Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.

type of blowing agent control the density of the foam
produced. Figure 2 shows the relationships for three of
today’s most commonly used blowing agents. The foam can
also be colored for aesthetics by the addition of color
concentrates (see also Colorants).
A large percentage of all foam sheet is thermoformed to
produce the final product. To achieve good postexpansion of
the sheet, the sheet should be allowed to age for 3–5 days.
This aging allows cell gas pressure to reach equilibrium.
Matched metal molds are normally used in the thermo-
forming process. Oven temperature control is critical for
consistent thermoforming. Scrap produced from the extru-
sion and thermoforming process can be reprocessed by
grinding the scrap and densifying it in an extruder.
ENVIRONMENTAL CONCERNS
Ecologically, polystyrene foam is very easily recycled, and
significant quantities are presently being reused. It also
conforms favorably to current forms of disposal, such as
landfill and incineration. In landfill, foam remains inert,
but it packs and crushes easily and there is no pollution of
underground water streams by decaying material. In
incineration the chief products of combustion are water,
carbon dioxide, and carbon monoxide, which are typical of
organic materials, and the Btu (British thermal unit)
value is high. If burned without enough oxygen, polycyclic
aromatic compounds, carbon mooxide, and carbon black
are produced. Use of polystyrene for food packaging has
been banned in some U.S. cities (San Francisco and Oak-
land are the latest to pass bans). Recycled polystyrene can
be used for park benches, flower pots, and toys.
BIBLIOGRAPHY
P. A. Wagner, ‘‘Foam Extruded Polystyrene’’ in: A. J. Brody and
K. S. Marsh, eds., The Wiley Encyclopedia of Packaging
Technology, 2nd edition, John Wiley & Sons, New York, 1997,
pp. 449–450.
Cited Publications
1. M. Xanthos and D. B. Todd, ‘‘Plastics Processing’’ in the Kirk–
Othmer Encyclopedia of Chemical Technology, Vol. 19, 5th ed.,
John Wiley & Sons, Hoboken, NJ, 2006.
2. C. I. Chung, Extrusion Principles and Practise, Carl Hanser
Verlag, Munich, 2000.
FOAM PLASTICS
K. W. SUH
M. H. TUSIM
The Dow Chemical Company,
Midland, Michigan
Foamed plastics, which are also known as cellular plastics
or plastic foams, have been important since primitive
Figure 2. Blowing-agent requirements for polystyrene foam.
HFC-152a (Formacel Z-2) data supplied by DuPont. To convert
lb/ft
3
to g/cm
3
, multiply by 0.0162.
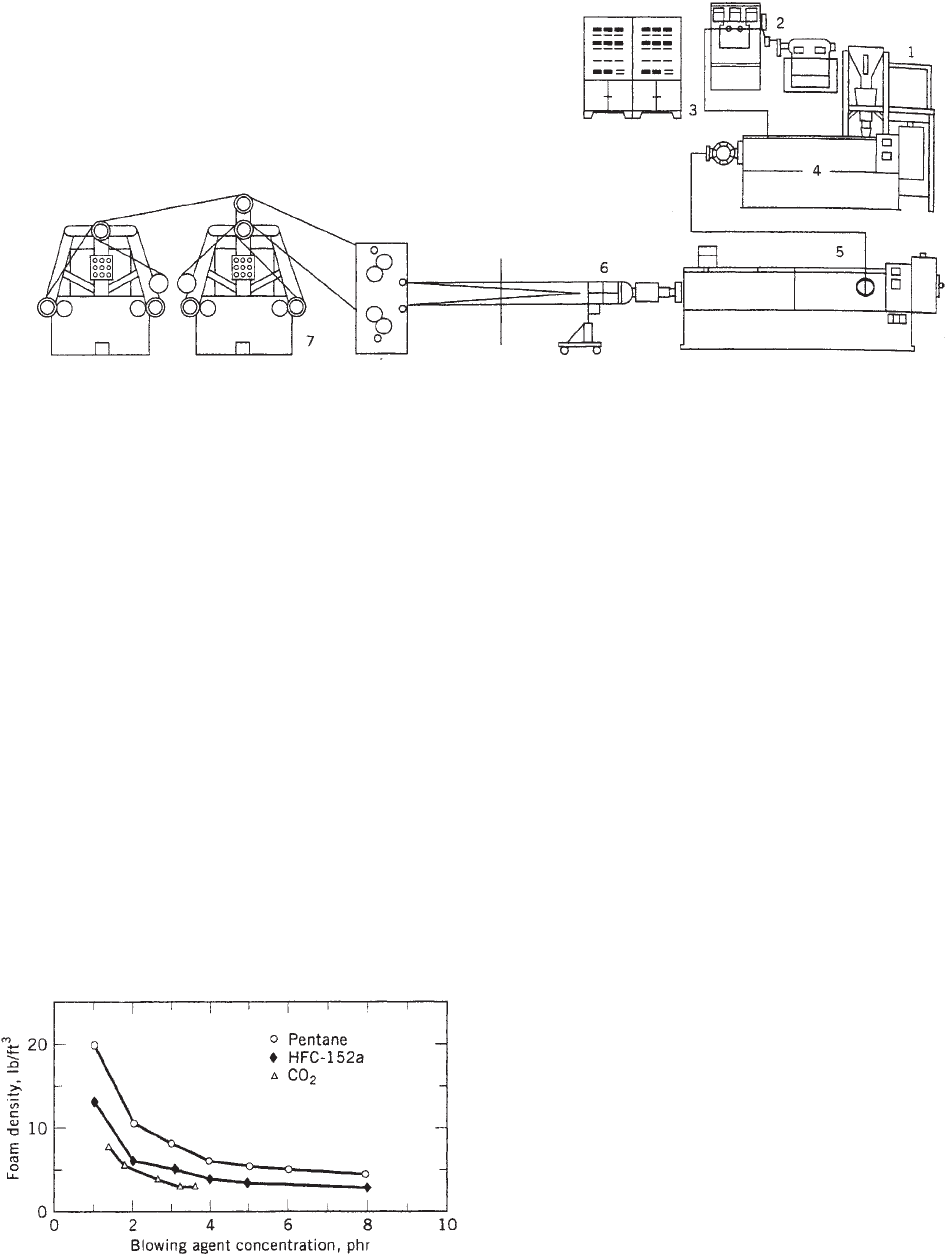
Figure 1. A two-extruder tandem system for the production of foamed sheet: (1) continuous feeding and blending system; (2) volumetric
pump system; (3) process control; (4) primary extruder; (5) secondary extruder; (6) annular die and cooling mandrel; (7) draw rolls and
winders.
518 FOAM PLASTICS

humans began to use wood, a cellular form of the polymer
cellulose. Cellulose is the most abundant of all naturally
occurring organic compounds, which represents approxi-
mately one third of all vegetable matter in the world. The
high strength:weight ratio of wood, the good insulating
properties of cork and balsa, and the cushioning proper-
ties of cork and straw have contributed to the development
of the board range of cellular synthetic polymers in use
today.Cellular polymers have been commercially accepted
for a wide variety of applications since the 1940s. The total
usage of foamed plastics in the United States rose from
about 2.1 10
6
tons in 1993. It is expected to rise to about
2.9 10
6
metric tons in 1998 (1).
NOMENCLATURE
A cellular plastic is defined as a plastic whose apparent
density is decreased substantially by the presence of
numerous cells disposed throughout its mass (2). In this
article, the terms cellular polymer, foamed plastic, ex-
panded plastic, and plastic foam are used interchangeably
to denote all two-phase gas–solid systems in which the
solid is continuous and composed of a synthetic polymer or
rubber.
THEORY OF THE EXPANSION PROCESS
Foamed plastics can be prepared by various methods. The
most widely used, which is called the dispersion process,
involves dispersion of a gaseous phase throughout a fluid
polymer phase and the preservation of the resultant state.
Other methods of producing cellular plastics include
sintering dispersed small particles and dispersing small
cellular particles in the plastic. The latter processes are
relatively straightforward techniques of lesser commercial
importance.
The expansion process has been the subject of extensive
investigation because it is the foundation of foamed plas-
tics (3–6). In general, the expansion process consists of
three steps: creation of small discontinuities or cells in a
fluid or plastic phase, growth of these cells to a desired
volume, and stabilization of the resultant cellular struc-
ture by physical or chemical means.
Bubble Initiation. The development of bubbles within a
liquid or polymer solution is generally called nucleation,
although the term actually refers only to those bubbles
that separate from the supersaturated liquid or polymer
solution in the presence of an initiating site such as a
surface irregularity. This process has several sources: (a)
dissolved gases that are normally present in the liquid or
polymer solution and forced into supersaturation by in-
creased temperature; (b) low-boiling liquids that are in-
corporated into the system as blowing agents and forced
into the gas phase by increased temperature and de-
creased pressure; (c) gases produced as blowing agents,
eg, by the water—isocyanate reaction used for CO
2
pro-
duction in polyurethane foams; and (d) chemical blowing
agents that decompose thermally to form a gas.
Bubble nucleation is affected by many conditions.
Physically, the effects of temperature, pressure, and in
some cases humidity are fairly obvious. Other important
parameters are surface smoothness of the substrate, sur-
face characteristics of filler particles, presence and con-
centration of certain surfactants or nucleators, size and
amount of second-phase liquid bubbles, and the rate of gas
generation.
In many cases, bubbles of gas and other contaminants
are already present in the liquid or polymer solution, and
these serve as sites into which the gas may diffuse. The
number and size of these gas bubbles may be another
important factor in bubble development.
Bubble Growth. The initial bubble is ideally a sphere
that grows as a result of the interaction of the differential
pressure (DP) between the inside and outside of the cell
and the interfacial surface tension (g). The radius (r) of the
bubble at equilibrium is related to these factors as shown
in equation (1):
DP ¼
2g
r
ð1Þ
The differential pressure is larger for a small bubble at
a fixed surface tension. Accordingly, smaller bubbles tend
to equalize these pressures by growing faster, breaking
the wall separating the cells, or by diffusion of the blowing
agent from the small to the large cells as indicated by
equation (2):
DP
1:2
¼ 2g
1
r
1
1
r
2
ð2Þ
where DP
1.2
is the difference in pressure between cells of
radius r
1
and r
2
. Therefore, the rate of growth of the cell
depends on the viscoelastic nature of the polymer phase,
the blowing-agent pressure, the external pressure on the
foam, the cell size, and the permeation rate of blowing
agent through the polymer phase.
Bubble Stabilization. Bubble stability during growth is
a function of the type and concentration of surfactant, rate
of bubble growth, viscosity of the fluid medium, differen-
tial pressure variations, and presence of cell-disrupting
agents such as solid particles, liquids, or gases. As the cell
walls are squeezed into polyhedra, a wall-thinning effect
takes place, and liquid is drained from cell-wall faces into
the lines of cell intersections to form ribs or struts, which
are typically triangular in cross section. This cell-wall
membrane thinning can continue to the point where the
cell walls collapse and the cells open. This becomes an
important characteristic of most plastic foams and affects
properties such as thermal conductivity, moisture absorp-
tion, breathability, and load bearing.
Ultimate stabilization occurs as a result of either
chemical reaction continuing to the point of complete
gelation or the physical effect of cooling below the sec-
ond-order transition point to prevent polymer flow. As the
final solidification is approached, the previously formed
bubbles may be distorted by the system flow or gravity,
FOAM PLASTICS 519
which thereby produce anisotropy in the cellular struc-
ture. This effect must be considered when evaluating the
physical properties of plastic foams by obtaining samples
oriented in specific directions to the process flow.
PROPERTIES
The data in Table 1 show the broad ranges of properties of
rigid foamed plastics (7); the manufacturer should be
consulted for the properties of a particular product. The
properties of some commercial flexible foamed plastics are
given in Table 2 (7). These values depend on several
structural variables and should be used only as general
guidelines (see also the Polymer properties article).
The properties of a foamed plastic depend on composi-
tion and geometry, which are often referred to as struc-
tural variables. Furthermore, they are influenced by the
foam structure and the properties of the parent polymer.
The polymer phase description must include the additives
present.
Cells. A complete knowledge of the cell structure of a
particular polymer would require the size, shape, and
location of each cell. Because this is impractical, approx-
imations are employed. Cell size has been characterized
by measurements of the cell diameter and as a measure-
ment of average cell volume. Mechanical, optical, and
thermal foam properties depend on cell size.
Geometry. Cell shape is governed predominantly by
final foam density and the external forces exerted on the
cellular structure before its stabilization in the expanded
state. In the presence of external forces, the cells may be
elongated or flattened. Cell orientation can influence
many properties.
Fraction open cells. An important characteristic of the
cell structure is the extent of communication with other
cells. This is expressed as fraction open cells.
Gas composition. In closed-celled foams, the gas phase
in the cells can contain blowing agent (so-called captive
blowing agent), air, or other gases generated during
foaming. Thermal and electrical conductivity can be pro-
foundly influenced by the cell-gas composition.
Mechanical Properties. In mechanical properties, rigid
foams differ from flexible foams. The tests used to char-
acterize them are therefore different, as are their applica-
tion properties. In the last two decades, a separate class of
high-density, rigid cellular polymers called structural
foams (density W0.3 g/cm
3
) have become commercially
significant.
Structural variables that affect the compressive
strength and modulus of a rigid plastic foam are, in order
of decreasing importance, plastic-phase composition, den-
sity, cell structure, and plastic state. The effect of gas
composition is minor with a slight effect of gas pressure in
some cases.
Structural foams or foams with integral skins are
usually produced as fabricated articles in injection-mold-
ing or extrusion processes. They have relatively high
densities, and cell structures are composed primarily of
holes in contrast to a pentagonal dodecahedron structure
in low-density plastic foams. Because structural foams are
generally not uniform in cell structure, they exhibit con-
siderable variation in properties with article geometry.
In flexible foams, which are used in comfort cushioning,
packaging, and wearing apparel, different mechanical
properties are emphasized than for rigid foams. The
compressive nature of flexible foams, both static and
dynamic, is their most significant mechanical property.
Other important properties are tensile strength and elon-
gation, tear strength, and compression set.
Thermal Properties. The thermal conductivity of cellu-
lar polymers has been thoroughly studied in heteroge-
neous materials and plastic foams (8). Heat transfer can
be separated into its component parts as follows:
k ¼ k
s
þ k
g
þ k
r
þ k
c
ð3Þ
where k = total thermal conductivity and k
s
, k
g
, k
r
, and k
c
represent solid conduction, gases conduction, radiation,
and convection, respectively.
As a first approximation, the heat conduction of low-
density foams through the solid and gas phases can be
expressed as the product of the thermal conductivity of
each phase and its volume fraction. In most cellular
polymers, the conduction through the solid phase is
determined primarily by the density and the polymer-
phase composition. Although conductivity through gases
is much lower than through solids, the amount of heat
transferred through the gas phase in a foam is usually the
largest component of a total heat transfer because of the
large gas-phase volume. Ordinarily, convection cannot be
measured in cells of diameter less than B4 mm (8).
Because most cellular polymers have cell diameters
of o4 mm, convection can be ignored. Radiant heat trans-
fer through cellular polymers has also been studied (8).
The increase in k at low densities is caused by increased
radiant heat transfer; at high densities, by an increasing
contribution of k
s
.
The thermal conductivity of most materials decreases
with temperature. When the foam structure and gas
composition are not influenced by temperature, the k of
the cellular material falls with decreasing temperature.
The thermal conductivity of a cellular polymer can change
on aging under ambient conditions if the gas composition
is influenced by aging. This is the case when oxygen or
nitrogen diffuses into cellular foams that initially have an
insulating fluorocarbon blowing agent in the cells (8). The
thermal conductivity of foamed plastics varies with thick-
ness (9). This has been attributed to the boundary effects
of the radiant contribution to heat transfer.
The specific heat of a cellular polymer is the sum of the
specific heats of each of its components. The contribution
of the gas is small and can often be neglected.
520 FOAM PLASTICS

Table 1. Physical Properties of Commercial Rig
id Foamed Plastics
Polyurethane
Polystyrene
Isocyanurate
Property
ASTM
Test
Cellulose
Acetate Epoxy Phenolic
Extruded Plank
Expanded Plank Extruded Sheet
PVC
Polyether Bun Laminate
Urea—
formaldehyde
Density, kg/m
3a
96–128 32–48 32–64 35
53 16 32 80
96 160 32 64
32–48 64–128 32 32
13–19
Mechanical
properties,
compressive
strength, kPa
b
at 10%
D1621 862
138–172 138–620 310 862
90–124 207–276 586–896 290
469 345 1,035 138–344
482–1896 210 117–206 34
Tensile strength,
kPa
b
D1623 1172
138–379 517
145–193 310–379 1020–
1186
2070–
3450
4,137 551 1,207 138–482 620–2000
250 248–290
6,900
Flexural strength,
kPa
b
D790 1104
172–448 1138
193–241 379–517
586 1,620 413–689 1380–2400
Shear strength,
kPa
b
C273 965
103–207 241
241
241 793 138–207 413–896
180 117
Compression
modulus, MPa
b
D1621 38–90 3.9
10.3
3.4–14
13.1 35 2.0–4.1 10.3–31
Flexural modulus,
Mpa
b
D790 38
41
9.0–26
10.3 36 5.5–6.2 5.5–10.3
Shear modulus,
MPa
b
C273
2.8–4.8 10.3
7.6–11.0
6.2 21 1.2–1.4 3.4–10.3
1.7
Thermal
properties,
thermal
conductivity W/
(m K)
C177 0.045–0.046 0.016 0.029
0.030
0.037 0.035 0.035 0.035
0.035 0.023
0.016–
0.025
0.022–
0.030
0.054 0.019 0.026
0.022 0.032
0.030
Coefficient of
linear
expansion,
10
5
/1C
D696
1.5 0.9 6.3 6.3
5.4–7.2 5.4–7.2
5.4–7.2 7.2 7.2
Maximum service
temperature,
1C
177
205–260 132 74
74–80 74–80 74–80 77–80
80
93–121 121–149 149 149
Specific heat, kJ/
(kg
K)
c
C351
1.1
B0.9
B0.9
B0.9
Electrical
properties
1.19–
1.20
o1.05
o1.05 1.02 1.02 1.02
1.27 1.28
1.05 1.1 1.4
Dielectric constant D1673 1.12
0.028–
0.031
o0.0004
o0.0004 0.0007 0.0007 0.0007
0.00011 0.00014
13
18
Dissipation factor
20
Moisture-
resistance
water
absorption,
vol%
C272 4.5
13–51 0.02 0.05 1–4
1–4 1–4
Moisture-vapor
transmission,
g/(m
s GPa)
b
E96
58
35
o120 35–120 23–35 86
56 15
35–230 50–120
230
1,610–2,000
Source:
Suh and Webb (7).
a
To convert kg/m
3
to lb/ft
3
, multiply by 0.0624.
b
To convert kPa to psi, multiply by 0.145.
c
To convert kJ/(kg
K) to Btu(lb
1F), divide by 4.184.

Table 2. Physical Properties of Commercial Flexi
ble Foamed Plastics
Polyethylene
Polypropylene
Polyurethane
Silicone
Property
ASTM
Test
Expanded
Natural Rubber
Expanded
SBR
Latex Foam
Rubber
Extruded
Plank
Extruded
Sheet Crosslinked Sheet Modified
Sheet
Standard
Cushioning
High-
Resilience
Type
Poly(vinyl
Chloride) Liquid Sheet
Density, kg/m
3a
56 320 72 80 130
35 43
26–28
64–96 10
24 40 56 112 272
160
Cell structure
Closed Closed Closed Open Open
Closed Closed Closed
Closed
Open Open Closed Closed
Open Open
Compression
strength 25%,
deflection, kPa
b
D33574
48
206
4.8
5.7 4.6
D3575
Tensile strength,
kPa
b
D33574
206 551 103
138 41
344
118 103 10.3 24 36
at
20%
Tensile elongation,
%
D33574
310
60 276 276–480
1380 138–275 205 160
227 310
Rebound
resilience, %
D3574
73
50
75
40 62
Tear strength, (N/
m)
c
10
2
D3574
10.5 26
4.4 2.4
Maximum service
temperature,
1C
70 70 70
82 82
79–93
135 121
350 260
Thermal
conductivity, W/
(m K)
C177 0.036 0.043 0.030
0.050 0.053 0.040–0.049 0.036–0.040
0.039 0.039
0.03 0.040 0.078 0.086
Source:
Suh and Webb (7).
a
To convert kg/m
3
to lb/ft
3
, multiply by 0.0624.
b
To convert kPa to psi, multiply by 0.145.
c
To convert kJ/(kg
K) to Btu/(lb
1
F), divide by 4.184.
522
The coefficients of linear thermal expansion of poly-
mers are higher than those of most rigid materials at
ambient temperatures because of the supercooled liquid
nature of the polymeric state. This large coefficient is
carried over directly to the cellular state. A variation of
this property with density and temperature has been
reported for foams in general (10).
Moisture Resistance. Plastic foams perform better than
other thermal insulation materials when exposed to
moisture, particularly when subjected to a combination
of thermal and moisture gradients.
Moisture absorption and freeze–thaw resistance of
various insulations and the effect of moisture on thermal
performance show that in protected-membrane roofing
applications, the order of resisting moisture pickup is
extruded polystyrene g polyurethaneWmolded polystyr-
ene (11). Water absorption values for insulation in use for
5 years were 0.2 vol% for extruded polystyrene, 5 vol% for
polyurethane without skins, and 8–30 vol% for molded
polystyrene. These values correspond to increases in k of
5–265%. For below-grade applications, extruded polystyr-
ene was better than molded polystyrene or polyurethane
without skins in terms of moisture and thermal
resistance.
Electrical Properties. Electrical insulation takes advan-
tage of the toughness and moisture resistance of polymers
along with the decreased dielectric constant and dissipa-
tion factor of the foams. In addition, because of the low
dissipation factor and rigidity, plastic foams are used in
the construction of radar domes.
Environmental Aging. The response of cellular materials
to light and oxygen is governed almost entirely by the
composition and state of the polymer phase. Expansion
into the cellular state increases the surface area; reactions
of the foam with vapors and liquids are correspondingly
faster than those of solid polymer. All cellular polymers
deteriorate under the combined effect of light or heat and
oxygen; this may be alleviated by additives.
Other Properties. Cellular polymers by themselves are
poor materials for reducing sound transmission. They are,
however, effective in absorbing sound waves of certain
frequencies. Materials with open cells on the surface are
particularly effective in this respect. The presence of open
cells in a foam allow gases and vapors to permeate the cell
structure by diffusion and convection, thus yielding high
permeation rates. In closed-cell foams, the permeation of
gases or vapors is governed by the composition of the
polymer phase, gas composition, density, and cellular
structure. The penetration of visible light through foamed
polystyrene has been shown to follow approximately the
Beer–Lambert law of light absorption (10).
The resistance to rot, mildew, and fungus is related to
moisture absorption. Therefore, open-cell foams support
such growth better than do closed-cell foams.
MANUFACTURING PROCESSES
Cellular plastics and polymers have been prepared by
processes that involve many methods of cell initiation,
growth, and stabilization. The most convenient method of
classifying these methods seems to be based on cell growth
and stabilization. According to equation (1), the growth of
the cell depends on the pressure difference between the
inside of the cell and the surrounding medium. Such
pressure differences may be generated by lowering the
external pressure, i.e., decompression; or by increasing
the internal pressure in the cells, i.e., pressure generation.
Other methods of generating the cellular structure are by
dispersing gas or solid in the fluid state and stabilizing
this cellular state, or by sintering polymer particles in a
structure that contains a gas phase (see Table 3).
Foamable compositions in which the pressure within
the cells is increased relative to that of the surroundings
are called expandable formulations. Both chemical and
physical processes are used to stabilize plastic foams for
expandable formulations. There is no single name for the
group of cellular plastics produced by the decompression
processes. The operations used are extrusion, injection
Table 3. Production of Cellular Polymers
Polymer Extrusion
Expandable
Formulation Spray
Forth
Foam
Compression
Mold
Injection
Mold Sintering Leaching
Cellulose acetate X X
Epoxy resin X X X
Phenolic resin X
Polyethylene X X X X X X
Polystyrene X X X X X
Silicones X
Urea–formaldehyde
resin
X
Urethane polymers X X X X
Latex foam rubber X
Natural rubber X X X
Synthetic elastomers X X X
Poly(vinyl chloride) X X X X X X
Ebonite X
Polytetrafluoroethylene X
FOAM PLASTICS 523

molding, and compression molding. Physical or chemical
methods may be used to stabilize the products.
EXPANDABLE FORMULATIONS
Physical Stabilization. Cellular polystyrene, poly(vinyl
chloride) (PVC), polyethylene, and copolymers of styrene
and acrylonitrile can be manufactured by this process.
Chemical Stabilization. This method has been used
successfully for more materials than the physical stabili-
zation process. It is generally more suitable for condensa-
tion polymers than the vinyl polymers because of the fast
yet controllable curing reactions. Chemical stabilization
can be used for foam formation from polyurethane, poly-
isocyanurate, phenolic, epoxy, and silicone resins.
DECOMPRESSION EXPANSION PROCESSES
Physical Stabilization. Cellular polystyrene, cellulose
acetate, polyolefins, and poly(vinyl chloride) can be man-
ufactured by this process.
Chemical Stabilization. Cellular rubber and ebonite are
produced by chemical stabilization processes.
Dispersion. In several techniques for producing cellular
polymers, the gas cells are produced by the dispersion of a
gas or solid in the polymer phase followed, when neces-
sary, by stabilization and treatment of the dispersion. In
frothing, a quantity of gas is mechanically dispersed in the
fluid polymer phase and stabilized. Latex foam rubber was
the first cellular polymer produced by frothing. In another
method, solid particles are dispersed in a fluid polymer
phase, the dispersion stabilized, and the solid phase
dissolved or leached, thus leaving the cellular polymer.
Cellular polymers called syntactic foams are made by
dispersing an already cellular solid phase in a fluid
polymer and stabilizing the dispersion.
USES
Concern over energy conservation and safety has stimu-
lated growth in applications for insulation and cushioning
in transport. A healthy economy is also expected to
increase the demand for cushioning in furniture, bedding,
and flooring as well as for packaging. Structural foams are
widely used as substitutes for wood, metal, or unfoamed
plastics. Table 4 shows the demand for plastic foams in the
United States.
Cushioning. The properties of significance in the cush-
ioning applications of cellular polymers are compression–
deflection behavior, resilience, compression set, tensile
strength and elongation, and mechanical and environ-
mental aging. The broad range of compressive behavior
of flexible foam is one of the advantages of cellular
polymers because the needs of almost any cushioning
application can be met by changing either the chemical
nature or the physical structure of the foam. Flexible
urethanes, vinyls, latex foam rubber, and olefins are
used to make foamed plastic cushioning for automobile
padding, seats, furniture, flooring, mattresses, and
pillows.
Thermal Insulation. Thermal insulation is the largest
application for the rigid materials because of their ther-
mal conductivity, ease of application, cost, moisture ab-
sorption, and transmission permeance. Plastic foams that
contain a captive blowing agent have much lower thermal
conductivities than do other insulating materials. The
low-thermal-conductivity polyurethane foams are used
in refrigerators and freezers. Polystyrene foam is popular
where cost and moisture resistance are important, and
polyurethane foams are used in spray applications.
Extruded polystyrene foam is found in residential
construction as sheathing, perimeter, and foundation in-
sulation. Both polystyrene and polyurethane foams are
highly desirable roof insulators.
Packaging. The entire range of cellular polymers from
rigid to flexible is used for packaging because of their low
cost, ease of application or fabrication, moisture suscept-
ibility, thermal conductivity, consumer appeal, and me-
chanical properties, especially compressive properties.
Extruded Polystyrene Foam Sheet. A polystyrene
foam sheet is made from a combination of poly-
styrene resin, blowing agents, nucleating agents, and
pigments in an extrusion process (12, 13). A polystyrene
foam sheet is used in many shapes and can usually be
characterized by its stiffness and low density, generally
2–12 lb/ft
3
(32–190 kg/m
3
). Sheet products are typically
0.015–0.150 in. thick.
Table 4. U.S. Cellular Polymer Market (million lb)
Foam 1990 1992 1993 1998
a
Annual Growth, 1993–1998
Polyurethane 2681 2752 2848 3509 4.3
Polystyrene 1417 1366 1416 1624 2.8
PVC 320 340 350 395 2.5
Polyolefins 100 105 110 135 4.2
Other 62 67 70 80 2.7
Total 4580 4630 4794 5743 3.7
Source: Courtes y of Business Communications Co., Inc.
a
Projected.
524 FOAM PLASTICS
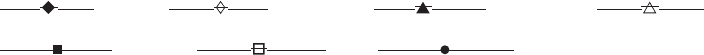
The major uses for foam sheets are for disposable
applications such as carryout cups and clam shells, plates,
bowls, egg cartons, and meat and poultry trays. New
market growth has come in the applications of a polystyr-
ene foam sheet for use in art boards, insulated boxes, and
bottle wraps. For applications that require extra stiffness
or modulus, films or paper may be laminated to the foam.
The foam sheet can be fabricated by die cutting and
scoring, in addition to thermoforming to shape and bend
the foam.
Typically, the foam-sheet process uses an annular die in
the production of thin sheets. Single-screw, twin-screw,
long-barrel single-screw, and tandem-extruder systems
can be used with the latter being most common.
Expanded Polystyrene Beadboard. Expanded polystyr-
ene (EPS) beadboard is produced with expandable poly-
styrene beads. These beads are impregnated with 5–8%
pentane and sometimes with flame retardants during
suspension polymerization. The beads are preexpanded
by fabricators with steam or vacuum and aged. Then they
are fed to steam-heated block molds where expansion and
fusion of beads continue. The molded blocks are sliced
after curing. Block densities range from 13 to 48 kg/m
3
,
with 24 kg/m
3
the most common for cushion packaging
and 16 kg/m
3
for insulation.
Expandable polystyrene bead-molding products ac-
count for the main portion of the drink-cup market and
are used in packaging materials, insulation board, and ice
chests.
Extruded Polystyrene Loose-Fill Packaging. Loose-fill
packaging particles are used in void-filling packaging
applications. The foamed particles provide cushioning for
lightweight, often fragile products. The foamed particles
are usually resilient and are low in density [bulk density
properties under o0.30 lb/ft
3
(4.8 kg/m
3
)] and can be
reused many times. Polystyrene loose-fill particles can be
recycled. The growth of this market has been affected by
the development of starch-based extrusion products.
Expandable polystyrene beads for loose-fill packaging
are produced by the extrusion process. Virgin and recycled
polystyrene with nucleating agents and additives are fed
into the extruder, and blowing agents are usually fed
downstream into the extrusion process. The blowing
agents used have progressed from chlorofluorocarbons
(CFCs) to hydrochlorofluorocarbons (HCFCs) to hydrocar-
bons or blends of hydrocarbons with carbon dioxide. Some
extrusion processes can use expandable beads (pentane/
isopentane blowing agent) for feedstock.
Loose-fill particles are produced by a foam-at-the-die
process, or by an expandable-bead process. In the foam-at-
the-die process, the molten gel is allowed to foam to a
density of approximately 1–2 lb/ft
3
. The foam is then cut at
the die face or downstream after some cooling has oc-
curred. The subsequent density reduction is accomplished
by steam treatment, followed by air aging. The foam may
be steamed and aged 2–3 times to decrease the density to
below 0.25 lb/ft.
3
The expandable-bead process produces an unfoamed
particle that has the blowing agent incorporated into the
bead. The expandable beads are produced at a central
extrusion location and then shipped to local expanders.
Local expansion facilities are needed because of the light-
weight nature of the final product. The expandable beads
are exposed to steam as done with the foam-at-the-die
process but will require an extra expansion step.
Polyolefin Foam Sheet and Plank. Polyolefin foams are
closed-cell foams that are tough, flexible, and resilient and
are used in applications such as cushioning, flotation,
furniture overwrap, water sports, surface protection, and
many other uses. The energy-absorbing characteristics of
polyolefin foams make them excellent foams for use in
cushioning applications. Polyethylene and polypro-
pylene foams are low-density products, generally less
than 10 lb/ft
3
(160 kg/m
3
), which are made by the extru-
sion process. These foams are made in many forms—
plank, sheet, rounds, and multiple strands—and can be
made with crosslinked or non–crosslinked formulation.
Polyethylene and polypropylene foams are usually
produced from a combination of virgin and recycled resins,
blowing agents, nucleating agents, and other additives
such as colorants, antistatic additives, and flame retar-
dants. Both polyethylene and polypropylene can be made
into sheet-type products, typically 0.06–0.625 in. thick
(2–15 mm) by up to 72 in. (1800 mm) wide. Because of the
low melt strengths typical of polypropylene, only poly-
ethylene is used in the manufacture of large cross sections
of foam of 2–4-in. (50–100-mm) thickness and 24–48-in.
(600–1200-mm) width. Planks of foam are often fabricated
into specific shapes to meet the packaging requirements of
electronic components such as computers and their moni-
tors, the handling requirements for water sports uses such
as boogie boards, or the part separation requirements of
multiple-use industrial applications.
Noncrosslinked polyolefin sheet foams can be made on
single-, dual-, and twin-screw extrusion lines that are
designed to mix in large quantities of blowing agents
and then extruding the supersaturated gel through an
annular die at the proper temperature, usually very near
the crystalline melting point. Figure 1 shows the theore-
tical relationship between the blowing-agent content and
the expected foam density. To produce a foam with a
density of B2 lb/ft
3
(32 kg/m
3
), the extrusion system
must be able to mix in 6–16 lb of blowing agent per
100 lb of resin, depending on the blowing agent selected.
The gel pressure at the die is also very important. If the
CFC-12 HCFC-22 HFC-134a
pentane
HFC-152a butane CO
2
Figure 1. Effect of blowing-agent concentration of theoretical foam density.
FOAM PLASTICS 525
pressure is too low, then the blowing agent will begin to
come out of solution and will allow the molten gel to begin
foaming prior to exiting the die lips. If this occurs, then
severe disruption of the skin surface will occur and a poor-
quality foam will result. Blowing agents are injected into
the molten resin gel downstream from the hopper. The
exact location will depend on the type of equipment being
used. To extrude sheet products that are very wide
and very thin, an annular die and forming mandrel is
used. The proper-size forming mandrel allows the ex-
truded tube to be stretched both radially and downstream
at the same time, and the mandrel provides a means to
cool the inner surface of the foam tube. The cooled tube is
then slit at one or two locations and subsequently wound
into rolls. Plank extrusion is usually through a more
rectangular-shaped die onto a conveyor belt or some other
means to move the foam away from the die. Plank extru-
sion is usually through a more rectangle-shaped die onto a
conveyor belt or some other means to move the foam away
from the die. Plank extrusion usually requires higher
rates than sheet because of the larger die opening and
the need for maintaining sufficient pressure to prevent
prefoaming.
Polyolefin foam manufacture generally uses physical
agents, which are materials that are a gas at the foaming
temperature (usually at the resin melting point). Chemi-
cal blowing agents are rarely used in noncrosslinked
applications. Blowing agents for polyolefin foam extrusion
are now typically hydrocarbons (14) or a blend of hydro-
carbon and carbon dioxide. Prior to the Montreal Protocol
agreements, CFC-114 was the main foaming agent used in
polyolefin foams. CFC-114 was an excellent foaming agent
because of its solubility and permeability characteristics.
Another strong attribute was its nonflammable character-
istic. With the Montreal Agreement, use of CFCs was
prohibited after 1991 in noninsulating applications. The
CFC-114 replacement list for foaming agents was not long.
Hydrocarbons (propane, butane, and isobutane), HCFCs
(22,142b), HFCs (152a, 134a), and carbon dioxide were
considered. Most of the replacement options included an
increase in flammability and an increase in cost to the
manufacturing operations. The use of carbon dioxide is
attractive in terms of cost, flammability, and environmen-
tal safety, but the processing aspect makes total substitu-
tion difficult. Because of the high vapor pressure of this
physical gas, maintaining the proper resin-blowing-agent
solution characteristics at the die is very difficult. The
Clean Air Act of 1990 (CAA) has since further reduced
the blowing-agent alternatives. The CAA has prohibited
the use of HCFCs, leaving only hydrocarbon and their
blends, HFCs (15), and carbon dioxide, or blends thereof.
Crosslinked foams offer higher temperature stability,
more flexibility, finer cell size, and better thermoforming
properties than do noncrosslinked foams. Crosslinking of
the olefin resin is accomplished by chemical crosslinking
(such as dicumyl peroxide or silanes) or radiation cross-
linking via X-ray or electron-beam exposure (7). Typically,
the polyethylene resin, additives, crosslinking agents, and
chemical blowing agents (such as azodicarbonamide) are
mixed together at temperatures below the activation
temperature of the blowing agents, and then extruded
into a flat sheet that can be rolled up, or into some other
profile, prior to expansion into a foam product.
Crosslinking occurs before the foam-expansion step.
Expansion is done by exposure of the crosslinked sheet
to hot (B2001C) air. Generally, chemical crosslinking is
used in the production of thick products, whereas radia-
tion crosslinking is used for thinner foams.
HEALTH AND SAFETY FACTORS
Flammability. Plastic foams are organic and therefore
combustible. All plastic foams should be handled, trans-
ported, and used according to manufacturers’ recommen-
dations and local and national regulations.
Virtually all plastic foams are blown with inert gases,
chemical blowing agents that release inert gases, hydro-
carbons containing three to five carbon atoms, chlorinated
hydrocarbons, and hydrochlorofluorocarbons such as
HCFC-22, HCFC-141b, and HCFC-142b.
Atmospheric emissions. Certain organic compounds
generate smog photochemically. Interaction with the total
environment must be considered in developing environ-
mentally acceptable blowing agents. The products of
combustion of plastic foams are usually carbon monoxide
and carbon dioxide with smaller amounts of many other
substances.
The presence of additives or unreacted monomer in
certain plastic foams can limit their use where food or
human contact is anticipated. The manufacturers’ recom-
mendations and existing regulations should be followed.
BIBLIOGRAPHY
1. H. Kibbel, P-120R, Polymeric Foams, A Huge Sub-Industry,
Business Communications Company, Inc., Norwalk, CT, 1994.
2. ASTM D883-80C, Definitions of Terms Relating to Plastics,
American Society for Testing and Materials, Philadelphia, PA,
1982.
3. K. C. Frisch and J. H. Saunders, Plastic Foams, Vol. 1, Parts 1
and 2, Marcel Dekker, New York, 1972 and 1973.
4. C. J. Benning, Plastic Foams, Vol. 1 and 2, Wiley-Interscience,
New York, 1969.
5. N. C. Hilyard and co-worker, Mechanics of Cellular Plastics,
Macmillan, New York, 1982.
6. D. Klempner and K. C. Frisch, Handbook of Polymeric Foams
and Foam Technology, Hanser Publishers, New York, 1991.
7. K. W. Suh and D. D. Webb, Encyclopedia of Polymer Science
and Engineering, Vol. 3, 2nd edition, Wiley, New York, 1985,
pp. 1–59.
8. R. E. Skochdopole, Chem. Eng. Prog. 57(10), 55 (1961).
9. B. Y. Lao and R. E. Skochdopole, paper presented at 4th SPI
International Cellular Plastics Conference, Montreal, Ca-
nada, SPI, New York, Nov. 1976.
10. J. D. Griffin and R. E. Skochdopole, ‘‘Plastic Foams,’’ in
E. Baer, ed., Engineering Design for Plastics, Reinhold, New
York, 1964.
11. F. J. Dechow and K. A. Epstein, ASTM STP 660, Thermal
Transmission Measurements of Insulation, ASTM, Philadel-
phia, PA, 1978, p. 234.
526 FOAM PLASTICS

12. U.S. Pat. 5,250,577 (Oct. 5, 1993), G. C. Welsh (to The Dow
Chemical Company).
13. U.S. Pat. 5,364,696 (Nov. 15, 1994), P. A. Wagner (to The Dow
Chemical Company).
14. U.S. Pat. 4,640,933 (Feb. 3, 1987), C. P. Park (to The Dow
Chemical Company).
15. U.S. Pat. 5,411,684 (May 2, 1995), M. H. Tusim and C. P. Park
(to The Dow Chemical Company).
FOIL, ALUMINUM
Foil Division, Aluminum
Association
Updated by Staff
INTRODUCTION
Aluminum foil is a thin-rolled sheet of pure or alloyed
aluminum, varying in thickness from about 0.00017
(4.3 mm) in. to a maximum of 0.0059 (150 m) in. (1). By
industry definition, rolled aluminum becomes foil when it
reaches a thickness less than 0.006 in. (152.4 mm) (see
Table 1). Aluminum foil is widely sold in the consumer
market in rolls of 50 cm in width and in varying lengths.
Aluminum, from which the foil is made, is a bluish
silver-white trivalent metallic element that is very malle-
able and ductile. Noted for its light weight, good electrical
and thermal conductivity, high reflectivity, and resistance
to oxidation, aluminum is the third most abundant ele-
ment in the earth’s crust (1).
Aluminum always occurs in combination with other
elements in mineral forms such as bauxite, cryolite, cor-
undum, alunite, diaspore, turquoise, spinel, kaolin, feld-
spar, and mica. Of these, bauxite is the most economical
mineral for production of aluminum. It can contain up to
60% alumina, which is hydrated aluminum oxide. It takes
about 4 kg of bauxite to produce 1 kg of aluminum (2).
Alumina is converted into aluminum at a reduction
plant or smelter. In the Hall–He
´
roult process, the alumina
is dissolved in a molten salt called cryolite. The action takes
place in steel boxes lined with carbon called pots. A carbon
electrode or anode is lowered into the solution, and electric
current of 50,000–150,000 A flows from the anode through
the mixture to the carbon-cathode lining of the steel pot.
The electric current reduces, or separates, the alumina
molecules into aluminum and oxygen. The oxygen com-
bines with the anode’s carbon to form carbon dioxide. The
aluminum, heavier than cryolite, settles to the bottom of
the pot from which it is siphoned into crucibles. The molten
aluminum is eventually processed into products.
FOIL MANUFACTURE
One of aluminum’s most common uses is as foil. Alumi-
num foil is generally produced by passing heated
aluminum-sheet ingot between rolls in a mill under
pressure. Ingot is flattened to reroll sheet gauges on sheet
and plate mills and finally to foil gauges in specialized foil-
rolling mills.
A second method for producing aluminum foil, rapidly
gaining popularity, involves continuous casting and cold
rolling. This method can eliminate the conventional en-
ergy-intensive and costly steps of casting ingot, cooling,
transporting to rolling plants, and then reheating and hot
rolling to various gauges. Some lubrication is needed
during the rolling stages, otherwise the foil surface be-
comes marked in a herringbone pattern. These lubricants
are sprayed on the foil before passing through the rolls.
Only oils approved for food contact must be used for foil
intended for food packaging (3).
First produced commercially in the United States in
1913, aluminum foil became a highly marketable com-
modity because of its protective qualities, economic pro-
duction capability, and attractive appearance. The first
aluminum foil laminated on paperboard for folding car-
tons was produced in 1921. Household foil was marketed
in the late 1920s, and the first heat-sealable foil was
developed in 1938.
World War II established aluminum as a major packa-
ging material. During the war, aluminum foil was used to
protect products against moisture, vermin, and heat da-
mage. It was also used in electrical capacitors, for insula-
tion, and as a radar shield.
Table 1. Physical Properties of Aluminum Foil
Property Value
Density 0.0976 lb/in.
3
(2.70 g/cm
3
)
Specific gravity 2.7 (approx.)
Melting range 1190–12151F (643–6571C)
Electrical conductivity 591 IACS. vol., 200% IACS
(approx.), weight
Thermal conductivity 53 W/(m K) at 251C
Thermal coefficient of linear
expansion
13.1 10
6
per 1F, 68–2121F
(23.6 10
6
per 1C, 29–1001C)
Reflectivity for white light,
tungsten filament lamp
85–88%
Reflectivity for radiant heat,
from source at 1001F
(37.81C)
95% (approx.)
Emissivity, at 1001F (37.81C) 5% (approx.)
Atomic number 13
Atomic weight 26.98
Valence 4
Specific heat at 201C 0.21–0.23
Boiling point 32001F (17601C)
Temperature coefficient of
resistance (representative
values per 1C)
0.0040–0.0036
at 201C
at 1001C 0.0031–0.0028
Low-temperature properties—
aluminum increases in
strength and ductility as
temperature is lowered,
even down to 3201F
(195.61C)
FOIL, ALUMINUM 527
