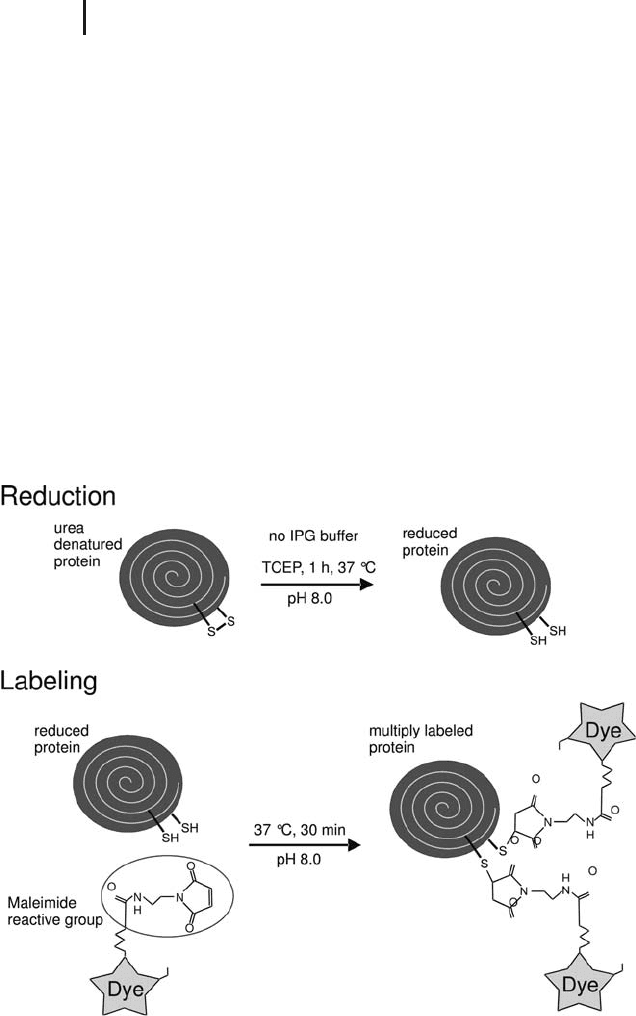
Cysteine labeling requires more work than lysine labeling. First the
disulfide bridges are cleaved with the reductant TCEP, thereafter the
However, there are bacteria
species, which do not contain a
single cysteine. Needless to say,
the method cannot be applied
for those.
Sitek B, Lttges J, Marcus K,
Klçppel G, Schmiegel W, Meyer
HE, Hahn SA, Sthler K.
Proteomics 5 (2005) 2665–
2679
1 Electrophoretic Techniques72
dye with the maleimide reaction group is added (see Figure 1.20).
The temperature is higher and the reaction time is longer than for
minimal labeling in order to achieve complete labeling of all
cysteines. To avoid bleaching of the dyes these steps are therefore per-
formed in the dark. Furthermore, some optimization for certain sam-
ple types may be necessary, because the quantitative amount of
cysteines in the protein mixture cannot be predicted. If more dye had
been applied than cysteines were available, non-specific labeling of
some lysines can be observed, indicated by a pI shift of some spots
towards the acidic side. If there was not enough dye for all the
cysteines, some proteins will partly have lower molecular sizes and
will migrate a little faster, resulting in vertically elongated spots or
vertical streaks. The optimal concentrations can be assessed by a
same–same experiment with different label concentrations (see also
Sitek et al. 2005). The applied ratio of reductant to dye is constant.
For this method only the two dyes Cy3 and Cy5 can be used,
because Cy2 does not work properly for this application.
Fig. 1.20: Simplified diagram of cysteine labeling for DIGE.
The more cysteinyls are available after reduction, the more dye
molecules will become attached to a protein molecule.
This technique is mainly
applied for very scarce samples.
Here is one of the major differ-
ences between minimal
labeling and cysteine labeling:
overlabeling causes vertical
streaks in minimal labeling, but
horizontal displacement in
cysteine labeling.
Cy2 shows too strong self-
quenching effects, when dye
molecules are in close proximity
to each other.
1.5 Two-dimensional Electrophoresis 73
For downstream analysis the spots could in principle be picked
directly from the image file (see spot picking below). However,
because the method is so sensitive, there will not be enough protein
material in a spot for mass spectrometry analysis. Therefore a pre-
parative 2-D gel of a reference proteome loaded containing about
300 mg protein has to be run. Also for this gel the cysteines must
carry a label; otherwise the spots to be excised cannot be matched
with the spots in the analytical gel. The selection of the optimal refer-
ence proteome, which should show the most similarities with the
analytical sample, is dependent on the type of sample. Labeling of the
preparative sample proteins is performed with Cy3.
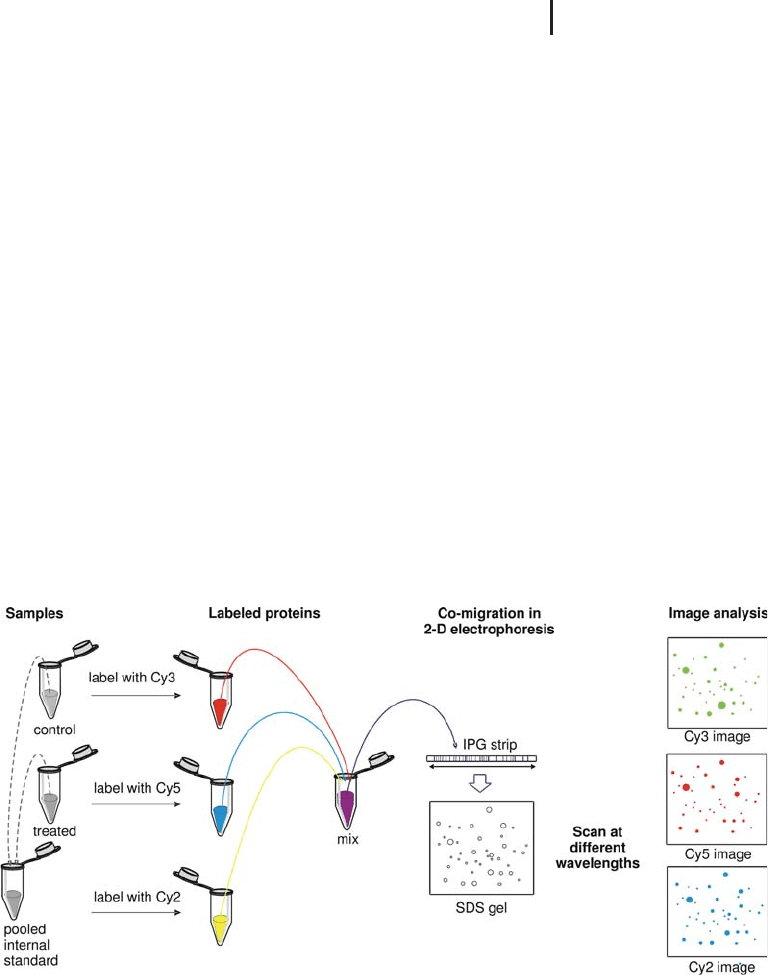
1.5.2.3 The Internal Standard
The multiplex feature of DIGE offers the unique possibility to reserve
one channel for an internal standard. Some papers are describing the
application of a mix of labeled commercial standard proteins. How-
ever, by far the greatest effect is obtained, when the internal standard
is a pool of aliquots taken from each sample of an experiment, and
labeling this mixture with one of the dyes. This concept is shown in a
simplified schematic diagram in Figure 1.21. Of course, “control” and
“treated” can be replaced by “healthy” and “diseased” or by “wild type”
and “mutant”.
Fig. 1.21: Simplified schematic representation showing how to
create a pooled internal standard in difference gel electrophoresis
(DIGE). The sample proteins are labeled with Cy3 and Cy5, the
internal standard with Cy2. From Westermeier and Scheibe (2007).
Because in this case all
cysteine-containing proteins are
modified, the molecular weight
added to the cysteines needs to
be entered into the database
search definitions, analogous to
alkylation of cysteines.
Westermeier R, Scheibe B. In
Posch A. Ed. Sample Prepara-
tion for 2D PAGE. Methods in
Molecular Biology, Humana
Press, Totowa, NJ (2007) in
press.

1 Electrophoretic Techniques74
In reality it makes no sense to run only two samples in an experi-
ment. As already mentioned in the introduction, in order to find sig-
nificant protein changes with acceptable statistical confidence, at least
three biological replicates are required. This means that the internal
standard will be composed of aliquots from six samples: three con-
trols plus three treated. It is important to create enough pooled inter-
nal standard to be able to apply it on every gel. With minimal labeling
each gel contains two samples and one standard, with saturation
labeling only one sample and the standard are run together.
Because the proteins are co-migrating in the gel, no spot matching
is needed within the gel. The sample spot volumes can directly be
compared to the spot volumes of the standard, which is the mixture
of all samples. The dedicated software, DeCyder, does this automati-
cally, employing a co-detection algorithm. The result is expressed in
spot volume ratios. For the comparison of several gels the spot match-
ing procedure is highly facilitated, because the images of the standard
are matched, which originate from the identical protein composition.
In this way it is possible to perform a fully automated evaluation of a
high number of images. Furthermore image analysis can be carried
out fully automatically with a batch processor, which reduces the user
bias to a minimum.
& Note: the result issued by DeCyder consists of
spot volume ratios to the internal standard
instead of absolute spot volumes. In this way
artifactual spot volume variations caused by gel-
to-gel variations are eliminated.
The use of the pooled internal standard makes the evaluation of the
results much easier and considerably more reliable:
.
Every protein in the population must appear on
the standard image in each gel, otherwise it will
not be included in the result.
.
The sample spot volumes are normalized against
the internal standard.
.
Variations of spot volumes due to gel-to-gel varia-
tions are eliminated.
.
Each sample is compared internally with the
same standard.
.
It enables fully automated accurate spot statis-
tics.
.
Gel-to-gel matching becomes much easier.
When minimal labeling is
employed, the standard is
always labeled with Cy2, for
saturation labeling it is Cy3.
All this will be described in
more detail in the chapter on
image analysis.
Artifactual spot volume varia-
tion can be caused by slight
different conditions during
sample application.

1.5 Two-dimensional Electrophoresis 75
1.5.2.4 Experimental Design
First of all, the pooled internal standard should be labeled with one
and the same dye within an experiment. For spot matching it is
important that the conditions for the internal standard are identical.
In order to obtain statistically significant results, the distribution of
the labels among samples has to be well planned. For instance, any
preferred labeling of certain proteins by one of the dyes must be sta-
tistically excluded. If the experiment consists only of a small number
of samples, it is recommended to run the samples two times, but
with inverse labeling. For larger experiments, no replicates are
required, but a “planned” randomization of the labels and sample
application is proposed:
.
The two available dyes should be evenly distribu-
ted between treated sample and control.
.
Treated sample and control from separate origins
should be applied to each gel.
An example for randomization of sample application and inverse
labeling is shown in Table 3.1 on page 309.
To minimize methodical variation all samples of an experiment
should be run together in one instrument in the first and also the sec-
ond dimension.
Typically the amount of three times 50 mg protein is applied, which
means 150 mg total sample load. Because the detection is very sensi-
tive, it is possible that a small spot does not contain enough protein
for mass spectrometry analysis. If more protein is required in a gel, it
is suggested to spike non-labeled pooled standard into the 150 mg
labeled sample to the desired protein amount.
It should also be mentioned that the DIGE technique is very useful
for checking the quality of sample preparation:
.
First the same sample is labeled with two differ-
ent dyes and separated in one gel;
.
In a second step three sample preparation repli-
cates of the same sample source labeled with
three different dyes are run in a gel.
1.5.2.5 Major Benefits of 2-D DIGE
In short the advantages of the DIGE approach over conventional 2-D
electrophoresis are ranked in the order of their importance to prac-
tice:
.
It saves time: differentially expressed proteins
are detected very rapidly.
For 3-color experiments the
standard is labeled with Cy, for
2-color experiments with Cy3.
The statistics algorithms of the
DeCyder software take these
measures in calculation.
Experiments with more than 12
gels at a time are more difficult
to evaluate.
The original suggestion to run a
separate preparative gel is less
practical.
Some samples contain protein
individuals with peculiar beha-
vior. Those are detected with
this strategy.
These points have been ranked
according the comments of an
adequate number of DIGE
users.
No gel replicates, automated
image analysis.

1 Electrophoretic Techniques76
.
It saves work: a smaller number of gels has to be
run, because samples are combined on gels, and
no gel replicates are necessary.
.
It eases handling of gels: the gels are analyzed
while they are still in the cassettes.
.
It simplifies the evaluation: image analysis with
DeCyder software runs automatically and is sim-
ple to carry out.
.
It improves the quality of the results: gel-to-gel
variations are eliminated from the result because
it measures spot volume ratios relative to the
pooled internal standard, which is included in
each gel.
.
It delivers quantitative values: the internal stan-
dard and the wide linear dynamic range of the
fluorescent dyes allow the quantitative measure-
ment of minor changes of expression levels with
high statistical confidence.
.
It affords very sensitive detection: with cysteine
labeling samples containing less than 1 mg total
protein can be analyzed with 2-D electrophor-
esis.
The DIGE concept is not limited to high resolution 2-D electrophor-
esis with immobilized pH gradients: it has also been successfully
applied on 2-D gels based on carrier ampholytes IEF (Sitek et al.
2006), blue native electrophoresis (Perales et al. 2005), and acidic elec-
trophoresis with cationic detergent (Helling et al. 2006).
1.5.2.6 Specific Labeling of Cell Surface Proteins
Usually the proteins of cell or tissue lysates are labeled. It is, however,
possible to label the proteins on the surface of the intact cell (Mayr-
hofer et al. 2006). In a GE Healthcare Application Note (2005) it is
shown that after cell lysis and 2-D electrophoresis only the specifically
labeled surface proteins are visualized. The proteins inside the cell do
not become labeled, because of unfavorable conditions: the pH value
in the cytosol of an intact cell is usually below pH 7.4; the labeling
time is short and performed on ice. This approach is much easier to
carry out than using the standard protocol of biotin labeling. As
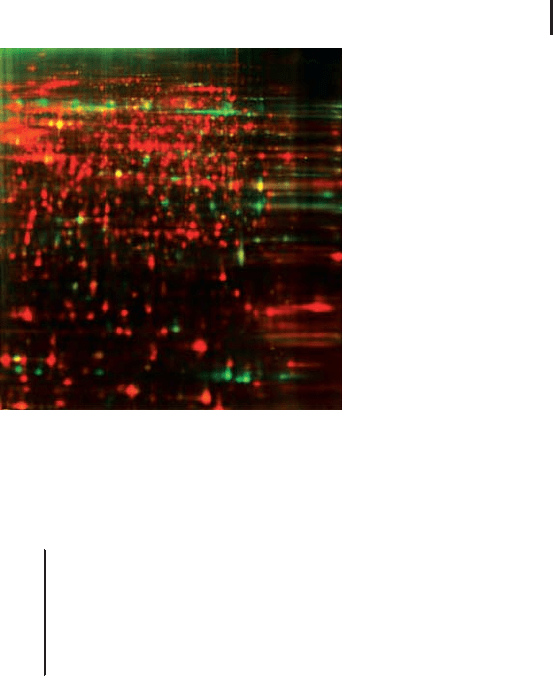
shown in Figure 1.22, the differential comparison of cell surface pro-
teins and whole cell lysate labeled with the second dye in the same
gel, is a very straightforward way to identify outer membrane proteins
against the background of the total proteins.
This reduces also costs for
consumables, equipment, and
labor.
Gels do not swell, shrink, or
break.
Image analysis is the bottle
neck in conventional 2-D elec-
trophoresis
In contrast replicate conven-
tional gels do not eliminate
these variations, they just
average them.
Note that those are still relative
quantitative results.
Sitek B, Scheibe B, Jung K,
Schramm A, Sthler K. In:
Proteomics in Drug Research
(M Hamacher et al. Eds.)
Wiley-VCH, Weinheim (2006)
pp 33–55.
Perales M, Eubel H, Heine-
meyer J, Colaneri A, Zabaleta
E, Braun H-P. J Mol Biol 350
(2005) 263–277.
Helling S, Schmitt E, Joppich
C, Schulenborg T, Mllner S,
Felske-Mller S, Wiebringhaus
T, Becker G, Linsenmann G,
Sitek B, Lutter P, Meyer HE,
Marcus K. Proteomics 6 (2006)
4506–4513.
Mayrhofer C, Krieger S,
Allmaier G, Kerjaschki D.
Proteomics 6 (2006) 579–585.
GE Healthcare Application
Note: Selective labeling of cell-
surface proteins using CyDye
DIGE Fluor minimal dyes.
(2005) 11-0033-92.
1.5 Two-dimensional Electrophoresis 77
Fig. 1.22: False color representation of 2-D gel images of a
CHO-K1 Cy5 cell-surface labeled sample (green spots) and a
Cy3 sample (red spots) labeled according to standard Ettan
DIGE protocol run in the same 2-D gel. From GE Healthcare
Application Note 11-0033-92.
& Note: The multiplex approach of DIGE has some
additional benefits for the conditions during
isoelectric focusing: the conductivity differences
between the strips are lower, because several
samples are mixed together. This leads to more
reproducible spot pattern across the gels.
1.5.3
First Dimension: Isoelectric Focusing in IPG Strips
IPG strips are available with many different lengths and gradients.
Optimally the resolution of the two-dimensional gel should be suffi-
cient to get every protein separated from each other. If a protein spot
contains more than one protein, the proteins can still be identified
and characterized with mass spectrometry, however quantitative
assessments are not possible. Therefore large gel formats are pre-
ferred. If the resolution of such a gel is insufficient, the sample can
be applied on several IPG strips with narrow, overlapping pH gradi-
ents. Most frequently wide gradients with pH 3 to 10 or 3 to 11 are
run. When higher resolution is required, a combination of an acidic
gradient pH 4 to 7 (or 3 to 7) and a basic gel pH 7 to 11 are employed.
IPG strips with printed serial numbers are very useful for tracing the
samples through the 2-D electrophoresis procedure.
When detection is performed
with western blotting, small
formats are preferred.
1 Electrophoretic Techniques78
For proteomics studies large formats with 18 or 24 cm strip lengths
are preferred because of several reasons:
.
To reach sufficient resolution.
.
To be able to load higher protein concentrations
for detection of as many spots as possible and
subsequent mass spectrometry analysis of spots.
.
To obtain sufficient evaluative gel area.
1.5.3.1 Rehydration
The dry IPG strips need to be reconstituted to their original thickness
of 0.5 mm with rehydration solution before use. There are two possi-
bilities:
.
Pre-rehydration of the strip without sample; the
sample is loaded either on the anodal or cathodal
end of the IPG strip with a cup or paper bridge.
.
Rehydration with the sample mixed into the
rehydration solution.
& Composition of the standard “rehydration
solution”:
8 mol/L urea, 0.5% (w/v) CHAPS,
0.2% (w/v) DTT, 1.25% (v/v) carrier ampholytes,
10% (v/v) glycerol, 0.002% Bromophenol blue.
.
Glycerol reduces electroendosmotic effects, pre-
vents drying of the gel, and urea crystallization.
It is not always necessary.
.
The concentrations of the additives are lower
than in the lysis buffer, in order to reduce
unwanted side effects like:
– Crystallization of urea;
– Instability of the patterns due to conflicts of
the buffering groups, caused by overloading
with carrier ampholytes;
– Formation of micelles between the different
detergents in the second dimension, which
can cause dark and dirty background with
some of the staining techniques (Gçrg et al.
1987a);
.
Bromophenol blue allows a good control of the
liquid distribution during rehydration.
When a sample has been extracted with urea and thiourea, the rehy-
dration solution must also contain thiourea.
Ideally one protein/spot.
Abundant proteins need more
space.
The smaller the gel, the higher
the fringe effect.
This solution is suitable for
samples without thiourea.
For electroendosmosis see
page 21 f or glossary.
See also page 79 for choice of
IPG buffers or carrier ampho-
lytes.
Gçrg A, Postel W, Weser J,
Gnther S, Strahler JR,
Hanash SM, Somerlot L. Elec-
trophoresis. 8 (1987a) 45–51.

1.5 Two-dimensional Electrophoresis 79
& Composition of the thiourea rehydration
solution:
7 mol/L urea, 2 mol/L thiourea, 0.5%
(w/v) CHAPS, 0.2% (w/v) DTT, 1.25% (v/v)
carrier ampholytes, 0.002% (w/v) Bromophenol
blue.
Samples without thiourea can also be applied to strips rehydrated
with such a solution.
Basic pH gradients It is not recommended to apply rehydration
loading to basic pH gradient strips. Obviously some proteins start to
aggregate in presence of high pH values. The samples are loaded at
the anodal end of the strip with a cup or paper bridge. As already
explained above in context with the reductant (see page 56 f), the best
remedy against horizontal streaking in basic gels is the rehydration
of IPG strips with HED (“DeStreak”) instead of a reductant.
& Composition of the DeStreak rehydration
solution:
7 mol/L urea, 2 mol/L thiourea, 0.5%
(w/v) CHAPS, 100 mmol/L DeStreak reagent,
1.25% (v/v) carrier ampholytes, 10% (v/v)
glycerol, 0.002% Bromophenol blue.
Ready-made rehydration solution containing the DeStreak reagent
and all other additives except the IPG buffer are available. This is
called “DeStreak solution”.
For the SDS electrophoresis in the second dimension the IPG
strips are equilibrated in the conventional way with SDS, Tris-Cl,
urea, glycerol, and reduced with DTT, followed by alkylation with
iodoacetamide.
Rehydration cassette In the original rehydration procedure, which is
still in use in some laboratories, the strips are placed into a glass cas-
sette, in order to control the gel thickness to prevent over swelling
(see Figure 1.23). An additional U-shaped gasket cut from 0.2 mm
thick film is added to achieve 0.5 mm thick gels on the 0.2 mm thick
film support.
With this procedure many strips can be rehydrated under identical
conditions. The disadvantages of the cassette method:
.
High volume of rehydration solution is needed.
.
Cassettes sometimes leak because of the urea
and the detergent in the liquid.
.
Rehydration loading of different samples is not
possible.
This is the more universal rehy-
dration solution.
At the anodal end of the strip
there are milder pH conditions
for the protein mixtures. Rehy-
dration loading on basic strips
leads to loss of proteins, which
stay outside of the gel.
This solution can also be used
for broad pH gradients, like pH
3–10 and pH 3–11.
The IPG buffer must be chosen
according to the used pH
gradient.
If this U-shaped film is not
added, the pore size of the
strips will be too small for high-
molecular-weight proteins to
enter the gel.
Some laboratories claim that
they obtain the best results
when they rehydrate the strips
with this method.
