Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.


2 Liquid Chromatography Techniques160
but to generate media with suitable particle sizes
for a range of applications (Table 2.4).
Tab. 2.4: Ion exchange matrices.
Product Form Mean particle size
MiniBeads Polystyrene/divinyl benzene 3 mm
MonoBeads Polystyrene/divinyl benzene 10 mm
SOURCE 15 Polystyrene/divinyl benzene 15 mm
MiniBeads is a matrix made from polystyrene, with divinyl benzene
as cross-linker, to produce highly spherical (monodispersed), very
small (3 mm), non-porous particles that facilitate micro-preparative or
analytical separations when extremely high resolution is more impor-
tant than high binding capacity or high flow rates.
MonoBeads and SOURCE are matrices made from polystyrene
with divinyl benzene to produce highly spherical (monodispersed),
small (10–15 mm), porous particles
Functional groups The functional groups substituted onto a chroma-
tographic matrix (Table 2.5) determine the charge of an IEX medium
i.e. a positively charged anion exchanger or a negatively charged
cation exchanger.
Tab. 2.5: Functional groups used on ion exchangers.
Anion exchanger Functional group
Quaternary ammonium (Q), strong –O–CH
2
N
+
(CH
3
)
3
Diethylaminoethyl (DEAE), weak –O–CH
2
CH
2
N
+
H(CH
2
CH
3
)
2
Cation exchanger Functional group
Sulfopropyl (SP), strong –O–CH
2
CHOHCH
2
OCH
2
CH
2
CH
2
SO
3
–
Methyl sulfonate (S), strong –O–CH
2
CHOHCH
2
OCH
2
CHOHCH
2
SO
3
–
Carboxymethyl (CM), weak –O–CH
2
COO
–
The terms strong and weak refer to the extent that the ionization
state of the functional groups varies with pH.
Strong ion exchangers show no variation in ion exchange capacity
with change in pH. These exchangers do not take up or lose protons
with changing pH and so have no buffering capacity, remaining fully
The most suitable matrix can
be selected according to the
degree of resolution, binding
capacity and flow rates desired
for the separation.
The terms strong and weak do
not refer to the strength with
which the functional groups
bind the proteins.

2.1 Basic Principles of Important Liquid Chromatography Techniques 161
charged over a broad pH range. Strong ion exchangers include Q
(anionic), S and SP (cationic).
There are several advantages to working with strong ion exchan-
gers:
.
Development and optimization of separations is
fast and easy since the charge characteristics of
the medium do not change with pH.
.
The mechanism of interaction is simple since
there are no intermediate forms of charge inter-
action.
.
Sample loading (binding) capacity is maintained
at high or low pH since there is no loss of charge
from the ion exchanger.
& The majority of proteins have isoelectric points
within the range 5.5 to 7.5, i.e. the first choice
would be to start with a strong anion exchanger
at physiological or slightly basic pH. Tris with a pK
of 8.3 is ideal for buffers with a pH between 7.2
and 9.0.
2.1.1.7 Binding Capacity and Recovery
The capacity of an IEX medium is a quantitative measure of its ability
to take up counter-ions (proteins or other charged molecules). The
total ionic capacity is the number of charged functional groups per
mL medium, a fixed parameter of each medium. Of more practical
relevance is the actual amount of protein which can bind to an IEX
medium, under defined experimental conditions. This is referred to
as the available capacity of a medium for a specific protein. If the
defined conditions include the flow rate at which the medium was
operated, the amount bound is referred to as the dynamic capacity for
the medium. The available and dynamic capacities depend upon the
properties of the protein, the IEX medium and the experimental con-
ditions.
The capacity of an IEX medium will vary according to the molecu-
lar size of the specific protein (which affects its ability to enter all the
pores of the matrix) and its charge/pH relationship (the protein must
carry the correct net charge at a sufficient surface density at the cho-
sen pH). With earlier ion exchange media, larger proteins had limited
access to the functional groups, significantly reducing the binding
capacity. Nowadays, state-of-the-art macroporous polymer-based ion
exchange matrices have exclusion limits for globular proteins in
excess of 110
6
and are therefore suitable for the majority of biomole-
cule separations. Binding capacities will still vary according to the
In practice, in order to achieve
best possible performance, a
protein load of 10 to 20% of
the dynamic capacity is recom-
mended.

2 Liquid Chromatography Techniques162
molecular size of the proteins. For example, a matrix with a high
degree of small pores will exhibit a higher binding capacity for smal-
ler molecules. Experimental conditions such as pH, ionic strength,
counter-ion, flow rate and temperature should all be considered when
comparing binding capacities of different IEX medium. Modern IEX
media show very low levels of non-specific adsorption so that sample
recovery under suitable separation conditions is very high, typically
between 90–100%.
2.1.2
Reversed Phase Chromatography
The following section on reversed phase chromatography (RPC) is
based on information material included in the GE Handbook
“Reversed Phase Chromatography” and modified to the requirements
of protein pre-fraction in proteome analysis.
In the presence of non-polar solvents proteins are more likely to
lose activity than peptides. The interaction of proteins or polypeptides
with a hydrophobic surface in the presence of organic solvents gener-
ally leads to some loss of tertiary structure, often giving rise to differ-
ent conformational states that may interact differently with an RPC
medium. However, denaturation and consequent loss of activity can
be minimized by returning the protein to conditions that favor the
native structure, as demonstrated by the widespread use of RPC for
large-scale purification of recombinant and synthetic proteins and
peptides, such as insulin and growth hormone.
Adsorption chromatography depends on the chemical interactions
between solute molecules and specifically designed ligands chemi-
cally grafted to a chromatography matrix. Over the years, many differ-
ent types of ligands have been immobilized to chromatography sup-
ports for biomolecule separation, exploiting a variety of biochemical
properties ranging from electric charge to biological affinity. An
important addition to the range of adsorption techniques for prepara-
tive chromatography of proteins has been reversed phase chromato-
graphy in which the binding of mobile phase solute to an immobi-
lized n-alkyl hydrocarbon or aromatic ligand occurs via hydrophobic
interaction.
Reversed phase chromatography has found both analytical and pre-
parative applications in the area of biochemical separation and purifi-
cation. Molecules that possess some degree of hydrophobic character,
such as proteins and peptides, can be separated by reversed phase
chromatography with excellent resolution and recovery. In addition,
the use of ion pairing modifiers in the mobile phase allows reversed
phase chromatography of charged solutes such as hydrophilic pep-
tides. Preparative reversed phase chromatography has found applica-
GE Handbook “Reversed Phase
Chromatography, Principles
and Methods”
GE Handbook “Hydrophobic
Interaction and Reversed Phase
Chromatography, Principles
and Methods”
Unless precipitation occurs,
denaturation is not a problem
when using RPC to separate
proteins or peptides for primary
structure determination and
proteome analysis.
2.1 Basic Principles of Important Liquid Chromatography Techniques 163
tions ranging from micropurification of protein fragments for
sequencing by mass spectrometry to process scale purification of
recombinant protein products.
2.1.2.1 Theory and Mechanism of Reversed Phase Chromatography
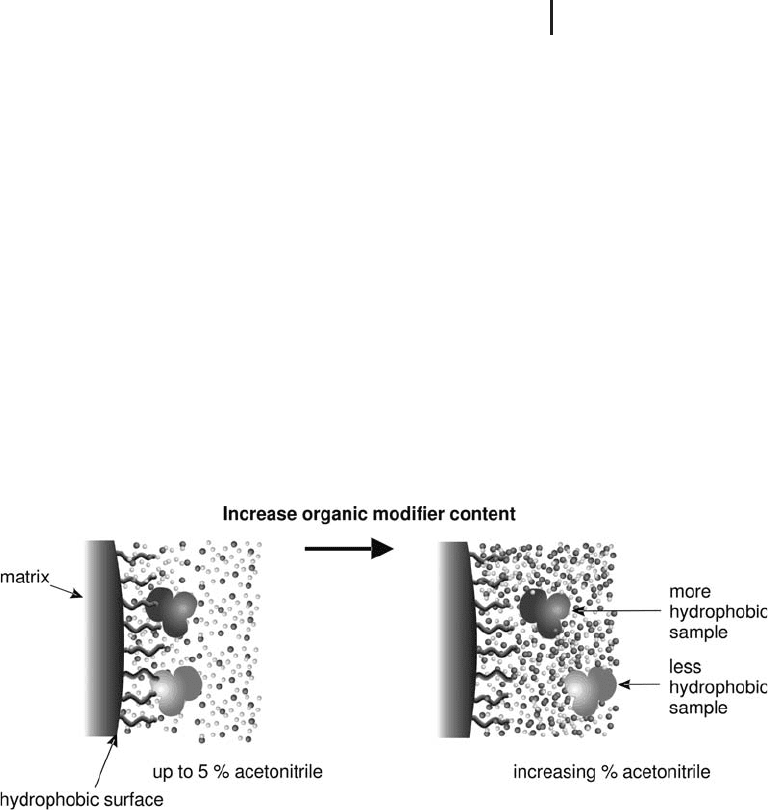
The actual nature of the hydrophobic binding interaction itself is a
matter of a heated debate, but the conventional wisdom assumes the
binding interaction to be the result of a favorable entropy effect. The
initial mobile phase binding conditions used in reversed phase chro-
matography are primarily aqueous which indicates a high degree of
organized water structure surrounding both the solute molecule and
the immobilized ligand (see Figure 2.4). As solute binds to the immo-
bilized hydrophobic ligand, the hydrophobic area exposed to the sol-
vent is minimized. Therefore, the degree of organized water structure
is diminished with a corresponding favorable increase in system
entropy. In this way, it is advantageous from an energy point of view
for the hydrophobic moieties, i.e. solute and ligand, to associate.
Fig. 2.4: Interaction of a solute with a typical reversed
phase medium. Water adjacent to hydrophobic regions is
postulated to be more highly ordered than the bulk water.
Reversed phase chromatography is an adsorptive process by experi-
mental design, which relies on a partitioning mechanism for small
peptides, in combination with an “on/off” adsorption effect for intact
bigger proteins to accomplish the separation. The solute molecules
partition (i.e. an equilibrium is established) between the mobile
phase and the stationary phase. The distribution of the solute
between the two phases depends on the binding properties of the
medium, the hydrophobicity of the solute and the composition of the
The separation mechanism in
reversed phase chromatography
depends on the hydrophobic
binding interaction between the
solute molecule in the mobile
phase and the immobilized
hydrophobic ligand, i.e. the
stationary phase.

2 Liquid Chromatography Techniques164
mobile phase. Initially, experimental conditions are designed to favor
adsorption of the solute from the mobile phase to the stationary
phase. Subsequently, the mobile phase composition is modified to
favor desorption of the solute from the stationary phase back into the
mobile phase. In this case, adsorption is considered the extreme equi-
librium state where the distribution of solute molecules is essentially
100% in the stationary phase. Conversely, desorption is an extreme
equilibrium state where the solute is essentially 100% distributed in
the mobile phase.
While proteins strongly adsorb to the surface of a reversed phase
matrix under aqueous conditions, they desorb from the matrix within
a very narrow window of organic modifier concentration. Along with
these high-molecular-weight proteins with their unique adsorption
properties, the typical biological sample usually contains a broad mix-
ture of proteins with a correspondingly diverse range of adsorption
affinities. The only practical method for reversed phase separation of
complex biological samples, therefore, is gradient elution. In sum-
mary, separations in reversed phase chromatography depend on the
reversible adsorption/desorption of solute molecules with varying
degrees of hydrophobicity to a hydrophobic stationary phase. The
majority of reversed phase separation experiments are performed in
several fundamental steps.
The first step in the chromatographic process is to equilibrate the
column packed with the reversed phase medium under suitable
initial mobile phase conditions of pH, ionic strength and polarity
(mobile phase hydrophobicity). The polarity of the mobile phase is
controlled by adding organic modifiers such as acetonitrile. Ion-pair-
ing agents, such as trifluoroacetic acid (TFA) or formic acid (FA), are
used in the majority of all protein and peptide separations with RPC.
The polarity of the initial mobile phase (usually referred to as mobile
phase A) must be low enough to dissolve the partially hydrophobic
solute yet high enough to ensure binding of the solute to the reversed
phase chromatographic column.
In the second step, the sample containing the solutes to be sepa-
rated is applied. Ideally, the sample is dissolved in the same mobile
phase used to equilibrate the stationary phase. The sample is applied
to the column at a flow rate where optimum binding will occur.
Once the sample is applied, the chromatographic column is
washed further with mobile phase A in order to remove any unbound
and unwanted solute molecules. Bound solutes are next desorbed
from the reversed phase medium by adjusting the polarity of the
mobile phase so that the bound solute molecules will sequentially
desorb and elute from the column.
In reversed phase chromatography this usually involves decreasing
the polarity of the mobile phase by increasing the percentage of
Reversed phase chromato-
graphy of proteins generally
uses gradient elution instead of
isocratic elution.

2.1 Basic Principles of Important Liquid Chromatography Techniques 165
organic modifier in the mobile phase. This is accomplished by main-
taining a high concentration of organic modifier in the final mobile
phase (mobile phase B). Generally, the pH of the initial and final
mobile phase solutions remains the same.
The gradual decrease in mobile phase polarity (increasing mobile
phase hydrophobicity) is achieved by an increasing linear gradient
from 100% initial mobile phase A containing little or no organic
modifier to 100% (or less) mobile phase B containing a higher con-
centration of organic modifier. The bound solutes desorb from the
reversed phase medium according to their individual hydrophobici-
ties.
The fourth step in the process involves removing substances not
previously desorbed. This is generally accomplished by changing
mobile phase B to near 100% organic modifier in order to ensure
complete removal of all bound substances prior to re-using the col-
umn.
The fifth step is re-equilibration of the chromatographic medium
from 100% mobile phase B back to the initial mobile phase condi-
tions.
The degree of solute molecule binding to the reversed phase med-
ium can be controlled by manipulating the hydrophobic properties of
the initial mobile phase. Although the hydrophobicity of a solute
molecule is difficult to quantitate, the separation of solutes that vary
only slightly in their hydrophobic properties is readily achieved.
Because of its excellent resolving power, reversed phase chromatogra-
phy is an indispensable technique for the high-performance separa-
tion of complex biological samples.
Typically, a reversed phase separation is initially achieved using a
broad range gradient from 100% mobile phase A to 100% mobile
phase B. The amount of organic modifier in both the initial and final
mobile phases can also vary greatly.
& For peptide separations routine percentages of
organic modifier are 5% or less in mobile phase A
and 80% or more in mobile phase B. For intact
protein separations starting with 25% organic
modifier in mobile phase A and 75% or more in
mobile phase B can be generally recommended. In
most cases, acetonitrile is the organic modifier of
choice.
The technique of reversed phase chromatography allows great flexibil-
ity in separation conditions so that the researcher can choose to bind
the solute of interest, allowing the contaminants to pass unretarded
through the column, or to bind the contaminants, allowing the
desired solute to pass freely. Generally, it is more appropriate to bind
Separation in reversed phase
chromatography is due to the
different binding properties of
the solutes present in the
sample as a result of the differ-
ences in their hydrophobic
properties.

2 Liquid Chromatography Techniques166
the solute of interest because the desorbed solute elutes from the
chromatographic medium in a concentrated state. Additionally, since
binding under the initial mobile phase conditions is complete, the
starting concentration of desired solute in the sample solution is not
critical allowing dilute samples to be applied to the column. Ionic
binding may sometimes occur due to ionically charged impurities
immobilized on the reversed phase chromatographic medium, espe-
cially for non- or insufficiently end-capped silica-based media.
The combinations of hydrophobic and ionic binding effects are
referred to as mixed-mode retention behavior. Ionic interactions can
be minimized by judiciously selecting mobile phase conditions and
by choosing reversed phase media with end-capping or based on poly-
mers which are commercially produced with high batch-to-batch
reproducibility and stringent quality control methods.
2.1.2.2 RPC of Peptides, Polypeptides and Proteins
The separation of peptides involves continuous partitioning between
the mobile and stationary phase. Polypeptides and proteins, however,
are too large to partition into the hydrophobic layer; instead, they
adsorb to the hydrophobic phase after entering the column and
remain adsorbed until the concentration of the mobile phase reaches
the critical concentration necessary to cause desorption. They then
adsorb and interact only slightly with the stationary phase as they
elute down the column.
& Desorption takes only place within a very narrow
window of organic modifier concentration. This
results in complete retention until the critical
mobile phase concentration is reached and
sudden desorption of the protein takes place. This
sudden desorption produces sharp peaks.
The “hydrophobic foot” of a polypeptide or protein which is responsi-
ble for the separation, is very sensitive to molecular confirmation. It
can be observed that proteins with intact tertiary structure elute ear-
lier than expected compared to denatured proteins because the
“hydrophobic foot” is involved in the interaction, while the rest of the
protein is in contact with the mobile phase. After desorption very lit-
tle interaction takes place between the protein and the reversed phase
surface and subsequent interactions have little effect on the separa-
tion, i.e. column can be kept short, as column length has little effect
on the separation. Short columns in combination with shallow gradi-
ents can be used very effectively to separate similar proteins. Because
proteins diffuse slowly, RPC results in broader peaks than obtained
with peptides.

2.1 Basic Principles of Important Liquid Chromatography Techniques 167
Column dimensions The adsorption/desorption of proteins respon-
sible for their separation takes place almost entirely near the top of
the column. Therefore, column length does not significantly affect
separation and resolution of proteins. Consequently, short columns
of £ 5 cm in length are often used for protein separation.
Mobile phase considerations
& Since decades, all in all acetonitrile as organic
modifier and TFA as ion-pairing reagent has
proven to be best compromise for reversed phase
chromatography of peptides and proteins. For
direct coupling of LC to mass spectrometry acetic
or formic acid are giving better overall results due
to significant higher ionization efficiency.
Propanol-2 is often used for large or very hydrophobic proteins. The
major drawback of propanol-2 is its high viscosity, especially in mix-
tures with water. To reduce the viscosity of propanol-2, while main-
taining its hydrophobic characteristics, it is recommended using mix-
ture of 50:50 with acetonitrile. Adding 1 to 3% propanol-2 to acetoni-
trile has shown to increase protein recovery in some cases.
Propanol-2 is the best solvent for retaining biological activity. Etha-
nol and methanol are slightly worse. Acetonitrile causes the greatest
loss of biological activity, however, for protein pre-fractionation of pro-
teomic samples this is of no importance, as long as the primary struc-
ture of the molecule remains unaffected and the recovery is suffi-
cient.
In rare cases trifluoroacetic acid (TFA) can be replaced by penta-
fluoropropionic acid (PFPA) or heptafluorobutyric acid (HFBA).
However, the effect is minimal to non-visible.
2.1.3
Affinity Chromatography
Affinity chromatography (AC) separates proteins on the basis of a
reversible interaction between a protein or a group of proteins and a
specific ligand coupled to a chromatography matrix, as illustrated and
described in full detail in the GE Handbook “Affinity Chromatrogra-
phy, Principles and Methods”.
The technique offers high selectivity, hence high resolution, and
usually high capacity for the protein(s) of interest. Enrichment can be
in the order of several thousand-fold and recoveries of active material
are generally very high.
There is no good reason to
deviate from TFA as the ion-
pairing reagent and invest time
and money to evaluate exotic
reagents. RPC coupled to MS
or MS/MS may be seen as an
exception and it is worth trying
other ion pairing reagents to
obtain a better performance in
MS.

2 Liquid Chromatography Techniques168
Affinity chromatography is unique in separation technology since
it is the only technique that enables the purification of a biomolecule
on the basis of its biological function or individual chemical struc-
ture. Purification that would otherwise be time-consuming, difficult
or even impossible using other techniques can often be easily
achieved with affinity chromatography. The technique can be used to
separate active proteins from denatured or functionally different
forms, to isolate, concentrate and enrich pure substances present at
low concentration in large volumes of crude sample and also to
remove specific contaminants, such as the depletion of high-abun-
dant proteins from plasma or serum, prior to proteome analysis.
2.1.3.1 Affinity Chromatography in Brief
Affinity chromatography separates proteins on the basis of a reversi-
ble interaction between a protein or a group of proteins and a specific
ligand coupled to a chromatography matrix.
The technique is ideal for concentration or enrichment steps as
well as for a depletion or elimination in a separation protocol and can
be used whenever a suitable ligand is available for the protein(s) of
interest. With high selectivity, hence high resolution, and high capa-
city for the protein(s) of interest, purification levels in the order of
several thousand-fold with high recovery of active material are achiev-
able.
Biological interactions between ligand and target molecule can be a
result of electrostatic or hydrophobic interactions, and/or hydrogen
bonding. To elute the target molecule from the affinity medium the
interaction can be reversed, either specifically using a competitive
ligand, or non-specifically, by changing the pH, ionic strength or
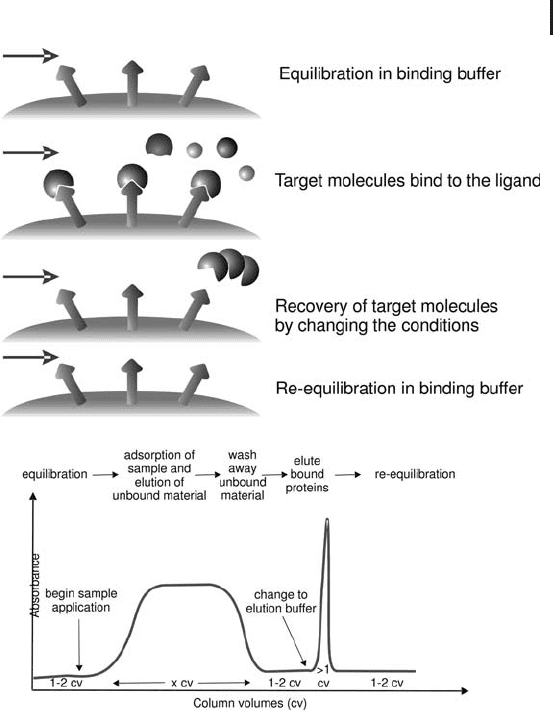
polarity. Figure 2.5 describes the mode of action of affinity chromato-
graphy. In a single step, affinity purification can offer immense time-
saving over less selective multi-step procedures. The concentrating
effect enables large volumes to be processed. Target molecules can be
separated from complex biological mixtures, native forms can be
separated from denatured forms of the same substance and small
amounts of biological material can be purified from high levels of
contaminating substances.
GE Handbook “Affinity Chro-
matography, Principles and
Methods” (2002) 18-1022-29
2.1 Basic Principles of Important Liquid Chromatography Techniques 169
Fig. 2.5: Schematic representation of the mode of operation of
affinity chromatography and a typical affinity chromatogram.
Successful affinity purification requires a biospecific ligand that can
be covalently attached to a chromatography matrix. The coupled
ligand must retain its specific binding affinity for the target mole-
cules and, after washing away unbound material, the binding
between the ligand and target molecule must be reversible to allow
the target molecules to be removed in an active form. Any component
can be used as a ligand to purify its respective binding partner. Some
typical biological interactions, frequently used in affinity chromato-
graphy for proteomics, are listed below:
.
Antibody–antigen: for efficient IgG and albumin
depletion from plasma or serum samples;
.
Lectins: selective glycoprotein enrichment;
