Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.

2 Liquid Chromatography Techniques210
Tab. 2.11: Examples of loading capacities for
different columns diameters.
Column i.d. Practical loading capacity
6.4 mm 200–400 mg
4.6 mm 100–200 mg
2.1 mm 20–40 mg
1.0 mm 4–8 mg
Because – for protein RPC – the loading capacity under high-reso-
lution conditions is not a function of the column volume, it is neces-
sary to increase the column diameter, if more loading capacity is
required.
2.4.5
Fraction Size and Number of Fractions
Ideally a biological sample is pre-fractionated in such a way that each
fraction contains only one protein and that each protein is present in
one fraction only. Although practically not at all achievable, this
would lead to as many fractions as proteins, far too much to manage
for a complex biological sample with tens to hundreds of thousands
of proteins.
However, it is feasible to aim for the presence of any given protein
in not more than two fractions with a negligible overlap to the pre-
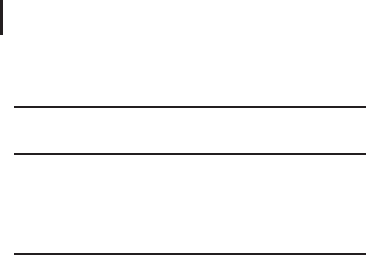
vious and subsequent fractions. An ideal chromatographic peak, as
shown in Figure 2.29, has a certain width at the base and at half
height that can be expressed in time or volume and can be calculated
and printed by state-of-the-art LC evaluation software packages.
For IEX this calculation results in some ten fractions: fraction 1 to
collect the flow-through of the unbound proteins, fraction 2 to 9 dur-
ing gradient elution and fraction 10 while washing down the remain-
ing proteins with high salt concentration.
In RPC, with its sharper peaks, some fifteen to twenty fractions
will be sufficient.
By applying the recommendations from the introduction in Section
2.2 – ideally 30 to 300 proteins per sample (=fraction) – a sample with
some 100,000 proteins would require approximately 300 to 400 frac-
tions, which is well in-line with the performance of the LC techniques
and the equipment used in protein pre-fractionation.
Naturally, less complex samples like yeast or E. coli with 4,000 to
6,000 proteins require a lower degree of protein pre-fractionation.
GraceVydac, The Handbook of
Analysis and Purification of
Peptides and Proteins by
Reversed-Phase HPLC, 3rd edn
(2002).
Decreasing the fraction size
further, results in too many
fractions, and an enormous
successive workload without
any tangible benefit.
2.5 Critical Review and Outlook 211
Fig. 2.29: In order to estimate the optimal fraction size for a protein
pre-fractionation experiment a fraction size of twice to three times the
peak width at half height can be recommended. If the apex of a peak is
in the middle of a fraction, this fraction contains almost the entire peak
volume. If a fraction change occurs at the peak maximum the peak is
split in equal parts in not more than two fractions.
2.5
Critical Review and Outlook
An internal comparison run with an E. coli sample (unpublished
data, work still in progress) has disclosed that identification rates and
sequence coverage raises significantly when using protein pre-fractio-
nation rather than applying shotgun approaches at peptide level only.
A preliminary interpolation claims that a complete analysis of all frac-
tions would give in the order of 2,000 identified proteins, a figure
matching the entire E. coli proteome.
Furthermore, protein pre-fractionation has additional benefits:
.
Subdivides complex protein mixtures into man-
ageable fractions;
.
Offers a wide dynamic range ;
.
Low m g to high mg initial sample load;
.
Very small to very large sample volumes;
.
Maintains the correlation between intact parent
proteins and their tryptic peptides;
2 Liquid Chromatography Techniques212
.
Facilitates convenient up-scaling of individual
fractions if more material is required, e.g. for
biomarker discovery;
.
Generates more and more confident protein
identification.
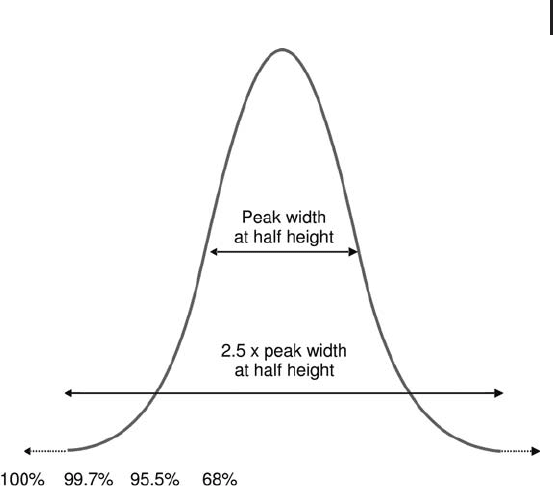
A sample that has undergone protein pre-fractionation, generally
does not require – after tryptic digestion – another multi-dimensional
separation at peptide level. Typically, a high-resolution one-dimen-
sional nanoRPC separation, followed by MS
n
analysis, will be the
method of choice. Figure 2.30 illustrates such a workflow.
Fig. 2.30: Complete LC-based workflow after the sample has
undergone a substantial de-complexification at protein level.
In the middle of the nineties of the last century there was hype for
high-throughput approaches in proteome analysis. Now, some ten
years later the success stories are still quite rare. Obviously, the chal-
lenges and requirements for meaningful proteomics have been
underestimated. It seems as if there are no quick achievements in
sight. However, time was not wasted as scientists have characterized

2.5 Critical Review and Outlook 213
and cataloged thousands of proteins in data bases, strong and undis-
putable foundations for the future work in proteomics.
The extra workload associated with running multiple replicates of
protein and peptide separations – electrophoresis or chromatography
– has not always been appreciated. However, recently several papers
can be found, which are requesting and proposing pre-fractionation
strategies and techniques in order to cope with the enormous com-
plexity of human samples for the deciphering of significant biomar-
kers; for instance the publications by Righetti et al., Guerrier et al.,
Wang and Hanash (all from 2005).
The strategies and application examples as outlined in the chapter
above, if performed properly, are somewhat more time consuming
compared to traditional concepts. It has proven that “quick and dirty”
approaches can only give access to the “low hanging fruit”. From a
rational and logical standpoint it appears more likely that more intel-
ligent sample preparation methods as e.g. protein pre-fractionation
will fractionate and de-complexify samples to such an extent that con-
secutive separation- and detection-devices will be enabled to analyze
the sample to the desired level. (This will – of course – require a cer-
tain amount of time and work effort.)
Righetti PG, Castagna A, Anto-
nioli P, Boschetti E. Electro-
phoresis 26 (2005) 297–319.
Guerrier L, Lomas L, Boschetti
E. J Chromatogr. 1073 (2005)
25–33.
Wang H, Hanash S. Mass Spec
Reviews 24 (2005) 413– 426.

215
3
Mass Spectrometry
This section discusses the use of mass spectrometry (MS) for proteo-
mics applications; protein identification, characterization and quanti-
fication. The section addresses key parameters for protein identifica-
tion and the systems available to perform the listed applications.
As explained in previous chapters, large format two-dimensional
gel electrophoresis enables the resolution of several thousand pro-
teins in a reproducible fashion in a relatively short period of time.
The instrumental developments in two-dimensional gel electrophor-
esis offered the momentum for proteomics. Similarly, technology
advancement for protein identification was essential if proteomics
was going to expand into the dominating field we know today. Edman
sequencing was the principal method of protein identification and
though it was used with considerable success for routine protein
identification, the method was relatively slow (one or two peptides
per day) and relatively insensitive (upper fmol to low pmol amounts;
Pappin et al. 1995). Developments in the field of mass spectrometry
addressed speed and sensitivity issues, which enabled biomolecular
analysis, particularly the analysis of peptides and proteins to be per-
formed routinely with confidence. Together with the development of
database search engines and the population of sequence databases,
researchers have employed MS with great success in proteomics. The
evolution of MALDI and ESI ionization sources has impacted sensi-
tivity significantly and mass analyzer development continues to
extend the boundaries of mass accuracy, resolution and functionality.
Mass spectrometry is an analytical technique that measures the
mass-to-charge ratio (m/z) of ions based upon their motion in an elec-
tric or magnetic field. Sample molecules are converted into ions in
the gas phase and separated according to their m/z ratio; positively
and negatively charged ions can be formed. This technique is per-
formed by a mass spectrometer, which typically consists of three com-
ponents (see Figure 3.1) and these components dictate the level of
performance and the type of analysis a mass spectrometer can per-
form.
Proteomics in Practice. A Guide to Successful Experimental Design 2
nd
Ed.
Reiner Westermeier, Tom Naven, and Hans-Rudolf Hçpker
Copyright 2008 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
ISBN: 978-3-527-31941-1
Pappin DJC, Rahman D,
Hansen HF, Bartlet-Jones M,
Jeffery W, Bleasby AJ. Mass
Spectrometry in the biological
sciences. (1996) 135–150

3 Mass Spectrometry216
Fig. 3.1: A schematic indicating the three regions of a mass
spectrometer: Ion source: the component which ionizes the
sample in question, i.e. ESI and MALDI. Mass analyzer: the component
that separate the ions according to their m/z ratio. Detector: the
final component which records the signal produced by an ion.
Ions produced in the source are separated in the analyzer according
to their mass-to-charge ratio (m/z) and the analyzer, to a large degree,
determines the MS performance and functionality. Resolution, and
mass accuracy are standard MS instrument specifications and tan-
dem mass spectrometry is an essential functionality. All three are key
for successful protein identification (ID) and characterization.
For instance, the higher the mass accuracy a mass spectrometer
can afford, then the greater the tolerance limits can be set for data-
base searches and ultimately the greater the confidence in the subse-
quent results. Resultantly, for protein identification and characteriza-
tion it is essential to assign the mass of a given peptide as accurately
as possible and as such a differentiation between the average and
monoisotopic mass needs to be made.
Average and Monoisotopic Mass Assignment Amino acids consist of
the elements carbon, hydrogen, nitrogen and oxygen, and to a lesser
extent sulfur. Each of these elements exists naturally as a mixture of
isotopes. For instance, carbon exists as a mixture of the
12
C isotope
(98.9%) and the
13
C isotope (1.1%). As such the abundance of the two
isotopes in a given compound will be reflected in a mass spectrum by
the isotope envelope of the compound. There are two types of mass
measurement for a given compound:
.
Average mass: the measurement which reflects
the contribution of the isotopes in an isotopic
envelope. The average mass is taken at the cen-
troid of the isotopic envelope.
.
Monoisotopic mass: is the sum of the masses of
the atoms in a molecule using the principle iso-
tope mass of each atom instead of the isotope
averaged atomic mass. In practice, this is the
217
mass of the first peak in a peptide isotopic envel-
ope. With respect to carbon, this is termed the
12
C peak.
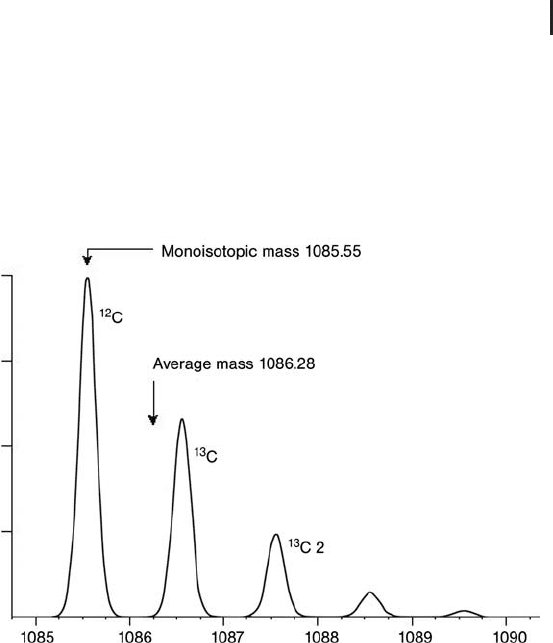
Figure 3.2 illustrates the predicted isotope distribution of the peptide
HLKTEAEMK at resolution 5000 (FWHM).
Fig. 3.2: Predicted isotope distribution of the peptide
HLKTEAEMK at resolution 5000 (FWHM).
The spectrum demonstrates the resolution of the
12
C and
13
C iso-
topes. As a result the monoisotopic mass,
12
C peak, can be clearly
assigned.
The mass difference between the average and monoisotopic masses
is 0.73 Da.
Resolution and mass accuracy specifications are determined in a
large part by the analyzer technology. Improvements in analyzer per-
formance have enabled the mono-istopic mass of a peptide, rather
than the average mass of a peptide, to be almost exclusively used for
protein identification, characterization and quantification. Mass mea-
surement accuracies of less than 2 ppm have been reported in the
routine analyzes of complex peptide mixtures and intact proteins. It
is possible to increase this further with internal mass calibration, as
described by Matthias Mann and co-workers, with mass accuracies of
less than 1 ppm (r.m.s.) reported.
3 Mass Spectrometry
The second peak in the
envelope represents the peptide
structure where one of the
carbon atoms in the peptide is
now a
13
C atom.
This is a significant difference
which can be capitalized on for
protein identification by redu-
cing the error tolerances for
database searches.
Yates JR, Cociorva D, Liao J,
Zabrouskov V. Anal Chem 78
(2006) 493–500.
Macek B, Waanders, L, Olsen
JV, Mann M. Mol Cell Proteo-
mics 5 (2006) 949–958.
Olsen JV, de Godoy LMF, Li G,
Macek B., et al.; Mol Cell
Proteomics 4 (2005) 2010–
2021.

3 Mass Spectrometry218
3.1
Ionization
The ion source is the region of the mass spectrometer where the gas
phase ions are produced from sample molecules. The method of pro-
ducing the ions is termed the ionization technique. Several ionization
techniques have been developed; the earliest incarnations include
electron impact and chemical ionization, which are useful for ioniz-
ing small molecular weight molecules, but less applicable for larger
(bio)molecules. The first example of ionizing larger biomolecules was
reported by MacFarlane and Torgerson in 1976 using the technique
plasma desorption. The introduction of fast atom bombardment in
1981 (Barber et al. 1981) enabled the ionization and detection of a
range of intact biomolecules with relatively good sensitivity. The FAB
source was coupled with a magnetic sector analyzer, enabling biomo-
lecule tandem mass spectrometry analysis to be performed with rela-
tively good sensitivity, high resolution for the first time.
However, FAB was supplanted by two ionization techniques devel-
oped in the late 1980s. Karas and Hillenkamp introduced matrix
assisted laser desorption ionization (MALDI) in 1988 as a technique
that could readily ionize (large) biomolecules in a very sensitive man-
ner. MALDI is a pulsed ionization technique which utilizes the
energy from a laser to desorb and ionize the analyte molecules in the
presence of a light absorbing matrix. In another breakthrough, Fenn
and co-workers demonstrated that electrospray ionization could also
ionize large biomolecules with high sensitivity.
3.1.1
Matrix Assisted Laser Desorption Ionization
Matrix Assisted Laser Desorption Ionization (MALDI) ions are cre-
ated by mixing the analyte with a small, organic molecule which
absorbs light at the wavelength of the laser, the matrix. The analyte
becomes incorporated into the crystal lattice of the matrix and is then
irradiated with a laser. The laser causes desorption and ionization of
the matrix and analyte, either by protonation or cationation (positively
charged ions) or by deprotonation (negatively charged ions; see Fig-
ure 3.3). The ions are then accelerated into the MS analyzer (see Sec-
tion 3.2)
& MALDI produces predominantly singly charged
ions.
McFarlane R, Torgerson DF.
Science 191 (1976) 920–925.
Barber M, Bordoli RS, Sedg-
wick RD, Tyler AN. Nature 293
(1981) 270–271.
These two ionization techniques
have become the standard for
ionization of protein and
peptide samples.
Fenn JB, Mann M, Meng CK,
Wong SK, Whitehouse C.
Science 246 (1989) 64–71.
Tanaka K, Ido Y, Akita S,
Yoshida Y, Yoshida T. 35-kai
Shitsuryo Bunseki Rengo
Toronkai, Yoshishu (1987)
22–23.
Karas M Hillenkamp F. Anal
Chem 60 (1988) 2299–2301.
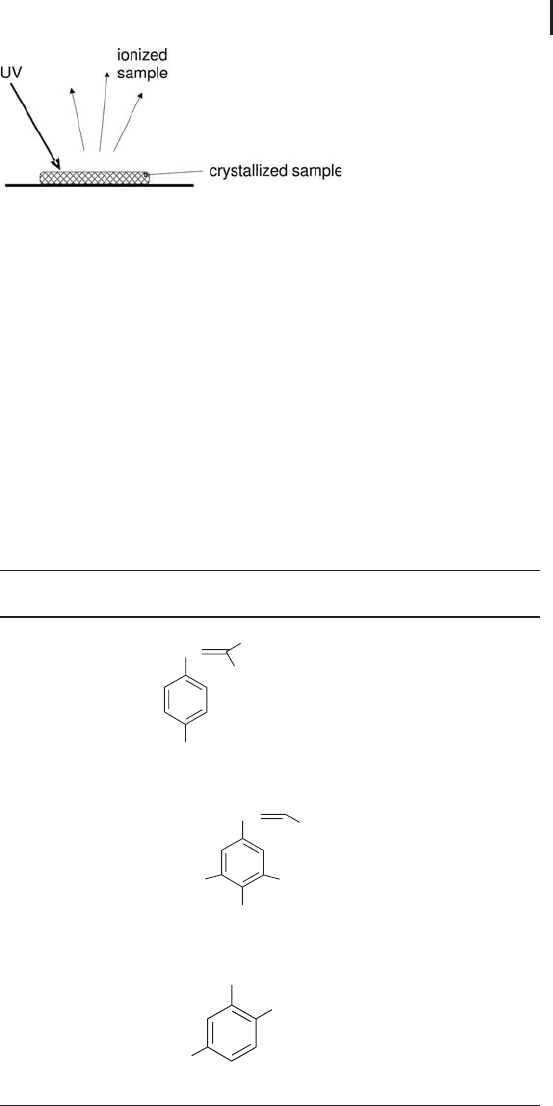
3.1 Ionization 219
Fig. 3.3: Schematic of the MALDI process.
For a tryptic digest, a-cyano-4-hydroxy cinnamic acid is typically the
standard matrix of choice (Beavis and Chait 1989). The matrix affords
high sensitivity for the detection of peptides (and proteins) and exhi-
bits negligible matrix adduction. A second matrix, 2,5-dihydoxyben-
zoic acid (DHB) initially described by Strupat et al. 1991; enables pep-
tide analysis, detection of high molecular weight proteins and the
analysis of oligosaccharides released from glycoproteins (Mock et al.
1991; Stahl et al. 1991; Harvey 1993).
Tab. 3.1: Common MALDI matrices used in biological applications.
Matrix Matrix Structure Application
a-Cyano-4-hydroxy-
cinnamic acid
OH
CH
CN
COOH
UV laser: peptide analy-
sis and protein digests.
Analytes <10 kDa
Sinapinic acid
(4-hydroxy-3,5-di-
methoxycinnamic acid)
CH
OMe
CH
3
O
OH
COOH
Analysis of large poly-
peptides and proteins
>10 kDa
2,5-Dihydroxybenzoic
acid (2,5 DHB)
COOH
OH
OH
UV laser: protein di-
gests and proteins, oli-
gosaccharides released
from glycoproteins
Beavis, RC, Chait BT. Rapid
Commun Mass Spectrom
3(1989) 432–435.
Strupat K, Karas M, Hillen-
kamp F. Int J Mass Spectrom
Ion Proc 111 (1991) 89–102.
Mock KK, Davy M, Cottrell JS.
Biochem Biophys Res Commun
177 (1991) 644–651.
Stahl B, Steup M, Karas M,
Hillenkamp F. Anal Chem 63
(1991) 1463–1466.
Harvey DJ. Rapid Commun
Mass Spectrom 7 (1993) 614–
619.
Beavis RC, Chaudhary T, Chait
BT. Org Mass Spectrom 27
(1992) 156–158
Beavis RC, Chait BT. Rapid
Commun Mass Spectrom 3
(1989) 432–435.
Strupat K, Karas M, Hillen-
kamp F. Int J Mass Spectrom
Ion Proc 111 (1991) 89–102.
