Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.


1 Electrophoretic Techniques100
1.5.4
Second Dimension: SDS Electrophoresis
1.5.4.1 Gel Types
Homogeneous gels
For 2-D separations mostly a homogeneous gel
with 12.5% T and 3% C is used; the proteins of major interest in the
size range from 10 to 100 kDa are optimally resolved. For special
cases a table with optimal gel concentrations for different protein size
ranges can be found in the GE Healthcare 2-D Handbook (2005).
However, as demonstrated by Langen et al. (1997), it is not easy to
predict the optimal acrylamide concentration according to a mathe-
matical function: In an example shown in this paper a group of pro-
teins with ca. 50 kDa, which were co-migrating in 9–16% T gradient
gels were best resolved in a gel with 7.5% T. This shows that a try and
error procedure must be applied for optimization, when certain pro-
tein groups need to be completely resolved.
Gradient gels Generally, in gradient gels the overall separation inter-
val is wider; also the linear relation interval between the logarithm of
M
r
and the migration distance is wider than for homogeneous gels.
Also the spots are sharper because the pore sizes are continuously
decreasing. This effect can be very useful for separating proteins,
which are highly glycosylated. However, when homogeneous gels are
run under optimal conditions, resolution and spot definition is high
enough: a gradient gel is not necessary.
Gradient gels have the following shortcomings:
.
The preparation of gradient gels is much more
work.
.
It is more difficult to obtain reproducible gel
properties than for homogeneous gels.
.
The gels swell unevenly during staining, and
show uneven background intensity.
Horizontal flatbed gels Because the IPG strip is just placed on the
surface of the horizontal gel, a stacking gel with low acrylamide con-
centration is necessary for optimal protein transfer and separation
(Gyenes and Gyenes, 1987). Resolving and stacking gel is usually
polymerized in one piece. These gels can only be cooled from one
side; rarely gels thicker than 0.5 mm are used. These gels are poly-
merized onto a support film.
Laboratory-made gels are usually run with liquid buffers, con-
nected to the gel with paper wicks. The electrodes are then placed
into the buffer tanks. The Laemmli buffers are used. Instructions and
GE Healthcare Handbook. GE
Healthcare Life Sciences 80-
6429-60 (2005).
Langen H, Rçder D, Juranville
J-F, Fountoulakis M. Electro-
phoresis 18 (1997) 2085–2090.
Gyenes T, Gyenes E. Anal
Biochem 165 (1987) 155–160.
Gçrg A, Weiss W. In Rabilloud
T, Ed. Proteome research: Two-
dimensional gel electrophoresis
and identification methods.
Springer Berlin Heidelberg New
York (2000) 107–126.

1.5 Two-dimensional Electrophoresis 101
recipes for preparing gels for flatbed systems in the laboratory can be
found in several books: Gçrg et al. (2000) and Westermeier (2004).
Vertical gels For 2-D electrophoresis in vertical systems the stacking
gel is not needed, because:
.
The proteins are already pre-separated by IEF
and will therefore not aggregate while they enter
the resolving gel.
.
The proteins migrate from a gel into another gel,
and not from a liquid phase into a gel.
Still a discontinuous buffer system is employed: the gel buffer com-
position is different from the running buffer. The spots are well
resolved, because there is a stacking effect happening between the
highly mobile chloride in the gel and the lowly mobile glycine in the
running buffer.
Omitting the stacking gel solves a few technical problems and
makes the procedure easier:
.
The edge between the stacking and resolving
gels contains incompletely polymerized acryla-
mide monomers and oligomers, which stick to
proteins and partly modify proteins.
.
Some proteins get caught between the stacking
and resolving gels.
.
One of the problems sources with reproducibil-
ity is abolished.
.
Ready-made gels cannot contain a stacking gel
with a buffer different from the resolving gel
because of diffusion.
.
The additional step of polymerizing a stacking
gel short before the run is abolished.
The standard thicknesses of SDS slab gels run in vertical equipment
are 1.0 mm and 1.5 mm. Thinner gels cannot be used, because the
IPG strip including the film support is 0.7 mm thick, and it swells
during equilibration with SDS buffer.
Tab. 1.4: Comparison of gel thicknesses.
Advantages of 1.0 mm gels: Advantages of 1.5 mm gels:
Faster separations possible, leading to sharper
spots
Higher loading capacity
Detection methods faster and more sensitive Higher mechanical stability
Protein / gel volume ratio more favorable for
tryptic digestions resulting in higher peptide yield
Westermeier R. Electrophoresis
in Practice. WILEY-VCH, Wein-
heim (2004).
Therefore the upper running
buffer must not contain any
chloride ions.
The IPG strip is placed onto the
SDS gel sideways.

1 Electrophoretic Techniques102
1.5.4.2 Gel Casting
First of all, it is very important to clean glass plates and equipment
very thoroughly. Mass spectrometry analysis is very sensitive, and
every contamination will show up in the mass spectrogram and
might lead to wrong results. Keratin stemming from hair and skin is
the most frequently found protein contamination; sometimes even in
reagents purchased from a supplier. It has become general practice in
most proteomics laboratories to filter each monomer and buffer solu-
tion through a membrane filter before use.
Also for the SDS PAGE step only high-quality chemicals should be
used. Particularly for such a sophisticated and multistep procedure
like 2-D electrophoresis the search for trouble causing sources is very
cumbersome and time-consuming.
When the sample proteins have been pre-labeled with fluorescent
dyes, the gels are scanned inside the cassette. Therefore only low-
fluorescent glass is used for DIGE experiments.
For accurate spot picking the gel have to be fixed to one of the glass
plates, the glass plate without the spacers is therefore treated with
Bind-Silane prior to assembling the cassettes. If the Bind-Silane treat-
ment is performed long time before the cassettes are assembled, non-
treated and treated glass plates should be kept separate. It has been
observed that Bind-Silane molecules diffuse towards the non-treated
glass surface and cause strong adherence of the gel to the second
glass plate.
& Meticulously cleaned instruments and glass plates
as well as high-quality reagents are of high
importance.
Gels for miniformat and medium size instruments can be cast as sin-
gle gels (see Figure 1.35) as well as in multicasters, which are built
similar to those used for large gels (see Figure 1.37).
Large format gels, which are usually used in proteomics, can only
be prepared in multicasters, because the glass plates of the cassettes
would bend out and form a bulbous gel with a higher thickness in
the center than along the edges.
For the handling of multiple gels it is not very practical to use glass
plates with separate spacers and clamps. Therefore the cassettes con-
tain fixed spacers and a hinge on one side, as shown in Figure 1.36.
However, when the gel is to be fixed to one of the glass plates with
Bind-Silane for spot picking, non-hinged cassettes are used, with the
spacers attached to the second glass plates.
The author of this chapter
could write a detective story on
pitfalls caused by dirty or old
reagents.
For a high throughput method
anyhow multiple gels are
needed.
For multiple gels it is practical
to insert a little piece of paper
with a key number for easy
identification of the sample.
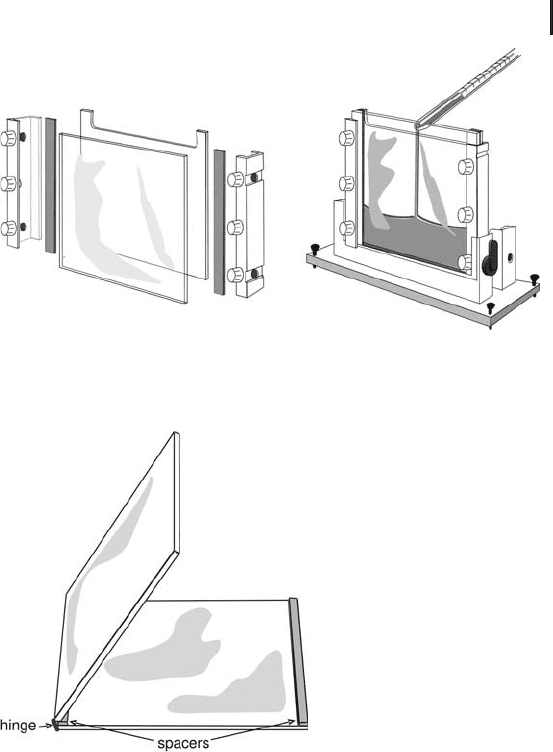
1.5 Two-dimensional Electrophoresis 103
Fig. 1.35: Casting single miniformat and medium size SDS
polyacrylamide gels for 2-D electrophoresis
Fig. 1.36: Glass cassette for multiple SDS gel casters
and instruments. Spacers are fixed; glass plates are
connected with a hinge.
Depending on how many gels to be cast at a time, different types of
multiple gel casters have been designed. During pouring the mono-
mer solution into the caster the liquid will not only flow between the
spacers, but also between the cassettes. For easy separation after the
polymerization plastic sheets have to be inserted between the cas-
settes. Originally they are designed to introduce the monomer solu-
tion into the cassettes from the bottom. This is necessary when gradi-
ent gels are prepared.
As already mentioned, the polymerization process is an exothermic
reaction. When multiple-gel casters are used, the composition of cata-
lysts in the polymerization solution is more critical than for single gel
Without the plastic sheets, the
cassettes would stick together
because of the polymerized gel
layers.
This practical hint is mentioned
here, because some instructions
incorrectly suggest the opposite
procedure.

1 Electrophoretic Techniques104
casting. If the polymerization runs too fast, very much heat is pro-
duced in the center of the multicasters, leading to thermal convec-
tion, which results in curved gel edges and irregular polymerization.
It is very practical to add the TEMED to the other constituents already
when the monomer solution is mixed together. It will not cause any
start of polymerization. The ammonium persulfate, however, must
not be added before pouring the gel.
It must be avoided that the start of polymerization begins while the
monomer solution is still flowing. Furthermore the multiple solu-
tions in the cassettes need a certain time to level out following the
principle of the communicating vessels. The monomer solution
should be degassed with a vacuum pump and then precooled in a
refrigerator at 4–8 C. This measure reduces the heat development
and delays the start of the polymerization. It has several advantages:
.
The solution has time enough to level out
between the cassettes and settle until the solu-
tion starts to polymerize: this prevents irregular
gel composition and variation in gel lengths.
.
Local overheating is avoided, which can result in
local variations of sieving properties of the gels.
.
Gels properties are more reproducible indepen-
dent of the seasonal temperatures in the labora-
tory.
When the room temperature is higher than 20 C, the multicaster
should – like the monomer solution(s) – be pre-cooled in a cold room
or a refrigerator.
For homogeneous gels it is quicker to fill the caster from the top by
pouring the solution directly into the cassettes (see Figure 1.37A).
Before the polymerization starts, 0.1% (w/v) SDS–water solution is
sprayed into the cassettes with a finger-pumped plant sprayer to
achieve straight upper gel edges (see Figure 1.37B). The very small
detergent solution droplets glide down on the glass surface very
quickly and will not mix with the monomer solution, because they
are very small and have a low density. This procedure gives much
straighter edges than pipetting water-saturated butanol or 70% (v/v)
isopropanol into the cassettes, as it is proposed in most instructions.
Pipetting produces larger drops, alcoholic solutions have a high affi-
nity to the vinyl spacer material and can cause bent surfaces.
& Note: It is very important to obtain a straight
gel surface, because the IPG strip is placed on
the gel edge-to-edge: in this direction the film
support is absolutely rigid and cannot be bent to
follow a curved edge.
Most of the stock solutions are
anyhow kept in the refrigerator.
When alcoholic overlay solu-
tions are used, they need to be
removed an hour later, because
they gnaw at the plastic surface
of the caster box. SDS solution
does not need to be removed.
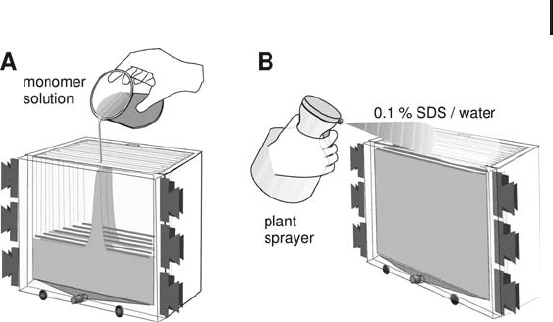
1.5 Two-dimensional Electrophoresis 105
Fig. 1.37: Casting multiple homogeneous SDS gels.
A. Pouring the monomer solution directly into the cassettes.
B. Overlaying the edges by spraying 0.1% (w/v) SDS–water
solution into the cassettes.
One should not forget that the quality and the age of chemicals, parti-
cularly the catalysts, greatly influence the polymerization efficiency of
the monomer solution. For instance: The TEMED can be stable for
several years, but it can suddenly decay within a week. Therefore it is
advisable to replace the TEMED every year with a new bottle.
Gradient gels For the preparation of gradient gels a gradient maker
has to be connected to the gel caster. Here there is no other way than
casting the gels from the bottom. The gradient solution has to be
delivered to the box slowly to avoid mixing – taking 5–10 minutes.
The speed can be adjusted by using a laboratory peristaltic pump, or
by selecting a certain level for the liquid beaker and reducing the dia-
meter of the tubing with a pinchcock clamp (see Figures 4.4 and 4.6
on pages 334 and 341).
Gels for spot picking After the 2-D patterns have been compared
with image analysis software, proteins of interest have to be identified
and characterized. For these further analyses the gel plugs containing
these proteins have to be cut out of the gel slab. For high accuracy,
reliability, and automation of this procedure robotic spot pickers are
employed (see pages 138 and 140 f). The most accurate and reliable
way is to use the x/y coordinates of the respective spots from the
image analysis results in the automatic spot cutter. It is, however,
important that the gel does not change its shape by shrinking or swel-
ling. Therefore gel slabs have to be fixed to a rigid film or glass plate
backing.
Note: The quality of the edge is
influenced by the quality of the
chemicals.
Using a pump means a lot of
work. Everything has to be set
up very quickly, because the
casting and the overlaying
procedure must be finished
before the gel solution starts to
polymerize. The casting proce-
dure must be as exact as
possible to prepare reproducible
gels.

1 Electrophoretic Techniques106
Gels fixed to a glass plate Because it is not easy to bind a gel to a
glass plate after staining, gels are covalently bound to a film support
or glass plate during polymerization. Laboratory made gels are
usually polymerized onto a glass plate, which has been treated with
Bind-Silane. Because staining solutions can diffuse into the gel only
from one side, the gel should be as thin as possible for fast and effi-
cient staining. These gels are usually 1.0 mm thick. Fluorescence
labeled or fluorescence stained gels must be attached to non-fluores-
cent glass. Because this type of glass is very expensive, the gel layer is
removed from the glass plate after all analysis has been done. When
the glass plates are reused, the gel layer is removed with a plastic scra-
per. Remaining gel pieces disappear after vigorous treatment with a
dishwashing brush.
Meanwhile a non-fluorescent support film material has been devel-
oped, which allows using ready-made gels for the combination fluor-
escent detection – spot picking.
Ready-made gels for vertical systems Ready-made gels are more
expensive than laboratory-cast gels. However, one should not forget
that gel casting is a lot of work – particularly for cleaning the equip-
ment – and working time costs money as well. Commercially pro-
duced gels are prepared according to GMP industry standards, and
they are quality controlled.
There are two different concepts:
.
Gels in glass or plastic cassettes. The handling does
not differ from that of laboratory-cast gels. The
glass cassettes are either sent back to the produ-
cer or they are disposed of.
.
Gels on film-support. The 1 mm thick gels are
inserted into specially designed re-usable cas-
settes (Figure 1.38). Conventional glass cassettes,
as shown in Figure 1.36, cannot be used,
because air pockets between the glass plate and
the backing develop mechanical pressure on the
gel, leading to an irregular front.
These 12.5% homogeneous polyacrylamide gels contain PPA-chloride
pH 7.0 instead of the standard Tris-chloride buffer pH 8.8, in order to
achieve a long shelf life. Because polymerization at pH 7.0 is more
efficient than at pH 8.8, the gels have a very high mechanical stability,
and the patterns are highly reproducible. Bis-Tris buffer containing
gels are a practical alternative (see above). The gels are packed airtight
in flexible aluminum bags.
It is not easy to produce these
vertical gels on a film support in
the laboratory, because the
polyester films are flexible. To
set up a casting system for film-
supported gels would mean a
big investment in equipment.
It is very annoying, when the
valuable sample is not sepa-
rated well, just because of a
little mistake occurring while
preparing gels.
The sieving properties of a
12.5% T PPA gel are compar-
able to those of 14% T poly-
merized with a Laemmli buffer.
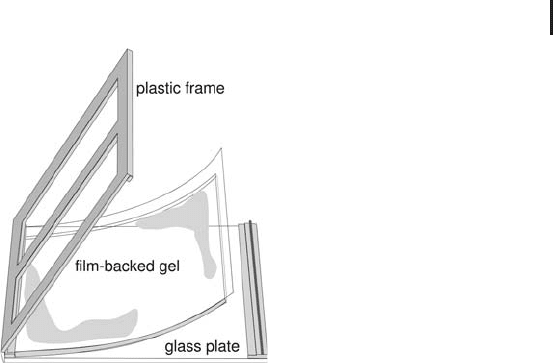
1.5 Two-dimensional Electrophoresis 107
Fig. 1.38: Support cassette and ready-made film-backed
vertical gel. The gel is shorter at the upper side to
accommodate the IPG strip.
Kits with buffer concentrates and agarose sealing solutions are
available. The kit for the film-backed PPA gels contains also a bottle
with the gel buffer. The best way to insert the gel into the cassette is
to apply a streak of 1 mL of the gel buffer onto the glass plate along
the spacer of the closing side, and placing the gel on the glass plate
first touching the buffer streak with the respective edge. The gel posi-
tion can be easily adjusted that the edge at this side is in contact along
the entire spacer and the lower gel edge is flush with the edge of the
glass plate. When the gel is lowered on the rest of the glass plate care-
fully, no air bubbles are caught between gel surface and glass plate.
The liquid near the closing side and some air bubbles – mostly close
to the hinged side – are squeezed out with a roller.
A narrow gap of less than 1 mm remains between one spacer and
the gel edge: this will be sealed with hot agarose solution after the
IPG strip has been inserted.
For Western blotting analysis or for high sensitivity fluorescent
staining the support film has to be removed. It can be cut off with a
simple instrument using a steel wire (see Figure 1.39). The large for-
mat gels are easy to handle also without the film-backing because
they are well polymerized.
This procedure works only with flexible support films, not with
rigid, Bind-Silane treated glass plates.
Of course, also these cassettes
have to be thoroughly cleaned
before and after each use to
avoid contaminations.
A liquid layer between gel
surface and glass plate has to
be avoided. It would cause
blurred spots.
Note: For those who have only
used film-backed gels, handling
of non-backed slab gels requires
some practice and skill.
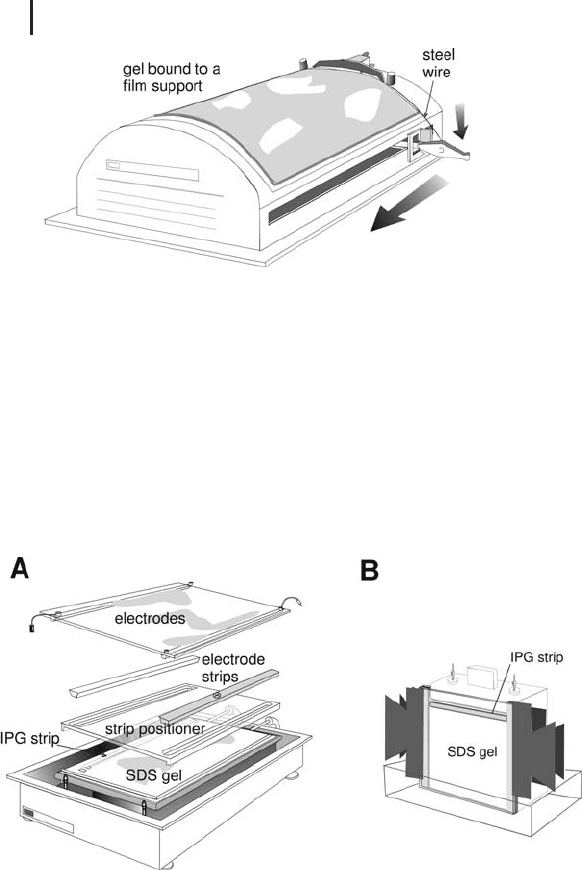
1 Electrophoretic Techniques108
Fig. 1.39: Film remover for complete removal of a flexible
film-backing from the gel.
1.5.4.3 Instrument and Gel Setup for the Second Dimension
Figure 1.40 shows a Multiphor flatbed chamber used as SDS PAGE
instrument and a mini vertical electrophoresis system for miniformat
gels. These two instrument types are not for high throughput and
thus mostly employed for optimization work.
Fig. 1.40: Flatbed and vertical mini electrophoresis apparatus for SDS PAGE.
In the flatbed apparatus the standard gel size is 2519 cm. The elec-
trode distances can be adjusted to shorter separation distances. For
example, SDS electrophoresis of three miniformat IPG strips with
7 cm length can be run together in one gel under identical conditions
(see Figure 1.41).

1.5 Two-dimensional Electrophoresis 109
Fig. 1.41: Three miniformat 2-D separations run in one flatbed gel.
IEF in three 7 cm IPG strips 4 to 7, SDS PAGE in a 2511 cm gel with
a stacking zone of 5% T and a homogeneous resolving area of
12.5% T, 0.5 mm thick. Silver staining.
Ready-made gels for vertical miniformat chambers are available from
several suppliers. An alternative is to run also several short IPG strips
together on a larger vertical gel. Some vertical systems can also be
equipped with shorter glass plates.
Tab. 1.5: Comparison of the properties of flatbed and vertical
systems.
Flatbed Systems Vertical Systems
One gel per instrument. Multiple gels runs are possible.
Can also be used for IEF in IPGs. Dedicated for SDS electrophoresis.
Gel thickness is limited, because cooling
is only possible from one side.
Higher protein loading capacity,
because thicker gels can be used,
which are cooled from both sides.
Very versatile for different gel sizes
and methods.
Gel sizes fixed by glass plate sizes.
Thin layer gels can be used, easy
application of the IPG strips.
Thin gels cannot be used, because
the IPG strip would not fit between
the glass plates.
Buffer strips (polyacrylamide or filter
paper) can be used instead of large
volumes of liquid buffers.
Blotting is easier because of higher
gel thickness.
The IPG strip has to be removed from
the SDS gel after 40 minutes because
of EEO effects.
The IPG strip does not have to be
removed.
Gçrg et al. (1995) have performed a systematic comparison of
results, obtained with a flatbed and a vertical system for the same
Up to 3 mm thick gels can be
used in vertical systems
Very thin gels show higher sensi-
tivity of detection, they are
easier and quicker to stain.
With flatbed systems there is
reduced chemical and radioac-
tive liquid waste.
EEO occurs in both systems,
therefore urea, glycerol in the
equilibration buffer and low
initial voltages are needed.
Gçrg A, Boguth G, Obermaier
C, Posch A, Weiss W. Electro-
phoresis 16 (1995) 1079–1086.
