Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.

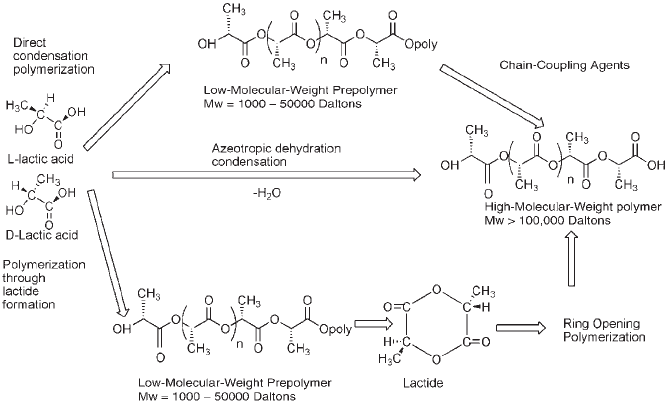
of PLA in the market. In this process, PLA is derived from
lactic acid existing in two stereochemical configurations
(i.e., L or D lactic acid), which is catalytically converted
under low pressure into LL-lactide, meso or LD-lactide,
and DD-lactide, and then it is ring open polymerized into
different structures of poly(L-lactic acid, D-lactic acid, or
LD-lactic acid). In the direct condensation and azeotropic
dehydration condensation polymerization, PLA is derived
from L and D-LA, directly. So, high-molecular poly(L,D, or
LD LA) can also be produced (2).
PLA resins between 92% L-LA and 98% L-LA, and
stereocomplex PLA (50/50 blend of PLLA and PDLA) can
be obtained commercially. These resin grades can be
processed by extrusion/thermoforming, injection molding,
biaxially oriented films, injection stretch blow molded
bottles, fiber melt spinning, and spunbond items. All these
resins are mainly targeted for particular application;
therefore, their degree of L-and D-lactide varies according
to the applications (4). The latest developed stereocomplex
resins are mainly used for fibers, films, and thermoform-
ing articles.
PROPERTIES
Structure
The principal peaks of PLA absorption in the infrared
range are summarized in Table 1. The strong infrared (IR)
band at 2997, 2946, and 2877 cm
1
are assigned to the CH
stretching region, n
as
CH
3
,n
s
CH
3
, and nCH modes, re-
spectively. The CQO stretching region is observed by a
large band at 1748 cm
1
. The region between 1500 and
1360 cm
1
is characterized by the 1456 cm
1
CH
3
bend.
The CH deformation and asymmetric bend appear at 1382
and 1365 cm
1
, respectively. The bands that appear at
1315 and 1300 cm
1
are caused by the CH bend modes. In
the region from 1300 to 1000 cm
1
, the C–O stretching
modes of the ester groups at 1225 cm
1
and the -O-C
asymmetric mode at 1090 cm
1
are presented. Between
1000 and 800 cm
1
, we can observe peaks at 956.36 and
921.94 cm
1
, which can be attributed to the characteristic
vibrations of the helical backbone, mixed with the CH
3
rocking modes. Two bands related to the crystalline and
amorphous phases of PLA are found at 871 and 756 cm
1
.
The peak at 871 cm
1
can be assigned to the amorphous
phase, and the one at 756 cm
1
to the crystalline phase.
The peaks below 300 cm
1
correspond mainly to CH
3
torsion modes and to the skeletal C-C torsions.
Semicrystalline PLAs have superior physical and
structural performance compared with amorphous PLAs.
Researchers have postulated that the improvements in
mechanical and barrier properties of semicrystalline poly-
mers are related directly to complex crystalline structures
and their distribution in the amorphous matrix (8–10).
The amorphous polymer region cannot be in the crystal
lattice of the semicrystalline polymer, and so it must
reside between the lamellar stacks (interlamellar) or in
the interfibrillar region within the spherulites (inter-
spherulitic). The variation in size and shape of the crystal-
lites, their spatial distribution, the crystalline morphology
(which depends on molecular weight, polydispersity index,
and processing conditions), the degree of crystallinity, and
the space and distribution available for the amorphous
region will all dictate the final physical, mechanical, and
barrier properties of PLA.
Depending on the preparation conditions, poly(L-lactic
acid) crystallizes in three forms (a-, b-, and g-) (2). The
stable a-form exhibits a well-defined diffraction pattern
with a space group orthorhombic P2
1
2
1
2
1
, with a unit cell
containing two antiparallel chains. The chain conforma-
tion is a 2-fold (15*2/7) helix distorted periodically form of
the regular s(3*10/7 helix). The lattice parameters are
a = 10.66 A
˚
; b = 6.16 A
˚
, and c (chain axis) = 28.88 A
˚
, with a
crystal density around 1.26 g/cm
3
. The b-form of PLA is
generally prepared at a high draw ratio and high drawing
temperature. The chain conformation is a left-handed
3-fold helix. The b-form of PLA has an orthorhombic
unit cell (containing 6 chains) that contains a 3 l (3 A
˚
rise/1 monomeric unit) polymeric helix. The lattice para-
meters of the unit cell are a = 10.31 A
˚
, b = 18.21 A
˚
, and c =
9.0 A
˚
. The a-structure is more stable than the b-structure,
with a melting point of 1851C compared with 1751C for the
Scheme 1. Synthesis methods for obtain-
ing high-molecular-weight polylactide and
poly(lactic acid); scheme is from Auras et
al. (2) by permission of Wiley-VCH Verlag
GmbH & Co. KGaA
968 POLY(LACTIC ACID)

b-structure. Chain packing in the b-form has recently
been termed a frustrated structure, where the crystal
structure comprises a frustrated packing of three 3-fold
helices in a trigonal unit cell of parameters a = b = 10.52 A
˚
,
c = 8.8 A
˚
, and space group P3
2
(11). The g-form is formed by
epitaxial crystallization and contains two antiparallel
s(3/2) helices in the pseudoorthorhombic unit cell
a = 9.95 A
˚
, b = 6.25 A
˚
, and c = 8.8 A
˚
, and it assumes the
known 3-fold helix of poly(lactic acid) (2).
Optical, Thermo-Physical, Rheological, Mechanical, and
Barrier
The optical, physical, mechanical, and barrier properties
of PLA compared with PS and polyethylene terephtalate
(PET) are shown in Table 2. PLA has similar density than
PET, and a similar semycrystalline structure can be
obtained during processing. Because the glass transition
temperature (T
g
) of PLA and its heat deflection tem-
peratures (HDT) are very close, PLA suffers heat deforma-
tion when it is exposed to temperatures higher than 501C.
The tensile strength values obtained for PLA are within
the range of values reported in the literature for PS under
the same testing conditions. However, when compared
with PET, PLA films showed lower values. Table 2 also
shows the water, oxygen, carbon dioxide, and aroma
barrier properties of PLA and these values are compared
with those of PS and PET when data were available. PLA
has a better barrier for carbon dioxide, oxygen, and water
vapor permeability when compared with PS but a lower
barrier when compared with PET. PLA has an aroma
barrier similar to PET, so it makes a good polymer for the
storage of products with high flavor. Theoretical predic-
tions based on the solubility parameters indicate that PLA
will interact with nitrogen compounds, anhydrides, and
some alcohol but will not interact with aromatic hydro-
carbons, ketones, esters, sulfur compounds, and water.
PROCESSING
PLA suffers hydrolysis, which is a reduction in molecular
weight caused by the presence of water at temperatures
near the glass transition temperature (T
g
B551C), so for
processing it must be dried below 250 ppm (12). Additional
drying should be conducted for processes that have long
residence time. Drying of semicrystalline PLA should be
conducted in the range of 80 to 1001C and below T
g
for
amorphous PLA (4). Because PLA degrades at elevated
temperatures and relative humidity (RH), the resins
should be protected from hot and humid environments
during storage. Henton et al. (12) reported that amor-
phous PLA can dramatically reduce its molecular weight
(M
w
) in less than a month when exposed to 601C and 80%
RH. More information about the drying condition of PLA
can be found elsewhere (4).
The main way to process PLA is through extrusion and
injection molding, blow molding, film blowing, and melt
spinning. To ensure that all the crystalline PLA phases
are melted and to achieve an optimal melt viscosity for
processing, PLA is generally extruded around 160 to
1951C. In general, commercial-grade PLA resins can
typically be processed using a conventional extruder.
Cast and film sheet of PLA are produced with a screw
diameter of 19.3 mm, compression ratio of 2:1, and length/
diameter ratio of 25. The recommended compression ratio
for PLA processing is in the range of 2:3 (13). High roller
temperatures (i.e., 25501C) are generally used to cast
PLA avoiding the building up of lactide around the die.
General equipment to process PET and PS can be used
and/or modified to process PLA. The physical properties of
PLA as for other polymers can be enhanced by orientation.
Resins with a higher D-lactide content allow a higher
drawing ratio. A blown film of virgin PLA resin is difficult
to obtain. So, blown PLA films contain additives, such as
viscosity enhancers, are to strengthen PLA’s melting
strength, and slip additives are used to reduce the coeffi-
cient of friction (4).
Thermoforming of PLA is normally conducted to pro-
duce PLA clamshells and containers. The properties of
thermoformed PLA containers have been compared with
PS and PET showing that PLA could out perform PET and
PS at lower temperatures (14, 15).
PLA bottles have been produced based on injection
stretch blow molding (ISBM) techniques. The ISBM pro-
cess produces biaxial-orientated PLA bottles and has
better properties than injection molding of amorphous
PLA (4). Most of these bottles are currently being used
for noncarbonated drinks.
PLA foaming has been gaining attention because of its
potential to reduce weight and reduce thermal conductiv-
ity. Foaming of PLA has been a niche application in tissue
engineering and medical implant applications. As the
price of PLA is dropping, PLA foam can find new applica-
tions in packaging, such as cups, bowls, and loose-fill
packaging materials (4). Most of the challenge in foaming
PLA is controlling the bubble size of the foam structure
(16). Another special category of growing interest for PLA
applications is fiber spinning and electrospinning of ultra-
fine fibers. The former application has mainly reached
market applications in textile production, and it is having
great acceptability because of lower water-barrier proper-
ties. However the electrospinng PLA ultrafine fibers is
mainly being developed for tissue engineering and biome-
dical applications (4).
Table 1. Infrared spectroscopy data: Peak band
assignments for PLA infrared spectra
Assignment
Peak Position,
cm
1
–OH stretch (free) 3571
–CH- stretch 2997 (asym),
2946(sym), 2877
–CQO carbonyl stretch 1748
–CH
3
bend 1456
–CH– deformation including symmetric
and asymmetric bend
1382, 1365
–CQO bend 1225
–C–O– stretch 1194, 1130, 1093
–OH bend 1047
–C–C– stretch 926, 868
POLY(LACTIC ACID) 969

Table 2. Optical, physical, mechanical, and barrier properties of PLA compared with PS and PET
PLA PS PET
Optical
Refractice index
a
1.35–1.45 1.591 1.54
Clarity Clear—yellow Clear Clear
Thermo-Physical
Density amorphous,
b
,kg.m
3
1250 1030 1330
Density 100% crystalline,
c
,kg.m
3
1490 1150 1465
Van der Waals volume (V
W
),
d
,cm
3
.mol
1
34.45 62.9 94.2
Molar volume of glassy amorphous (Vg),
d
,cm
3
.mol
1
55.12 99.0 144.5
Molar volume of semycrystalline polymer (Vc),
d
,cm
3
.mol
1
49.44 92.0 130.0
Solubility parameter (dp), 251C,
e
J
0.5
cm
1.5
19–20.5 19.4 22.9
Glass transition temperature (T
g
)
f
1C 55–65 105 73–80
Melting temperature (T
m
),
f
,1C 130–180 Atactic 245–265
Initial decomposition temperature (T
d,0
)
g
1C 335 326 378
Half decomposition temperature (T
d,1/2
),
g
, 1C 395 363 450
Average energy of activation (E
act
),
g
, kJ.mol
1
205–297 230 —
Enthalpy (DHm),
h
, 100%, J.g
1
93, 135 N/A 135.8
Crystallinity,
g
, % 0–40 N/A 10–40
Heat deflection temperature,
i
, 1C 55–65 88 70
Vicat penetration temperature,
i
, 1C 59 101 76
Thermal conductivity 10
4
,
j
, cal.cm
1
.s
1
.C
1
2.9 4.3 5.7
Heat capacity,
j
cal.g
1
.1C
1
0.39 0.54 0.44
Thermal expansion coefficient 10
6
,
j
, 1C
1
70 70 70
Surface tension,
k
, dyn.cm
1
42.0 40.7 44.6
Oxygen index (OI),
l
— 0.185 0.21
Friction Coefficient,
m
0.37 0.38 0.25
Melt Flow Index,
n
, g. min
1
0.85 0.28 7.2
Electrical, Magnetic, and Acoustic
Dielectric constant,
o
, e N/A 2.25 2.9/3.2
Magnetic susceptibility, (c),
o
, 10
6
,cm
3
.g
1
N/A 0.705 0.505
Sound velocity, m
L
,
o
, m.s
1
N/A 2400 N/A
Sound velocity of propagation of transverse waves, m
Sh
,
o
, m.s
1
N/A 1150 N/A
Rheological
Intrinsic viscosity, n,
p
, dl.g
1
— — 0.72270.029
Mark-Houwink constants
K 10
4
,
p
, DL.g
1
8.5
b
— 7.44
a
a
p
0.66
b
— 0.648
a
Mechanical
Tensile strength @ yield,
q
, MPa 0.88 36–51.7 48–72
Elastic modulus,
q
, GPa 8.6 2.3–3.3 2.8–4.1
Elongation at break,
q
, % 3–30 1.2–2.5 30–300
Flexural strength,
r
, MPa 70 83 70
Flexural Modulus,
r
,GPa 3.8 — 2.4–3.1
Unnotech Izod Impact,
r
,J.m
1
106 — —
Notched Izod Impact,
r
,J.m
1
26 16 101
Rockwell hardness
r
, 88 — 106
Impact strength Poor Poor Good
Barrier
Oxygen 10
18
,
s
, kg.m.m
2
.s
1
.Pa
1
@251C 1.2170.07 27 0.19
Oxygen activation energy,
s
, kJ.mol
1
[25–451C] 41.4373.5 – 30.30
Carbon dioxide 10
17
,
t
, kg.m.m
2
.s
1
.Pa
1
@251C 2.7770.05 15.5 0.17
Carbon dioxide activation energy,
t
, kJ.mol
1
15.6570.63 — —
Nitrogen 10
19
,
u
, kg.m.m
2
.s
1
.Pa
1
468 — 0.68
Nitrogen activation energy,
u
, kJ.mol
1
———
Water 10
14
,
v
, kg.m.m
2
.s
1
.Pa
1
@251C 1.7570.05 0.67 0.11
Water activation energy,
v
, kJ.mol
1
9.7370.27 0–8.0 —
d-limonene 10
19
,
w
, kg.m.m
2
.s
1
.Pa
1
o1.0 — 9
d-limonene activation energy,
w
, kJ.mol
1
———
Ethyl acetate 10
19
,
w
, kg.m.m
2
.s
1
.Pa
1
5.34 — —
Ethyl acetate activation energy,
w
, kJ.mol
1
N/A — —
970 POLY(LACTIC ACID)
Blends of PLA with other polymers have been re-
searched to improve PLA films bursting strength, elonga-
tion and tear strength, and impact resistance properties.
Some main materials that have been blended with PLA are
thermoplastic starch, poly(ethylene glycol), triacetin, tri-
butyl citrate, poly(hydroalkanoates), and poly(3-hydroxy-
loctanoate) (4). Cost reduction is achieved when the
blending counterpart has a lower cost. Besides PLA blend-
ing, compounding of PLA with inorganic or organic parti-
cles has investigated to enhance the barrier, thermal, and
mechanical properties of the PLA composite. Special cate-
gories of compounding are nanocomposites which have
been reinforced into PLA. Among the main nanocompo-
sites used are layered silicates, carbon nanotube, hydro-
xyapaite, layered titanate, aluminum hydroxide, and
cellulose nanowiskers (4). Layered silicate clays such as
montmorillonite MMT have been researched extensively in
the industry to produce PLA with higher mechanical and
flexural properties, elevated heat distortion temperature,
enhanced barrier properties, and accelerated biodegrada-
tion. Additional promise has been created with cellulose
nanowiskers to produce PLA microwavable packages (4).
End-of-life Scenario
PLA can be recycled and has been certified as a compo-
stable polymer according to ASTM D6400 and EN13432
by the main international certifying entities (7, 17). The
composting of PLA is enhanced by hydrolysis because of
the absorption of water resulting in the hydrolysis of the
ester linkages. The rate of degradation is highly affected
by temperature, moisture, and pH conditions of the
(Table footnote Continued)
N/A: Not available
a
Refractive index values for PLA were calculated by Gladstone and Dale, Vogel, and Lloyenga methods according to Ref. (20). PS and PET values are
experimental values obtained from Ref. (20).
b
Density of the amorphous PLA was obtained from Ref. (21), PS and PET values are from Ref. (20).
c
Density of 100% PLA, PET, and PS was calculated according to the group contribution method (20).
d
PLA, PET, and PS values were calculated using the group contribution method (20).
e
PLA, PET, and PS values were obtained from Ref. (2).
f
T
g
and T
m
of PLA, PS, and PET were obtained from Ref. (1).
g
Td,0 and Td,1/2 of PLA was obtained from Ref. (18); PS and PET values were obtained from Ref. (20).
h
PLA and PET values obtained from Ref. (22).
i
PLA, PS, and PET values were obtained from Ref. (23).
j
PLA, PS, and PET values were obtained from Ref. (4).
k
PLA value was obtained from Ref. (2); PS and PET values were obtained from Ref. (20).
l
PLA value was obtained from Ref , PS and PET values were obtained from Ref (20).
m
PLA, PET, and PS values were obtained from Ref. (2).
n
PLA and PS, PET values were measured at 2001C and 5 kg according ASTM D1238.
o
PS and PET values were obtained from Ref. (20).
p
PLA and PET, PS values were measured according to ASTM D445 and D446 (PLA values were determined in Tetrahydrofuran at 30 1C, PET values were
determined in phenol and 1, 1, 2, 2-tetrachloroethane (60:40 by volume) at 25 1C.
q
PLA, PET, and PS values were obtained from Ref. (2).
r
PLA, PS, and PET values were obtained from Ref. (23).
s
PLA, PET, and PS values were obtained from Ref. (1, 22); oxygen activation energy are reported for temperatures between 25 and 451C.
t
PLA, PET, and PS values were obtained from (1); carbon dioxide activation energy is reported for temperatures between 25 and 451C.
u
PLA values were obtained from Ref. (24); PET and PS values were obtained from Ref. (20).
v
PLA, PS, and PET values were obtained from Refs. (1, 22); water activation energy is reported for temperatures between 101C and 37.81C.
w
PLA, PS, and PET values were obtained from Refs. (25, 26) (et hyl acetate values of PLA and PET are at 3030 PA, and 301C and 9435 Pa and 301C,
respectively; d-limonene of PLA and PET are at 245 Pa and 451C and 45 Pa and 231C, respectively).
0
10
20
30
40
50
60
70
80
010203040506070
Time (days)
%Mineralization
CS
PLA
PET
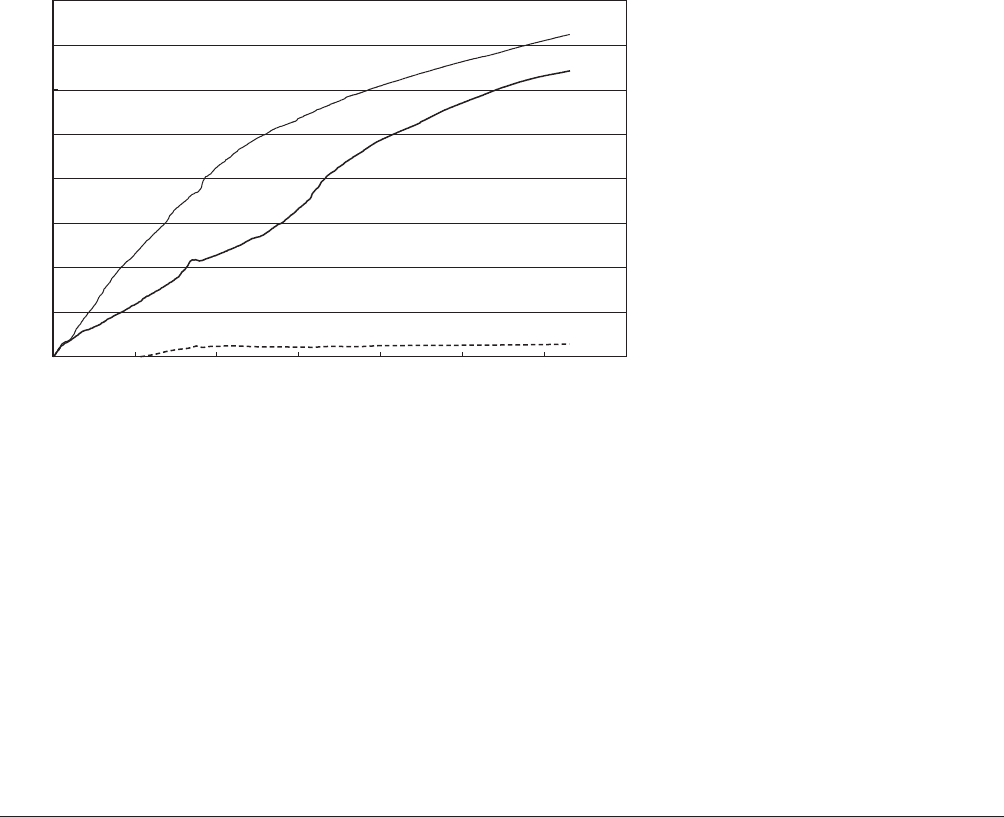
Figure 1. Percentage of mineralization of
PLA, PET, and corn starch at 58721C and
approximately 5575% RH in yard waste com-
post (reproduced from Kijchavengkul et al.
(27)) by permission from Elsevier B.V.
POLY(LACTIC ACID) 971
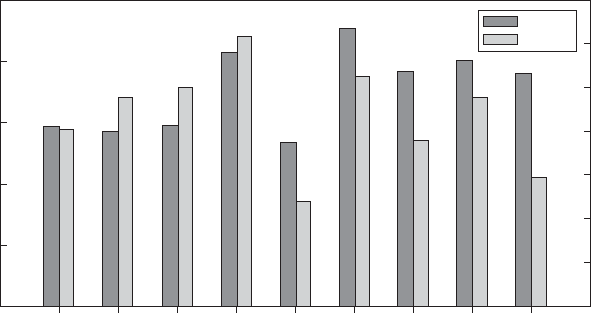
compost (18, 19). PLA polymers with lower L-lactic
acid content degrade faster in a compost environment.
Figure 1 shows the mineralization of PLA bottles com-
pared with PET bottles as a negative control in yard waste
compost.
Figure 2 shows the energy used and amount of green-
house gases emitted in CO
2
equivalents, from producing
1 kg of polymer resin (cradle-to-gate boundary). According
to Figure 2, in general, production of these biodegradable
polymers, except poly(caprolactone) (PCL), uses less en-
ergy than production of many nonbiodegradable plastics,
such as PS, polypropylene (PP), PET, and polyethylene
(PE). The production of PLA and PHA (polyhydrox-
yalkanoate) produces greenhouse gases in similar
amounts as those of PS, PP, and PET. More research is
necessary regarding the end-of-life scenario of these new
biopolymers.
BIBLIOGRAPHY
1. R. Auras, B. Harte, S. Selke, and R. Hernandez, ‘‘Mechanical,
Physical, and Barrier Properties of Poly(lactide) Films,’’ J.
Plastic Film Sheet. 19, 123–135, (2003).
2. R. Auras, B. Harte, and S. Selke, ‘‘An Overview of Polylactides
as Packaging Materials,’’ Macromolec. Biosci. 4, 835–864,
(2004).
3. R. Auras, J.J. Singh, and S.P. Singh, ‘‘Performance Evaluation
of PLA Existing PET and PS Containers,’’ J. Test. Eval. 34,
530–536, (2006).
4. L.T. Lim, R. Auras, and M. Rubino, ‘‘Processing Technologies
for Poly(lactic acid),’’ Progr. Polymer Sci. 33, 820–852, (2008).
5. H. Tsuji, ‘‘Poly(lactide) Stereocomplex: Formation, Structure,
Properties, Degradation, and Applications,’’ Macromol Biosci.
5, 569–597, (2005).
6. R.E. Conn, J.J. Kolstad, and J.F. Borzelleca, et al., ‘‘Safety
Assessment of Polylactide (PLA) for Use as a Food-Contact
Polymer,’’ Fd Chem Toxic. 33, 273–283, (1995).
7. T. Kijchavengkul, and R. Auras, ‘‘Compostability of Poly-
mers,’’ Polymer Internat. 57, 793–804, (2008).
8. B. Bonavoglia, G. Storti, and M. Morbidelli, ‘‘Modeling of the
Sorption and Swelling Behavior of Semicrystalline Polymers
in Supercritical CO
2
,’’ Indust. Engin. Chemi. Res. 2006.
9. M. H. Klopffer, and B. Flaconneche, ‘‘Transport Properties of
Gases in Polymers: Bibliographic Review,’’ Oil Gas Sci. Tech-
nol. 56, 223–244, (2001).
10. L. Mandelkern, Physical Properties of Polymers: The Crystal-
line State, 3rd edition, Cambridge, University Press, New
York, 2004.
11. J. Puiggali, Y. Ikada, H. Tsuji, L. Cartier, T. Okihara, and B.
Lotz, ‘‘The Frustrated Structure of Poly(L-lactide) + ,’’ Poly-
mer. 41, 8921–8930, (2000).
12. D. E. Henton, P. Gruber, J. Lunt, and J. Randall, ‘‘Polylactic
Acid Technology,’’ In: Mohanty AK, Misra M, Drzal LT, eds.
Natural Fibers, Biopolymers, and Biocomposites, Taylor &
Francis, Boca Raton, FL, 2005, pp. 527–577.
13. B. Glasbrenner, NatureWorks(R) PLA—Commerical Develop-
ment of Biopolymers on a World Scale, GPEC. Atlanta, GA,
2005.
14. E. Almenar, H. Samsudin, R. Auras, B. Harte, and M. Rubino,
‘‘Postharvest Shelf Life Extension of Blueberries Using a
Biodegradable Package,’’ Food Chemi. 110, 120–127 (2008).
15. R. Auras, S.P. Singh, and J.J. Singh, ‘‘Evaluation of Oriented
Poly(lactide) Polymers vs. Existing PET and Oriented PS for
Fresh Food Service Containers,’’ Packag. Technol. Sci. 18,
207–216, (2005).
16. L.M. Matuana, ‘‘Solid State Microcellular Foamed Poly(lactic
acid): Morphology and Property Characterization,’’ Biore-
source Technol. 99, 3643–3650, (2008).
17. G. Kale, T. Kijchavengkul, R. Auras, M. Rubino, S.E. Selke,
and S.P. Singh, ‘‘An Overview of Compostability of Bioplastic
Packaging Materials,’’ Macromole. Biosci. 7, 255–277,
(2007).
18. G. Kale, R. Auras, and P. Singh, ‘‘Comparison of Degradabil-
ity of Poly(lactide) Packages in Composting and Ambient
Exposure Conditions,’’ Packag. Technol. Sci. 19, 49–70,
(2007).
19. G. Kale, R. Auras, S.P. Singh, and R. Narayan, ‘‘Biodegrad-
ability of Polylactide Bottles in Real and Simulated Compost-
ing Conditions,’’ Polymer Test. 26, 1049–1061, (2007).
PLA
0
20
40
60
Energy used (MJ/kg polymer)
80
100
PHA PH(3B) PCL
Starch+PCL
PS
PP
Polymer
PET PE
0.0
0.5
1.0
1.5
2.0
GHG emission (kg CO
2
eq./kg polymer)
2.5
3.0
Energy
GHG
3.5
Figure 2. Energy used and amount of greenhouse gases (GHG) emission in CO
2
equivalent from producing 1 kg of polymer resin at the
resin production plants (reproduced from Kijchavengkul and Auras (7) by permission from Wiley-VCH Verlag GmbH & Co. KGaA.)
972 POLY(LACTIC ACID)
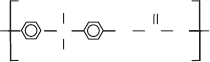
20. D. W. VanKrevelen, Properties of Polymers. 3rd edition, Else-
vier Science B.V. Amsterdan, The Netherlands, 1997.
21. D. R. Witzke, Introduction to Properties, Engineering,
and Prospects of Polylactide Polymers [Doctor of Philo-
sophy], Michigan State University, East Lansing, MI,
1997.
22. R. Auras, B. Harte, and S. Selke, ‘‘Effect of Water on the
Oxygen Barrier Properties of Polyethylene Terephthalate and
Poly(lactide) Films,’’ J. Applied Polymer Sci. 92, 1790–1803,
(2004).
23. R. Bhardwaj, A. K. Mohanty, ‘‘Advances in the Properties of
Polylactides Based Materials: A Review,’’ Biobas. Materi.
Bioenergy. 1, 191–209, (2007).
24. H. J. Lehermeier, J. R. Dorgan, and D .J. Way, ‘‘Gas Permea-
tion Properties of Poly(lactic Acid),’’ J. Membr. Sci. 190, 243–
251, (2001).
25. R. Auras, M. L. Trevor, ‘‘Solubility of Gases and Vapors in
Polylactide Polymers,’’ in Thermodynamics, Solubility and
Environmental Issues, Elsevier, The Neatherlands, 2007,
pp. 343–368.
26. R. Auras, R. Harte, S. Selke, Sorption of ethyl acetate and d-
limonene in poly(lactide) Polymers,’’ J. Sci. Food and Agricul.
86, 648–656, (2006).
27. T. Kijchavengkul, R. Auras, M. Rubino, M. Ngouajio, and R.
Thomas Fernandez, ‘‘Development of An Automatic Labora-
tory-Scale Respirometric System to Measure Polymer Biode-
gradability,’’ Polymer Test. 25, 1006–1016, (2006).
POLYCARBONATE
Updated by Staff
INTRODUCTION
O
CH
3
C
C
O
n
CH
3
O
Polycarbonate (PC) is an amorphous resin that does not
require orientation to achieve its full property profile. The
molten resin can be extruded by the blown or cast pro-
cesses (see Extrusion), injection-molded (see Injection
molding), or blow-molded by the extrusion-blow or injec-
tion-blow techniques (see Blow molding). As polycarbo-
nate cools into film, sheet, or containers, it exhibits
excellent dimensional stability, rigidity, impact resistance,
and transparency over a wide range of temperatures and
loading rates (see Table 1). Because PC is amorphous, its
wide softening range and added strength in thermoform-
ing (see Thermoforming) operations provides deep-draw
capabilities.
Polycarbonate is now the second largest engineering
plastic produced, behind crystalline nylon. Leading world
producers are BASF, General Electric, and The Dow
Chemical Company (1). Trade names include Lexan
(GE), Makrolon (Bayer), Calibre (Dow), Panlite (Teijin),
and Iuplon (Mitsubishi).
Polycarbonate is prepared by the Schotten–Baumann
condensation reaction of phosgene and an aromatic diol
using an amine catalyst or a based-catalyzed transester-
ification of bisphenol and a monomeric carbonate (2).
PROPERTIES
Polycarbonate provides an excellent combination of tensile
strength and flexural modulus at high temperatures (see
Table 2). Its heat resistance, combined with superior
impact resistance at both high and low temperatures,
makes it an excellent structural layer in coextruded or
laminated packaging for hot fill at 180–2101F (82–991C),
retorting at 2501F (1211C), autoclaving at 270–2801F
(132–1381C), and frozen-food packaging. In addition, PC
can be sterilized with both gamma and electron-beam
irradiation with good stability.
Polycarbonate has light-transmittance values of 88–
91% as compared with 92% for clear plate glass. It has a
haze factor of less than 1%, and it maintains these values
throughout the temperature scale. Its high gloss and easy
colorability and printing contribute to distinctive package
design. Polycarbonate has high resistance to staining by
tea, coffee, fruit juices, and tomato sauces, as well as
lipstick, ink, soaps, detergents, and many other common
household materials. Its relatively dense composition
makes it immune to odors, and its hard, smooth surface
facilitates easy removal of foodstuffs.
Some of the most significant attributes of polycarbo-
nate stem from its very low water absorption. Added
weight increase after 24-h immersion at room tempera-
ture is only 0.15%. This low absorption level helps account
for the resin’s excellent dimensional stability and stain
resistance. It also indicates that the resin, itself tasteless
and odorless, is unlikely to pick up food odors. PC is
available in grades that meet FDA and USDA regulations
and is recognized as safe for food-contact applications.
PACKAGING APPLICATIONS
Refillable Bottles
Polycarbonate is the material of choice for use in reusable
bottles, particularly 5-gal (19-L) water bottles, which
represent the resin’s chief packaging application. These
bottles take advantage of polycarbonate’s toughness (to
resist breakage) and clarity (to see the contents). The fact
that PC is much lighter than glass provides fuel savings as
well as productivity improvements, since several bottles
can be carried at once. Systems have been developed to
wash polycarbonate and provide clean bottles for reuse
with minimum impact on trippage. Lexan (now sold under
SABIC Innovative Plastics) can be washed and filled 50
times while maintaining excellent taste, neutrality, and a
transparent glasslike appearance (3).
POLYCARBONATE 973

Medical-Device Packaging
Polycarbonate meets many requirements of medical-
device packaging (see Healthcare packaging). It is clear
and tough, and it can be sterilized by commercial sterili-
zation techniques: ethylene oxide (ETO), radiation,
and autoclave sterilization (see Radiation, effects of).
The development of coextrusion technology has afforded
opportunities in all sterilization systems. Special grades
have been developed to maintain clarity and resistance to
yellowing upon gamma radiation (2). In thin films,
PC can be coextruded with polyolefin heat-seal layers
(see Coextrusions for flexible packaging; Coextrusions
for semirigid packaging) to produce a cost-effective
alternative to laminations based on oriented films (see
Films, plastic). Because it is amorphous, heat sealing
does not shrink or embrittle the film (see Sealing, heat).
This virtually eliminates puckering, which can lead to
hairline cracks and shattering upon opening. A soft blister
package with good puncture resistance can be produced
by thermoforming heavier-gauge film. For increased
stability in the autoclave, a polycarbonate copolymer,
poly(phthalate carbonate), can be incorporated into the
structure. Properties of monolayer PC films are listed in
Table 3.
Food Packaging
Coextrusions of polycarbonate are being evaluated for use
in several segments of the food-packaging market. The
snack-food industry, along with other users of flexible-
packaging materials, is discovering ways to use PC in
coextrusions to replace laminated films in a portion of a
structure or to replace the entire structure (see Multilayer
flexible packaging). It is polycarbonate’s toughness with-
out orientation that makes it a good candidate for coex-
trusion. PC is used for food containers and baby bottles (4).
For frozen foods, the resin’s low-temperature impact
strength adds durability to dual-‘‘ovenable’’ trays. In
coextrusions, it can add toughness to crystallized polye-
ster or polyetherimide trays. In cases where the wall
thickness is determined by impact-strength requirements,
gauge reductions up to 33% are possible.
In high-barrier multilayer containers, polycarbonate
offers the rare combination of dimensional stability at
retort and hot-fill temperatures, along with crystal clarity
(see Retortable flexible and semirigid packages; Multi-
layer plastic bottles). To produce a 1% distortion in PC
requires 3000 psi (20.7 MPa) stress. A similar distortion in
polypropylene occurs at less than 500 psi (3.4 MPa) stress.
Polycarbonate is about three times as expensive, but
dimensional stability can be translated into value by
making lighter containers with thinner walls by faster
and more reliable closing a hot-fill temperatures, higher
Table 2. Tensile Strength and Modulus of Polycarbonate
over a Temperature Range
Temperature, 1F(1C) Tensile Strength,
psi (MPa)
Flexural Modulus,
psi (MPa)
73 (23) 10,000 (68.9) 320,000 (2,206)
212 (100) 5,800 (40) 233,600 (1,610)
270 (132) 4,000 (27.6) 211,200 (1,456)
Table 1. Typical Property Values for Polycarbonate
a
Melt Flow Indexes
Property
ASTM Test
Method 22 16 10 6 PC Copolymer
Specific gravity D792 1.20 1.20 1.20 1.20 1.20
Light transmittance, 0.125 in.
(3.2 mm), %
D1003 89 89 89 89 85
Haze, 0.125 in. (3.2 mm), % D1003 1 1 1 1 1–2
Deflection temperature at 264
psi (1.8 MPa) 1F(1C)
D648 260 (127) 265 (129) 270 (132) 270 (132) 325 (163)
Flammability rating,
b
UL 94, at
0.060 in. (1.5 mm)
V-2 V-2 V-2 V-2 HB
Tensile strength, yield, psi (MPa) D638 9,000 (62) 9,000 (62) 9,000 (62) 9,000 (62) 9,500 (65.5)
Tensile strength, ultimate,
psi (MPa)
D638 9,500 (65.5) 10,000 (68.9) 10,000 (68.9) 10,500 (72.4) 11,300 (77.9)
Elongation, rupture, % D638 120 125 130 135 78
Flexural strength, psi (MPa) D790 13,500 (93.1) 14,000 (96.5) 14,000 (96.5) 14,200 (97.9) 14,100 (97.2)
Flexural modulus, psi (MPa) D790 335,000
(2,310)
340,000
(2,340)
340,000
(2,340)
340,000
(2,340)
338,000
(2,330)
Izod impact strength, notched,
1
8
in: (3.2 mm) thick, ft lbf/in.
(kJ/m)
D256 12 (0.64) 13 (0.69) 15 (0.80) 17 (0.94) 10 (0.53)
Tensile impact, ft lbf/in.
2
(J/cm
2
) D1822 180 (37.8) 225 (47.3) 275 (57.8) 300 (63.0) 275 (57.8)
a
Properties shown are average values that can be expected from typical manufacturing lots and are not intended for specification purposes. These values are
for natural color only. Addition of pigments and other additives may alter some of the properties.
b
This rating is not intended to reflect hazards of this or any other material under actual fire conditions.
974 POLYCARBONATE

retort temperatures with less critical overpressure con-
trol, and enhanced container-design flexibility.
An unexpected benefit from the use of PC in retort
applications is an increase in the effectiveness of the
barrier material EVOH (see Ethylene–vinyl alcohol). In
a retort, moisture is driven into all layers of a plastic
container. When the outside skin layer is polypropylene,
the polypropylene prevents moisture from entering the
structure, but it also prevents moisture trapped in the
EVOH layer from escaping. Because polycarbonate has
poorer moisture-barrier properties than polypropylene, it
allows much more rapid drying of the EVOH layer and
longer shelf life.
BIBLIOGRAPHY
J. M. Mihalich and L. E. Baccaro, ‘‘Polycarbonate’’ in The Wiley
Encyclopedia of Packaging Technology, 1st edition, John
Wiley & Sons, New York, pp. 510–511; in EPT, 2nd edition,
pp. 740–741.
Cited Publications
1. K. L. Ring, H. Janshekar, and G. Toki, ‘‘Polycarbonate Resins’’
in Chemical Economics Handbook, SRI Consulting, Menlo
Park, CA, 2005.
2. D. J. Brunelle, ‘‘Polycarbonates’’ in Kirk–Othmer Encyclopedia
of Chemical Technology, Wiley, Hoboken, NJ, 2006.
3. ‘‘Lexan’’ www.geplastics.com, accessed April 2008.
4. O. G. Piringer and A. L. Baner, Plastic Packaging, 2nd edition,
Wiley-VCH, Weinheim, 2008.
POLYESTERS, THERMOPLASTIC
Updated by Staff
INTRODUCTION
Poly(ethylene terephthalate) (PET) was first developed
by a British company, Calico Printers, in 1941 for use
in synthetic fibers. The patent rights were then acquired
by DuPont and Imperial Chemical Industries (ICI),
which in turn sold regional rights to many other compa-
nies. Polyester fibers have since made a considerable
impact on the textile industry. The second principal
application of PET was film. In 1966, PET became avail-
able for the manufacture of injection-molded and extruded
parts.
The amazing growth of PET in beverage packaging
began in the early 1970s with the technical development
of biaxially oriented PET bottles (see Blow molding) and
with the introduction of the first 2-L PET beverage bottle
in 1976. The total world market for PET botttle resin is
still growing at a rapid rate. The total devoted to carbo-
nated soft drink or PET bottles is very large and the
United States is the largest consumer, with Europe next.
China is now advancing with a potential of 25–30 billion
PET beer bottles per annum. The annual output of all
plastic packaging materials including PET bottle resins
was 9.5 million tons in 2005 and is expected to reach 13.7
Table 3. Polycarbonate Film Properties, 1 mil (25.4 lm)
Property ASTM Test Value
Specific gravity D792 1.20
Yield, in.
2
/(lb mil) (m
2
/(kg mm) 23,100 (1,294)
Haze, % D1003 0.5
Optical clarity D1746 86–88
Tensile strength, psi (MPa) MD 10,735 (74)
XD 10,009 (69)
Elongation, % D882 MD 91
XD 92
Secant modulus, psi (MPa) MD 185,000 (1,275)
XD 196,000 (1,351)
Tear strength, gf/mil (N/mm)
Initial
a
D1004 454 (175)
Propagating
b
D1922 16 (6.32)
Tensile impact, S type, ft lbf/in.
2
(J/cm
2
) D1822 225–300 (47.3–63.0)
Bursting strength, 1 mil, psi (kPa) D774 27.4 (189)
Water absorption at 24 h and 731F (231C), % D570 0.15
Folding endurance
c
, cycles D2176 11,000
Heat-seal temperature at 40 psi (276 kPa) for 3 s, 1F(1C) 400–420 (204–216)
Oxygen permeability at 771F (231C) and 0% rh, cm
3
mil/
(100 in.
2
day atm) [cm
3
mm/(m
2
day kPa)]
D1434 240 (933)
Water-vapor transmission rate at 1001F (381C) and 90% rh,
g mil/(100 in.
2
day) [g mm/m
2
day)]
E96-66 6.5 (2.6)
Coefficient of friction
Static D1894 0.570
Kinetic 0.542
a
Graves.
b
Elmendorf.
c
Double folds.
POLYESTERS, THERMOPLASTIC 975
million tons in 2008. PET bottle demand seems to be
assured because of its balance of properties and the ability
to be recycled. There is a strong demand for packaging
mineral waters in Europe and there is a large hot-fill
market in Japan. A market survey of the United States
beverage container market for 2002–2007 shows that the
market for plastic bottles (largely PET) is still growing,
while glass and metal metal cans are static or in slight
decline (see Table 1) (1).
PET has the following formula:
HOCH
2
CH
2
OC
O
O
OH
COCH
2
CH
2
n
where n=100–200.
MANUFACTURE OF PET
Raw Materials
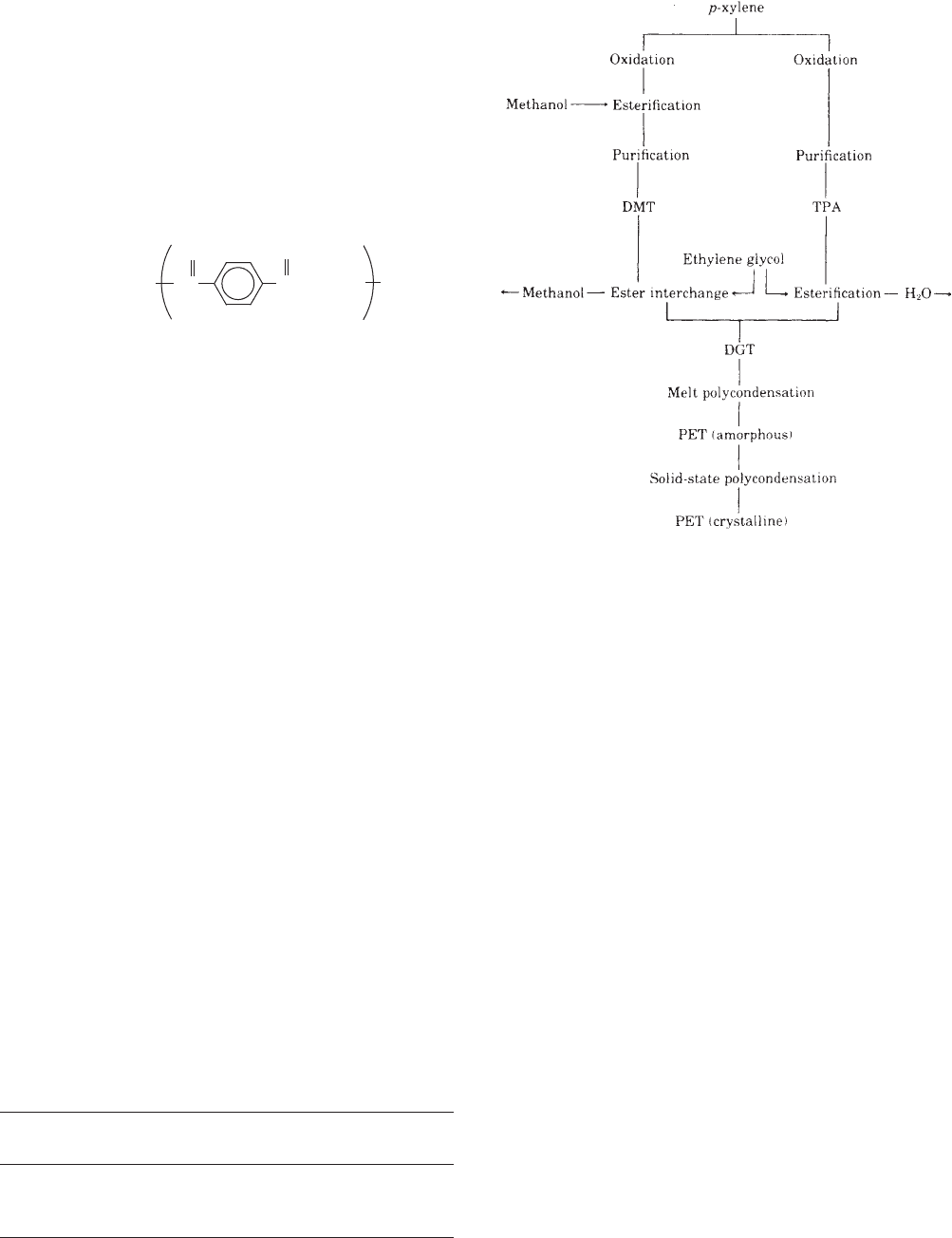
The raw materials for PET (see Figure 1) are derived from
crude oil. Para-xylene, one of the two starting materials, is
part of the naphtha feedstock that used to be fully avail-
able for chemicals because it was a byproduct of limited
value to the oil refiner. Now it has become the source of
additives that replace lead in unleaded gasoline in many
countries. Of the mixed xylenes, because they come from
the reformer, only p-xylene is suitable for building
straight polymer chains, and the straight configuration
is necessary to give the polymer its fiber- and film-forming
(or bottle-wall-forming) characteristics and ultimately its
high tensile strength. The other raw material for polyester
is ethylene, which is contained in the crude oil’s gas
fraction. It is converted to ethylene glycol by oxidation
and hydrolysis. All of these raw materials are now derived
from oil, but it is technically feasible to produce them from
coal-tar distillation.
Intermediate Products
Two different routes are used to manufacture PET: One
route is by way of dimethyl terephthalate (DMT), and the
other is by way of terephthalic acid (TPA). Both are dibasic
acids. Figure 1 shows both routes. The plants for inter-
mediate and end products are shown together, but they
are normally physically separated. The process of making
DMT is relatively simple: One end of p-xylene is first
oxidized with air and esterified with methanol to yield a
half-ester that is subsequently converted at the other end.
The resulting DMT is purified by distillation and repeated
crystallization to remove isomers and other impurities.
The TPA route is similar. TPA can be produced by oxida-
tion of p -xylene in solution and purified by solvent
extraction.
Melt Polycondensation
Both batch- and continuous-polycondensation processes
are used. The continuous process inherently allows better
product uniformity; the batch process is preferred for
small quantities of specialty resins. On the DMT route,
DMT and ethylene glycol (EG) are continuously metered
into the ester interchanger, where the methyl end groups
of the DMT are replaced by ethyl end groups to form
diethylene glycol terephthalate (DGT), the monomer of
PET. In this step, EG is consumed while methanol is
evaporated and collected to be returned to the DMT plant.
On the other route, TPA is esterified with EG to DGT, and
water is removed as a byproduct. Subsequent polyconden-
sation is the same for both routes. In EG takeoff, excess
EG is removed and sent to the distillation plant for
recovery. Polycondensation takes place in vacuum reac-
tors designed to evaporate EG, the condensation bypro-
duct, thereby shifting the equilibrium toward long
polymer chains. The final step includes extrusion of the
melt as strands or ribbon, quenching in water, and cutting
to the desired chip size.
Table 1. U.S. Beverage Container Market Survey a
Material
1997,
Billion Units
2002,
Billion Units
2007,
Billion Units
Plastic 35 52 70
Metal 99 100 102
Glass 28 26 30
Source: Reference 2.
Figure 1. Manufacture of PET.
976 POLYESTERS, THERMOPLASTIC
Solid-State Polycondensation
Melt polycondensation produces amorphous PET as used
in most fiber and film applications. Unfortunately, this
product is not suitable for the injection molding of food
containers because the inherently high acetaldehyde level
would affect the taste of some foods. A final solid-phase
polymerization before stretch molding must be performed
to remove the acetaldehyde (1). There are other impurities
that might promote degradation during the injection-
molding process; and amorphous resin tends to fuse and
form lumps in the drying hopper. The polymer must be
upgraded by the solid-state polycondensation process.
Various agitation devices and polymer chip treatments
have been described to prevent sticking (1). The chips are
crystallized to avoid later sticking and then dried to
reduce hydrolysis at high processing temperatures. So-
lid-state polycondensation takes place in the reactor,
where the chips are subjected to high temperatures under
vacuum (batch process) or in a nitrogen or dry-air stream
(continuous process). The product’s intrinsic viscosity (IV)
is normally between 0.70 and 1.0. (Intrinsic viscosity is a
method for the characterization of the average length of
the molecule chains in PET.) High-viscosity resin is rela-
tively expensive because its production is lengthy. High-
viscosity PET (i.e., having longer molecule chains), offers
better mechanical properties than the average-viscosity
resin. These properties compensate for certain deficiencies
of molded articles; for example, excessive volume expan-
sion of beverage bottles can be limited by higher-viscosity
resins that creep less under load. The final step is cooling,
since the resin should not be exposed to moist air while it
is hot.
HOMOPOLYMERS AND COPOLYMERS
PET is a homopolymer made from one part dibasic acid
(i.e., TPA or DMT), and one part EG. A copolyester
(copolymer) is made from more than one dibasic acid
and/or glycol. Copolymers remove processing limitations
and provide increased physical properties at elevated
temperatures. In addition to DMT or TPA, isophthalic
acid (IPA) can be used as a comonomer to reduce the
rate and degree of crystallization to an extent that de-
pends on its dosage. This broadens the processing para-
meters of food-container manufacturing machines.
Glycols offer several opportunities for modification. Dur-
ing polycondensation, EG reacts with itself to some extent
to form diethylene glycol (DEG). Higher amounts of DEG
affect many polymer properties. There are other glycols
available as partial substitutes for EG (e.g., neopentyl
glycol, cyclohexane dimethanol). All these modifications
lead to desired polymer property changes (i.e., reduction
of the crystallization rate, melting point, etc.). Cyclohexane
dimethanol can react with a mixture of terephthalic and
isophthalic acids in order to increase the melt strength of
the polymer for extrusion processes (3).
On the other hand, some injection-molding (see Injec-
tion molding) and thermoforming (see Thermoforming)
applications require accelerated crystallization rates in
order to set up crystallization in the article, which prevents
physical deformation at elevated temperatures. This objec-
tive can be achieved by nucleation, which involves the
addition of other ingredients to the polymer. Inert, non-
soluble substances (e.g., mica, talc), organic substances
(e.g., aromatic alcohols), and certain polymers (e.g., PP,
PE) can be used as nucleation ingredients to increase
crystallization rates.
The use of PET and optional added substances for food-
packaging applications is governed by FDA Regulation
No. 177-1630 of March 16, 1977 (revised April 1, 2007).
Homopolymers and copolymers and additives must con-
form to this regulation.
PACKAGING APPLICATIONS
Homopolymers
By strict definition, most PET resins are modified homo-
polymers. These homopolymers are used to manufacture
containers (i.e., bottles), by injection blow molding or
injection-stretch blow molding (see Blow molding). Con-
sumption of bottles for carbonated soft drinks runs into
the billions of units in the United States alone. A main
factor for bottle use is the permeability of the bottle wall to
oxygen and carbon dioxide. Carbon dioxide pressure must
remain during storage and oxygen from the air must not
diffuse in. PET is semipermeable. Some early PET bottles
had coatings of impermeable polymers, but the process
involves extra steps and the bottles are not readily
recyclable (1). The most marketable is plain PET with
reduced permeability. PET bottles are also used for liquor,
wine, food, toiletries, and pharmaceuticals, as well as for
beer in some countries. Homopolymers cannot be pro-
cessed by extrusion blow molding because of insufficient
melt strength. For bottles containing beer and wine, the
demands are far more severe than for carbonated soft
drinks. Here it is important to keep oxygen out of the
bottle. Oxygen permeability is a problem with PET. For
high-barrier one-trip bottles, people are looking to coated
or multilayer bottles to reduce oxygen permeability. There
is much activity in Europe at the moment for plastic beer
bottles. Ten percent of beer produced in Europe comes in
PET bottles. In the United States, Anheuser Busch and
Coors have started supplying their beers in 16-oz multi-
layer bottles (1).
Biaxially oriented PET film (see Film, oriented polye-
ster) is usually manufactured by polycondensation and
subsequent continuous casting of the film plus direct
biaxial orientation; that is, molecule chains of the resin
become biaxially oriented. Nonoriented PET film and
sheet are manufactured by melting PET resin in an
extruder and casting the melt through a flat die with
subsequent calendering.
PET is also used for ‘‘ovenable’’ trays for frozen food and
prepared meals. These trays are thermoformed from cast
PET film and crystallized. Crystallization heat-sets the
article
to prevent deformation
during cooking and serving.
The main advantages of PET for this application include
suitability for both conventional and microwave ovens,
POLYESTERS, THERMOPLASTIC 977
