Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.

1 Electrophoretic Techniques110
sample: The spot patterns are very similar. It is demonstrated that
sharper spots are obtained in thinner gels, which can be employed in
the flatbed system, but not in a vertical setup.
Design of vertical instruments The following functions are needed in
the instrument for the second dimension:
.
High throughput;
.
Large gel cassettes;
.
Efficient cooling for fast runs with straight front;
.
Leakage free;
.
Reproducible results independent from gel posi-
tion;
.
Different gel thickness should be possible;
.
Easy handling;
.
Buffer consumption not too high.
The first high-throughput chamber had been developed by Anderson
and Anderson (1978): the DALT box, where the gel cassettes are tilted
by 90 degrees and inserted between insulating rubber flaps (see Fig-
ure 1.42). In contrast to conventional vertical systems, the migration
direction of the proteins is from left to right instead of from the top
to the bottom. Handling of the instrument is easy. As only one single
tank is used, there is no problem of buffer leakage from an upper to a
lower tank.
Fig. 1.42: Schematic drawing of the traditional DALT chamber
for multiple SDS PAGE runs. The migration direction is from the
left to the right. Insulating rubber flaps prevent current flow
between the cassettes.
The gels are usually run overnight, because the cooling efficiency
is limited. The entire box is filled with Tris-glycine buffer. The buffer
Anderson NG, Anderson NL.
Anal Biochem 85 (1978)
341–354.
The buffer volume of 20 L is
rather high.
1.5 Two-dimensional Electrophoresis 111
should not be used more than once, because it will become enriched
with chloride ions from the gel buffer. When the Tris-glycine buffer
contains chloride ions, the initial stacking effect is disturbed – result-
ing in a loss of resolution – and the running time is extended.
A limitation is the design of the buffer tank: the cathodal and ano-
dal buffer reservoir is identical. This makes peptide separations with
Tris-tricine buffer very expensive. And – the stable PPA buffer system
cannot be applied, because it would require two completely separate
tanks for the anodal and the cathodal buffer.
For vertical instruments with completely separate tanks, several
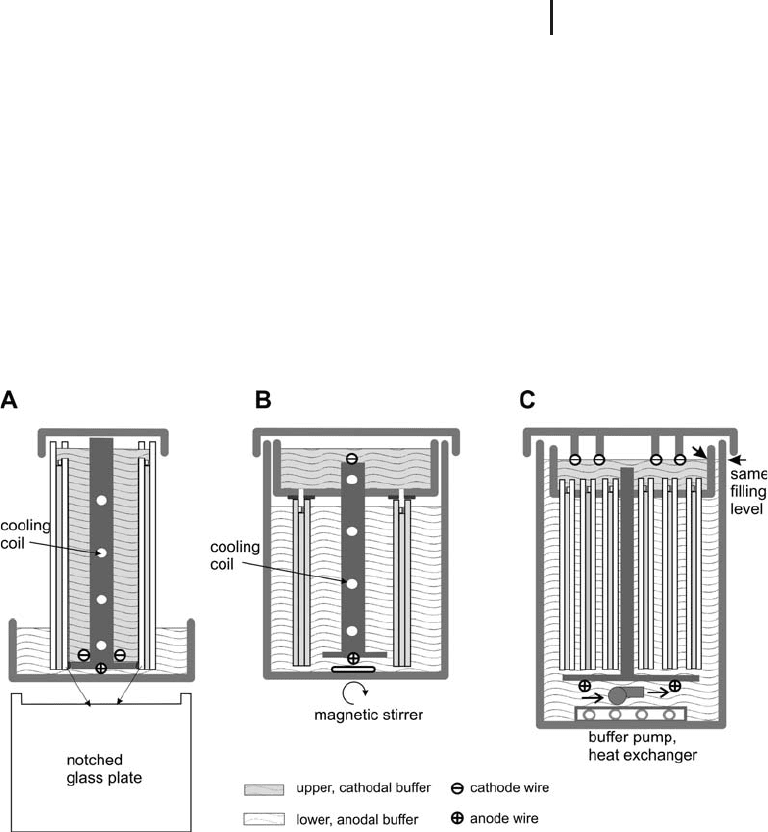
construction concepts are possible. Figure 1.43 shows three different
designs.
Fig. 1.43: Three different designs of vertical chambers.
All of them can be cooled.
A. Buffer-backed chamber.
B. Chamber with tight upper buffer tank.
C. Chamber for fast multiple runs.
Vertical electrophoresis chambers
.
In the buffer-back chambers (Figure 1.43, type
A) the gels are cooled via the cathodal buffer.
Notched glass or aluminum oxide ceramics
plates are used to enable the contact between the
cathodal buffer and the gel.
When the buffer tanks are
completely separate, the
anodal buffer can be used
repeatedly, because chloride
ions do not disturb there.
Maximal two gels can be run;
therefore the principle is mostly
applied on miniformat appa-
ratus.
1 Electrophoretic Techniques112
.
The medium sized chamber with tight upper
buffer tank (Figure 1.43, type B) can accommo-
date up to four gels at the same time, when
notched glass plates are inserted between each
two gel pairs. The gels are cooled via the anodal
buffer.
.
Multiple runs with large gels can be performed
in chambers of type C in Figure 1.43. It is, how-
ever, important to fill upper and lower buffer to
the same level to avoid buffer mixing. The heat
removal in this chamber type is very efficient,
because the cooling anodal buffer is vigorously
pumped around in the lower tank.
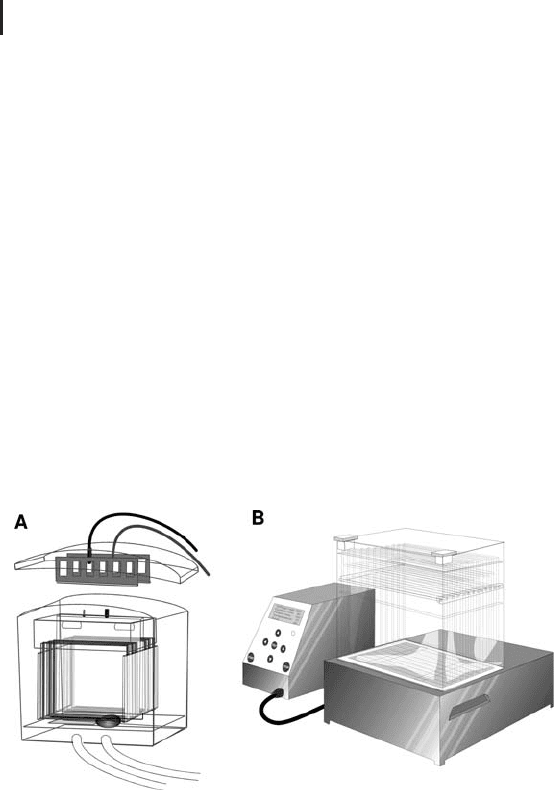
The concept of pumping the anodal buffer around the cassettes is
employed for large gel systems: stand-alone instruments (Figure
1.44A) and in integrated systems for running up to 12 gels (Figure
1.44B).
Fig. 1.44: Electrophoresis instruments for large cassettes.
A: Stand-alone instrument for up to six gels.
B: Integrated apparatus Ettan DALT twelve for multiple 2-D PAGE.
The programmable power supply controls also the Peltier cooling
system and the pump for circulating the lower buffer.
At the rear of the separation unit of the integrated system sits a
draining valve for convenient removal of used buffer. At the back
panel of the control unit of the Ettan DALT twelve there is a serial port
for possible software updates and to connect a computer or serial
printer to the instrument for a report on the electric conditions after
every five minutes of the run. With this interface the instrument can
In this concept the edges of the
glass plates must be absolutely
intact to prevent leakage of the
upper tank.
The buffer concentration for the
upper tank must usually be
doubled to prevent depletion of
the cathode buffer.
1.5 Two-dimensional Electrophoresis 113
be integrated into the laboratory workflow system and allow proce-
dures according to GLP (good laboratory practice).
& Note: for optimally reproducible 2-D patterns all
gels of an experiment should be run together in
one chamber.
The following hint goes for all vertical chambers with separate tanks:
When the lower – anodal – buffer is mixed with the used upper –
cathodal – buffer after each run, the Tris concentration is high
enough for repeated use as anodal buffer. This saves work and
reagent costs. Of course, the cathodal buffer must be new for each
electrophoresis.
1.5.4.4 Equilibration of IPG Strips
Prior to the run in the second dimension the strips have to be equili-
brated with SDS buffer to transform the focused proteins into SDS–
protein complexes, which are completely unfolded and carry negative
charges only. The following conditions have been optimized by Gçrg
et al. (1987b).
& Equilibration stock solution: 2% (w/v) SDS, 50
mmol/L Tris HCl pH 8.8, 0.01% (w/v)
Bromophenol blue, 6 mol/L urea, 30% (v/v)
glycerol
Equilibration is performed twice on a shaker:
15 min 10 mL equilibration stock solution Plus 1% (w/v) DTT
15 min 10 mL equilibration stock solution Plus 2.5% (w/v) iodoacetamide
Sodium dodecyl sulfate With the amount of 2% SDS there is usually
sufficient SDS for preparative protein loads. Some laboratories use 3–
6% SDS to facilitate the migration of focused hydrophobic proteins.
Tris HCl buffer pH 8.8 In former protocols it was proposed to add the
stacking gel buffer pH 6.8, because the use of a stacking gel was stan-
dard procedure. However, it is better to add the more basic resolving
gel buffer with a pH of 8.8 to improve the alkylation of the cysteines.
Bromophenol blue This tracking dye allows a control of the running
conditions, the shape of the migration front, and the separation time.
Running gels of one experiment
in different chambers makes
spot matching more difficult.
The Tris moves from the anodal
to the cathodal buffer. Glycine
moves from the cathodal to
anodal buffer, it is not needed
in the anodal buffer.
Gçrg A, Postel W, Weser J,
Gnther S, Strahler JR,
Hanash SM, Somerlot L. Elec-
trophoresis 8 (1987b) 122–124
Note: with iodoacetamide the
solution can easily become
acidic. This would disturb the
formation of the SDS–protein
complexes.

1 Electrophoretic Techniques114
Urea and glycerol These additives have been introduced to keep
electroendosmotic effects (see above) as low as possible. Urea is also
supporting the solubility of hydrophobic proteins.
Dithiothreitol After the proteins have been focused they have to be
treated with the reductant again. When very high protein loads are
analyzed, the concentration of DTT needs to be increased.
Iodoacetamide The alkylation agent has several functions:
.
Complete alkylation of the cysteines, to avoid
partial modification by acrylamide for increased
spot sharpness and improved protein identifica-
tion with mass spectrometry.
.
Elimination of point streaking as described by
Gçrg et al. (1987b).
.
Avoidance of the artifactual horizontal lines
across the SDS gel in the size range of 40–
50 kDa.
When the cysteines had been pre-labeled with saturation dye for
DIGE, the iodoacetamide step is omitted.
Equilibration time Two times 15 minutes seems to be a rather long
time, and some might fear considerable losses of proteins due to dif-
fusion. As already mentioned on page 51, immobilized pH gradients
keep proteins back like a weak ion exchanger. Thus, only a little
amount of proteins – those from the surface – are washed out.
The long equilibration time is necessary for the complete formation
of SDS–protein complexes, because the negatively charged SDS is
repelled by the negative charges on the carboxylic groups of the
strips. It has been observed that a too short equilibration leads to ver-
tical streaks and losses of high molecular weight proteins.
Modified equilibration Although Hedberg et al. (2005) claim that
after running the first dimension with DeStreak under oxidative con-
dition equilibration can be shortened and both, equilibration and the
second-dimension run can be performed under oxidative conditions,
it is not recommended. This procedure is limited just to a few pH
gradients. Practice has shown that this method does not give well
resolved spot patterns for all sample types.
There are cases where increased SDS content facilitates the transfer
of hydrophobic proteins from the first to the second dimension. One
example is described by McDonough and Marbn, where 10% (w/v)
SDS in the equilibration buffer was the only way to move a highly
hydrophobic protein into the second-dimension gel.
This is valid also after running
IEF with DeStreak.
The iodoacetamide functions as
scavenger of the excess reduc-
tant.
However, the equilibration
time must not be extended too
far, because this leads to losses
of low molecular weight
proteins due to diffusion.
Hedberg JJ, Bjerneld EJ, Cetin-
kaya S, Goscinski J, Grigorescu
I, Haid D, LaurinY, Bjellqvist B.
Proteomics 5 (2005) 3088–
3096.
McDonough J, Marbn E.
Proteomics 5 (2005) 2892–
2895.

1.5 Two-dimensional Electrophoresis 115
1.5.4.5 Transfer of the IPG Strip and Running the SDS Gel
In order to make examination and evaluation of 2-D maps easier, the
first and second dimensions should always be set together in a stan-
dardized way. The majority of the scientific world agrees that it makes
sense to orientate the gel in the style of a Cartesian coordinate sys-
tem: the low values are located at the left bottom and the high values
at the right top.
& The IEF gel should always be placed on the SDS
gel in a standard orientation: low pH to the left,
high pH to the right.
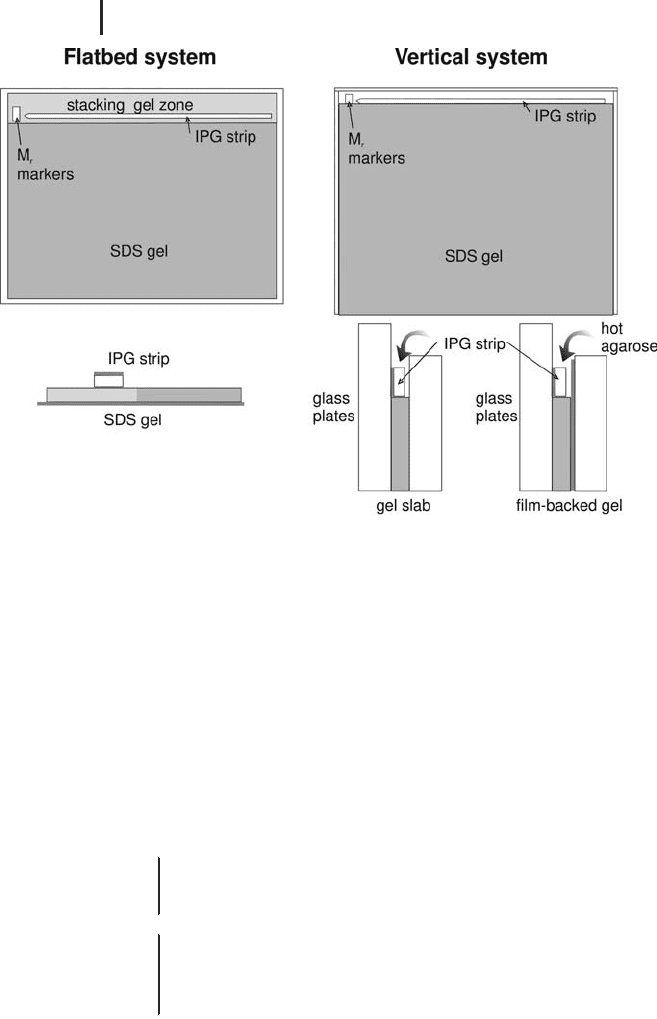
Flatbed system
It is highly recommended to use the strip positioner
plate as shown in Figure 1.40A for:
.
Improved alignment of the electrode strips;
.
Preventing the electrode strips to slide away;
.
Reproducible positioning of the electrode strips
and the IPG strip on the SDS gel;
.
Achieving a straight front, because the gel sur-
face is covered.
The gels are placed directly on a cooling plate, which is connected to
a thermostatic circulator. Usually the running temperature is 15 C.
After equilibration of the IPG strip, the excess buffer is removed by
blotting it slightly with clean filter paper. The strip is placed – gel sur-
face down – parallel to and 5 mm apart from the cathodal electrode
strip. The acidic end of the strip points to the left side.
M
r
marker proteins are applied with IEF sample application pieces
as shown in Figure 1.45.
After 40 minutes of low voltage – for sample entry – the IPG strip
is removed from the gel; the cathodal electrode strip is moved from
its original position to the contact area of the IPG strip. This measure
prevents drying out and burning of the SDS gel along this contact
line. The separation can now be continued with higher voltage set-
tings.
No agarose sealing solution is
needed.
In the flatbed system EEO has
stronger effects, because the
water is transported out of the
SDS gel surface, causing drying
of the SDS gel.
1 Electrophoretic Techniques116
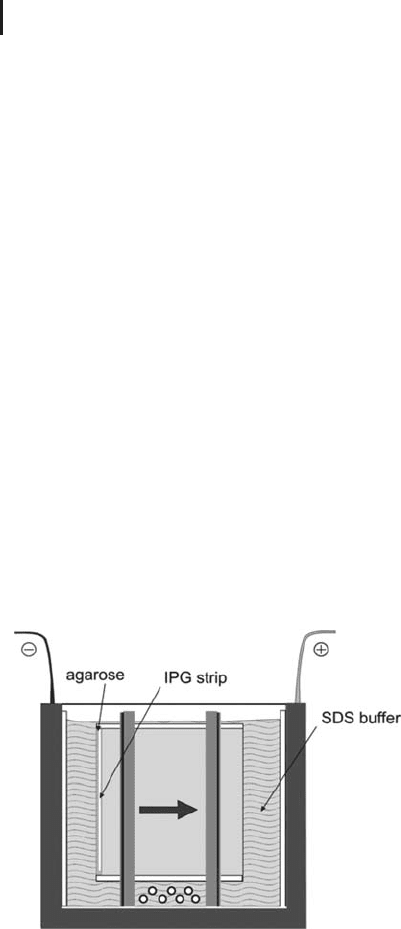
Fig. 1.45: Application of the IPG strip on the different types of
second-dimension gels.
Vertical system The IPG strip is placed onto the edge of the SDS gel
sideways, with its support film touching one glass plate. Hot agarose
embedding solution, pipetted onto the strip has several functions:
.
Achieve a gel continuity;
.
Prevent air bubbles between the gels;
.
Avoid floating of the strip in buffer solution;
.
Reduce electroendosmotic effects at very high
and very low pH values of the IPG strips;
.
Seal the gaps between cassettes spacers and the
ready-made gels.
& Note: The agarose solution should not be hotter
than 60 C; it could cause carbamylation of some
proteins because of the presence of urea.
& Composition of the agarose embedding
solution:
0.5% (w/v) agarose, SDS cathode
buffer (1 concentration), 0.01% (w/v)
Bromophenol blue
Ca. 2 mL of agarose embed-
ding solution is necessary per
gel.

1.5 Two-dimensional Electrophoresis 117
Molecular weight marker proteins.
& Tip: Best results are obtained when the
molecular weight marker proteins have the same
start conditions like the proteins from the IPG
strips: a IPG strip is rehydrated in the marker
protein solution, cut into small pieces, which can
be stored in a deep freezer or applied directly to
the SDS gel together with the IPG strips of the
first dimension.
The alternative is to apply the markers to an IEF sample application
piece directly, in a volume of 15–20 mL and let it dry.
1.5.4.6 Running Conditions for Vertical Gels
Electric conditions
Also vertical gels must be run with low voltage at
least for the first two hours to reduce the electroendosmosis effects
(see page 21). When immobilized pH gradient gels are equilibrated
in basic buffers, they become deprotonated: The carboxylic groups
become negatively charged, the amino groups are neutral. Thus the
IPG strips acquire negative net charges during equilibration with the
SDS buffer. It has been observed already during the first approaches
utilizing immobilized pH gradients in 2-D electrophoresis that this
can cause electroendosmotic effects (Westermeier et al. 1983). Those
result in protein losses due to a water flow towards the cathode. The
water transport is minimized by adding urea and glycerol to the equi-
libration solution and reducing the electric field during the phase of
protein transfer into the second-dimension gel (Gçrg et al.1985,
2003):
& Phase 1: apply only 5 mA (» 0.2 W) per gel for
2 hours
& Phase 2: continue with normal SDS
electrophoresis settings for fast or overnight runs
In discontinuous buffer systems, first the conductivity is high,
because the gel contains a lot of chloride ions, which have high mobi-
lity. When more of the glycine or tricine ions – with low mobilities –
are migrating into the gel, the conductivity decreases, and the field
strength increases. In order to avoid the production of too much Joule
heat at the end of the run, the gels are usually run at low current set-
tings. A possibility to speed up the separation is to adjust the current
setting from time to time.
Westermeier R, Postel W, Weser
J, Gçrg A. J Biochem Biophys
Methods 8 (1983) 321–330.
Gçrg A, Postel W, Gnther S,
Weser J. Electrophoresis 6
(1985) 599–604.
Gçrg A, Drews O, Weiss W. In
Simpson RJ, Ed. Purifying
proteins for proteomics: A
laboratory manual. Cold Spring
Harbor Laboratory Press, Cold
Spring Harbor, New York
(2003) 391–430.

1 Electrophoretic Techniques118
Ideally the power supply offers the feature to run the separation at
constant power. Then the current is high at the beginning and low at
the end without overheating the gels and the buffers. In this way a
separation can be run faster than under conventional conditions.
Temperature Multiple gels produce a substantial amount of Joule
heat, which has to be removed. If this is not efficient enough the gels
show the “smiling effect”: faster migration in the center than on the
lateral sides. It was the general opinion that SDS gels should be
cooled to 10–15 C to remove the heat efficiently enough to obtain a
straight front. Systematic studies in chambers with more powerful
buffer circulation have shown that better results and faster runs are
obtained when the gels are run at 25 C. Of course there are also cer-
tain limits how much temperature can be removed from the gels and
the buffers.
Benefits of quick runs Fast separations of large gels – within a few
hours instead of overnight – result in less diffused spots compared to
overnight runs. This is particularly obvious in the low molecular
weight area. Less diffused spots have three advantages:
.
Increased spatial resolution in homogeneous
gels;
.
Increased detection sensitivity;
.
Improved enzyme kinetics during tryptic in-gel
digestion.
1.5.4.7 Molecular Weight Determination
For the determination of molecular weights in 2-D electrophoresis
gels two different procedures are used.
Method 1. Co-running molecular weight standard proteins Molecular
weight marker proteins can be applied as a separate track to the 2-D
gel. The molecular weights of the proteins in the 2-D map are then
interpolated with the molecular weight curve obtained from the posi-
tions of the marker proteins. This procedure is usually carried out
with the image analysis software as “1-D calibration”.
The problem with this method is its limited accuracy. The markers
have to be applied at the lateral sides of the gels. Often the space is
very limited and the bands are curved because of the edge effects in
SDS PAGE. Furthermore, the estimation of molecular weights with
SDS PAGE is not accurate at all.
The high settings are applied
after the 2 hours protein
transfer.
Less need for gradient gels.
In comparative experiments
over 50% more spots could be
detected in these gels.
Because of the higher protein
concentration in the gel plug.

1.5 Two-dimensional Electrophoresis 119
Method 2. Interpolation between identified sample proteins with known
M
r
values Prominent spots showing up in each 2-D map of a sam-
ple type can be analyzed for identification and amino acid sequence
information. The theoretical M
r
values can then be used as keystones
for interpolating the M
r
values of the other proteins. The second pro-
cedure is performed with the image analysis software as “2-D calibra-
tion”. This method is much more exact than the method described
above.
1.5.5
Detection of Protein Spots
There is a demand list of properties required for the ideal spot detec-
tion technique in 2-D gels in proteomics: it should:
.
Be sensitive enough for low copy number
proteins;
.
Allow quantitative analysis;
.
Have a wide linearity;
.
Have a wide dynamic range;
.
Be compatible with mass spectrometry;
.
Be non-toxic;
.
Be environment-friendly;
.
Be affordable.
Unfortunately there is no method that affords all these features
together. Lately some reviews on detection techniques in proteomics
have been published: a “minireview” by Westermeier and Marouga
(2005), and a very elaborate compilation by Miller et al. (2006). The
following table gives an overview over currently used detection princi-
ples and their features. Several different protocols exist for most of
these methods. Those of major importance are quoted in the table.
1.5.5.1 Comparison of Detection Methods
Tab. 1.6: Detection methods for protein spots.
Method Advantages Disadvantages
Coomassie brilli-
ant blue staining
(colloidal)
Steady state method, good
quantification, inexpensive,
mass spectrometry
compatible
Medium sensitivity: LOD
ca. 20 ng of BSA; the more
sensitive, the slower
Westermeier R, Marouga R.
Bioscience Reports 25 (2005)
19–32.
Miller I, Crawford J, Gianazza
E. Proteomics 6 (2006) 5385–
5408.
Neuhoff V, Arold N, Taube D
and Ehrhardt W. Electrophor-
esis 9 (1988) 255–262.
