Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.


structure due to variations in polymer blending or adhe-
sion of various layers in a laminate can often be extremely
complex, time-consuming, or impractical for commerciali-
zation. The development of a successful perforation pro-
tocol for these films is usually driven by knowledge of the
interaction of laser energy with film structural compo-
nents, and then whether or not the perforated product can
be marketed as a packaging solution at a cost that would
suit the utilizing industry.
BIBLIOGRAPHY
1. S. Ben-Yehoshua, S. Fishman, S. Jayanty, N. Mir, and R. M.
Beaudry, ‘‘Modified Atmosphere Packaging, Controlled Atmo-
sphere Packaging, and Seal Packaging’’ in S. Ben-Yeshoshua,
ed., Post Harvest Physiology, Sheffield Academic Press, Shef-
field, UK, 2005, pp. 61–112.
2. Y. Mizutani, Ind. Eng. Chem. Res. 32, 221–227 (1989).
3. G. Peiser and T. V. Suslow, Perish. Handl. Q. 95, 17–19 (1998).
FILM, PLASTIC
ALLEN G. KIRK
E. I. Du Pont de Nemours, Inc.,
Wilmington, Delaware
Films are continuous membranes that can separate one
area from another. These membranes can vary in thick-
ness, ranging from less than that for rigid containers, to
sheeting, to film, and even to thin coatings. Usually films
are considered self-supporting and less than B10 mils
(250 mm) in thickness. No definition is given for a mini-
mum thickness, which can vary depending on the material
of construction of the film; however, thicknesses of o1 mil
(25 mm) are common.
An important feature of most films discussed in this
article is heat sealability, which refers to the thermoplastic
property of the film, or coating on the film, which allows it
to be fashioned into pouches, bags, and overwrapped
packages by virtue of its ability to make a hermetic seal
to itself. Heat sealing is accomplished by heating up the
film areas then applying the hot areas to each other under
pressure. Sometimes these operations occur simulta-
neously. During heat sealing, the polymer molecules be-
come entangled; the better the intermingling, the stronger
the seal. The time allowed for heat sealing in a typical
high-speed food-packaging machine is less than 1 s, during
which time heat-seal strengths of very high values can be
achieved, i.e., several pounds per inch of seal width.
FILM USES
Because of their ability to keep two areas (or volumes)
separated from each other, films have come to have a
multitude of commercial uses over the years, particularly
in the area of food packaging. Films do provide protection
to the foodstuffs being displayed, and in doing so they can
provide a multitude of other functional attributes. They
can function as barriers for gases, vaporous flavor compo-
nents, or moisture from escaping from the food, thus
preserving its freshness. They can also protect the food-
stuff from attack by undesirable outside agents such as air
(oxygen), moisture, or sunlight, thus ensuring that the
food does not become rancid or soggy. The film material
can serve as a partial gas barrier, thus allowing some
gases to escape but not others; the choice of packaging film
Stand 10.05.2007 CO
2
Systems – The Perforation People
Focus on Fine Solutions
-1 -
PERFOLAS
®
Details - Perforation Area
Individual adjustment
of focus
16 beams with
independent shutters
Individual adjustment
of head position
High-power exhaust system with filter
Laser safety class 1 protection housing
Individual selection of the number of active
perforation heads
Figure 1. Laser system for manufacture of perforated film.
488 FILM, PLASTIC
to protect fresh vegetables and fruit can be very selective,
especially if the vegetables and fruit are already cut or
sliced for customer convenience. For fresh produce, the
film must allow CO
2
to easily leave the package while
allowing only low levels of air (oxygen) to enter the
package for longer shelf life.
Films also provide a billboard for information about the
contained product, directions on how to open the package,
recommended dose levels, warnings about toxic contents,
nutritional information, and universal product code (UPC)
symbols for pricing and inventory control. This informa-
tion can be displayed in a multicolor and aesthetically
pleasing way on the packaging film so that it ‘‘sells’’ itself
and acts as an impulse-buying aid. In addition, the
transparency and high clarity of the film can provide a
window in the package to allow the consumer to judge
desirability of the product before buying or to judge
remaining quantity in the package after purchase and as
it sits in the consumer’s home.
Film applications can be large, as in the case for some
construction uses. Films or sheeting materials are used to
wrap houses and buildings under construction, thus ser-
ving as moisture and wind barriers in the finished con-
struction. Film products can be used in highway
construction, as leakage barriers in municipal water
reservoirs and as protective liners for municipal solid-
waste landfill sites. Film applications can be small, such
as the push-through blister pack for individually pack-
aged medical pills. Packaging film uses can be categorized
as follows.
Overwrap. This type of packaging usually involves
unwinding a roll of flat film into the packaging machine,
where it is folded around the object, the film sealed to
itself, and the finished package removed by cutting the
film. Typical products packaged by this method include
small packs of snack crackers and cookies. The machine
is commonly called a ‘‘horizontal make-and-fill machine.’’
Products such a potato chips, corn chips, and bags of candy
are packaged in vertical form/fill/seal machines in which a
roll of flat film feeds into the machine over a forming collar
where the film is formed into a tube. This tube is sealed
along the back with a vertical heat seal, and then the
bottom cross seal is made. The open-top tube is filled with
the product and indexed down, and the top cross seal is
made simultaneously as the bottom seal is formed for the
next pouch to be filled and as the filled pouch is removed
from the packaging machine. Coated cellophane, coated
oriented polypropylene (OPP) film, and laminated films
typically are used for this packaging application.
Skin Packaging. As the term implies, this type of packa-
ging involves forming a skin of film over the object being
packaged and sealing the skin film tightly to a heat-seal-
coated display card. This is accomplished by placing the
object to be packaged, usually a hardware item, onto the
coated display card, then draping a hot sheet of film over
the entire assembly, and applying vacuum to the backside
of the permeable display card. The vacuum pulls the
cooling hot film down over the object and applies pressure
to assist in making the seal to the display card. Typically
ionomer, low-density polyethylene (LDPE), or polyvinyl
chloride (PVC) resin films are used for this packaging
applications, and the board coating is LDPE or ethylene-
vinyl acetate (EVA) (See the Skin packaging article).
Blister Packaging. Preformed blisters are filled with the
object, the coated paperboard is placed over the filled
blister, and the assembly is heat sealed. Small hardware
items and small notions are commonly packaged in this
manner.
Shrink Packaging. This type of packaging refers to the
use of a film manufactured in such a way that when it is
heated it will contract in both directions, reducing its
surface area. When this type of film is wrapped around
an object and sealed around its edges, then sent through a
shrink tunnel where large volumes of heated air wash
over it, the film will react to the heat and contract down to
the object, thus making an attractive skin-tight package.
A vent hole must be cut into the film to allow the trapped
air to escape from the package as is undergoes film
shrinkage in the hot-air tunnel. Shrink-film resins are
polyethylenes, polypropylenes, PVC, polyesters, and some
coextruded structures. Articles packaged in this manner
are numerous and can include frozen poultry, pizzas,
stationery, toys, cassettes, and compact disks.
Stretch Packaging. This packaging technique is useful
for bundling objects together for convenience in shipping
or handling. Resins used in this application include linear
LDPE (LLDPE), LDPE, EVA, PVC, and some copolymers
of polypropylene. Typically, when stretch wrap is used for
pallet bundling, the tacky film is wound around the pallet
many times to ensure tightness, the film cut, and the film
end stuck down on the wrapped pallet. There is no need to
heat seal or use mechanical fasteners with this packaging
technique.
PLASTIC-FILM RESINS
It would not be possible to cover all resins that have been
fabricated into film in this article. What are presented
here are typical resins used over the years as packaging
films.
Food packaging has successfully employed films for
many years, starting in the 1920s in the United States
with the introduction of cellophane film, which was used
initially to package loaves of bread that had been pre-
viously overwrapped in paper. Because it was transpar-
ent, cellophane allowed the bread to be viewed by the
customer before purchase. Uncoated cellophane is com-
posed
of regenerated cellulose
, a softening agent, and
water as a plasticizer. Although it is not moistureproof,
it is impermeable to gases when the film is kept dry.
Additional slip, antistatic, and antifog properties can be
provided by special formulations applied as coatings to the
base sheet. Cellophane is not a thermoplastic and does not
have a melting point, heat sealability is possible only by
applying a heat-seal coating such as nitrocellusose (NC) or
polyvinylidene chloride (PVDC) to the base sheet.
FILM, PLASTIC 489
By the early 1950s, LDPE resin was developed and
began serving as the first major human-made (synthetic)
thermoplastic packaging resin. Because this resin has a
melting point and is thermoplastic, the packaging indus-
try developed thermal extrusion equipment to extrude
thin films of this new resin for packaging. Polypropylene
(PP), which is another popular packaging resin, was first
produced during the 1950s. However, it did not develop
commercially until the 1970s, when new high-efficiency
catalysts were discovered for its production. PP competed
effectively for many of the cellophane packaging applica-
tions. Although cellophane had attractive properties of
clarity, stiffness, gas barrier, and heat resistance, it also
had disadvantages of limited shelf life because of the loss
of volatile plasticizer. This loss of plasticizer caused the
cellophane to embrittle to the point where the packaged
product was no longer protected. PP has the inherent
attributes of clarity, toughness, thermoplasticity, and heat
sealability after being coated by PVDC. PP also has the
advantage of having a density lower than that of cello-
phane; thus, more square inches of packaging film are
possible from a pound of PP than from a pound of
cellophane film—a definite economic advantage. As PP is
a thermoplastic resin, it did not have the thermal resis-
tance to provide fail-safe high-temperature sealing protec-
tion against substrate burn through on the packaging
machine. Additionally, the lower modulus PP could not
be fed easily through the push-feed packaging equipment
developed years earlier for the stiffer cellophane. Feeding
problems for PP were exacerbated by its greater tendency
to generate and hold static charge, which caused clinging
and feeding jams in the packaging machine. These serious
packaging problems were eventually eliminated by ma-
chinery redesign and by improvements in PP base-sheet
and coating technology. Plastic-film resins can be categor-
ized by the chemical process used to form the molecules in
the polymer resin backbone.
Addition Polymerization (Homopolymers). Polymers in
this category include LDPE, HDPE, PP, polybutylene
(PB), and polystyrene. During the polymerization or
building of the polymer molecules, the individual mono-
mer units (ethylene, propylene, and butylene, respec-
tively) are chemically connected together in the pressure
reactor in the presence of high temperature and an
appropriate catalyst. By control of the residence time in
the reactor, the number of these monomer connections can
be varied. Long reactor time, longer polymer chain length,
and higher molecular weight result. By controlling mole-
cular weight, the manufacturer can control polymer resin
properties such as viscosity, which, in turn, can control
final film properties such as toughness. These resins,
called aliphatic resins, are prepared from monomers that
consist of only carbon and hydrogen atoms. The character-
istic chemical structure in the polymer backbone is a
carbon-to-carbon bond (—CH
2
—CH
2
—) that links the
individual monomer units together. Aliphatic ethylene
and propylene homopolymer resins are nonpolar, have
good clarity, and have relatively low melting points that
range from B1051C for LDPE to 1251C for high-density
polyethylene (HDPE), and 1651C for PP. Polystyrene does
not have a melting point but does exhibit a Vicat softening
point at T r 1061C. Because their monomers are derived
relatively easily from petroleum feedstocks and the corre-
sponding resins are used in large-volume packaging ap-
plications, resin cost/pound is not high, particularly for
LDPE, HDPE, and PP. LDPE resins can be found, for
example, as premade flexible bags and pouches and other
large-volume packaging applications. HDPE, which is
stiffer and tougher, is used as grocery bags and multiwall
bags and bag liners. PP, as described above, has replaced
cellophane film in many food-packaging applications.
Some of these applications include overwrap for cigarette
packs, snack-food packs, and pouches and bags for potato
and corn chips. PB has found some applications as a meat-
packaging film and as an additive for hot-melt adhesives.
Addition Polymerization (Copolymers). Polymers pre-
pared from combinations of two or more monomers can
give rise to plastic resins with a wide range of properties
not possible from homopolymers. Resins of ethylene/propy-
lene (E/P), ethylene/butylene (E/B), ethylene/hexene
(E/H), and ethylene/octene (E/O) are possible combinations.
The last three mentioned resins are gaining popularity as
new aliphatic polymers with a much broader range of
thermal and mechanical properties than previously possi-
ble. These resins, which are designated metallocene poly-
ethylenes (mPEs) are prepared using the relatively new
constrained geometry catalysts that allow the tacticity and
branching of the polymer molecule to be better controlled,
which yields polymers with narrower molecular distribu-
tions and lower densities. mPE resins are aliphatic copoly-
mers somewhat endowed with the properties of the higher
polarity copolymers described below.
Addition copolymers can also be prepared with ethy-
lene and a polar second monomer. These polar monomers
can include acrylic acid (AA), methacrylic acid (MAA),
ethyl acrylate (EA), and vinyl acetate (VA). Because these
monomers contain oxygen atoms in addition to carbon and
hydrogen and because oxygen is a heavier atom and rich
in electrons, the polymerization of these polar monomers
results in a copolymer resin with higher polarity than
carbon- or hydrogen-containing aliphatic resins. Resins of
E/AA, E/MAA, E/EA, and E/VA are characterized not by
an unusual chemical linkage between the repeated mono-
mer units but rather by the polar nature of the copolymer
in the polymer chain with the ethylene monomer. The
E/AA and E/MAA resins, which are also called acid
copolymer resins, have good clarity, low haze, and lower
melt and sealing temperatures, and they can adhere
strongly to polar substrates such as paper, foil, and some
highly polar film resins. Thus, they have found use more
in specialty packaging applications and in association
with multilayer structures for meat, cheese, snack foods,
and medical items. E/VA resins typically contain between
5% and 40% VA comonomer, with VA contents above about
25% used largely in hot-melt adhesive applications.
Packaging-film applications employ resins with lower VA
contents ranging from 5% to about 18%. These composi-
tions are not too sticky, and films of these compositions can
be handled on typical packaging machines; however, often
slip and antiblock additives must be incorporated into the
490 FILM, PLASTIC
resin to ensure the proper slip level. Because of the low
crystallinity, toughness properties at low temperatures,
and low melting points, E/VA resins are used as a poultry
and meat wrap, bag-in-box for liquid packaging, stretch
film, and as ice bags.
Because E/AA and E/MAA resins contain free-acid
groups, they can be partially neutralized to form a class
of resins called ionomers. The ionomers serve some of the
same market applications and have excellent hot-tack
sealability, the ability to seal through contamination,
and grease/oil resistance. These polar copolymer resins
find use in packaging meat, cheese, breakfast cereals, and
many other wet and dry products. They also are used as
skin-packaging films for hardware products.
Condensation Polymerization. Some chemically reactive
polar monomers can react with each other or with a second
monomer to form a polymer chain and a small volatile
second molecule. Many times that second molecule is
water; thus, water seems to ‘‘condense’’ from the reaction.
Removal of this second molecule leaves the relatively
clean polymer behind. Resins made by this condensation
polymerization route include polyamides or nylon, and
polyester. Nylon resins are made from diamines and
dibasic acids and are characterized by the amide group
(—CONH—) in the polymer backbone. Nylon resins are
identified by the number of carbon atoms in the mono-
mers. Thus, homopolymer nylon 6 can be prepared from a
monomer with six carbons in a straight line with reactive
end groups on each end. Also nylon 6/6, 6/10, and 6/12 can
be prepared from two monomers with the differing num-
ber of carbon atoms. Polyamides are regarded as semi-
crystalline resins with high melting points (175–2751C).
As packaging films, they offer toughness, chemical resis-
tance, resistance to oils and greases, and moderate gas
barrier. Food products packaged in polyamide films in-
clude meats and cheeses.
Polyesters similarly are formed by reaction between an
aromatic diacid such as terephthalic acid or the dimethyl
ester of terephthalic acid and a polyalcohol such as
ethylene glycol. These resins are characterized by the
ester group (—CO—O—) linking the monomer units in
the polymer backbone. Thus, these resins contain a stiff
aromatic molecule within the polymer backbone as well as
a flexible aliphatic portion. This stiff resin can be extruded
into oriented high-tensile-strength film, metallized, and
heat-seal coated to yield a packaging film in a thin gauge.
In combination with other sealant coatings or lamina-
tions, polyester films are used as boil-in-bag pouches and
for processed-meat packaging.
Chlorinated Vinyl Addition Polymerization. Polyvinyl
chloride resin for film applications is prepared by suspen-
sion polymerization and employs the vinyl chloride
(CH
2
QCHCl) monomer. A homopolymer resin is polymer-
ized from this monomer by a free-radical process. After
purifying and removing excess water, this rigid resin must
be blended with softeners or plasticizers to reduce its
glass-transition temperature and render it a usable resin.
For a more detailed discussion on how to modify melt
temperature and glass-transition temperature in poly-
mers, refer to Nielsen (1). Other additives such as lubri-
cants and thermal stabilizers are blended into the resin as
well. Packaging applications for PVC film include fresh
meat, shrink film, and blister packs. For more details on
how packaging film choices are made for different food-
stuffs, refer to Jenkins (2).
MULTILAYER FILMS
Laminated film structures refer to multilayered structures
composed of many monofilm layers. By effecting adhesion
between the layers, more sophisticated film properties can
be obtained in a single film. Heat-sealable films can provide
the outside layers and can cover interior layers of non-
thermoplastic materials such as foil, metallized film, or
paper. Previously made films that contain light-blocking
levels of TiO
2
can be printed before being laminated with a
clear glossy, heat-sealable film, thus yielding an attractive
multilayered film that can be heat sealed, providing protec-
tion of the product from ultraviolet (UV) degradation, and
exhibiting striking printed panels. Because each monofilm
of the laminate must have been previously prepared in-
dividually as a self-supporting film, the final multilayered
laminates can end up thick and costly.
Coating of monofilms also provides a higher level of
property sophistication to monofilms. Frequently, solvent
coatings are applied to films to provide heat sealability or
an enhanced gas and moisture barrier to the film. Vacuum
metallization can be considered a form of coating. This
technique enhances light opacity and gas- and moisture-
barrier properties of the film. Individual coating thick-
nesses are usually less than 1 mil (25 m m); thus, coating a
monofilm does not substantially add to its thickness.
Coextrusion describes a process in which a multi-
layered film is prepared by adhering several individual
film layers within the body of the extrusion die. This
process can prepare as many as nine separate layers of
film within the same final film. Each resin may have its
own extruder feeding a melt stream into the complex feed
block attached to the die. Sometimes, one extruder will
feed melt into two or more separate flow streams by the
use of a stream divider within the feed block. This practice
is fairly common for the extruder supplying the tie layer
resin, which is the resin used to adhere two dissimilar
resin types in the final multilayered film. Many factors
must be considered when choosing the tie layer resin for a
coextruded structure (3). Obviously, the initial cost for
coextrusion equipment is high. However, this technique is
popular and can provide outstanding film property combi-
nations at a reasonable cost.
BASIC FILM–FORMING PROCESSES
Thermoplastic resins such as ethylene and propylene
homopolymers and copolymers, and the condensation
polymers described above, by definition have a melting
temperature above which they are not rigid solids but
FILM, PLASTIC 491

rather viscoelastic materials. As such, they are soft plastic
melts that can be formed into shapes convenient for
packaging and other commercial uses. Several typical
thermal processes are available for effecting this conver-
sion from the small pellet supplied in the resin bag, box,
hopper truck, or hopper railroad car to the final film
structure. The following descriptions describe the pre-
paration of monofilms; however, keep in mind that the
coextrusion process is also possible for all these extrusion
processes. Refer to Figure 4, which relates typical EVA
resin melt flow requirements to film manufacturing pro-
cess type.
Extrusion-Cast Film. In this process, the plastic resin
pellets are introduced into the feed hopper of the extruder,
where they are funneled down into the extruder barrel
and onto the rotating screw (see Figure 1). As the screw
rotates, it drives the pellets deeper along the hot barrel,
compressing and heating them and applying force or shear
so that the air spaces between the pellets are driven out
and the pellets are heated to the melt temperature or
softening temperature of the resin. The screw, which is
driven by a powerful electric motor, is typically composed
of three separate regions: (a) the compression zone, which
is closest to the feed hopper; (b) the transition zone; and
(c) the last region, which is called the metering or blending
zone. Each zone occupies about one third of the screw
length. The compression zone heats up the pellets driving
out the air, the transition zone describes the length of the
screw where the transformation from solid pellet to
melted resin takes place, and the metering zone allows
the melt pool to become homogenized so that a uniformly
mixed melt stream is presented to the heated die. The die
serves the purpose of fixing the dimensions of the final
shape of the polymer. Thus, most dies have a circular cross
section to the melt inlet, which then gradually transforms
to the thin and wide cross section of the film. As the flat,
thin sheet of polymer melt exits the die, it is quenched to a
lower temperature below its melting point, thus solidify-
ing it into the final and desired shape. Often the quench-
ing is accomplished with water-cooled metal drums or
rolls with a high-gloss chrome-plated and smooth finish
so that the quenched surface on the film is flat and
blemish-free. By controlling the takeoff speed of the
quench roll at a faster rate than what the melt exits the
die, the film thickness can be reduced and controlled. This
differential in speed also impacts some final film proper-
ties, such as toughness, stiffness, and film clarity or haze.
Basically, this stretching of the film melt as it is cooling
causes the polymer molecules to become better aligned or
oriented in the long direction of the film machine direction
(MD). This molecular alignment is responsible for en-
hance stiffness and toughness properties. The film is
then accumulated as rollstock on the windup turret of
the cast-film line. Film widths of r in 120 in. are possible
by this process. The length of the film is defined by how
much is wound on the roll, and usually as one film roll is
being completed, the windup turret can be indexed around
so that a new roll can be started without interrupting the
continuous resin flow through the extruder and die. The
rolls are then removed from the windup turret and slit to
the width desired by the customer. Property-enhancing
lubricants, stabilizers, or colorant additives can be added
to the resins pellets at the feed hopper. They will become
mixed with the melt stream by the extruder screw. These
additives can be added as powders to the feed hopper or
more likely will be added as a preblended concentrate in
pellet form.
Extrusion-Blown Film. The blown-film process (Figure 2)
is somewhat similar to the cast-film process in that a
plasticating screw extruder is employed to convert the
solid resin pellets into a uniform plastic melt. The blown-
film process employs a circular or annular die to form the
film, which is then stretched over and around a captured
bubble of air. The film is usually air quenched, collapsed as
a flat tube, and wound up on the windup stand. Some-
times, the bubble is slit in the vicinity of the nip rolls so
that two sheets of film may be fed down to the windup
stand. This bubble process can be oriented so that the film
extrudes upward, downward, or in the horizontal direc-
tion. The bubble may be long but typically is 20–30 ft long.
Bubble diameters may be 6 ft or more. The resin used in a
blown film process must have a high melt strength to
sustain the relatively high hoop stress and MD tension.
This process requires that the melt emerge from the die
then quickly flare out or inflate around the captured air
Figure 1. Cast-film line.
Figure 2. Blown-film line.
492 FILM, PLASTIC

bubble while it is being cooled. Thus, the polymer melt is
being cooled and biaxially oriented at the same time. The
ratio between the annular die diameter and the final
bubble diameter defines the ‘‘blowup ratio’’ (BUR) for the
film from this process. The die gap opening and BUR
define the final film thickness. Because the blown film
process requires such high polymer melt strengths, higher
molecular-weight polymers must be used and are reflected
in the typical melt index values of o1–5 dg/min for poly-
olefin resins (see Figure 3). Coextrusion-blown-film pro-
cesses are useful for preparing multilayer films. Care
must be taken to ensure that the tacky heat-seal layer
ends up as the outside of the bubble and not the bubble
inside, where it might stick to itself as the warm bubble is
collapsed at the nip. One advantage of the blown-film
process is that it yields a film tube and not a flat film with
thick edge beads. Thus blown-film products can be more
efficiently used because the thicker edge beads are not
present and do not need to be removed or recycled.
Extrusion Coating. As this name implies, the extrusion
coating process is employed to apply a thin layer of polymer
film onto an existing film structure (see Figure 4). Thus, it
is not usually used to make structural monoextruded or
coextruded films. It is often used to apply a thin sealant
layer to paper, foil, or plastic film. As Figure 4 shows, this
process resembles a cast-film process. Extrusion coating
requires that the exiting melt curtain quickly contact and
adhere to the substrate film. Thus, the proper compatibility
and adhesion between the substrate and the extruded resin
is important as is the need for the resin to have a relatively
high melt flow value (see Figure 3). Typical extrusion
coating resin melt flow values can range from about 8 dg/
min up to values of several hundred decigrams per minute.
The adhesion between the melt curtain and the substrate is
aided by the pressure on the nip roll. Because the high-
melt-flow polymer is hot and quickly contacts both to the
coated stock and the quench roll, the quench-roll surface is
usually machined or treated to reduce adhesion. Chill-roll
release additives can also be incorporated into the resin to
prevent sticking (see the Extrusion coating article).
BIBLIOGRAPHY
1. L. E. Nielsen, Mechanical Properties of Polymers, Reinhold,
New York, pp. 27, 31.
2. W. A. Jenkins, Packaging Foods with Plastics, Technomic
Publishing, Lancaster, PA, 1991.
3. B. A. Morris, TAPPI J. 75(8), 107–110 (1992).
General References
J. H. Briston, Plastic Films, 2nd edition, Godwin, London, 1983.
F. W. Billmeyer, Jr., Textbook of Polymer Science, Wiley, New York,
1965.
FILM, RIGID PVC
H. J. G. VAN BEEK
R. G. RYDER
Klo
¨
ckner-Pentaplast of
America, Inc., Gordonsville,
Virginia
Poly(vinyl chloride) (PVC) is extraordinarily adaptable to
custom compounding for desired performance. This diver-
sity of physical properties at relatively low cost has been
the driving force behind its popularity in the packaging
industry.
HOMOPOLYMER PVC RESIN
Commercial PVC resin is a dry free-flowing powder pro-
duced by the polymerization of vinyl chloride monomer
[see Poly(vinyl chloride].
Molecular Weight and Viscosity
The fundamental property of PVC is its molecular weight:
a measure of its means polymer chain length and a
parameter proportional to the resin’s viscosity. In general,
higher-molecular-weight (higher-viscosity) resins require
higher processing temperatures and yield film or sheet
with higher heat-distortion temperatures, impact resis-
tance, and stiffness than do resins of lower average
molecular weight. The processing equipment for generat-
ing film and sheet for the packaging industry demands
Figure 3. Resin meet flow vs. extrusion process.
Figure 4. Extrusion coating line.
FILM, RIGID PVC 493
low- to medium-viscosity PVC resins with relative viscos-
ities of 1.75–2.10, in contrast to extrusion pipe and con-
struction markets that typically use medium- to high-
molecular-weight resins.
Heat Stability
All PVC resins are subject to thermal degradation during
processing and must be compounded with appropriate
heat stabilizers to minimize discoloration. These stabili-
zers serve to scavenge free radicals that perpetuate de-
gradation, as well as hydrogen chloride, the principal
degradation product. The highly complex thermal degra-
dation mechanism proceeds by an ‘‘unzippering’’ process
whereby liable allylic chlorides act as reaction sites for the
liberation of hydrogen chloride and the formation of
conjugate-bonding systems. When the conjugation exceeds
six bonds in length, color development begins and, if
unchecked by ample and appropriate use of heat stabili-
zers, will progress from a very subtle yellow tint to amber
and, finally, to black. Commerical processing methods
occasionally generate ‘‘burned’’ material that has reached
these initial stages of degradation. It is important to
recognize that such thermal degradation is both time-
and temperature-dependent and that although stabilizers
retard the rate of degradation during processing, they do
not prevent it. The stabilizers also help protect the film or
sheet during subsequent processing (e.g., thermoforming)
and during the lifetime of the package itself.
COMPOUNDING FOR PROPERTIES
Among all polymers used in the packaging industry, PVC
is widely regarded as the most versatile and suitable for
custom compounding to deliver special properties (see
Additives, plastics). It may be compounded for high clarity
and sparkle or for maximum opacity and it accepts a full
range of custom colorants (see Colorants). Properly com-
pounded, PVC film and sheet are approved for food and
drug contact and are available with residual vinyl chloride
monomer (VCM) levels below 10 ppb. Examples of what
custom compounding can produce are shown in Table 1.
After identification of such desired properties for the
specific packaging application, the compounder selects a
suitable resin viscosity. If the film is to be approved
for food or drug contact, the resin’s VCM level must be
low enough before processing to ensure that the resulting
film will meet all customer requirements on residual
VCM. A heat stabilizer must then be selected. Tin mer-
captides are frequently chosen because of their high
efficiency, excellent early color, good light stability, and
excellent crystal clarity in the product (1). Some of these
stabilizers (octyl tins) are cleared for food and drug
contact. Uncleared options include lead stabilizers, which
are limited to opaque systems, and combinations of
barium, cadmium, and zinc. Although there are a few
calcium/zinc systems in limited use in food packaging,
the tin stabilizers dominate the packaging field. Octyl tins
are the principal systems used in food and pharmaceu-
tical packaging (2). Stabilizers are also available that
impart improved UV resistance. All of the stabilizers
mentioned above are used in rigid PVC at only very small
loadings.
In contrast, impact modifiers may constitute up to 15%
of the product’s weight. As a result, the proper selection
and loading of impact modifier is an important compound-
ing decision. Clear packaging films typically contain MBS
impact modifiers because of their superior clarity, heat
stability, and room-temperature efficiency. ABS modifiers
are good for opaque products, and chlorinated polyethy-
lene (CPE) and acrylics (see Acrylics) are often selected for
outdoor applications and/or low-temperature environ-
ments in opaque systems. Pigments may then be added
to provide custom color, and titanium dioxide is generally
used at levels as high as 15% to provide the desired level of
opacity. Fillers may be used for cost reduction in opaque
systems and, in many cases, to improve such physical
properties as impact strength, stiffness, and heat-distor-
tion temperature. Present in very low levels are a variety
of proprietary lubricants and processing aids that are
necessary to facilitate processing and to provide desirable
properties such as slip, denesting, and improved thermo-
formability for the film processor/packager. Flame retar-
dants, antioxidants, coupling agents, antistatic agents,
phosphite stabilizers, and a host of additional additives
may be included if necessary. Because of this tremendous
facility for custom compounding, and the variety of pro-
ducts that result from it, Table 2 must be considered only
as a general guide to typical rigid PVC properties.
FILM AND SHEET PRODUCTION METHODS
Extrusion (see Extrusion) and calendering are the princi-
pal methods of producing rigid PVC for the packaging
industry. Extrusion is used to produce very thin blown
films (see Film, flexible PVC) as well as heavy-gauge
sheeting nearly 1 in. (2.54 cm) thick produced by sheet-
die methods. Calendering requires a much greater capital
investment, but it offers much greater production rates,
superior gauge control (cross direction and machine direc-
tion) (75%), superior cosmetic quality including clarity,
and much wider versatility in accommodating gauge and
Table 1. Examples of Custom Compounding
Higher heat-distortion temperatures for hot-fill packaging
Ethylene oxide (ETO) sterilizable film without water-blush for
medical devices
Improved low-temperature impact resistance for drop tests of
shipping cartons
Improved UV resistance
Improved outdoor weatherability
Improved sealability (impulse heat, RF, ultrasound)
Denesting formulations for machine-fed blisters
Static-resistant formulations
Optimum performance in laminating to other materials (PVDC,
PE, etc.)
Formulations for vacuum metalizing
Improved printability
Absence of ‘‘white break’’ or crease whitening
494 FILM, RIGID PVC

width changes. Calendered film and sheet generally have
better dimensional stability, which provides thermoform-
ing consistency throughout a given lot. Rigid calendered
PVC is available in thickness of 2–45 mil (51–1143 mm)
with gloss, matte, or embossed surfaces, either in rolls or
in sheets up to about 60 in. (1.5 m) wide. Calendering is
the principal means of processing rigid PVC film for
packaging.
Calendering
In a modern rigid PVC calendering operation, compound-
ing is done by computer-controlled electronic scales
that supply precise amounts of each ingredient to a
high-intensity mixer designed to incorporate all liquids
into the resin particles and to secure uniform distribution
of all powdered ingredients. Blending is generally
done for a specific time period and to a specific tempera-
ture. The still-dry, free-flowing blend is then charged to a
feed hopper where it is screw-fed into a continuous mixer
such as an extruder or kneader. Under the action of this
mixer’s reciprocating screw in the confined volume of the
mixing chamber, the blend begins to flux or masticate
into the plastic state. It is then forced out of the barrel
of the mixing chamber. The continuous strand may be
chopped into small fist-sized buns of hot material or
simply exit as a continuous rope. This material may
then be directly conveyed to the calender, or it may
first pass through a two-roll mill. The calender is a large
unit, typically consisting of four or five heated rolls
designed to process masticated PVC buns into a con-
tinuous web of designated width and thickness (see
Figure 1).
Table 2. Typical Physical Properties of Rigid PVC (Clear)
Property Test Method
a
Units Values
Specific gravity
b
D1505 1.30–1.36
Yield (1.30 sp gr) D1505 in.
2
lb (cm
2
/g)
7.5 mil (0.19 mm) 2850 (40.5)
10.0 mil (0.25 mm) 2130 (30.3)
12.0 mil (0.30 mm) 1780 (25.3)
15.0 mil (0.38 mm) 1420 (20.2)
20.0 mil (0.51 mm) 1070 (15.2)
Tensile strength (yield) D882 psi (MPa) 6500–7800 (44.8–53.8)
Tensile modulus D882 psi (MPa) 2.5–4.0 10
5
(1723–2757)
Elongation (break) D882 % 180–220
Izod impact (1/4 in. or 6.4 mm) D256 ft lbf in (J/m) 0.5–20.0 (26.7–1068)
Gloss, 201 D247 120–160
Heat-distortion temperature (264 psi or 1.82 MPa) D648
c
F(1C) 158–169 (70–76)
Cold-break temperature D1790
c
F(1C) 14 to 40 (10 to 40)
WVTR (381C, 90% rh) DIN53122 g (100 in.
2
24 h)
[g (m
2
day)]
7.5 mil (0.19 mm) 0.30 [4.7]
10.0 mil (0.25 mm) 0.20 [3.1]
Surface resistance DIN53482 O 10
9
–10
13
Specific resistance DIN40634 O cm 10
13
–10
15
Dielectric strength DIN40634 kV/mm 60–70
Specific heat (201C) kJ/(kg K) 0.8
Thermal conductivity W/(m K) 0.16
Linear thermal expansion K
1
7.0–8.0 10
5
Infrared absorption
c
(3–18 mm) Various intensities
a
Ds are ASTM test methods, and DINs are German (Deutsche) Industr ial Norm test methods.
b
Indirectly related to amount of impact modifier. Increased opacity may raise to 1.40.
c
20 mil (508 mm) unmodified.
Figure 1. PVC calender.
FILM, RIGID PVC 495
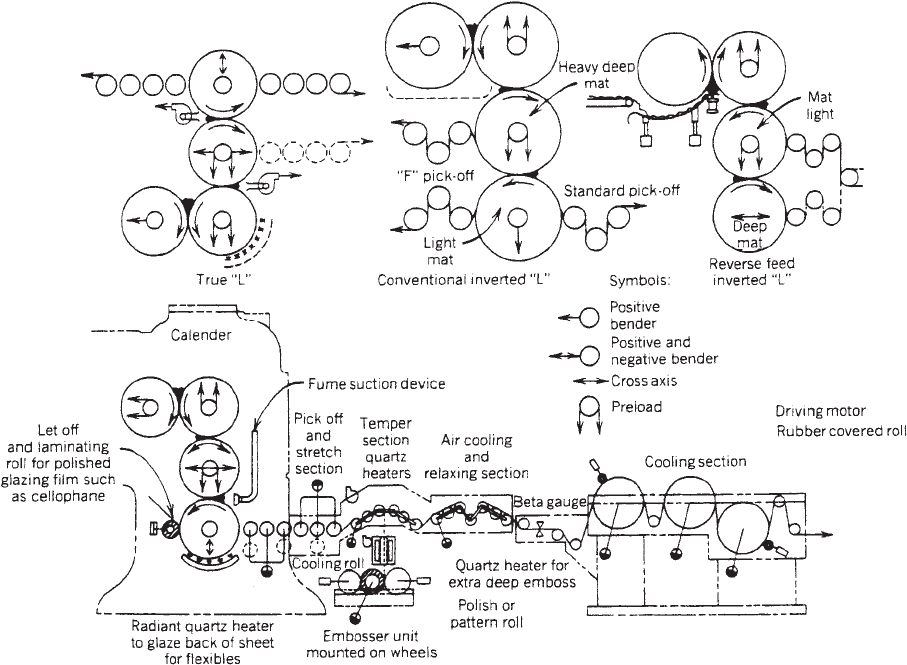
Figure 2 illustrates the typical ‘‘L’’ and ‘‘inverted L’’
configurations generally used for rigid and flexible pro-
duction, respectively (3). The calender rolls have separate
temperature and speed controls as well as roll bending
and crossing capabilities to control profile across the web.
Proper use of these controls, along with speed and stretch
in the takeoff train, allows the production of an extremely
flat sheet with a profile tolerance of less than 75% across
and down the web. Such control is maintained by contin-
uous beta scanning equipment that traverses the web
constantly and calls for adjustments in nip openings,
skew, and/or roll bending. Such constant inprocess mon-
itoring and continuous profile adjustments is a significant
advantage of calendering over other processing methods.
By using special grit-blasting techniques, the third and
fourth calender rolls may be custom-surfaced to generate
a uniform two-sided matte product. Alternatively, one or
more downstream embossing stations may be utilized to
produce a custom surface on one or both sides of the film.
Antistatic and or de-nesting slip agents may be applied to
the surface(s) of the web after separating from the last
calender roll. Finally, after the cooling section, the web is
cut in-line into finished sheets or wound about a core into
a master roll for subsequent custom slitting. Typical slit
widths are made to the nearest 1/32 in. (0.8 mm) on 3- or
6-in. (7.6- or 15.2-cm) cores with roll diameters of 14–40 in.
(36–102 cm).
PACKAGE PRODUCTION BY THERMOFORMING
Most commercial PVC packages are the result of thermo-
forming rollstock into custom blisters. In those cases
where further enhancement of PVC’s own oxygen and/or
moisture barrier properties are required, barrier materi-
als (see Barrier polymers) such as PE, PVDC, or fluor-
opolymer film (see Film, fluoropolymer) may first be
laminated to the PVC web prior to thermoforming. Ther-
moforming processing conditions are generally dictated by
the PVC material itself regardless of lamination. Since
PVC is an amorphous material (see Polymer properties), it
softens over a large temperature range and has no sharp
melting point. There are two temperatures ranges in
which rigid PVC can most readily be formed (see Figure
3). It must be emphasized that these temperatures are
actual film temperatures that must be measured with
thermocouples located directly on the surface of the film
(heating-element temperatures are very much hotter).
The first plateau at 221–2751F (105–1351C) is the
elastic area best for most high-speed form/fill/seal pres-
sure-forming machines (see Thermoform/fill/seal). In this
area, the film has sufficient hot strength to elongate or
stretch to the conformation of the mold. Between 2751F
and 3381 F (1351C and 1701 C) is a region of inadequate
elongation for proper forming; and attempts to process in
this area may result in blowing holes, tearoffs, and poor
Figure 2. PVC calender operation.
496 FILM, RIGID PVC
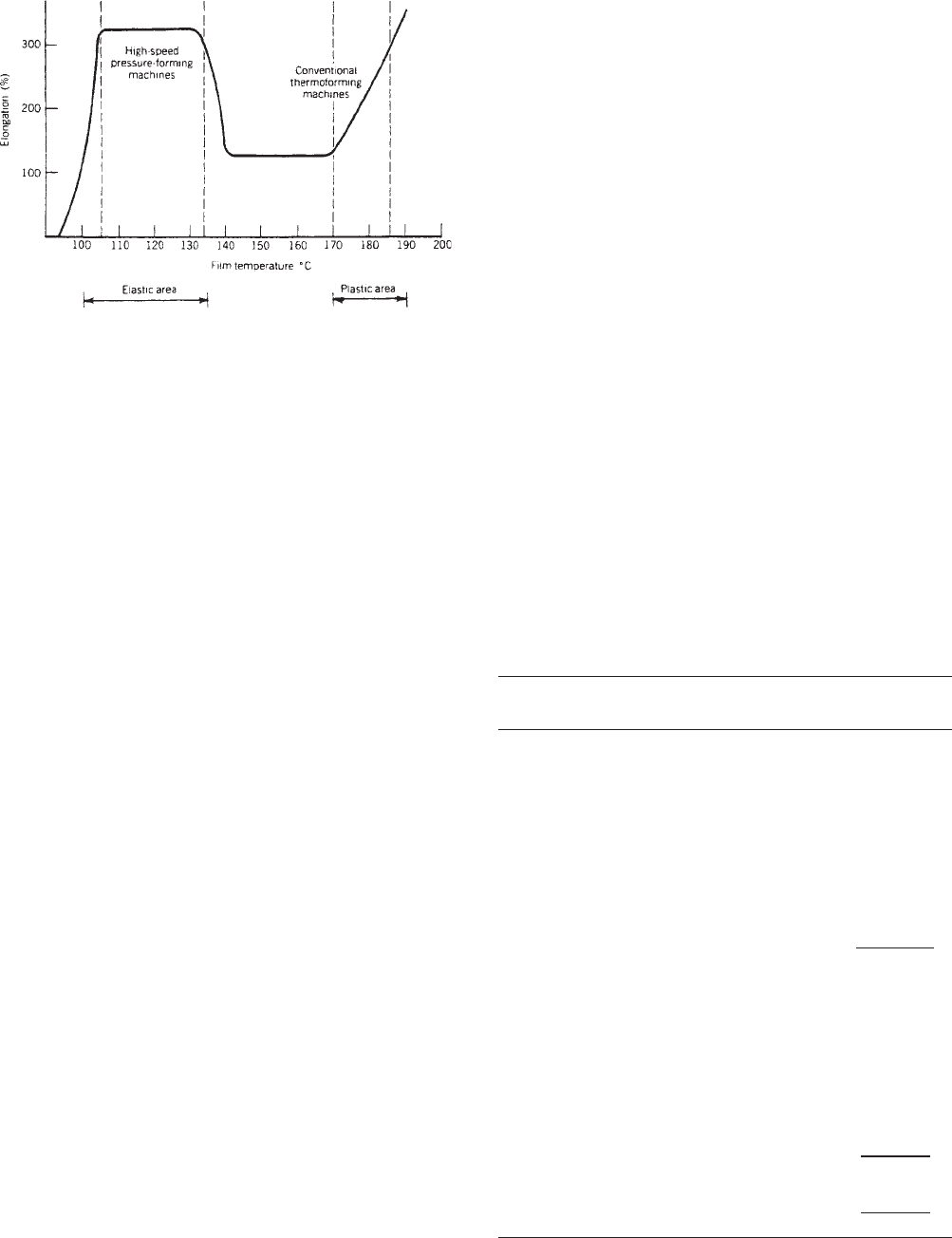
definition. Failure to respect this ‘‘no man’s land’’ is among
the most common reasons for thermoforming problems
seen in the field by PVC technical service representatives.
The elongation quickly increases in the film at 3381F
(1701C), and excellent forming is possible up to about
3651F (1851C). This is the range for optimum performance
for conventional commercial thermoforming machines
(see Thermoforming). Beyond 3651F (1851C), the material
will sag excessively, resulting in webs, wrinkles, holes, or
thin areas. In general, rigid PVC film thermoforms best
when the film temperature is slightly above 3381F
(1701C), with relatively cool molds (as low as 50–601For
10–161C), and when plug assist temperatures are about
1941C (901C). Mold design should avoid exceeding a 1:1
draw ratio, and a few extra degrees of draft and extra
radius on corners will help avoid problems.
Film Shrinkage for Optimum Forming
Since distinct types of equipment are used in each of
the two forming regions, it is particularly important that
the PVC-film supplier have a thorough understanding
of the appropriate shrinkages to be put into the feedstock
for each type of machine. PVC destined for pressure-
forming over the lower temperature region should
have its shrinkage controlled by the producer at 2841F
(1401C), a temperature that will release all plastic mem-
ory relevant to the processing range of 221–2751F
(105–1351C). Typically, best results are achieved with
PVC film having 2841F (1401C) shrinkages of 2–5% in
the machine direction and 0–1% in the transverse direc-
tion. Such very slight growth in the transverse direction
is desirable to compensate for the repeating necking-in
that may otherwise occur across each line of blisters such
as those used for press-through packaging of ethical
drugs.
In the higher temperature range for conventional
thermoforming, film shrinkages should be controlled by
the film supplier at 3501F (1771C), a temperature that will
release all plastic memory relevant to this higher proces-
sing range. Best results are typically achieved with PVC
film having 3501F (1771C) shrinkages of 4–8% in the
machine direction and 1 to +1% in the transverse direc-
tion. Excessive shrinkages may cause the film to pull out
of the chains, clips, or frame. Insufficent machine direction
shrinkage and/or excessive transverse growth may led to
webbing. Thermoforming machine operators often re-
spond to such webbing problems by reducing operating
temperatures to prevent excessive sag, but this step can
result in blowing holes and poor wall distribution because
it takes the film out of the optimum thermoforming
temperature range and elongation falls off rapidly. Careful
shrinkage control is critical to successful thermoforming
of rigid PVC film with all types of forming equipment.
Proprietary process controls are available in modern PVC
calendering technology to custom produce film with the
proper shrinkages for optimum thermoformability within
each temperature region and on each design of forming
equipment.
PACKAGING MARKET
U.S. consumption of calendered rigid PVC film and sheet
in 1994 is estimated at 523
10
6
lb (237,000 tons), not
including an additional 30% for extruded sheet. About
63% of this calendered production was used in packaging
(see Table 3). Total 1994 consumption of calendered rigid
PVC film and sheet for packaging was 330 10
6
lb
(150,000 tons).
Figure 3. Temperature ranges for PVC thermoforming.
Table 3. U.S. Markets for Calendered Rigid PVC Film
and Sheet
Market 10
6
lb
(10
3
tons)
Packaging (FDA-grade)
Food 25 (11.4)
Pharmaceuticals 30 (13.6)
Medical devices and supplies 10 (4.5)
Total packaging (FDA grade) 65 (29.5)
Packaging (general-purpose)
Thermoforming—blisters, clam
shells, etc.
225 (102.3)
Vinyl boxes and lids 35 (15.9)
Static control thermoforms 5 (2.3)
Total general-purpose and specialty
packaging
265 (120.5)
Specialty and industrial applications
Printing and stationery 90 (40.9)
Cooling tower and wastewater fill
media
40 (18.2)
Artificial Christmas trees 30 (13.6)
Furniture lamination 10 (4.5)
Construction and flooring 10 (4.5)
Floppy disks 8 (3.6)
Litho mask 5 (2.3)
Total specialty and industrial
applications
193 (87.6)
Total packaging (61.5%) 330 (150.0)
Total nonpackaging (36.5%)
193 (87.7)
Total rigid PVC film and sheet 523 (237.7)
Source: Klo
¨
ckner-Pentaplast of America, Inc.
FILM, RIGID PVC 497
