Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.

2 Liquid Chromatography Techniques200
2.4.3.1 IEX under Denaturing Conditions
As the most efficient sample extraction takes place under denaturing
conditions it is a natural choice to maintain these conditions also dur-
ing ion-exchange chromatography. Inter and intra protein interac-
tions are widely suppressed and solubility of proteins is improved.
The addition of protein inhibitors reduces protease activities and
maintains the integrity of the sample. Low percentages of organic
modifier (e.g. 6% propanol-2) or the presence of DTT might be advan-
tageous (Wiegand et al. 2002). Ion exchange chromatography (IEX) is
the natural choice for the first dimension in protein pre-fractionation.
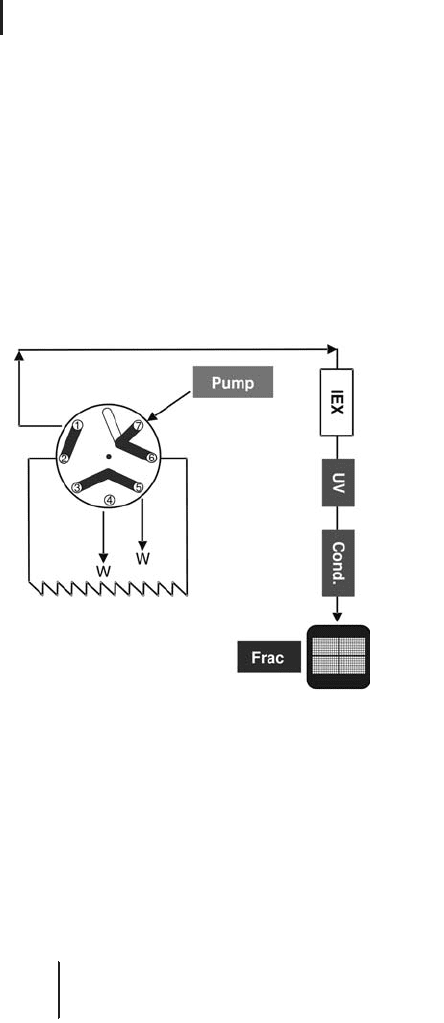
Figure 2.21 illustrates the schematic flow path for the IEX separation.
Fig. 2.21: IEX (first dimension) flow path with single sample injection.
Even small IEX columns have a significant loading capacity for high
protein amounts in the range of 5–10 mg/mL for best possible perfor-
mance. The volume loading capacity in principle is unlimited, i.e.
even large volumes of 100 mL and more of diluted sample can be
applied.
Applications of high sample volumes, of samples with high protein
concentrations or samples with high viscosity have to be handled
with care.
& During sample introduction it is recommended to
considerably reduce the flow rate in order not to
exceed the pressure limit of the column.
The adsorbed proteins are eluted from the column with an ascending
salt gradient and collected in a fraction collector. A fraction size
Wiegand G, Parbel A, Seifert
MHJ, Holak TA, Reuter W. Eur
J Biochem 269 (2002) 5046–
5055.
IEX is a retentive technique
where proteins can be effec-
tively concentrated.

2.4 Practical Considerations and Application of LC-based Protein Pre-fractionation 201
approximately three times the base peak width of a typical protein
peak is a good initial setting.
& Try the following setup, before wasting time with
buffer development or column testing.
Columns
RESOURCE Q 1 mL (strong anion exchanger, SAX);
or Mono Q 5/5, slightly better resolution;
or Mini Q 4.6/5.
Buffer composition
A: 20 mmol/l Tris, 8 mol/L urea, 6% (v/v) propanol-2, pH 8.5
(HCl).
B: 20 mmol/L Tris, 8 mol/L urea, 6% (v/v) propanol-2, 1 mol/L
NaCl, pH 8.5 (HCl).
Degas both buffers prior to use.
As it is recommended to use denaturing conditions for protein
extraction, it is natural and good practice that the consecutive step
also takes place in the presence of chaotropic agents. In contrast to
electrophoresis thiourea is not suitable in chromatography, because it
makes UV detection troublesome. The use of propanol-2 as an addi-
tive is intended to eliminate unspecific interactions between station-
ary phase and hydrophobic proteins and maintains its solubility.
The flow-through fraction, before the gradient starts, contains (the-
oretically) the basic proteins with a pI > 8.5. Although they do not
bind to the column they are not lost and will be transferred as all the
other fractions to the next dimension.
2.4.3.2 The Tandem IEX Approach
Depending on the origin and composition of the sample the distribu-
tion between acidic, neutral and basic proteins can differ consider-
ably. In order to obtain a preferably even pI distribution of proteins in
the different fractions it might be beneficial to apply the tandem
arrangement of anion- and cation-exchanger. At a given pH, e.g. 8.5,
negatively charged proteins, anions, bind to the anion-exchanger,
while the other proteins will pass the column with no interaction.
While the adsorbed proteins will be concentrated the other proteins
in the flow-through fraction are even more diluted than before sam-
ple application. By connecting an additional cation-exchanger after
the anion-exchanger all proteins, also the positively charged cations
will be adsorbed. Only the species with a net charge of zero, i.e. with
a pI equal or near the pH of the buffer, will not bind to either col-
umn.
Cost effective, try this first.
The industry standard.
Highest possible resolution for
small sample amounts.
Dissolving urea in water takes a
long time; a heated magnetic
stirrer may help to accelerate
this job.
No filtering through
membranes due to the risk of
introducing PEG into the
sample.
Prepare fresh at least once a
week.
With this approach most of the
proteins will be adsorbed and
concentrated, and the number
of proteins in the flow-through
fraction will be significantly
reduced.
2 Liquid Chromatography Techniques202
In principle every suitable HPLC system, preferably bio-inert, that
is capable to control two additional pressure- or motor-actuated six-
port Rheodyne
- or Valco
-valves, can be used. Figure 2.22 illustrates
the schematic flow path.
In general, there is no need for an autosampler for the first dimen-
sion, as in IEX only one sample can be run at a time (a third INV
valve will be needed if the autosampler is skipped). It is more impor-
tant to cover a wide dynamic range of injection volumes and to be
capable of introducing higher volumes (£150 mL) of diluted protein
extracts.
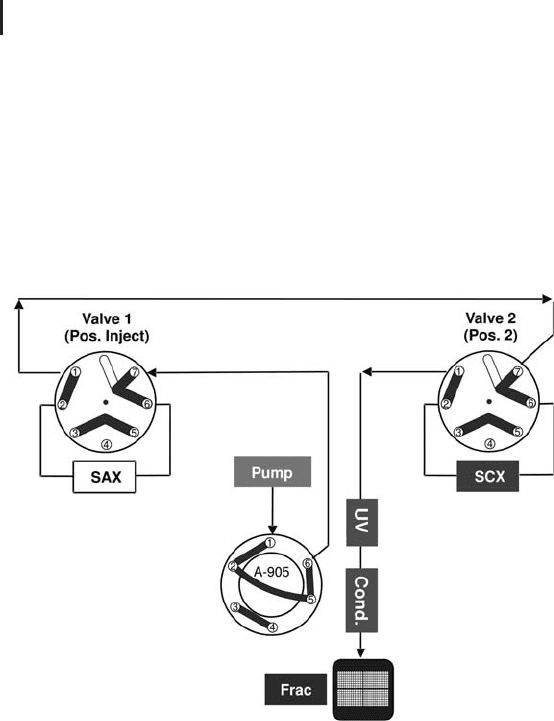
Fig. 2.22: Tandem IEX (first dimension).
Step 1: Sample injection – loading on stacked SAX/SCX columns.
During sample injection both IEX columns are connected in series.
In principle the order of columns does not matter. However, as the
tandem column approach is derived from the single column SAX lay-
out, the SCX column is installed after the SAX column. As it is not
possible, or at least not so easy to compose a buffer that is equally
good for both modes, it has proven to be acceptable to take the same
buffer as for the SAX column, knowing that it is sub-optimal for the
SCX column. After a sufficient long wash period, until both UV and
conductivity have leveled out, both columns will be separated from
each other by the switching of valve 2 (see Figure 2.23).
The flow path, as outlined
below, has been developed on
Ettan LC controlled by
UNICORN
software.
2.4 Practical Considerations and Application of LC-based Protein Pre-fractionation 203
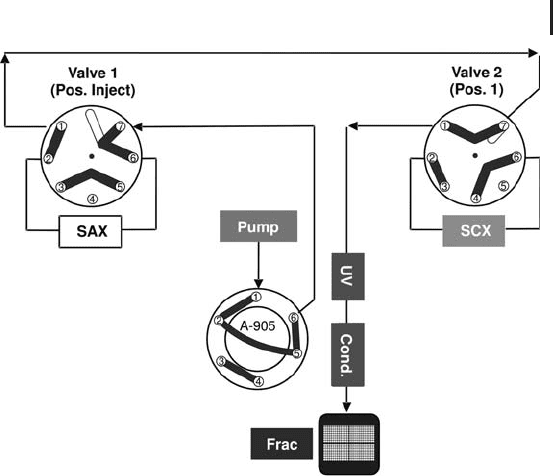
Fig. 2.23: Tandem IEX (1st dimension). Step 2: SAX gradient elution.
Now, only the SAX column is in the flow path, while the SCX column
is “parking” in a stand-by position.
In order to achieve a good separation and a minimum of band
broadening at a comparably low flow rate and a shallow salt gradient
will be applied and the eluting proteins are collected preferably in
micro-titer plates, which are directly compatible with the autosampler
for the consecutive 2
nd
dimension RPC separation. After a full cycle,
including washing, regeneration and re-equilibration, until both UV
and conductivity have leveled out again, both valves switch and con-
nect the SCX column in the flow path, while the SAX column
remains in a parking position (see Figure 2.24).
Now, the SCX column is in the flow path, while the SAX column is
idling in a stand-by position. The same gradient as for the SAX col-
umn will be applied and the eluting proteins are collected in the frac-
tion collector. When the cycle is completed, before another sample
can be loaded, it is recommended to remove remaining proteins and
contaminants from the system and the columns by washing accord-
ing to the data sheet supplied with the columns. After washing out
the cleaning reagents quantitatively the system is ready for the next
sample. Figure 2.25 shows two typical chromatograms.
Best separation at a minimum
of band broadening is achieved
at comparably low flow rate
and a shallow gradient.
2 Liquid Chromatography Techniques204
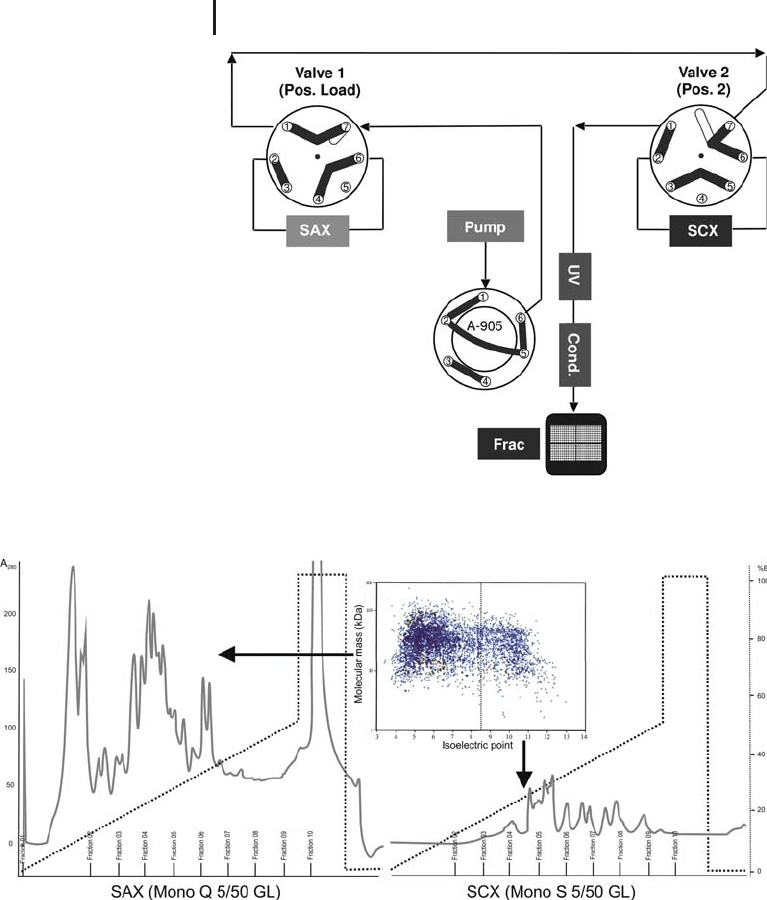
Fig. 2.24: Tandem IEX (1st dimension). Step 3: SCX gradient elution.
Fig. 2.25: Elution profile of the SAX and SCX column, using a
10 mg sample of E. coli in 8 mol/L urea. Please note the good
representation of the expected theoretical ratio between acidic
and basic proteins in the two chromatograms.
In order to cope with the desired productivity it is convenient to have
one system dedicated to the first dimension IEX and another system
with for the 2
nd
dimension RPC separation. Both systems can easily
be controlled from the same computer.

2.4 Practical Considerations and Application of LC-based Protein Pre-fractionation 205
2.4.4
Reversed Phase Chromatography and Protein Pre-fractionation
Fundamental information on this topic can be found in a chapter by
Carr and Moritz in the laboratory manual edited by Richard Simpson
(2003).
Reversed phase chromatography is the ideal subsequent step for
any technique that release samples with a high salt concentration,
such as the fractions that have been processed by ion exchange chro-
matography before. The early eluting peaks (flow-through) contain
no or very little salt, whereas the late eluting peaks contain typically
up to 500 mmol/L NaCl (or more) and all together huge amounts of
urea.
All the very hydrophilic, inorganic buffer constituents as well as
the urea do not interact with the stationary phase and pass through
the column, while the proteins of interest bind to the column – espe-
cially under aqueous, acidic conditions – and get concentrated again
in a sharp narrow zone on the top of the column. In contrast to the
separation mechanism of peptides on an RPC column, which is
believed to be a mixed mode between partition and adsorption phe-
nomena, proteins behave more in a “digital” on/off mode, where des-
orption just takes place in a very narrow range of organic modifier
present. This observation implies that column length does not really
matter.
In practice there is no significant difference in resolution between
a 150 and a 50 mm column.
In addition to shorter separation times also the washing and re-
equilibration periods can be minimized in order to achieve a better
overall throughput. Furthermore, the exposure time of proteins with
the stationary phase is shorter, which also yields in a better recovery.
In general, the life time of RPC columns in protein pre-fractiona-
tion is fairly high. This might be due to the fact that the sample has
already passed the IEX column(s) in the first dimension where
unwanted contaminations and impurities have been removed.
Ideally, the trap column is packed with exactly the same RPC media
as the separation column and has the same inner diameter (i.d.), but
can be much shorter (£ 5 mm). Figure 2.26 shows the flow path of a
system that can process all the fractions from a previous IEX or tan-
dem run in a fully automated and unattended way. After RP desalting
and separation the fractions are collected preferably in microtiter
plates, ready for lyophilization or evaporation and tryptic digestion.
In principle every suitable HPLC system, preferably bio-inert or
bio-compatible, that is equipped with an autosampler, fraction collec-
tor and capable to control two additional pressure- or motor-actuated
six-port Rheodyne- or Valco-valves can be used. The detailed flow
Carr CD, Moritz RL. in
Simpson RJ, Ed. Purifying
proteins for proteomics: A
laboratory manual. Cold Spring
Harbor Laboratory Press, Cold
Spring Harbor, New York
(2003) pp 179–208.
An RPC column deals with
such samples in a perfect dual
way, the combination of
desalting and high-performance
separation.
For protein pre-fractionation it
is recommended to select the
shortest column available.
The installation of a so called
trap column on top of the
separation column can further
extend the life time and robust-
ness of the whole setup.
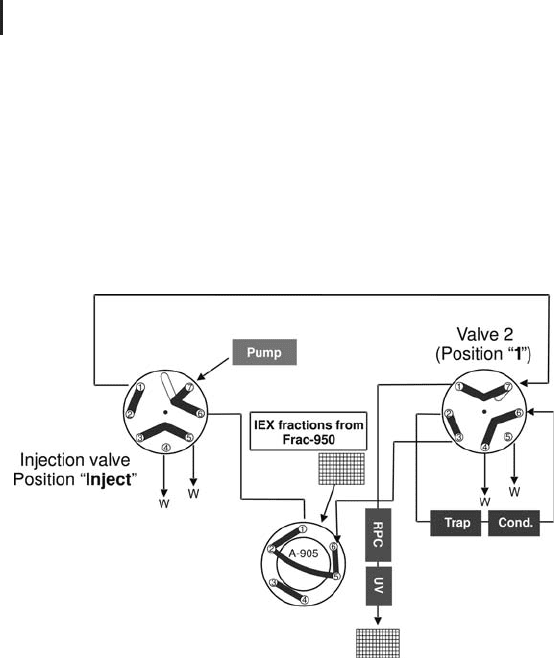
2 Liquid Chromatography Techniques206
path diagrams as outlined below have been operated on Ettan LC con-
trolled by UNICORN.
During step 1 the pump is pumping acidic eluent A that contains
no organic modifier through the sample loop of the autosampler
(A-905) to the trap column. The proteins bind to the top of the col-
umn while salts and urea pass through the column via the conductiv-
ity monitor to waste (W). The conductivity signal confirms to the
operator whether the desalting process was successful (or not). In
this constellation the RPC separation column is by-passed.
Fig. 2.26: RPC (2nd dimension). Step 1: Sample loading and desalting.
During step 2 (see Figure 2.27) both valves switch to the next position
and after a short, but sufficient wash period the gradient elution
starts in order to desorb the proteins. Now the trap column is flown
through in reverse direction, i.e. the proteins are transported on the
shortest possible way – with a minimum of band broadening – to the
connected separation column followed by the UV monitor and the
fraction collector. In order to minimize the gradient delay volume
and to get shortest possible cycle times from sample injection to sam-
ple injection, the whole autosampler with the sample loop is by-
passed.
After washing and re-equilibration the system switches back to
position 1 and the next cycle can start again and again until all the
IEX fractions have been desalted, separated by RPC and collected
(typical examples are shown in Figure 2.28.
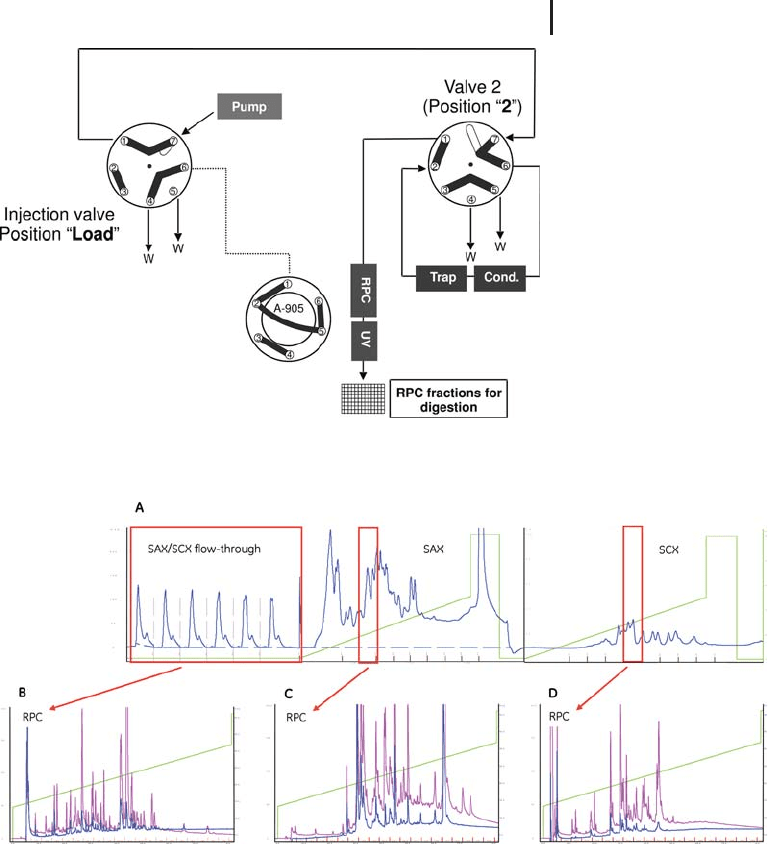
2.4 Practical Considerations and Application of LC-based Protein Pre-fractionation 207
Fig. 2.27: RPC (2
nd
dimension). Step 2: Sample elution – reversed flow.
Fig. 2.28: A: Stitched SAX/SCX chromatograms.
B: Typical RPC chromatogram of the SAX/SCX flow-through fraction.
The flow-through fraction is a pool from six consecutive injections of a
total of 10 mg of E. coli lysate.
C: typical RPC chromatogram of SAX fraction no. 3,
same conditions as B.
D: typical RPC chromatogram of SCX fraction no. 5,
same conditions as B.
RPC columns: BioBasic4, 1100 mm, trap column, 110 mm,
flow rate 40 mL/min, gradient 25%B to 75%B in 84 min
(blue: 280 nm; magenta: 214 nm).
Please note the excellent peak
shape and resolution of this
intact protein RPC separation.
From: Hoepker HR, Renlund S,
Edblad N, Wadensten H,
Mascher E, strçm, Fenyç D.
Protein pre-fractionation:
Passing phase or forward-
looking approach? HUPO 4th
Annual World Congress,
Munich (2005).

2 Liquid Chromatography Techniques208
Although high-performance columns and media should be preferred,
an acceptable compromise between resolution and recovery should
be envisaged.
There is some evidence that polymer based resins, especially
macro-porous resins, offer superior recovery – compared with wide-
pore silica based stationary phases – in combination with a fairly
good separation power and resolution. Furthermore, polymer-based
columns are resistant to very extreme pH values, a prerequisite for
efficient column cleaning in order to avoid unwanted memory effects
caused by sticky contaminations between the different samples.
Recently, very impressive protein separations on monolytic col-
umns were published. Peak shapes are extremely narrow and run
times are short. Up to now there are no long term data about life-
times and recoveries available; loadability normally is (too) low for the
application of protein pre-fractionation, where a reasonable loading
capacity is required.
Columns The following columns have been used successfully and
can be recommended:
.
RESOURCE RPC 1 ml (desalting/trap column);
.
RESOURCE RPC 6 ml (separation column);
.
SOURCE 5RPC, 4.6 , with custom-packed trap
columns;
.
BioBasic
4, available in 1.0, 2.1 and 4.6 mm
i.d., including trap columns.
& If, no stationary phase with exactly the same
selectivity is available for custom-packed trap
columns, it might be beneficial to have a trap
column with slightly higher retention
characteristics.
Although, not specifically tested for the purpose of protein pre-frac-
tionation columns from other manufacturers like GRACE-Vydac,
Agilent, Merck, Dionex, etc. are also suitable.
In the column market, especially for RPC, the offering is over-
whelming. If in doubt what product to buy, or before spending/wast-
ing valuable time for column testing it is recommended to screen the
literature; or – even better – ask an experienced colleague in your
neighborhood for good advice.
Eluent composition
A: 0.065% (v/v) TFA, sequential grade, in water.
B: 0.050% (v/v) TFA, sequential grade, in 84% (v/v) acetonitrile and
16% water.
In protein pre-fractionation
recovery should have a higher
priority than resolution.
For micro-scale protein separa-
tions monolytic columns might
be a promising alternative.
Cost-effective, try this combina-
tion first.
Same selectivity as RESOURCE
RPC, resolution comparable to
silica-based material.
Excellent resolution, suspect to
reduced recovery.
Prepare fresh at least once a
week.
These eluents will provide a
smooth and stable baseline
even at low wavelengths (210
to 220 nm) and high sensitivity.

2.4 Practical Considerations and Application of LC-based Protein Pre-fractionation 209
& Note: 84% acetonitrile in water is an azeotropic
mixture, i.e. if this mixture evaporates, e.g. in
summer in a non-air conditioned laboratory, the
composition of the mixture remains widely
unchanged (Handbook of Chemistry and Physics
1997).
Column dimensions and loading capacity With complex samples
such as proteins from cell cultures, tissues or even plasma it is
impossible to separate all the proteins. However, it is essential that
individual proteins are not spread over too many fractions. Keeping
the overlap small is of paramount importance, i.e. it is required to
perform the chromatography under high-resolution conditions in
order to avoid peak broadening and distribution of proteins over too
many fractions. According to a simulation it can be achieved that
under ideal conditions an individual protein is present in not more
than three adjoining fractions.
In order to be able to publish large numbers, many column manu-
facturers specify loading capacities as dynamic loading capacity for a
single standard protein. Under such conditions the column is com-
pletely filled with protein. However, such data are not at all useful for
high-performance separations, where overloading must be avoided.
For example, although dynamic capacity is very high (e.g.
10 mg/ml for albumin to 50 mg/ml for insulin) these figures are of
limited use for high-resolution conditions. As a rule of thumb, load-
ing capacity under high-resolution conditions is at least ten times
lower than the dynamic capacity.
When increasing amounts of protein are loaded on an RPC col-
umn, the peak width remains nearly constant. After a certain point
peak width and asymmetry start to increase and retention times
become shorter. The loading limit of a column is normally defined as
the maximum amount of sample that can be chromatographed with
not more than 10% increase in peak width. This point is called the
“overload point”. In practice the overload point will vary for different
samples. If the peaks are distributed over the effective elution range
of the gradient, more sample material can be injected, and each peak
may have a similar loading capacity compared to standard proteins.
While the effective elution range of the gradient reaches for pro-
teins between 25 to 75% of organic modifier only, the effective peak
capacity per column is finite.
Carried forward to the application area of protein pre-fractionation
the following practical loading capacities, as shown in Table 2.11, can
be expected. It can be assumed that the capacity for multi-peak sam-
ples is higher.
Handbook of Chemistry and
Physics, 78
th
edition, Lide DR
(Ed. in chief) CRC press, Boca
Raton, Florida (1997).
Serious manufactures give
figures for the practical loading
capacity which often is just
10% of the theoretical capa-
city.
The higher the molecular mass
of a protein, the lower the
capacity.
