Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.


151
2
Liquid Chromatography Techniques
This chapter gives inspiration, hints and ideas on how to tackle the
challenges in proteomics by applying the tools of liquid chromatogra-
phy (LC), a complementary approach to the already well established
gel-based workflow.
It is obvious that the capabilities of liquid chromatography are
investigated (Link et al. 1999). There is evidence that LC-based meth-
ods can close some of the gaps and deliver supplementary informa-
tion if we state that 2D electrophoresis is the established standard
method. However, we have always to keep in mind that protein
separation is not just a tool by itself rather than an important inter-
mediate step towards protein identification and characterization by
mass spectrometry.
2.1
Basic Principles of Important Liquid Chromatography Techniques
Proteins are separated using chromatography techniques according
to differences in their specific properties, as shown in Table 2.1. The
following chapter is based on information for protein purification
and adapted to the requirements of protein pre-fractionation in pro-
teome analysis.
Tab. 2.1: Separation principles in protein chromatography.
Property Technique
Charge Ion exchange chromatography,
chromatofocusing
Size Gel filtration
Hydrophobicity Reversed phase chromatography,
hydrophobic interaction chromatography
Biorecognition (ligand specificity) Affinity chromatography
Proteomics in Practice. A Guide to Successful Experimental Design 2
nd
Ed.
Reiner Westermeier, Tom Naven, and Hans-Rudolf Hçpker
Copyright 2008 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
ISBN: 978-3-527-31941-1
In order to overcome some of
the existing limitations of 2D
electrophoresis the need for
complementary approaches
becomes apparent.
Link AJ, Eng J, Schieltz DM,
Carmack E, Mize GJ, Morris
DR, Garvik BM, Yates JR III.
Nature Biotech 17 (1999)
676–682.
GE Healthacre “Protein
Separations’ Handbook Collec-
tion”

2 Liquid Chromatography Techniques152
Knowing that there exist numerous methods for protein separa-
tion, the following chapters cover only the most relevant LC techni-
ques for protein pre-fractionation.
Besides personal preferences for one or the other chromatography
technique there are more objective selection criteria available (see
Table 2.2). It is not only important to look at the performance of a sin-
gle technique, but also it is even more important to plan and consider
how to best interconnect the methods in order to bypass avoidable
intermediate steps that bare the risk of sample losses that lead to an
unwanted reduction in protein recovery.
Tab. 2.2: Selection criteria of LC methods for protein pre-fractionation
in proteomics.
IEX RPC AC GF
Enrichment High High Very high Dilution
Loadability Very high Medium Very high Low
Peak capacity Medium High N/A Low
Desalting capability N/A Yes N/A Yes
Raw sample compatibility Very good Limited Excellent Good
Running costs (column/ eluents) High/low Moderate/
high
Low/low to
excessive/
high
High/low
Volatile buffer No Yes No Possible
Unless there are meaningful affinity columns or media available
(e.g. high-abundancy protein depletion for serum, selective glyco- or
phospho-protein enrichment) ion exchange chromatography is the
first choice to apply the initial sample.
No matter what sample concentration, volume or amount, IEX
works perfectly at any scale. Columns in all different dimensions
packed with the same identical material are available to enable seam-
less up- or down-scalability. Diluted samples are efficiently concen-
trated and separated with high resolution and reasonable peak capa-
city. The method can easily be performed under native or denaturing
and/or reducing conditions, depending on the requirements of the
sample.
& In proteome analysis, the preservation of
biological activity could be of minor importance,
as long as recovery is high.
Due to its triple functionality RPC concentrates diluted samples and
efficiently releases samples from salt, urea or other additives prior to
Originally developed for the
purpose of protein purification,
these methods are also of para-
mount importance to pre-frac-
tionate and de-complexify
crude samples in proteome
analysis.
Ion exchange chromatography
is the first choice.
Columns for protein separa-
tions are somewhat cost inten-
sive, but with a favorable life
time if handled with care.

2.1 Basic Principles of Important Liquid Chromatography Techniques 153
separating the proteins at highest possible resolution and is therefore
an excellent consecutive step for all salt containing fractions. Further-
more, due to the use of volatile solvents RPC fractions can directly be
evaporated or lyophilized, prior to tryptic digestion, in the same vessel
or microtiter plate without unnecessary sample transfer steps.
Affinity chromatography is one of the most selective and specific
separation techniques. Numerous ligands, both commercial and self-
made, are available to isolate or remove a variety of proteins and pro-
tein classes. Dealing with this subject in detail would go beyond the
aim of this book. Some dedicated proteomics applications, as the
enrichment of phospho- and glyco-proteins or peptides as well as the
depletion of high-abundant plasma proteins are described below.
Gel filtration is probably the most simple to perform LC technique.
But, due to its low loadability, low resolution and peak capacity, in
combination with its diluting rather than concentrating character it is
not one of the first methods to consider in proteome analysis. Never-
theless, under special conditions it might be worth to consider.
2.1.1
Ion Exchange Chromatography
Ion exchange chromatography (IEX) for the separation of proteins
was introduced in the 1960s and continues to play a major role in the
separation of proteins. Today, IEX is one of the most frequently used
techniques for the separation of proteins and peptides offering high
resolution and loading capacity. The technique is capable of separat-
ing molecular species that have only minor differences in their
charge properties, for example two proteins differing by one charged
amino acid. These features make IEX well suited for concentration,
enrichment, isolation and fractionation.
This chapter provides a general introduction to the theoretical prin-
ciples that underlie every ion exchange separation. An understanding
of these principles will enable the separation power of ion exchange
chromatography to be fully appreciated.
Practical aspects of performing IEX separations related to proteome
analysis are covered in Chapter 2.3.
2.1.1.1 Charge on Proteins
The charge on proteins arises from some of the amino acid side-
chains, as well as the carboxyl and amino termini, bound ions, and
some prosthetic groups.
The charge on amino acid side-chains depends on the pH of the
solution and the pK
A
of the side-chains. It is also affected by the
microenvironment around a side-chain. We assume the following:
IEX is used from micro-scale
separations and analysis
through to fractionation of
micrograms, milligrams, grams
of sample and more.
IEX separates molecules on the
basis of differences in their net
surface charge.
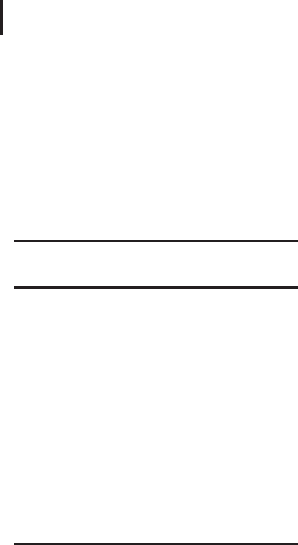
2 Liquid Chromatography Techniques154
pK
A
values for ionizable groups on the protein and that the side-
chains will have the same pK
A
values regardless of their environment
within the protein (Table 2.3). We also assume that the separation is
based on the total net charge of the protein. Therefore, a protein with
a charge of –15 will bind more tightly to an anion exchanger than a
protein with a charge of –10.
Tab. 2.3: pK
A
values of charged amino acids, C- and N-terminus.
Group pK
A
Acids
Carboxyl terminus 3.1
Aspartate 4.4
Glutamate 4.4
Cysteine 8.5
Tyrosine 10.0
Bases
Amino terminus 8.0
Lysine 10.0
Arginine 12.0
Histidine 6.5
The charge on these proteins can be calculated. In brief, when the
pH is less than the pK
A
of a group, the protonated form of the group
predominates. This leaves the acidic side-chains with a charge
approaching 0 and the basic side-chains with a charge approaching a
limiting value of +1. Conversely, when the pH is greater than the pK
A
of a group, the deprotonated form predominates, giving acidic side-
chains a charge approaching –1 and basic side-chains a charge
approaching 0. The charge on the protein is the sum of the charges
on the individual amino acid side-chains.
Molecules vary considerably in their charge properties and will
exhibit different degrees of interaction with charged chromatography
media according to differences in their overall charge, charge density
and surface charge distribution. The charged groups within a mole-
cule that contribute to the net surface charge possess different pK
A
values depending on their structure and chemical microenviron-
ment.
Since all molecules with ionizable groups can be titrated, their net
surface charge is highly pH dependent. In the case of proteins, which
are built up of many different amino acids, containing weak acidic
and basic groups.
Each protein has its own unique net charge versus pH relationship
which can be visualized as a titration curve. Their net surface charge
2.1 Basic Principles of Important Liquid Chromatography Techniques 155
will change gradually as the pH of the environment changes as pro-
teins are amphoteric. The titration curve reflects how the overall net
charge of the protein changes according to the pH of the surround-
ings. Figure 2.1 illustrates some theoretical protein titration curves
(these curves can be generated using an isoelectric focusing gel, but
in practice titration curves are rarely used).
Fig. 2.1: Theoretical protein titration curves, showing
how net surface charge varies with pH.
IEX chromatography takes advantage of the fact that the relationship
between net surface charge and pH is unique for a specific protein.
In an IEX separation reversible interactions between charged mole-
cules and oppositely charged IEX media are controlled in order to
favor binding or elution of specific molecules and achieve separation.
However, at a pH above its isoelectric point, a protein will bind to a
positively charged medium (anion exchanger) and, at a pH below its
pI to a negatively charged medium (cation exchanger). In addition to
the ion exchange interaction, other types of binding may occur. These
effects are mainly due to van der Waals forces and non-polar interac-
tions, which occurs frequently with hydrophobic proteins and pep-
tides.
2.1.1.2 Steps in an IEX Separation
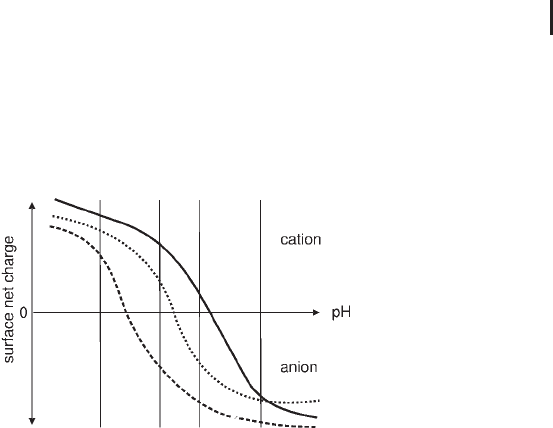
An IEX column comprises a matrix of ideally spherical particles sub-
stituted with ionic groups that are negatively (cationic) or positively
(anionic) charged (see Figure 2.2).
The matrix is usually porous to give a high internal surface area. In
small- and micro-scale separations also non-porous media are used.
The medium is filled into a column to form a packed bed. The bed
is then equilibrated with buffer which fills the pores of the matrix
and the space in between the particles.
A protein that has no net
charge at a pH equivalent to its
isoelectric point (pI) will not
interact with a charged
medium.
2 Liquid Chromatography Techniques156
Fig. 2.2: Principles of an anion exchange separation.

2.1 Basic Principles of Important Liquid Chromatography Techniques 157
The pH and ionic strength as well as the other composition of the
equilibration buffer are selected to ensure that, when sample is
loaded, proteins of interest bind to the column. The proteins which
bind are effectively concentrated onto the column while proteins that
do not have the correct surface charge pass through the column with
the flow of buffer, eluting during or just after sample application.
& Note: The condition of the sample is very
important in order to achieve the most effective
high-resolution separations and make the most
of the high loading capacity. Ideally, samples
should be in the same condition as the starting
buffer.
When the entire sample has been loaded and the column washed so
that all non-binding proteins have passed through the column (i.e.
the UV signal has returned to baseline), conditions are altered in
order to elute the bound proteins.
As ionic strength increases, the salt ions (typically Na
+
or Cl
–
) com-
pete with the bound proteins for charges on the surface of the med-
ium and one or more of the bound species begin to elute and move
down the column. The proteins with the lowest net charge at the
selected pH will be the first ones eluted from the column as ionic
strength increases. Similarly, the proteins with the highest charge at
a certain pH will be most strongly retained and will be eluted last.
By controlling changes in ionic strength using different forms of
gradient, proteins are eluted differentially in a purified, concentrated
form. A wash step with very high ionic strength buffer removes most
tightly bound proteins and impurities at the end of an elution. The
column is then re-equilibrated in start buffer before applying another
sample in the next run.
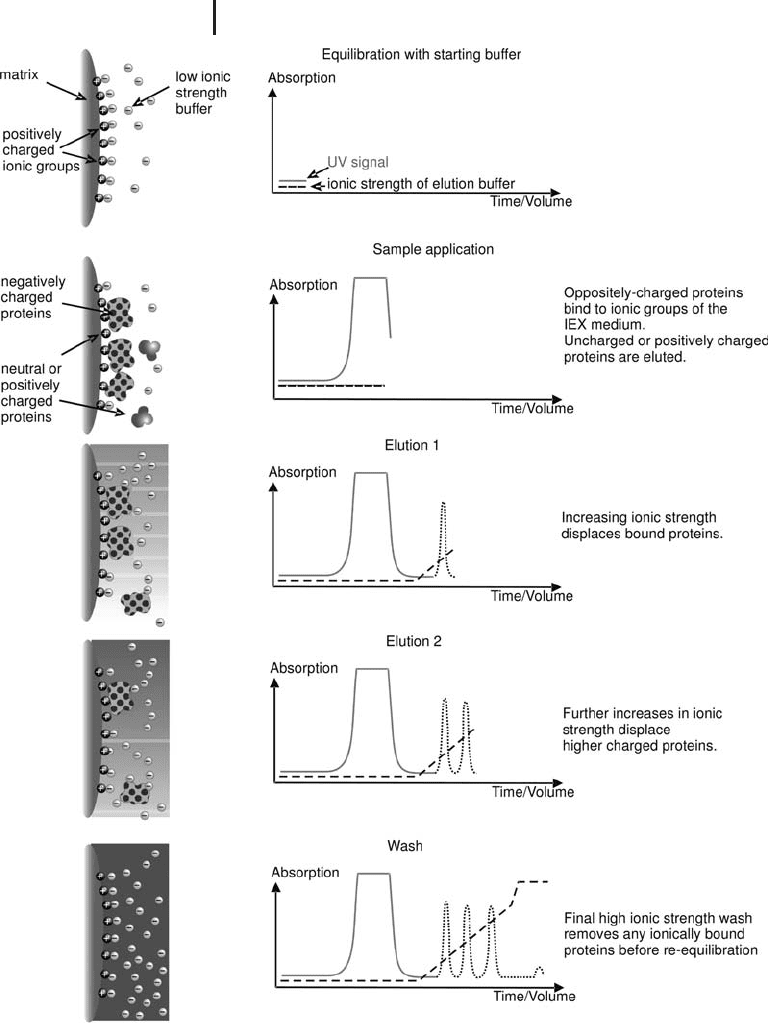
2.1.1.3 Selectivity
Good selectivity (the degree of separation between peaks) is a more
important factor than high efficiency in determining resolution (see
Figure 2.3) and depends not only on the nature and number of the
functional groups on the matrix, but also on the experimental condi-
tions, such as pH (influencing the protein charge), ionic strength and
elution conditions. It is the ease and predictability with which these
experimental conditions can be manipulated, when using a suitably
designed chromatography medium, which gives IEX the potential of
extremely high resolution.
Most frequently, proteins are
eluted by increasing the ionic
strength (salt concentration) of
the buffer.
The higher the net charge of the
protein, the higher the ionic
strength that is needed for
elution.
2 Liquid Chromatography Techniques158
Fig. 2.3: Effect of selectivity and efficiency on resolution.
From GE Healthcare handbook: “Hydrophobic Interaction
and Reversed Phase Chromatography. Principles and Methods”.
2.1.1.4 Selectivity and pH
In protein separation good selectivity is achieved by performing IEX
separations at pH values carefully selected to maximize the differ-
ences in net charge of the components of interest. Optimum selectiv-
ity can be expected at a pH where there is maximum separation
between the titration curves for the individual proteins (i.e. the differ-
ence in net charges between the species is greatest) and when using
an ion exchanger with a charge opposite to the charge of the proteins
at the particular pH. The order in which proteins are eluted cannot
always be predicted with absolute certainty since a titration curve
(produced in practice by measuring electrophoretic mobility in a gel)
reflects the total net charge on a protein and IEX chromatography
depends on the net charge on the surface of the protein.
For the purpose of protein pre-fractionation where in principle all
proteins must be isolated, which in practice is impossible, a pH is
selected with regard to protein stability and recovery for all, or at least
the majority of the proteins.
2.1.1.5 Selectivity and Elution
The figures below illustrate the most common forms of IEX separa-
tion in which proteins are eluted by increasing the ionic strength of a
buffer (typically with NaCl) using linear gradient or step elution. The
UV absorbance and conductivity traces show the elution of protein
peaks and the changes in salt concentration, respectively, during elu-
tion.
A pH interval of plus/minus
1.5 pH unit apart from the
physiological pH seems to be
reasonable.

2.1 Basic Principles of Important Liquid Chromatography Techniques 159
Buffer volumes used during sample application, elution, washing
and re-equilibration are expressed in column volumes, for example 5
CV=5 mL for a column with a 1 mL bed volume.
Using column volumes to describe a separation profile facilitates
method development and transfer of methods to columns of different
dimensions when scaling-up or scaling-down.
Gradient elution is used when starting with an unknown sample
(as many components as possible are bound to the column and eluted
differentially to see a total protein profile) and for high-resolution
separation that are required in protein pre-fractionation.
2.1.1.6 Components of Ion Exchange Media
Chromatography media for ion exchange are made from porous or
non-porous matrices, chosen for their physical stability, their chemi-
cal resistance to stringent cleaning conditions and their low level of
non-specific interaction. The matrices are substituted with functional
groups that determine the charge of the medium.
Matrix
.
High porosity offers a large surface area covered
by charged groups and so ensures a high bind-
ing capacity. High porosity is also an advantage
when separating large proteins. Non-porous
matrices are preferable for extremely high-reso-
lution separations when diffusion effects must
be kept to a minimum.
.
An inert matrix minimizes non-specific interac-
tions with sample components.
.
High physical stability ensures that the volume
of the packed medium remains constant despite
extreme changes in salt concentration or pH,
thus improving reproducibility and avoiding the
need to repack columns.
.
High physical stability and uniformity of particle
size facilitate high flow rates, particularly during
cleaning or re-equilibration steps, to improve
throughput and productivity.
.
High chemical stability ensures that the matrix
can be cleaned using stringent cleaning solu-
tions if required.
.
Modern IEX media use polymer-based matrices
to fulfill not only the requirements for high
binding capacity, chemical and physical stability,
