Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.

products. At present, post-metallocene catalysts are being
developed. Salicylaldimine complexes of group IV metals
may show higher activity and the possibility of further
refinement of molecular structure.
CHARACTERIZATION OF POLYETHYLENE
The properties of products made from polyethylene are
dependent on some basic characteristics of polyethylene
itself. Several of the terms commonly used to describe
polyethylene are discussed in the following paragraphs.
Melt Index
The melt index (MI) of a polymer is used as an empirical
measure of its molecular weight. To measure melt index
according to ASTM D1238, a polymer sample is melted
and forced through a small orifice of fixed size under a
fixed pressure. The weight of polymer that is extruded in
10 min under 44 psi (303 kPa) of pressure is called the melt
index. When the pressure is increased to 440 psi
(3030 kPa), the weight of polymer extruded in 10 min is
the flow index (FI). Since a polymer with very high
molecular weight will be very viscous and resistant to
flow, it will not pass through the small orifice quickly and
the weight obtained in 10 min (or melt index) will be low.
Melt index is therefore inversely proportional to molecular
weight. Typical low-density polyethylene melt indices
range from 0.2 to over 150 dg/min. In general, products
in the lower-melt-index range are used for film extrusion
and the higher-melt-index products for molding and ex-
trusion coating. Within any fabrication process, the use of
a lower-melt-index resin will result in a stronger product,
although usually with some sacrifice in extrudability.
Melt-Flow Ratio
The melt-flow ratio (MFR) is a rough estimate of the
molecular-weight distribution (MWD) of a resin. Since
all polymer chains in a given resin are not exactly the
same length, an MWD measurement will describe how
dissimilar the chains are from each other. Melt-flow ratio
is the ratio of the flow index to the melt index. The higher
the ratio, the broader the molecular-weight distribution
and the more dissimilar the chains are from each other. A
polymer with every chain exactly the same length would
have a very narrow molecular-weight distribution and a
very low melt-flow ratio. Melt-flow ratios of commercial
low-density polyethylenes vary from about 20 (very nar-
row) to about 100 (very broad). Polymers with narrow
molecular-weight distributions give stronger products, but
are more difficult to extrude than those with broad
molecular-weight distributions.
Density
The density of a polymer is a measure of its crystallinity.
Density measured according to ASTM D1505 consists of
taking a small sample of polymer that has been molded in
a carefully prescribed manner and dropping it into col-
umns with solutions of different viscosities. The position of
the unknown polymer is the column is then compared to
standard samples of known density. The density of a film
or molded article is only partly controlled by the density of
the resin used to make the product. The rate at which the
product is cooled also plays an important role. The faster a
film or molded article is cooled, the less time there is for
the polymer chains to crystallize and the lower the density
of the final product. For example, the density of a sample
cut from a blow-molded polyethylene bottle was 0.945 g/
mL. The same polyethylene resin, when compression-
molded into a plaque and cooled according to ASTM
D1505, measured 0.954 g/mL. Product properties such as
stiffness, rigidity, environmental stress-crack resistance
(ESCR), and moisture vapor transmission rate (MVTR)
are affected by density. The lower the density of a product,
the more limp and flexible it is. A 0.918-g/mL polyethylene
product will have better ESCR and higher MVTR than a
product with a density of 0.930 g/mL.
LDPE PROCESS
LDPE is made by the high-pressure polymerization of
ethylene (2, 8). In either a tubular or autoclave reactor,
ethylene is pressurized to more than 20,000 psi (138 MPa)
and heated to W150 1C. Small amounts of an initiator,
typically oxygen or peroxide, are added to start the poly-
merization process. Comonomers such as vinyl acetate or
ethyl acrylate can be added to make EVA and EEA
copolymers, respectively. Critical molecular characteris-
tics such as molecular weight, molecular-weight distribu-
tion, and density are controlled by reaction temperature,
ethylene pressure, and the concentration of chain-transfer
agents. Constraints on the viscosity of the polymer solu-
tion in the reactor limit the rate at which products with
high molecular weight and/or high density can be made in
the high-pressure process. It has been found that the
tubular high-pressure process gives products that differ
subtly in the type and degree of branching from products
made in the autoclave process. Density depression occurs
in all types of polyethylene because of the presence of
short branches along the backbone of the polyethylene
chain. These short branches, typically one to five carbon
atoms long, prevent the long polyethylene chains from
folding together and forming crystals. However, early in
the development of LDPE, it was found that the presence
of these short branches did not totally explain some of the
rheological properties of LDPE. It was then hypothesized
and proved that LDPE also contains low levels of long-
chain
branching (9). These
long chains, which can be over
1000 carbon atoms in length, have a very small effect on
the density of LDPE but have major impact on the
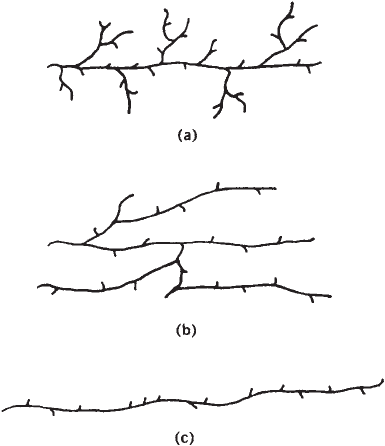
processing and properties of LDPE (Figure 1). The tubular
reactor makes LDPE with a large number of long-chain
branches. The branches are of relatively short length,
however. The autoclave reactor, on the other hand, gives
products with low levels of long-chain branches of extre-
mely long length. Because of the difference in long-chain
branch type and frequency, some specialization in product
applications has occurred with these products. The auto-
clave reactors produces products that are especially useful
988 POLYETHYLENE, LOW-DENSITY
in high-speed extrusion coating and in film applications
requiring toughness. The tubular reactor gives products
with optimal clarity and processing characteristics.
LLDPE PROCESS
Low-pressure processes for the polymerization of ethylene
were developed in the 1950s (2, 8). These processes used
organometallic catalysts to polymerize ethylene to form
HDPE at moderate pressures (approximately 300 psi) and
temperatures (approximately 100–200 1C). Three basic
types of low pressure systems developed: solution, where
the polymer was completely dissolved in a solvent at high
temperatures; slurry, where the solid polymer particles
were physically suspended in a solvent at lower tempera-
tures; and gas phase, where the solid polymer was in
contact with only the polymerization gases. Again, be-
cause of process constraints in the reactor, molecular
weights and densities were thought to be limited, espe-
cially in the solution and slurry systems. The density
limitations of the low-pressure and high-pressure pro-
cesses were thought to form incompatible regions, with
the low-pressure process making polythylenes above 0.935
density and the high-pressure process making polyethy-
lene below 0.935 density.
The first commercial production of LLDPE was made in
a solution process by DuPont Canada in 1960 (10). A large
market for this new polymer did not develop until 1977,
when Union Carbide started licensing their gas-phase
process for the manufacture of linear low-density poly-
ethylene. Since that time, several other polyethylene
manufacturers have announced the conversion of low-
pressure HDPE processes or even high-pressure LDPE
processes to make LLDPE and some of these processes are
available for licensing.
The structure of these new linear low-density polymers
is very different from the LDPE made from the high-
pressure process. In LLDPE, there is no long-chain
branching. Density is controlled by the addition of como-
nomers such as butene, hexene, or octene to the ethylene.
These comonomers give rise to short-chain branches of
different lengths, two carbon atoms for butene, four for
hexene, and six for octene. The length of the short-chain
branches determines some of the strength characteristics
of LLDPE. The absence of long-chain branches in LLDPE
plays a significant role in the difference in extrusion
characteristics between LLDPE and LDPE, as discussed
in the sections on specific application areas.
LOW-DENSITY POLYETHYLENE PROPERTIES
Low-density polyethylene is one of the most widely used
packaging materials in the market. Its utility in a variety
of different applications is due not to some single out-
standing property or characteristic, but usually to a
combination of properties. The low price of polyethylene
compared to wood, metal, and other polymers has accel-
erated its penetration into many applications. In addition
to the low cost of polyethylene, the excellent toughness,
flexibility, moisture barrier, chemical resistance, electrical
insulation, and light weight of polyethylene films, bottles,
pipes, cables, and other articles make them superior to
articles made from conventional materials of construction.
Since the introduction of LLDPE into the market, the
superior properties of LLDPE have led to its use in new
applications for polyethylene as well as the replacement of
LDPE and/or HDPE in some areas. Compared to LDPE,
LLDPE at the same melt index and density offers better
toughness, rigidity, stress-crack resistance, elongation,
melting point, and moisture barriers. (See article on
polymer properties for more details.)
MARKETS/APPLICATIONS
Low-density polyethylene was the first thermoplastic
polyolefin used commercially. The free-radical initiated
polymerization process leads to a polymer with large
amounts of long chain branching. This imparts an unu-
sual rheological behavior in both shear and extension.
LDPE is used in a variety of applications such as film,
coating, molding, and wire and cable insulation.
The global market for polyethylene is undergoing ex-
tensive changes. Increases are concentrated in China and
the Middle East. Consumption growth of LDPE is rather
strong, despite the fact that a decline was expected with
the advent of LLDPE to the market (11).
In 2006, 7.853 10
6
lb of LDPE were produced in the
United States (12). Film applications are by far the largest
market for LDPE, accounting for 55% of world use in 2006.
Film demand is split between packaging and nonpacking
uses. Extrusion coating is the second largest market for
LDPE accounting for 9.9% of total in 2006.
Figure 1. Molecular structures for LLDPE and HP-LDPE: (a)
HP-tubular process; (b) HP-autoclave processes; (c) LLDPE.
POLYETHYLENE, LOW-DENSITY 989
Film and Sheet
About 50% of all the low-density polyethylene used in the
world today goes into the film and sheet area. These
applications include garbage bags, grocery sacks, garment
bags, heavy-duty sacks, shrink film, stretch film, carrier
film, pond liners, construction and agricultural film, and
food packaging. Food applications include packaging for
baked goods, dairy products, frozen food, meat and poul-
try, cookies, and candy. Polyethylene used in the film area
range in melt index from 0.2 to 6 and in density from 0.915
to 0.935. The advantages of LDPE/LLDPE include low
cost, flexibility, toughness, and chemical- and moisture-
barrier properties (Tables 1–3). This has been the market
most rapidly penetrated by LLDPE in the United States.
This rapid penetration is due, in part, to the outstanding
physical properties of LLDPE film (Table 4). Dart drop and
Elmendorf tear measure the toughness of film in a high-
speed test and give an indication of the failure behavior of
film under catastrophic conditions. Puncture and tensile
tests are done at relatively slow speeds and give an
indication of intrinsic properties and failure modes that
can occur with long-term use.
The differences in molecular structure between LLDPE
and LDPE (linear vs branched molecules and molecular-
weight distributions) affect the rheology of the two mate-
rials. LLDPE is more viscous at extrusion shear rates and
requires more power to extrude. In addition, it is neces-
sary to use a wide dip gap to avoid melt fracture when
extruding 100% LLDPE. Therefore, in the early days of
LLDPE introduction, minor modifications to the screw
and die gap had to be made in order to extrude LLDPE
on extruders designed for HP-LDPE and obtain optimum
film properties Most extruders today have been designed
to handle the different rheology of LLDPE, and such
modifications are not necessary. In addition, there are
additives now available that allow the extrusion of con-
ventional LLDPE through narrow die gaps without melt
fracture. The new generation of LLDPE film products,
designed to be ‘‘drop-in’’ for LDPE products, will not
require any modifications to extrusion equipment.
LDPE remais the resin of choice for shrink wrap and
clarity films (13).
Injection Molding
This market includes lids, buckets, wash basins, house-
wares, toys, freezer containers, and general housewares.
The advantages of low-density polyethylene include
good low-temperature properties, low cost, light weight,
and flexibility (Table 5). Low-density polyethylene in the
melt-index range of 2–150 and density range of 0.920–
0.930 is commonly used in injection-molding applications.
LLDPE has made significant penetration into certain
markets such as lids and housewares, where property
advantages, such as higher stiffness and improved envir-
onmental stress-crack resistance and heat-distortion
resistance, can be coupled with higher melt indices to
result in faster cycle times and downgauged articles
(Table 6) (14).
Extrusion Coating
In this application, polyethylene is used as a coating on
another material such as paper, aluminum foil, and card-
board. These coated products are used for liquid packa-
ging, such as milk and juice, and for the new aseptic
packages of nonrefrigerated juices. The polyethylene
serves as an adhesive, moisture barrier, seal layer, prin-
table surface, and/or barrier to tear. Because of its mole-
cular structure, that is, the type of long chain branching,
polyethylene made in tbe high-pressure autoclave reactor
is the most successful in this market. Resins with melt
indices of 4–10 and densities of 0.920–0.930 are commonly
used. Because of its linear structure, LLDPE has not
penetrated the extrusion coating market, except as blends
with LDPE.
Blow Molding
High-density polyethylene is the preferred material for
this market because of its rigidity and barrier properties.
Table 1. HP-LDPE Homopolymer Film Resins
Type: HP-LDPE HP-LDPE HP-LDPE HP-LDPE HP-LDPE HP-LDPE
Comonomer: None None None None None None
Melt index: 0.2–0.8 1.5–2.0 1.5–2.0 1.2–2.0 1.0–2.0 5.0–10.0
Density (g/cm
3
): 0.919–0.923 0.922–0.925 0.930–0.935 0.918–0.924 0.923–0.927 0.917–0.930
Molecular-weight
distribution:
Broad Broad Broad Broad Broad Broad
Process: Blown Blown Blown and/or
cast
Blown Cast Cast
Applications: Shipping sacks;
heavy-duty
applications
Bread and
bakery;
general-
purpose
packaging
Overwrap General-
purpose
packaging
Bread and
bakery;
general-
purpose
packaging
Extrusion
coating
Critical properties: Toughness Clarity Clarity; stiffness Extrudability;
toughness
Extrudability;
good tear
Drawdown
adhesion;
pinhole
resistance
990 POLYETHYLENE, LOW-DENSITY

Low-density polyethylene may be used in those segments
where flexibility and excellent stress-crack resistance are
required, such as squeeze bottles, toys, and drum liners.
Fractional-melt-index polyethylene (density 0.920–0.935)
is typically used in this market. LLDPE offers better
stiffness and improved stress-crack resistance compared
to LDPE.
SAFETY AND HEALTH
Polyethylene is generally recognized as a safe packaging
material by the Food and Drug Administration. Resin
suppliers will state which of their resins comply with
regulations governing polyethylenes used in food-contact
applications. These regulations are covered in the U.S.
Food, Drug, and Cosmetic Act as amended under Food
Additive Regulation 21 CFR 177.1520. Polyethylene is a
very stable polymer. However, proper material handling
procedures are required to control dust and smoke and to
provide adequate ventilation during extrusion. Copoly-
mers containing EEA, EMA, and EVA should be used
selectively on food contact applications. Resin suppliers
will provide Material Safety Data Sheets and Materials
Handling Guides for polyethylene resins on request.
Table 2. HP-LDPE Copolymer Film Resins
Type: HP-LDPE HP-LDPE HP-LDPE HP-LDPE
Comonomer: 2–5% VA
a
3–5% VA
a
7% VA
a
15–18% EA
b
Melt index: 1.5–2.0 0.2–0.4 0.2–4.0 2.0–6.0
Density (g/cm
3
): 0.925–0.930 0.923–0.927 0.927–0.945 0.927–0.940
Molecular-weight
distribution:
Broad Broad Broad Broad
Process: Blown Blown Blown; extrusion coating Blown; extrusion coating
Applications: Frozen food Ice bags Sealing layer; liquid
packaging
Disposable gloves; ID cards
Critical properties: Clarity; low-temperature
properties
Low-temperature
properties
Adhesion; low-temperature
properties
Low stiffness; adhesion to polar
substrates
a
Vinyl acetate.
b
Ethyl acrylate.
Table 3. LLDPE Film Resins
Type: LLDPE LLDPE LLDPE LLDPE LLDPE
Comonomer: Butene Hexene; octene Hexene; octene Hexene; octene Hexene; octene
Melt index: 0.8–2.5 2.0–4.0 2.0–5.0 0.7–1.5 0.8–1.5
Density (g/cm
3
): 0.917–0.922 0.912–0.919 0.928–0.935 0.924–0.928 0.917–0.923
Molecular-weight
distribution:
Narrow Narrow Narrow Narrow Narrow
Process: Blown Cast Cast Blown Blown
Applications: General-purpose
packaging
Stretch wrap Bread and bakery;
overwrap
Grocery sack Blending; ice bags
Critical properties: Extrudability;
toughness
Puncture and tear
resistance
Stiffness; moisture
barrier
Stiffness; tear
resistance
Excellent
toughness; Low-
temperature
properties
Table 4. Blown-Film Properties
a
of LLDPE and HP-LDPE
Property: ASTM test HP-LDPE HP-LDPE LLDPE LLDPE LLDPE
Melt index: 2.5 0.2 1.0 1.0 1.0
Density (g/cm
3
): 0.921 0.923 0.918 0.918 0.920
Comonomer: None None Butene Hexene Octene
Dart drop, gf/mil (N/mm): D1709 0.75 (29) 185 (71.4) 100 (38.6) 200 (77.2) 250 (96.5)
Puncture energy, in. lbf/mil (kJ/m): 6 (26.7) 5 (22.2) 16 (71.2) 17 75.6)
Elmendorf tear, gf/mil (N/mm): D1922 MD 160 (61.8) 90 (34.7) 140 (54) 340 (131.2) 370 (142.8)
XD 110 (42.5) 100 (38.6) 340 (131.2) 585 (225.8) 800 (308.8)
Tensile strength, psi (MPa): D882 MD 2900 (20) 2800 (19.3) 5000 (34.5) 5200 (35.9) 6500 (44.8)
XD 2700 (18.6) 3000 (20.7) 3800 (26.2) 4700 (32.4) 5100 (35.2)
Tensile impact strength, ft lbf/in. (MPa): MD 440 (36.4) 500 (41.4) 1200 (99.3) 1930 (159.7)
XD 650 (53.8) 1050 (86.9) 900 (74.5) 1760 (145.6)
a
All properties measured on 1.5-mil (38-mm) film produced at 2:1 blowup ratio.
POLYETHYLENE, LOW-DENSITY 991

ENVIRONMENTAL IMPACT
In recent years there has been quite a bit of adverse
publicity around the use of plastics and the impact of
litter on the environment. In the 1980s, the decision of fast
food restauraants to discontinue the use of the expanded
polystyrene clamshell burger container is one example of
how plastics growth and use can be changed by public
opinion. Many U.S. states have considered or passed
regulations specifying the amount of recycled plastic
that must be incorporated into new plastic articles. This
has had a large impact on the polyethylene industry since
polyethylene, including LDPE, is one of the more recycl-
able plastics being used today. The recycling effort has
been hampered by the higher cost of collecting, separating,
and cleaning the polyethylene containers compared to the
cost of the virgin polymer.
Photo- and biodgradable LDPE copolymers have been
offered as a solution to the litter problem. LDPE copoly-
mers containing carbon monoxide have been produced.
These photodegradable copolymers have been used to
make beverage carrier rings, but have not been used in
other commodity applications. The search for a biodegrad-
able LDPE continues. Blends of starch-like fillers improve
biodegradation, but at a significant sacrifice of physical
properties. A new biodegradable/compostable technology
has been developed, such that biodegradability can be
triggered by heat, light, or stress. Products made with this
technology are being marketed in Europe and South
America (13).
BIBLIOGRAPHY
Norma J. Maraschin, ‘‘Polyethylene, Low-Density’’ in the Wiley
Encyclopedia of Packaging, 2nd edition, John Wiley & Sons,
New York, 1997, pp. 752–758.
Cited Publications
1. British Patent 471,590 (September 6, 1937) and U.S. Patent
2,153,553 (April 11, 1933), E. W. Fawcett and coworkers (to
ICI, Ltd.).
2. A. Renfrew and P. Morgan, eds., Polyethylene, Interscience
Publishers, New York, 1960, Chapter 2.
3. Chem. Week, 12 (May 5, 1993).
4. Plast. News, 22 (March 8, 1993).
5. Chem. Week, 8 (June 12, 1991).
6. Chem. Market. Reporter, 5 (December 23, 1991).
7. Chem. Eng. News, 20 (September 18, 1989).
8. R. A. V. Raff and J. B. Allison, Polyethylene, Interscience
Publishers, New York, 1956, Chapter 3.
9. J. J. Fox and A. E. Martin, Proc. Roy. Soc. London Ser.A175,
211, 216 (1940).
10. A. W. Anderson, U.S. Patent 4,076,698 (to E. I. DuPont de
Nemours & Co., Inc.), February 28, 1978.
11. A. Borruso, ‘‘Low-Density Polyethylene Resins,’’ Chemical Eco-
nomics Handbook, SRI Consulting. Menlo Park, CA, Oct. 2007.
12. Year End Statistics, American Chemistry Council, December
2007.
13. N. J. Maraschin, ‘‘Polyethylene, Low-Density’’ in Kirk-Othmer
Encyclopedia of Chemical Technology, 5th edition, Vol. 20,
John Wiley & Sons, Hoboken, NJ, 2006.
Table 5. HP-LDPE and LLDPE Molding Resins
Type: HP-LDPE HP-LDPE HP-LDPE LLDPE LLDPE LLDPE
Melt index: 6–10 15–25 35–50 0.8–1.2 12–30 50–150
Density (g/cm
3
): 0.924–0.926 0.914–0.918 0.923–0.925 0.918–0.922 0.920–0.926 0.926–0.935
Molecular-weight
distribution:
Broad Narrow Narrow Broad Narrow Narrow
Process
a
: IM IM, BM, PE IM BM, IM, PE IM IM
Applications: Bottle closures Drug bottles;
aseptic
packaging
Lids Drum liners;
irrigation
tubing; spouts
Industrial
containers;
Speciality
housewares
Lids;
housewares
Critical properties: ESCR;
b
stiffness ESCR;
b
low
modulus
Cycle time ESCR;
b
processability
ESCR;
b
low-
temperature
impact
Cycle time
a
Key; IM, injection molding; BM, blow molding; PE, profile extrusion.
b
ESCR, environmental stress-crack resistance.
Table 6. Comparison of LLDPE and HP-LDPE Molded Properties
Type: LLDPE LLDPE LLDPE HP-LDPE HP-LDPE
Melt index: 20 50 100 23 49
Density (g/cm
3
): 0.924 0.926 0.931 0.924 0.924
Dishpan impact, ft lbf (J) at – 20 1C: 30 (40.7) 24 (32.5) 3 (4.1) 9 (12.2) 3 (4.1)
Failure mode: Ductile Ductile Ductile Shatter Shatter
ESCR
a
(F
50
h)
b
: 150 3 o2 o1
c
a
Environmental stress-crack resistance.
b
At 50 1C, 100% Igepal, no slit, ASTM D1693.
c
F
100
h.
992 POLYETHYLENE, LOW-DENSITY
POLYMER PROPERTIES
RUBEN J. HERNANDEZ
Michigan State University, East
Lansing, Michigan
The properties of a polymer result from its chemical
nature, morphology, formulation, processing, and even
use conditions. Intrinsic polymer properties depend pri-
marily on the chemical nature of the polymer; but since
most polymers are polymorphic materials, their intrinsic
properties may also depend on the polymer’s morphology.
Morphological changes at room temperature, however, are
very slow and highly time-dependent. Formulation (com-
pounding with additives) and processing have a direct
impact in the final properties of a polymeric material.
Furthermore, storage and use conditions (e.g., humidity
and aging) may affect a plastic’s performance.
Intrinsic properties of a polymer can be classified as
either molecular or bulk. Intrinsic molecular properties
depend mainly on the chemical structure of the polymer’s
constitutional units and, to a lesser degree, on its macro-
molecular character. The chemical nature of a constitu-
tional unit, which results from the type and number of
atoms, existing side groups, charge distribution, and type
of secondary molecular forces, controls important proper-
ties. These properties are cohesive energy, molecular
packing density, molecular relaxation (including glass-
transition temperature), barrier, mechanical strength,
frictional forces, surface tension, and adhesion character-
istics. In PE, for example, the –CH
2
– group yields a set of
properties values quite different from the constitutional
unit –OC(CH
2
)
5
NH– of nylon 6. The latter unit is larger
and more polar than the first, and it tends to develop
strong intermolecular hydrogen bonding.
Intrinsic bulk properties such as stiffness, melting
temperature, heat-sealing temperature range, melt-flow
index, and viscosity are largely influenced by the molecu-
lar mass and distribution, as well as the architecture of
the polymer chain. Molecular mass and molecular distri-
bution include the average molecular mass, dispersion
index, and single- or multimodal distribution. The ‘‘archi-
tecture’’ of a polymer refers to the monomers layout in the
polymer chain. In this respect, polymers can be linear,
branched, or crosslinked, have different tacticity (e.g.,
atactic, isotactic, and syndiotactic polypropylene), and
show various copolymer arrangements such as random,
block, alternating, or graft.
Phase morphology affects, in varying degrees, the
physical, mechanical, and optical properties of a polymer.
Polymers can be isotropic (amorphous); that is, they are
glassy (hard and brittle) below the glass-transition tem-
perature and are rubber-like (soft and elastic) above it. As
temperature increases, amorphous polymers become true
liquids without any thermodynamic discontinuity. Poly-
mers that are nonisotropic can crystallize in several
arrangements; even two different crystal structures can
coexist, depending on the values of temperature and
pressure. Sometimes a crystalline polymer remains a
liquid below the melting temperature, thus producing a
supercooled material. In semicrystalline polymers, crys-
talline and noncrystalline regions may coexist, forming
microcrystalline or paracrystalline regions. Polymers may
also have a one- or a two-dimensional molecular order in
the liquid state that forms thermotropic or lyotropic liquid
crystalline mesophases. A simple example of how the
morphology affects the properties is illustrated by the
density of PE; the density of PE may range from 0.90 to
0.97 g/mL as the percent of crystallinity increases. Besides
density, there are other properties proportional to the
degree of crystallinity: rigidity, heat resistance, barrier,
abrasion resistance, gloss, shrinkage, and parting-line
difference. However, stress-crack resistance, clarity, fold-
ing endurance, impact strength, and parison sag decrease
as crystallinity increases.
Process operation, on the other hand, can alter the
morphology of a material. Film orientation, for instance,
directly affects the polymer morphology by producing a
slightly more compact molecular packing and a more
transparent film. Similarly, a rapid cooling process will
increase the amorphousness of a supercooled phase, while
slow cooling will increase crystallinity. Thus, process
conditions may affect properties such as the heat of fusion
of a material and may also affect operations such as
thermoforming operation and heat sealing. Since trans-
parency (or opacity) is directly controlled by the crystal-
linity, morphological changes can influence the optical
properties of a polymer. When additives are incorporated
into a resin, however, they can substantially alter the
resin’s original properties.
DETERMINATION OF POLYMER PROPERTIES
Polymer properties can be measured experimentally
(usually according to standardized methods) or be esti-
mated from semiempirical correlations. Many intrinsic
properties related to the molar constitutional unit can be
estimated from group contributions or increment meth-
ods. Some polymer properties that can be estimated by
group contribution include density, thermal expansion
coefficient, thermal conductivity, specific heat, specific
entropy of fusion, melting temperature, glass-transition
temperature, cohesive-energy density, solubility para-
meter, surface tension, viscosity coefficient, dielectric con-
stant, magnetic susceptibility, specific shear modulus,
specific bulk modulus, and sound velocity (1).
The most common properties of polymers related to
packaging applications are described next. For easy re-
ference they are grouped under these headings: (1) density
and thermophysical properties, (2) mechanical properties,
(3) solubility and degradation properties, (4) barrier prop-
erties, (5) surface and adhesion properties, (6) electrical
properties, and (7) optical-appearance properties.
DENSITY AND THERMOPHYSICAL PROPERTIES
Density. The density of a plastic is directly proportional
to its crystallinity: d = d
a
+ C(d
c
d
a
)/100, where d is the
density, C is percent volumetric crystallinity, and d
a
and
POLYMER PROPERTIES 993
d
c
are the amorphous and crystalline density, respectively.
For example, d
a
= 0.855 g/mL while d
c
= 1.0 g/mL for PE
(2). Polymer density values vary from 0.87 g/mL for poly-
propylene up to 1.86 g/mL for PVDC (3). The standard
ASTM D1505 (4) describes the density gradient method to
evaluate the density of films and resins.
Glass-Transition Temperature. The glass-transition tem-
perature T
g
, which is actually a relaxation temperature, is
an important property in amorphous polymers. As a
relaxation temperature, T
g
is associated with the onset
of the rotation and mobility of chain segments involving
several monomers. Rather than a single-point tempera-
ture, T
g
in semicrystalline polymers is a range of tem-
perature values. The broadness of this range depends on
the sample morphology. When the polymer is at a tem-
perature below T
g
, it becomes glassy and stiff, while above
T
g
it shows a plastic or rubbery behavior (5). The T
g
values
for polymers range from about 251C(1251C is also
commonly reported) in polyethylene to 3651C for thermo-
plastic polyimide (1, 6). Both the T
g
and the melting
temperature set the application temperature range of a
plastic container. For instance, a polypropylene container
may become brittle at freezing temperature if its T
g
is
near 01C; ‘‘crystal’’ polystyrene is brittle at room tempera-
ture because it has a T
g
of B801C and does not contain
plasticizer. On the other hand, a container made of a
low-melting-temperature (T
m
) plastic will soften if heat-
sterilized. Both differential thermal analysis (DTA) and
differential scanning calorimetry (DSC) are used to deter-
mine T
m
and T
g
(these methods are described in ASTM
D3418). Van Krevelen (1) showed that polymers have
T
g
/T
m
ratios ranging from 0.5 to 0.76. Most polymers,
however, have the T
g
/T
m
ratio centered at 0.67.
Heat Capacity. Heat capacity (or specific heat), c, is the
amount of energy needed to change a unit of mass of a
material one degree of temperature. The heat capacity of
plastics, which are obtained at constant pressure, are
temperature-dependent, especially near the glass-transi-
tion temperature. In a semicrystalline polymer, the heat
capacity of the amorphous phase is greater than the heat
capacity of the crystalline phase implying that the c values
depend on the percent of polymer’s crystallinity. The heat
capacity values of polymers at 251C vary from 0.9 to 1.6 J/
(g K) for amorphous polymers and from 0.96 to 2.3 J/
(g K) for crystalline polymers. Reliable data of the heat
capacity of amorphous and crystalline phases are avail-
able for only a limited number of polymers (1, 2). Values of
c for polymers can be found in the review by Wunderlich
(7). The usual techniques to measure c are differential
thermal analysis (DTA) and differential scanning calori-
metry (DSC).
Heat of Fusion. The heat of fusion, DHm, is the energy
involved during the formation and melting of crystalline
regions. For semicrystalline polymers, the heat of fusion is
proportional to the percent of crystallinity. Amorphous
polymers or amorphous polymer regions do not have heat
of crystallization, since amorphous structures have a
smooth transition from the liquid amorphous state to the
liquid state. Experimental values of crystalline heat of
fusion for common packaging plastics vary from 8.2 kJ/mol
for polyethylene to 43 kJ/mol for nylon 6,6 (1). ASTM
D3417 describes a method for measuring the heat of fusion
and crystallization of a polymer by differential scanning
calorimetry (DSC).
Melting Temperature. The melting temperature T
m
is a
true transition temperature. This means that at T
m
both
the liquid and solid phases have the same free-energy
value. Most semicrystalline polymers, instead of having a
sharp melting point, show a melting temperature range,
and for amorphous polymers there is no T
m
. Similar to T
g
,
T
m
can be estimated from contribution groups and em-
pirical relationships between T
g
and T
m
(as indicated
above). Values of T
m
range from as low as 275 K (for
polyisobutylene), to up to 728 K (for polyethylene ter-
ephthalamide) (1, 6). ASTM D2117 and D3418 describe
methods for measuring T
m
.
Thermal Conductivity. Thermal conductivity k is the
parameter in Fourier’s law relating the flow of heat to
the temperature gradient. In practical terms, k is a
measure of the ability of a material to conduct heat. The
thermal conductivity of a polymer is the amount of heat
conducted through a unit of thickness per unit area, unit
time, and degree of temperature. Thermal conductivity
values control the heat-transfer process in applications
such as plastic processing, heat sealing, cooling and heat-
ing a package, and sterilization processes. The k values for
plastics are much lower than those for metals; for in-
stance, thermal conductivity for plastics ranges from
3 10
4
for PP to 12 10
4
cal/(s cm 1C) for HDPE,
while for aluminum k = 0.3 cal/(s cm 1C) and for steel
k = 0.08 cal/(s cm 1C) (8). Plastic foams show k values
much lower than those for the nonfoamed plastics because
of the presence of air trapped within the cellular struc-
ture. Enhanced by low thermal conductivity of air, foams
make excellent insulating materials and good cushioning
packaging materials. Plastic fillers may increase the
thermal conductivity of plastics. Methods for measuring
k are given in ASTM D4351, C518, and C177.
Thermal Expansion. The coefficient of linear (or volume)
thermal expansion is the change of length (volume) per
unit of length (volume) per degree of temperature change
at constant pressure: b = (1/L)(dL/dT) and a = (1/V)
(dV/dT). Units of a and b are K
1
or 1F
1
(reciprocal
degrees Kelvin and Fahrenheit). Compared to other ma-
terials, polymers have high values of thermal expansion
coefficients. While metals and glass have values in the
range 0.9–2.2 K
1
polymers range from 5.0 to 12.4 K
1
(8).
Thermal expansion coefficients can be measured by ther-
momechanical analysis (TMA). ASTM D696 gives a
method using a quartz dilatometer, and ASTM E831
describes the determination of the linear thermal expan-
sion of solid materials. Volume contraction of a container
from the molding operation temperature down to room
temperature is called shrinkage, and its measurement is
described in ASTM D955, D702, and D1299-55.
994 POLYMER PROPERTIES
MECHANICAL PROPERTIES
The response of polymers subjected to mechanical forces is
determined by the polymer’s isoentropic elastic and viscoe-
lastic behavior. Ideally, elastic behavior should be reversi-
ble for small deformations. Two types of small deformation
are possible: tensile and shear. Hooke’s law describes the
response of plastics to tensile forces by relating tensile
stress to elongation, while the law of shear deformation
relates shear stress to both shear modulus and angle of
shear. Viscoelastic behavior is observed in large deforma-
tions of solid polymers and polymer melts (9). In a creep
experiment, the elongation of solid sample increases slowly
as the weight hangs from the sample. When the force is
released, the sample partially recovers its shape, decreas-
ing the elongation. This behavior is due to the viscoelasti-
city behavior of the polymers: It partially recovers because
it is elastic, and it creeps because it is viscous. This behavior
is due to the continuous-chain molecular rearrangement
that takes place at all temperatures. The time lapses
associated with molecular rearrangement are much larger
below than above the glass-transition temperature. The
viscoelastic behavior affects almost all mechanical proper-
ties of a polymer and should be carefully considered in
packaging design. Polymer strength increases with increas-
ing molecular mass and with increasing intermolecular
forces, but it decreases with the presence of plasticizers.
For example, at the same molecular mass, polyamides,
polyesters, or polyacrylonitriles are stronger than polyole-
fins, and plasticized PVC is weaker than rigid PVC.
Bursting Strength. This is the hydrostatic pressure,
usually in pascal (or psi), required to produce rupture of
the material when the pressure is applied at a controlled
increased rate through a circular rubber diaphragm of
30.48 mm (1.2 in.) in diameter. ‘‘Points bursting strength’’
is the pressure expressed in psi. The measurement of the
bursting strength of plastic films is described in ASTM
method D774.
Dimensional Stability. Dimensional stability refers to
the capability of a structure to maintain its dimensions
under the changing conditions of temperature and humid-
ity. Machine and transverse directions may produce dif-
ferent changes in dimensional stability, which is
important in any flexible material converting process.
During printing, for example, even small changes in
dimensions may lead to serious problems in holding a
print pattern. ASTM D1204 describes a standard method
for linear dimensional changes of flexible thermoplastic
films and sheets at elevated temperature.
Folding Endurance. This is a measure of the resistance
of the material to flexure or creasing. Folding endurance is
greatly influenced by the polymer’s glass-transition tem-
perature and the presence of plasticizers. ASTM D2176
describes the procedure to determine the number of folds
necessary to break the sample film.
Impact Strength. Impact strength is the material’s re-
sistance to breakage under a high-velocity impact. Widely
used impact tests are Izod (ASTM D256A) and Charpy
(ASTM D256) for rigid materials, and for dart-drop impact
(ASTM D4272) and pendulum-impact resistance (ASTM
D3420) for flexible structures. A free-falling dart method
for polyethylene films is described in ASTM D1709. Unlike
low-speed uniaxial tensile tests, the pendulum-impact test
measures the resistance of film to impact puncture simu-
lating high-speed enduse applications. Dart drop mea-
sures the energy lost by a moderate-velocity blunt
impact passing through the film. Both pendulum and
impact tests measure the toughness of a flexible structure.
Melt-Flow Index. Most polymer melts follow a pseudo-
plastic behavior, which implies that the viscosity de-
creases with shear rate. In common extrusion processes,
the shear-rate value varies from 100 to 100,000 s
1
(per
second). It follows that a complete rheological description
of an extrusion process should include a wide range of
shear-rate values at suitable temperature range. The
melt-flow index (MFI) is a widely used test to measure
polymer flow properties, but it provides flow values at only
one shear-rate value. MFI, also called extrusion plast-
ometer test, is described in ASTM D1238. The melt-flow
index measures the mass of polymer extruded during
10 min. Values of MFI vary between 0.3 and 20 g/10 min,
corresponding to shear-rate values ranging from 1 to
50 s
1
. Variables that affect the MFI of a resin include
average molecular mass, distribution of molecular mass,
branching, and temperature (10).
Pinhole Flex Test. Pinhole flex resistance is the property
of a film to resist the formation of pinholes during re-
peated folding. A related test is the folding endurance.
Film having a low value of pinhole flex resistance will
easily generate pinholes at the folding line, under re-
peated flexing. The test is described by the standard
ASTM F456.
Poisson’s Ratio. Poisson’s ratio is the ratio of lateral
strain to longitudinal strain measured simultaneously in
a creep experiment. Its value varies from 0.5 (for totally
deformed but nonelastic liquids), to 0 (for pure elastic
incompressible solid materials). Typical values for rubber
materials range from 0.49 to 0.499; and for plastic, from
0.20 to 0.40. This shows that when a polymer sample is
elongated, its volume will increase. Because polymers are
viscoelastic materials, the Poisson-ratio value is morphol-
ogy- and time-dependent (1).
Tensile Properties. The mechanical behavior of a poly-
mer can be evaluated by its stress–strain tensile charac-
teristics. The stress is measured in force/area, which can
be given in pascals or psi, and the strain is the dimension-
less fractional length increase. Modulus of elasticity, E,is
the elastic ratio between the stress applied and the strain
produced, giving the material’s resistance to elastic defor-
mation. The tensile modulus also gives a measure of the
material stiffness; the larger the modulus, the more brittle
the material. For example, E of LDPE is 250 mPa, while
for ‘‘crystal’’ PS it is 2500 mPa. Comparatively, values of
tensile modulus in polymers (1.9 10
3
mPa for nylon) are
POLYMER PROPERTIES 995
much lower than for glass (60 10
3
mPa) or mild steel
(220 10
3
mPa) (6). Elastic elongation is the maximum
strain under elastic behavior. Ultimate strength or tensile
strength is the maximum tensile stress the material can
sustain. Ultimate elongation is the strain at which the
sample ruptures. Toughness is how much energy a film
can absorb before rupturing, and it is measured by the
area under the stress–strain curve. Brittleness is the
lack of toughness (11). Amorphous and semicrystalline
polymers become brittle when cooled below their glass-
transition temperature. Tests for tensile properties are
described in ASTM D882, for flexural strength in ASTM
D790, and for flexural modulus in ASTM D790 M.
Tear Strength. Tear-strength measurement considers
the energy absorbed by the film sample in propagating a
tear. Standard methods available are (a) ASTM standard
D1004, which describes the measurement for initial tear
resistance, and (b) ASTM D1922 and ASTM D1938, which
refer to the energy absorbed by a test specimen in propa-
gating the tear that has already been initiated. The value
of tear strength in one film depends on the orientation
stretching ratio, whether the measurement is performed
along the crossdirection or along the machine direction
(CD or MD).
Viscosity. Viscosity, a fundamental rheological property
of fluids, is a measure of the molecular resistance to shear
fluid deformation, generated by the action of external
forces. When the external action is given by the shear
stress and the shear flow deformation given by the rate of
strain or shear rate, the absolute viscosity is equal to the
ratio of shear stress/shear rate. The viscosity of a polymer
will increase with the length of the polymer chain, with
smaller dispersion index (narrow distribution), and with
increasing intermolecular forces. An increase of tempera-
ture, however, will decrease the viscosity. The rheological
behavior of thermoplastics is, in most cases, Newtonian or
pseudoplastic (10). The viscosity of Newtonian fluids is
constant at any shear rate value. This behavior is char-
acteristic of fluids with low molecular mass. In pseudo-
plastic materials, the viscosity decreases with shear rate;
that is, the viscosity depends on the value of shear rate.
Pseudoplastic behavior is characteristic of polymer melts.
The viscosity of polymer melts ranges between 100 and
10
7
N/(s m
2
). Viscosity is also known as either kinematic
viscosity or intrinsic viscosity. Kinematic viscosity is the
absolute viscosity divided by the density. Intrinsic viscos-
ity (IV) can be obtained as follows: (1) Solutions of differ-
ent concentrations c of the polymer in a solvent (having
viscosity Z
0
) are prepared; (2) the viscosity Z of the solu-
tions are measured; and (3) from the plot of viscosity
number, [(Z Z
0
)/Z
0
c], versus concentration, the IV is
obtained as the value of the viscosity at concentration
zero (11). Therefore, intrinsic viscosity, is a measure of the
polymer’s capacity to enhance the viscosity of the solvent.
The units of IV are mL/g. For PE, the relation between
molecular weight (MW) and IV is accepted to be as follows:
MW = 5.3 10
4
(IV)
1.37
(8). An IV of 5, for example, corre-
sponds to an MW of 500,000. Intrinsic viscosity values of
polymers can be estimated by group contribution in terms
of molar intrinsic viscosity (1). ASTM has published
several methods for measuring viscosity.
SOLUBILITY AND CHEMICAL DEGRADATION
Plastics are chemically resistant to many gases, liquids,
and solid products within a wide range of pH. Never-
theless, polymers are not inert materials. Given the right
conditions (temperature, time, and concentration), poly-
mers can be depolymerized, transformed by chemical
reactions, and penetrated by solvents and vapors. They
can be affected by environmental agents such as visible
and UV radiation, oxygen, microbes, solvents, and organic
compounds (12). The extent of the transformation depends
on the thermodynamic and kinetics parameters, which, in
turn, depend heavily on temperature. In hydrophilic
polymers (e.g., nylons, EVOH, and PET) water acts as a
plasticizer, consequently lowering the glass-transition
temperature and changing oxygen permeability and other
mechanical properties (13). Water can dissolve certain
polymers such as PVOH. Hydrophobic polymers such as
polyolefins, on the other hand, are not affected by water.
Chemical Degradation. Chemical degradation of poly-
mers results in the fragmentation of large chains into
smaller ones and, eventually, into atomic elements. One
important type of polymer chemical degradation is oxida-
tion (12). Often oxidation is promoted or induced by
electromagnetic radiation (photodegradation) and by ther-
mal energy at high temperature (thermal oxidation).
Polymers such as PE, PP, and PVC are particularly prone
to oxidation during melt processing. Tensile strength,
elasticity, and impact strength are drastically affected by
thermodegradation processes. Polymers may also be de-
graded by acids and alkalis. Test for resistance to acids,
bases, and solvents is described in ASTM D543. Plastic
resistance to grease and oil is covered in ASTM D722. The
ASTM D756 test covers the effect of atmospheric humidity
and temperature on plastics, and ASTM D570 covers the
sorption of water of immersed plastics.
Environmental Stress Cracking. A plastic material may
be resistant to a chemical compound in no-stress condi-
tions. Nevertheless, the plastic may crack when subjected
to a mechanical stress during exposure to that compound.
Almost all plastics can show environmental stress crack-
ing (ESC), when exposed to gases, liquids, or solids while
under stress. The mechanism by which a plastic shows
ESC is often complex. A practical evaluation of a plastic’s
resistance to liquids can be done by immersing a polymer
sample in the liquid and then checking for the appearance
of crazing, cracks, or even total failure of the sample. Also,
a plastic container can be tested by filling it with the
liquid, causing ESC. Stress conditions applied during
testing may be of two types: constant stress or constant
strain. The selection of the test method should correspond
with the intended use of the container. Common ESC tests
for PE bottles include bottle stress crack (ASTM D2561),
top-load stress crack, and internal pressure (ASTM
996 POLYMER PROPERTIES
D2561). For some applications, the temperatures recom-
mended by ASTM for these tests may be too mild (14).
Flammability. The flammability behavior of a material
is a broad term related to the easiness to ignite, burn,
produce smoke, and endure burning. A flammable mate-
rial ignites easily and has a rapid flaming combustion
process. The initial step is decomposition (or pyrolysis),
which is an endothermic process (the material absorbs
heat). After ignition, products from pyrolysis are com-
busted and consequently, heat Q is generated. Some
flame-retardant agents tend to minimize Q. The flamm-
ability characteristics of a polymer may be predicted from
its chemical structure (1), and a method to measure flame
resistance is the oxygen index (15). ASTM test D3713
describes the response of solid plastics to ignition by a
small flame, and the ASTM D4100 method covers the
gravimetric determination of smoke-particulate matter
produced from combustion or pyrolysis of plastic materi-
als. Testing plastics for practical fire situations is very
complex and depends on many variables other than just
chemical structure (16, 17).
Photodegradation. Sunlight is the source of energy for
polymer photochemical degradation. Near-ultraviolet and
visible radiation (290–710 nm) carry enough energy to
break single covalent bonds such as C–C, C–O, or C–N.
The absorption of these radiations is sometimes attributed
to impurities in the polymer. Photodegradation processes
generate chain ruptures and free-radical formation, re-
sulting in color changes and increasing fragility (because
of chain crosslinking and chain scissions). UV light stabi-
lization of PP and other polyolefins can be achieved by the
addition of hindered amine light stabilizers (HALSs) (18).
These additives reduce the rate of photoinitiation and
chain-reaction propagation by their ability to trap free
radicals. ASTM D4674 describes the accelerated testing
for color stability of plastics exposed to indoor fluorescent
lighting and window-filtered daylight.
Solubility Parameter. Polymer solubility properties in
organic liquids are controlled by the polymer’s cohesive
energy. The cohesive energy of a solid (or liquid) is the
total energy necessary to remove one molecule from
the bulk of the solid (or liquid). It is also an indication of
the molecular internal pressure. The solubility of a poly-
mer is given by its solubility parameter d, which is the
square root of the cohesive-energy density (CED),
d = (CED)
1/2
. In addition to cohesive energy density, poly-
mer morphology and molecular mass also strongly affect
the tendency of a polymer to dissolve. Crystalline struc-
tures, on the other hand, are insoluble below their melting
temperature. Polymers have solubility parameter’s ran-
ging from 12 to 29 (J/cm
3
)
1/2
. Substances that have similar
delta values [o0.5 (J/cm
3
)
1/2
apart] will tend to mutually
dissolve. This substantiates the rule of thumb that ‘‘like
dissolves like.’’ Solubility parameters can be determined
by solvent swelling (19) or intrinsic viscosity methods (20).
Values of CED and solubility parameter of a polymer
can be estimated by group contribution methods, based
on the contribution of intermolecular forces: polar forces,
dispersion forces, and hydrogen bonding (1). Extensive
tables of d are presented by Barton (21).
Solubility in Organic Solvents. Many organic compounds
interact with polymers to produce slight swelling or
staining, or total dissolution, depending on the polymer
molecular mass, crystallinity, and solubility parameter.
ASTM D2299 gives a method to assess the susceptibility of
plastic staining by incidental contact with organic pro-
ducts. Common solvents for plastics are boiling xylene and
trichlorobenzene; solvents for polyolefins, are styrene
polymers, vinylchloride polymers, and polyacrylates; the
solvent for polyamides and polyvinyl alcohol derivatives is
formic acid; tetrahydrofuran is the solvent for all uncros-
slinked polymers; nitrobenzene and m-cresol are the
solvents for PET; water is the solvent for polyacrylamide
and polyvinyl alcohol; and dimethylformamide is the
solvent for polyacrylonitrile. In addition, polyvinyl acetate
is dissolved by benzene, chloroform, methanol, acetone,
and butyl acetate (22). ASTM D543 covers the testing of
all plastic materials for resistance to chemical reagents.
Thermodegradation. Thermal degradation of polymers
is the breaking of chain bonds by heat, in the absence of
oxygen. Heat-stabilizing additives help prevent degrada-
tion, but impurities may have the contrary effect. Ther-
mally stable polymers resist thermal degradation and will
have high values of bond dissociation energy (1). As the
temperature increases, chemical bonds with the lowest
values of dissociation energy will be the first to break.
Thermodegradation affects PE, PP, and PVC (18). The
poor thermal stability of the PVC resin is caused mainly
by dehydrochlorination. For each HCl eliminated from the
polymer chain, a double bond is created, which, in turn,
promotes oxygen chemical degradation. Heat stabilizers
include inorganic and organic compounds. In the case of
flexible PVC films, barium–zinc and calcium–zinc stabili-
zers are used, while organotin compounds are commonly
used in PVC bottle containers.
BARRIER PROPERTIES
The barrier property of a material indicates its resistance
to diffusion and sorption of foreign molecules. A high-
barrier polymer has low values of both diffusion (D) and
solubility (S) coefficients. Since the permeability coeffi-
cient P is a derived function of D and S, a high-barrier
polymer has low permeability. The diffusion coefficient
indicates how fast a penetrant will move within the
polymer, while the solubility coefficient gives the amount
of the penetrant taken (or sorbed) by the polymer from a
contacting phase. Both diffusion and solubility can be
applied to the reverse process of sorption—that is, the
migration of compounds from the polymer to a surround-
ing medium. Several factors influence the effective value
of the diffusion and solubility coefficients in polymers:
(1) chemical compositions of the polymer and permeant,
(2) polymer morphology (since diffusion and sorption occur
mainly through the amorphous phase and not through
crystals), (3) temperature (as temperature increases,
POLYMER PROPERTIES 997
