Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.


2 Liquid Chromatography Techniques190
The first step in protein pre-fractionation can still be regarded as sam-
ple preparation for the purpose of eliminating irrelevant proteins
such as albumin and immuno-globulins and other high-abundancy
proteins in plasma or serum where affinity chromatography is the
method of choice. The flow-through fraction is then subject to intelli-
gent multidimensional protein pre-fractionation and sample de-com-
plexification, while the remaining proteins on the column will be
stored for other investigations or simply discarded during column
equilibration and cleaning.
And that is going to require development of new technologies as
well as the refinement of already existing tools, like e.g. affinity chro-
matography. It can be efficiently used to simplify protein samples by
enriching subsets of the proteome such as phosphorylated proteins
by IMAC, glycoproteins on lectin columns, just to mention the most
frequently used applications. In this case the retained proteins are of
main interest and the flow-through fraction is less important but
might be stored for continuative investigations.
Samples that do not contain any major contaminating proteins can
be processed directly. For reasons of simplicity and clarity the follow-
ing explanations are covering two-dimensional separations only,
which in practice will be relevant in most cases.
& Note: Too many steps can result in an
unacceptable loss of proteins. In this case less
can be more. As much separation as necessary,
but as little as possible. Sticking to the “keep it
simple” rule can be a good advice and a
consideration for success.
See also Figure 6 on page 9.
Even with the most sophisticated strategy, and the best analysis
techniques, it will – for the foreseeable future – not be possible to iso-
late all proteins in a complex proteomics sample.
In order to identify and characterize proteins present at very low
concentrations, it is inevitably to start with large sample amounts
and/or volumes in order to assure their presence in sufficient concen-
tration. One of the most exiting samples is probably blood. Dealing
just with complex samples is already difficult enough; however, if the
composition of a sample is similar to plasma, the challenges are
increasing exponentially. About 90% of the amount of plasma pro-
teins is albumin and IgG and just 25 high-abundant proteins repre-
sent 99% of the total amount. The remaining thousands (or mil-
lions?) of proteins are distributed over a huge concentration range
(see Figure 2.16).
Post-translational modifications
are perhaps the largest frontier
for proteomics.
Sample complexity and
dynamic range
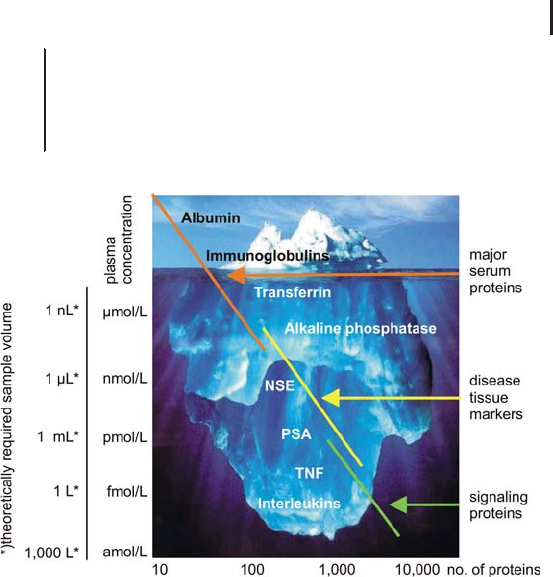
2.3 Liquid Chromatography Techniques and Applications in Proteome Analysis 191
& Plasma is the largest and deepest version of the
human proteome: Largest, i.e. most proteins
(estimates range from >30,000 to >10,000,000).
Deepest, i.e. widest dynamic range (>10
9
). See
paper by Anderson and Anderson (2002).
Fig. 2.16: The analogy with an iceberg illustrates the challenges in
proteomics in terms of sensitivity and dynamic range. In order to
detect a biomarker present at a concentration of 1 fmol/L, theoretically
a sample volume of 1L is required, if the LOD of the MS equipment
is 1fmol.
Currently there is research ongoing for developing strategies on how
to tackle this demanding dynamic range and sensitivity challenge.
Key to this task is to get rid of as many of the low-interest high-
abundant proteins as possible (tip of the iceberg), without losing too
many of the free low-abundant proteins and those bound to carrier
proteins like e.g. albumin. High-abundant protein affinity depletion
followed by a 2DLC (IEX, RPC) strategy on protein level as described
further down seems to be the most promising approach. As much as
possible a sample load combined with high-recovery/low-loss separa-
tion steps and a high-sensitivity mass spectrometer are essential pre-
requisites for the successful deciphering of at least parts of the
human plasma proteome.
A simple calculation may help to highlight the current limits and
limitations. Let us assume that a state-of-the-art mass spectrometer
can routinely operate at the low femtomole level. If you start with
1 milliliter of sample you theoretically can come to the 10
–12
mmol/L
Anderson NL, Anderson NG.
Mol Cell Proteomics 1 (2002)
845–867.
2 Liquid Chromatography Techniques192
range in the iceberg picture, providing a 100% recovery, which is very
unlikely, since complex separations cannot be performed quantita-
tively. Although this is a question of simple mathematics, it is not
always appreciated. A possible solution might be to apply more sam-
ple, if possible or available, or improve techniques, methods, and
skills in order to attack the atto- or even zeptomolar range. Such
results are published these days and achievable by the top research
labs of the world.
Theoretically, applying 1 L of serum or a zeptomolar sensitivity
mass spec, or better both, would provide the desired performance of a
12 to 15 orders of magnitude dynamic range.
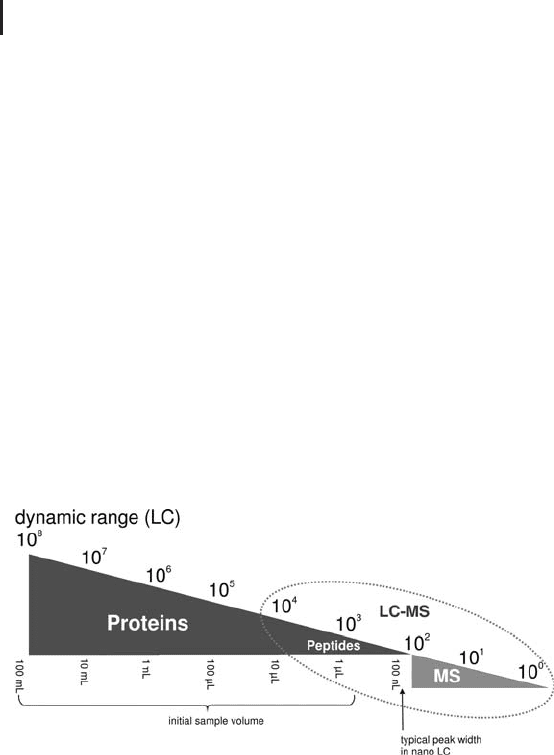
LC-MS manufactures claim that the combination of nanoLC and
nanoESI at peptide level covers a dynamic range of 10
4
(see Figure
2.17). A typical peak volume at this scale is ideally in the range of
100 nL. Closing the gap with the desired dynamic range is only possi-
ble if more sample can be acquired at the very beginning and concen-
trated in an intelligently designed workflow with high recovery. The
more we (can) load, the more we see.
Fig. 2.17: Expanding the overall dynamic range by increasing
the initial sample load.
The HUPO organization currently promotes a series of different
initiatives to work on the proteomes of plasma (discussed above),
brain, liver, and more are expected to follow soon. For all these com-
plex samples in principle – with specific and dedicated sample pre-
paration – very similar strategies can be applied. A schematic general
workflow is shown in Figure 2.18.
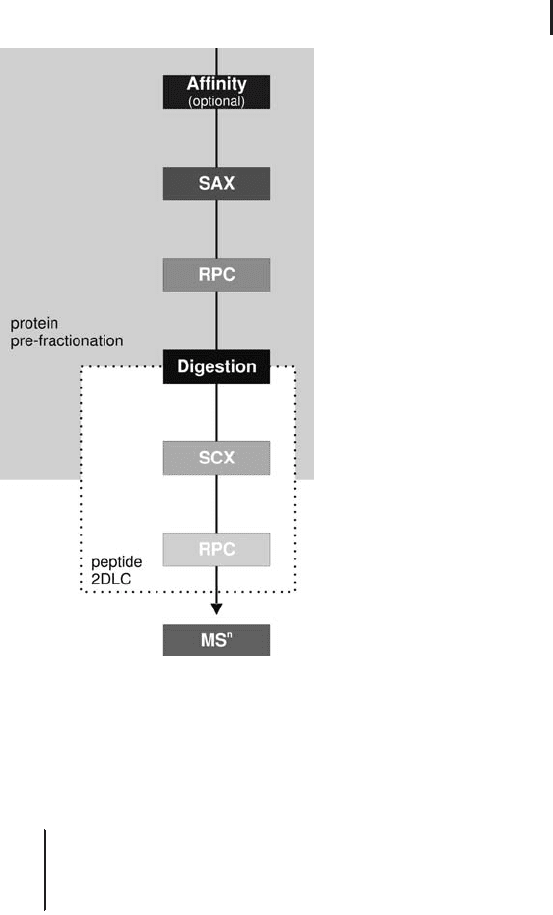
2.3 Liquid Chromatography Techniques and Applications in Proteome Analysis 193
Fig. 2.18: Schematic flow diagram showing the separation
steps on protein and peptide level.
Peak capacities and separation performance in protein
pre-fractionation
& Today, even with the most sophisticated tools and
intelligent separation strategies it is impossible to
achieve high purity for each protein in a
proteomics sample.
Human plasma consists of at least some 100,000 proteins, post trans-
lational modifications, splice variants and isoforms not yet included.
A pre-fractionation into 1,000 fractions still results in more than 100
proteins per fraction in average, but each single fraction probably
simple enough to be identified and characterized after digestion by
peptide 2DLC-MS. Literature reports of capacities for just 1D RPC of
200 to 500 peaks; however, in practice under normal lab conditions

2 Liquid Chromatography Techniques194
one can be happy if 10% of the theoretical values will be achieved.
Table 2.9 shows expected peak capacities for real samples in practice.
Tab. 2.9: Expected peak capacities on intact protein level under
practical laboratory conditions.
Affinity chromatography >2
Ion exchange chromatography ~20
Gel filtration ~10
Reversed phase chromatography ~50
2.4
Practical Considerations and Application of LC-based Protein
Pre-fractionation
In contrast to the well established electrophoresis techniques, the
application of liquid chromatography in proteomics is a – surpris-
ingly – rather young discipline. Fortunately, especially for the widely
used separation of tryptic peptides, both for one- and two-dimen-
sional separations, most researchers worldwide rely more or less on
the same recipe of running conditions with a very few minor – indivi-
dual – modifications only.
In protein pre-fractionation – a very promising approach still in its
infancy – we have not yet achieved any kind of standardization or
standard operation procedures (SOP). Based on the well founded and
long lasting history with chromatography in general and protein pur-
ification in particular, it was not too difficult to come up with a range
of suitable approaches and technical solutions as described by Apffel
(2003) and Dixon et al. (2006).
Although the purpose of protein purification is somewhat different
compared to protein pre-fractionation (PPF), as shown in Table 2.10,
by far most of the well established rules and basic considerations are
applicable for this new strategy in proteomics, too. Although the
same equipment, columns and expertise is used, the objective of each
approach is different.
Apffel A. In Simpson RJ, Ed.
Purifying proteins for proteo-
mics: A laboratory manual.
Cold Spring Harbor Laboratory
Press, Cold Spring Harbor,
New York (2003) pp. 75–100.
Dixon SP, Pitfield ID, Perrett
D. Biomed Chromatogr 207
(2006) 508–529.
2.4 Practical Considerations and Application of LC-based Protein Pre-fractionation 195
Tab. 2.10: The differences between protein purification and pre-
fractionation.
Protein purification „ protein pre-fractionation
The aim of protein purification is to
obtain a single fraction with a single
pure protein for continuative studies.
The protein pre-fractionation procedure
in proteomics results in very many
fractions with each still containing tens
to hundreds of proteins
During the purification process the
number of target protein containing
fractions decreases.
During the pre-fractionation process the
number of fractions increases exponen-
tially.
The fractions containing contaminant
proteins are discarded.
All fractions are of the same high value.
Knowing that within the next future a lot of new and probably even
more successful approaches will enter the scene, this section will
only cover one protein pre-fractionation workflow as a typical exam-
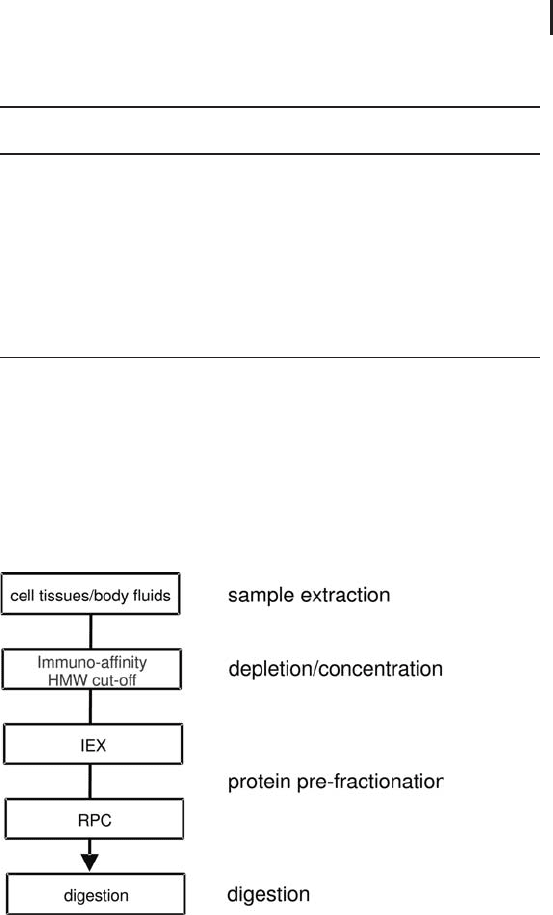
ple. This universal two-dimensional workflow combines ion-exchange
with reversed phase chromatography (see Figure 2.19). With some
individual modifications only, it can be used for a variety of samples.
Fig. 2.19: Schematics of the protein part of the LC-based
proteome analysis workflow.

2 Liquid Chromatography Techniques196
2.4.1
Sample Extraction and Preparation
In principle exactly the same methods for sample preparation as
described in Section 1.5.1 can be applied. As liquid chromatography
deals with UV detection the purity requirements for the reagents can
be significantly higher compared to electrophoresis.
& Note: Very hydrophobic proteins, e.g. membrane
proteins, are not solubilized with this type of
buffers and are thus not accessible with the
methods described below.
2.4.1.1 Plasma, Serum, CSF, and other Body Fluids
A special sample preparation step for the removal of the highest-
abundant proteins is required in most of the cases. Such a depletion
step can be performed by applying immuno-affinity techniques.
Despite its unique selectivity immuno-affinity chromatography does
not meet the requirements for loading capacity or economy. Just a
few microliters of plasma can be loaded and the running costs are
high. In addition, caused by unavoidable leaking effects, small
amounts of antibodies can be released into the elution buffer that
might result in ambiguous protein identification further downstream
in the workflow.
Recently, a hypothesis is under discussion that claims that the low-
molecular-weight region of the blood proteome, which is a mixture of
small intact proteins plus fragments of the large proteins, represents
all classes of proteins. As stated by Liotta et al. (2003) and Tirumalai
et al. (2003) this fraction is expected to be the ideal source for biomar-
ker discovery.
Such an approach, described by Tanaka et al. (2006), suggests to
cut-off the high-molecular-weight and concentrate the low-molecular-
weight proteins by dedicated membrane or hollow-fiber devices and
to continue the analysis with the low-molecular-weight fraction only.
Another remarkable paper by Wagner et al. (2002) describes the
application of protein mapping of biological samples of human
hemofiltrate for the analysis of proteins and peptides with a molecu-
lar weight below 20 kDa.
Once the sample has been extracted and prepared in a suitable way,
a two-dimensional protein pre-fractionation – ion-exchange chroma-
tography in combination with reversed phase chromatography – will
follow.
Liotta LA, Ferrari M, Petricoin
E. Clinical proteomics: written
in blood. Nature 425 (2003)
905.
Tirumalai RS, Chan KC, Prieto
DA, Issaq HJ, Conrads TP,
Veenstra TD. Mol Cell Proteo-
mics 2 (2003) 1096–1103.
Tanaka Y, Akiyama H, Kuroda1
T, Jung G, Tanahashi K, Sugaya
H, Utsumi J, Kawasaki H,
Hirano H. Proteomics 6
(2006) 4845–4855.
Wagner K, Miliotis T, Marko-
Varga G, Bischoff R, Unger KK.
Anal Chem 74 (2002) 809–
820.

2.4 Practical Considerations and Application of LC-based Protein Pre-fractionation 197
& Before starting with the most valuable and
difficult sample it is strongly recommended to
learn and establish the new method in the
laboratory by using a well known simple standard
sample first. This period can take or save several
weeks and months but is a basic prerequisite for
the application of each new technique.
2.4.2
Experimental Setup
2.4.2.1 Scale of Operation
As mentioned earlier in Section 2.2 it is advantageous to start with a
higher initial amount of protein in order to extend the overall
dynamic range, enable access to lower-abundant proteins and last but
not least to compensate for the inevitable losses in each of the indivi-
dual steps in the entire workflow until the sample finally enters the
mass spectrometer.
Protein pre-fractionation as described here permits the separation
of initial sample loads between » 100 mg and » 100 mg of protein.
Protein pre-fractionation at an even higher, industrial scale has been
reported.
2.4.2.2 Type of Instrumentation
In principle any kind of biocompatible HPLC system equipped with a
fraction collector can be used to perform protein pre-fractionation.
However, for the efficient separation of proteins and peptides it is
very often necessary to use buffers with high salt concentrations at
very acidic or basic pH that are extremely corrosive to stainless steel
components which are commonly used in conventional HPLC equip-
ment. Besides contaminating the sample with corrosion products
that are released from the metal surfaces it may also result in a mod-
ifications of the proteins and finally in increased maintenance and
replacement costs for the instrumentation.
& It is recommended to use a dedicated
biocompatible, high-performance bio molecule
separation system. Such systems have a
completely bio-inert flow path and can withstand
the even most aggressive buffers, solvents and
high salt concentrations (like 8 mol/L urea).
Rose K, et al. Proteomics 2004,
4, 2125–2150.
Conventional HPLC systems
are intended for small molecule
analytical rather than bio mole-
cule preparative use.

2 Liquid Chromatography Techniques198
For convenience and ease-of-use the fraction collector should be com-
patible with all different kind of vessels including microtiter plates.
KTA and Ettan LC have proven to be suitable for protein pre-
fractionation and can be recommended without any restriction or
limitation.
2.4.2.3 Choice of Chromatography Column
The number of HPLC columns on the market is huge and unma-
nageable. It is recommended to use columns that have proven to per-
form well for protein separations. Except for RPC it is essential that
the columns are made of biocompatible material such as glass,
PEEK
or other suitable plastic material.
& Packing material that is based on silica is not
recommended as it is unstable at pH above 7.5. In
order to avoid cross-contamination from one
sample to another the column must survive a
cleaning-in-place (CIP) process, i.e. the column
must tolerate wash steps with 1 mol/L NaOH or
HCl. A column packed with porous polymer based
material is recommended.
Non-porous (NPS) columns show superior resolution but offer only
low loading capacity. Besides a good separation performance protein
recovery is a key feature.
2.4.2.4 Monitoring Each Separation Step, Monitoring Each Dimension
In general, but especially during the method development process it
is beneficial to monitor progress and performance of each protein
pre-fractionation step by analyzing each, or representative fractions
by SDS electrophoresis, and – if appropriate or necessary – optimize
the procedure, as outlined in the poster of K. Marcus et al. (2005).
2.4.3
Ion Exchange Chromatography and Protein Pre-fractionation
An ideal technique to deal with samples of crude protein extracts –
small to large amounts or volumes, native, denaturing or reducing
conditions – is ion exchange chromatography. This technique can be
operated at any scale, from micro to macro scale. Due to its adsorptive
mode diluted protein solutions can be concentrated and separated
with high resolution in less than an hour. Its ease of use and its logi-
Good batch-to-batch reproduci-
bility and long-term availability
is best with big and well estab-
lished manufactures.
K. Marcus, et al. “A novel
approach towards differential
proteomics with multidimen-
sional intact protein prefractio-
nation of brain samples” Poster
at the HUPO 4th Annual
World Congress Munich
(2005).
2.4 Practical Considerations and Application of LC-based Protein Pre-fractionation 199
cal, directly adaptable theory made IEX the most widely used techni-
que for protein separations.
In order to benefit from the concentration effect all proteins should
bind to the column. In theory this can be achieved by loading the
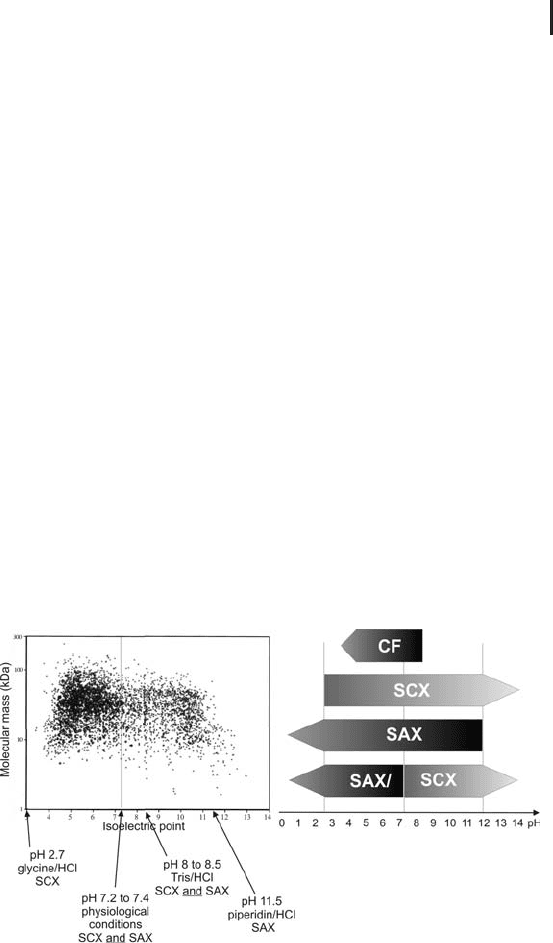
sample at very low pH (e.g. glycine/HCl buffer) to a cation, or at very
high pH (e.g. piperidine/HCl buffer) to an anion exchange column.
However, these extreme pH conditions are not compatible with pro-
tein stability. Physiological pH or slightly basic conditions (Tris/HCl
buffer) are more favorable conditions for high protein recovery.
In cases, where AIEX does not give satisfying results, or in the tan-
dem IEX approach, see below in Figure 2.20, the application of CIEX
should be considered.
Recently, another technique based on charge, or more precisely, on
isoelectric point (IEP, or pI) had its renaissance in the area of protein
pre-fractionation: chromatofocusing (CF). CF can be regarded as the
LC analogue to isoelectric focusing in electrophoresis, however, with-
out achieving its separation performance. Although the theory is very
convincing, in practice this technique is suffering from a very
restricted pH range, protein precipitation at the isoelectric point,
high running costs and the necessity to remove the pH gradient
creating ampholytic reagents (Polybuffer) after the separation. It is
quite unlikely that CF will have a major breakthrough in the near
future.
Fig. 2.20: Potential operating area for charge- and pI-based protein
separation (theoretical spot map modified from Link et al. 1997).
Experience from several
decades indicates that anion
exchange chromatography is
the first choice for the separa-
tion of intact proteins.
In general anion-exchange-
chromatography is the first
choice for the separation of
intact proteins.
Link AJ, Robison K, Church
GM. Electrophoresis 18 (1997)
1259–1313.
