Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.


principle of identifying the CCP (critical control point).
Documentation and verification are integral to hazard
analysis and establishing CCP. Flowing from this basic
concept, HACCP is simply a methodical and systematic
application of the appropriate science and technology to
plan, control, and document the safe production of foods.
HACCP covers three types of potential food safety
hazards that could lead to a health risk—biological,
physical, and chemical—whether they are naturally oc-
curring in the food, contributed by the environment, or
generated by something in the manufacturing process.
Chemical hazards will be of primary interest to packagers,
although, of course, chemicals are used at numerous
points in the food production chain (see Table 1). While
these types of chemicals do not usually represent health
hazards when used properly, some of them are capable of
causing illness or even death when used improperly.
Another potential hazard of interest to packagers is a
physical hazard such as glass shards associated with the
use of glass bottles as packaging.
PRINCIPLES OF HACCP
NACMCF (8) recommends that the HACCP system should
be built upon a solid foundation of prerequisite programs
(e.g., Good Manufacturing Practices, Sanitation Standard
Operating Procedures, supplier approval program). In
preparation for developing an HACCP plan, NACMCF
recommends that five preliminary tasks be accomplished
to facilitate the application of HACCP principles to a
specific product and process. These preliminary tasks
are: (a) assemble the HACCP team, (b) describe the food
and its method of distribution, (c) identify the intended
use and consumers of the food, (d) develop a flow diagram
that describes the process, and (e) verify the flow diagram.
According to the NACMCF 1997 document (8), HACCP
is a systematic approach to food safety consisting of seven
priciples:
Principle 1: Conduct a Hazard Analysis
W ith information from the preliminary tasks, the HACCP
team conducts a hazard analysis to (a) determine significant
food safety hazards associated with the product and (b)
identify appropriate control mesaures for those significant
hazards. The hazard analysis consists of two stages: hazard
identification and hazard evaluation. In the hazard identi-
fication stage, the HACCP team prepares a list of potential
biological, chemical, and physical hazards that may be
introduced, enhanced, or controlled at each of the steps in
the flow diagram, including potential hazards associated
with ingredients and packaging materials. The HACCP
team then conducts a hazard evaluation, based on the
severity of the potential hazard and its likelihood of occur -
rence, to determine which of the potential hazards are
significant and must be addressed in the HACCP plan. In
other words, the hazard anlysis identifies the steps in the
process where hazards of potential significance can occur
and thus warrants control in the HACCP plan. Table 2
enumerates examples of packaging questions that may be
considered during the hazard analysis.
For inclusion in the list of significant hazards and thus
the HACCP plan, a hazard must be of such a nature that
its prevention, elimination, or reduction to an acceptable
level is essential to the production of a safe food (8, 14).
Potential hazards that are of a low severity and/or not
likely to occur (i.e., low risk potential hazards) would not
require further consideration when developing the
HACCP plan. Low-risk hazards, however, should not be
dismissed as insignificant to the operation and may need
to be addressed by other means, for example, by the total
quality management (TQM) team.
The HACCP team must then consider what control
measures, if any, exist that can be applied for control of
each significant hazard. If a significant hazard is identified
in the hazard analysis but no control measure exists in the
process, the HACCP team must modify the process and
product so that a control measure can be applied. Control
measures are physical, chemical, or other factors that can
be used to control an identified significant health hazard.
More than one control measure may be required to control a
specific hazard. More than one hazard may be controlled by
a specified control measure. Potential chemical hazards
Table 1. Examples of Chemicals Used in Food Production
and Processing
Point of Use Types of Chemicals
Growing crops Pesticides, herbicides, defoliants
Raising livestock Growth hormones, antibiotics
Production Food additives, processing aids
Plant maintenance Lubricants, paints
Plant sanitation Cleaners, sanitizing agents, pesticides
Packaging Adhesives resins, surfactants,
defoaming agents, slimicides, polymers
Table 2. Examples of Packaging-Related Questions to Be Considered in a Hazard Analysis
Does the method of packaging affect the multiplication of microbial pathogens and/or the formation of toxins?
Is the package clearly labeled ‘‘Keep refrigerated for safety’’ if this is needed for safety?
Does the package include instructions for the safe handling and preparation of the food by the end user (e.g., validated cooking
instruction)?
Is the packaging material resistant to damage, thereby preventing the entrance of microbial contamination?
Are tamper-evident packaging features used?
Is each package and case legibly and accurately coded?
Does each package contain the proper label?
Could the packaging materials contribute any potential physical hazard (e.g., glass) to the product during processing?
Could the packaging materials contribute any chemical compound to the product that may make the product unsafe to consume?
568 HAZARD ANALYSIS AND CRITICAL CONTROL POINTS
applicable to packaging that the HACCP team may consider
during the analysis include the inks, indirect additives, and
prohibited substances in packaged ingredients and packa-
ging materials if the intended use is food packaging. The
points of control may be, for example, prior to receipt, and
the control measures may be compliance with specifications ,
letters of guarantee, vendor certification, and/or approved
uses listed in the Code of Federal Regulations. In a food
plant producing canned salmon, a significant biological
hazard is a pathogen such as Clostridium botulinum in
the salmon. Retorting (a point of control) using the appro-
priate scheduled process is a control measure that can be
used to eliminate the pathogen hazard. In the packaging
step (a point of control), proper double seaming will prevent
recontamination of the product with potential pathogens
from the processing plant environment, as well as during
distribution, storage, and retail and in the consumer’s
home. The control measure is forming a proper double
seam to prevent leakage and recontamination.
Principle 2: Determine the Critical Control Points (CCPs)
A critical control point is defined as a step in the process at
which control can be applied and is essential to prevent or
eliminate a food safety hazard or reduce it to an accpetable
level. The HACCP team determines CCPs based on the
outcomes of the hazard analysis. An ideal CCP has the
following characteristics: (a) critical limits that are sup-
ported by scientific data from research and the technical
literature; (b) critical limits that are specific, quantifiable
or otherwise measurable, and provide the basis for a yes/
no decision on acceptability of product; (c) technology for
controlling the process at a CCP that is readily available
and at reasonable cost; (d) adequate monitoring (prefer-
ably continuous) and automatic adjustment of the opera-
tion to maintain control; and (e) historical point of control.
All significant hazards identified by the HACCP team
during the hazard analysis must be addressed with at
least one CCP.
Examples of CCPs may include cooking, chilling, acid-
ification, packaging of processed foods, labeling, specific
sanitation procedures, product formulation control, pre-
vention of cross-contamination, and certain aspects of
employee and environmental hygiene.
Principle 3: Establish Critical Limits
A critical limit is defined as maximum and/or minimum
value to which a biological, chemical, or physical parameter
must be controlled at a CCP to prevent, eliminate, or reduce
to an acceptable level the occurrence of a food safety hazard.
As HACCP evolves, a critical limit can also include the
presence of an attribute that is not necessarily a quantita-
tive value (e.g., products passing through a functioning
metal detector). Each CCP will have one or more control
measures that must be properly controlled to assure pre-
vention, elimination, or reduction of hazards to acceptable
levels. Each control measure has associated critical limits
that serve as boundaries of safety for each CCP. Critical
limits may be set such as temperature, time, physical
dimensions, sealing conditions , humidity, moisture level,
water activity (a
w
), pH, titratable acidity, salt concentration,
available chlorine, viscosity, presence or concentration of
preservatives, and sometimes attributes such as presence of
an intact screen of appropriate pore sizes and a functioning
metal detector with appropriate sensitivity (14). Critical
limits may be derived from sources such as regulatory
standards and guidelines, literature review, experimental
studies, and experts. The food industry is responsible for
engaging competent authorities to validate that the critical
limits will control the identified hazard.
Canning operations offer an example of a critical limit
for packaging. The Low Acid Canned Food Regulations
(Title 21, Code of Federal Regulations Part 113) require
that cans should be examined for presence of visual
defects, and cans should be periodically removed from
the line and torn down to verify that can seam measure-
ments are within specifications (15). If the HACCP team
determines that recontamination of product by pathogens
due to seam defect and leakage is a hazard reasonably
likely to occur after retoring, then a control measure must
be identified to address the hazard, for example, in the
form of proper seams on cans to protect consumers from
foodborne illness, and a CCP would be established at the
sealing machine. The monitoring activities are the visual
observation and the seam teardown and double-seam
measurements. The seam measurement would be com-
pared to the minimum acceptable dimensions (critical
limits) specified by the container supplier. Another exam-
ple of a critical limit is the time and temperature for the
retorting of canned salmon. At the retorting CCP, the
process must be designed to eliminate the heat-resistant
spore-forming pathogen, Clostridium botulinum, which is
reasonably expected to be present in the incoming raw
salmon. Technical development of the appropriate critical
limit(s) requires accurate information on the operation of
the retort, heating rate of the product, and heat resistance
of Clostridium botulinum.
A partial hazard analysis for canned salmon is shown
in Table 3, which also illustrates how the first three
principles of HACCP may be documented by the team.
Principle 4: Establish Monitoring Procedures
In the monitoring process a planned sequence of observa-
tions or measurements is conducted to assess whether a
CCP is under control and to produce an accurate record for
future use in verification. Monitoring serves three main
purposes:
1. Monitoring is essential to food safety management
in that it tracks the operation of the system. If
monitoring indicates that there is a trend toward
loss of control (i.e., exceeding a target level), then
action can be taken to bring the process back into
control before a deviation from a critical limit occurs.
2. Monitoring is used to determine when there is loss of
control and a deviation occurs at a CCP–that is,
exceeding or not meeting the critical limit. When a
deviation to a critical limit occurs, corrective action
must be taken.
3. Monitoring provides written documentation for use
in verification of the HACCP plan.
HAZARD ANALYSIS AND CRITICAL CONTROL POINTS 569

Examples of monitoring activities include:
. Visual observations
. Temperature
. Time
. pH
. Moisture level
. Can teardown and seam dimensions
Principle 5: Establish Corrective Actions
The HACCP system for food safety management is de-
signed to identify health hazards and to establish strate-
gies to prevent, eliminate, or reduce their occurrence.
However, ideal circumstances do not always prevail, and
deviations from established processes may occur. Correc-
tive actions are designed to prevent products that may be
hazardous from leaving the process facility. When mon-
itoring indicates that there is a deviation from an estab-
lished critical limit, corrective actions must be taken. The
first step is to bring the process and the CCP under control
by determining and correcting the cause of noncompli-
ance. Second, put all product on hold until determination
has been made on the safety of the product to facilitate
appropriate product disposition. A third component is to
prepare records that document the corrective actions
taken. Because of the diversity of possible deviations
in a food process, specific corrective action procedures
cannot always be developed for every incident. However,
a plan should be developed that addresses the process, the
product, and the recordkeeping components, with proce-
dures developed in advance for those deviation scenarios
that are likely to occur or are expected. The actions taken
must demonstrate that the CCP has been brought under
control. Individuals who have a thorough understanding
of the process, product, and HACCP plan are to be
assigned responsibility for taking corrective actions. The
corrective action plan should be documented in the
HACCP plan.
Principle 6: Establish Verification Procedures
Verification is defined as those activities, other than mon-
itoring, that determine the validity of the HACCP plan and
that the HACCP system is operating according to the plan.
Monitoring is a distinct activity from verification. For
example, verification may be calibration of a thermometer
once per day while an example of monitoring is continuous
chart recording of the temperature of the scheduled pro-
cess. The NAS (4) pointed out that the major infusion of
science in a HACCP system centers on proper identifica-
tion of the hazards, critical control points, critical limits,
and instituting proper verification procedures. An example
of a verification activity is the scientific or technical
process to ensure that critical limits at CCPs are adequate
and effective. This is sometimes referred to as validation
of the HACCP plan. Verification activities should take
place during the development and implementation of the
HACCP plan (14). There are a few aspects involved in the
verification process. An important aspect of verification is
the initial validation to determine that the HACCP plan is
scientifically and technically sound and that the HACCP
plan, when properly implemented, will be effective in
controlling food safety hazards. The validation process
ensures that the HACCP plan in the facility is functioning
effectively. Another aspect of verification is evaluating
whether the HACCP system is implemented according to
the HACCP plan. This aspect includes ongoing verification
of CCPs that involves evaluating that day-to-day compli-
ance of the monitoring and other activities specified in the
HACCP plan at each CCP as well as a periodic HACCP
system compliance audit. A third aspect of verification
consists of periodic validations or reassessments, indepen-
dent of audits or other verification procedures, that must
be performed to ensure the accuracy and adequancy of the
Table 3. Partial Hazard Analysis, CCP and Critical Limits for Canned Salmon
a
Critical Control Point (CCP) Hazard(s) to Be Addressed
in HACCP Plan
Control Measures Critical Limits
Processing—processes
designed to provide
commercial sterility
C. botulinum Retorting Minimum 2501F for 3 min for product
Other critical factors as specified by
processing authority
Sealing of containers
b
C. botulinum due to
recontamination
c
Creating and maintaining
hermetic seal
No visible cutovers, seam sharpness,
false seams, deadheading, product
overlapping the flange, product
leakage, or other visual defects
Cover hook: minimum 0.70 in., body
hook 0.072–0.088 in.; width: maximum
0.125 in.; thickness 0.052–0.058 in.;
tightness minimum 70%
Cooling of containers—applies
to water-cooled cans only
Pathogens due to
recontamination
Chlorination Measurable residual chlorine at
discharge
a
Note: The partial HACCP plan in this table is for illustrative purposes only and it is not intended to replace the processor’s hazard analysis and HACCP plan
development.
b
Source for crticial limits at this CCP: FDA’s Fish and Fishery Products Hazards and Controls Guidance, 3rd edition (June 2001), except that the tightness
value (i.e., the percent of the cover hook that is free of wrinkles) is based on an industry guideline.
c
Some processors may conclude in their hazard analysis that recontamination with C. botulinum due to can defects is a hazard likely to occur in the absence of
control, in light of at least two botulism outbreaks associated with recontaminated canned salmon. In this case, critical limits are established for the CCP at
the sealing step to control the hazard.
570 HAZARD ANALYSIS AND CRITICAL CONTROL POINTS
HACCP plan. A fourth aspect of verification deals with the
government’s regulatory responsibility and actions to en-
sure that the HACCP system in the facility is functioning
satisfactorily. FSIS and FDA conduct risk-based inspec-
tions of facilities producing meat and poultry products and
seafood and juice products, respectively, which include
verification of the HACCP plan.
Principle 7: Establish Record-Keeping and Documentation
Procedures
Record-keeping provides written confirmation of the ha-
zard analysis, the HACCP plan, and the implementation
of the HACCP plan. HACCP records provide written data
for review, and records must be maintained for the
required period of time. Well-maintained records sub-
stantiate that the HACCP plan is being followed and
that requirements in the HACCP plan are being met
during production; HACCP records help document the
safety of the products being produced.
The approved HACCP plan and associated records
must be on file at the food establishment. Generally,
HACCP records include the following:
1. Summary of the hazard analysis.
2. The HACCP plan. NACMCF (8) recommends that
HACCP plan records include:
a. A list of the HACCP team and assigned
responsibilities.
b. A description of the product, its distribution,
intended use, and customer.
c. A verified flow diagram for the manufacturing
process with CCPs indicated.
d. A HACCP plan summary table that contains
information tabulated in Table 4, including the
CCPs, the hazards to be addressed in the HACCP
plan at each CCP, critical limits, monitoring
procedures, corrective action plan, verification
procedures, and record-keeping.
3. Supporting documentation.
4. Daily operational records.
HACCP AND PACKAGING
HACCP plan requirements in the manufacture of packa-
ging materials cannot be known until a hazard analysis
has been conducted. Methodical steps of the HACCP
process will prove most helpful in assessing the safety
versus the quality needs of the manufacturing plant. The
supplier may need records for its buyers that such an
assessment has been conducted even if no hazards have
been found during the hazard anlysis. Critical steps in
quality may come under different programs—for example,
ISO. Grocery Manufacturers Association and the National
Fisheries Institute favor interpreting HACCP as a pro-
gram that focuses only on food safety hazards. Thus, in the
case of milk carton manufacture, paperboard stock is
Table 4. HACCP Plan Master Sheet
Critical
Control Point
(CCP)
Hazard to be
Addressed in
HACCP Plan
Critical Limits
for Each
Control
Measure
Monitoring
Corrective
Action
Verification
Activities
Record-
Keeping
ProceduresWhat How Frequency Who
Source: Scott and Stevenson (14).
HAZARD ANALYSIS AND CRITICAL CONTROL POINTS 571
required by the Pasteurized Milk Ordinances (16) to have
no more than 50 bacterial colonies per 8 square inches. By
our interpretation, this would be a quality specification
and not a HACCP CCP, if historical data show that these
are spoilage microorganisms rather than pathogens that
are likely to cause illness.
End use of the packaging materials may lead to an
exception to the above rule that HACCP is not needed in
package manufacture. Some examples follow:
. Active packaging is now a very viable area of re-
search, and much of that research has focused on
incorporating into the material types of antimicro-
bials that are efficacious against microorganisms.
If such a compound/product is used in commercial
packaging and is itself an allergen to some popula-
tions, the labeling step may become a CCP, if the
HACCP team concludes in their hazard analysis that
the undeclared allergen is a hazard likely to occur.
In this case, the CCP is the label, the hazard is
the undeclared allergen, and the critical limit is
the proper notation on the label with visual monitor-
ing by the label operator or label scanner for the
correct label each time the labels are loaded. In some
cases, the HACCP team may conclude that the
hazard of an undeclared allergen is not reasonably
likely to occur due to a labeling prerequisite program
in place; in this case, no CCP would be needed.
. Gas combinations in modified atmosphere packaging
(MAP) may also prove critical in outgrowth of patho-
gens because oxygen concentrations below 2–3%
allow or enhance the growth of anaerobic organisms
such as Clostridium botulinum. Oxygen concentra-
tion would be a CCP in the MAP-packaging of mush-
rooms where growth of Clostridium botulinum is a
hazard reasonably likley to occur, for example. A
special case in fresh seafood packaging is vacuum
packaging. As noted in the Seafood Hazards Guide
(13), packaging material with an oxygen transmis-
sion rate (OTR) of 10,000 cc/m
2
/24 h has permeability
sufficient to allow aerobic spoilage to occur before
toxin can be produced. Selection of proper gas com-
binations and OTRs are CCPs in the cases of certain
commodities, where growth of anaerobic pathogens
such as Clostridium botulinum is a hazard reason-
ably likely to occur in the absence of control.
SUMMARY
HACCP is a systematic approach to assuring the safety of
food products based on the concept of prevention. The
HACCP program is widely used in the food industry in the
United States as well as worldwide. Although the seven
HACCP principles are well-developed and application
guidelines have been developed and revised based on
experience gained during HACCP implementation in the
last several decades, there is no universal formula for
putting together the specific details of a HACCP plan. The
plan must be specific to each facility and be dynamic,
allowing for modifications to production, substitution of
new ingredients and new materials (including specialized
packaging materials and packaging steps), and develop-
ment of new products. The plan is participatory at all
levels of management, both in formulating and managing
the plan. The strength in an HACCP program is in
providing a risk management framework that a company
can use effectively to organize and manage the safety of
the products which are produced.
BIBLIOGRAPHY
1. D. A. Corlett, Jr., HACCP User’s Manual, Aspen Publishers,
Gaithersburg, MD, 1998.
2. DHEW, Proceedings of the 1971 National Conference on Food
Protection, U.S. Dept. Health, Education, and Welfare, Public
Health Service, Washington, DC, 1971.
3. Pillsbury Company, Food Safety through the Hazard Analysis
Critical Control Point System, Contract No. FDA 72-59, Re-
search and Development Department, The Pillsbury Company,
Minneapolis, MN, 1973.
4. National Academy of Sciences, An Evaluation of the Role of
Microbiological Criteria for Foods and Food Ingredients, Na-
tional Academy Press, Washington, DC, 1985.
5. M. D. Pierson and D. A. Corlett, Jr., eds., HACCP, Principles
and Applications, Van Nostrand Reinhold, New York, 1992.
6. NACMCF (National Advisory Committee on Microbiological
Criteria for Foods), HACCP Principles for Food Production,
USDA, FSIS, Washington, DC, 1989.
7. NACMCF, ‘‘Hazard Analysis and Critical Control Point Sys-
tem,’’ Int. J. Food Microbiol. 16, 1 (1992).
8. NACMCF, ‘‘Hazard Anlaysis and Critical Control Point Prin-
ciples and Application Guidelines,’’ J. Food Prot. 61, 762–775
(1998).
9. Codex, ‘‘Hazard Anlaysis and Critical Control Point (HACCP)
System and Guidelines for Its Application,’’ Annex to the
Recommended International Code of Practice General Princi-
ples of Food Hygiene, FAO/WHO Codex Alimentarius Commis-
sion, Rome, 2003.
10. U.S. Department of Agriculture/Food Safety and Inspection
Service (FSIS), ‘‘Hazard Analysis and Critical Control Point
(HACCP) Systems; Final Rule,’’ 9 CFR Part 417, Federal
Register 61, 38868 (July 25, 1996).
11. U.S. Department of Health and Human Services/Food and
Drug Administration (FDA), ‘‘Procedures for the Safe and
Sanitary Processing and Importing of Fish and Fishery
Products; Final Rule,’’ 21 CFR Part 123, Federal Register
60, 65197 (December 18, 1995).
12. U.S. FDA, ‘‘Hazard Analysis and Critical Control Point
(HACCP); Procedures for the Safe and Sanitary Processing
and Importing of Juice; Final Rule,’’ 21 CFR Part 120, Federal
Register 66, 6197 (January 19, 2001).
13. U.S. FDA/Center for Food Safety and Applied Nutrition,
Fish & Fisheries Products Hazards & Controls Guidance,
3rd edition (online), Chapter 13, http://www.cfsan.fda.gov/
~comm/haccp4.html, 2001.
14. V. N. Scott and K. E. Stevenson, eds. HACCP: A Systematic
Approach to Food Safety, A Comprehensive Manual, 4th
edition, Food Products Association, Washington, DC, 2006.
15. U.S. FDA, Thermally Processed Low-Acid Foods Packaged in
Hermetically Sealed Containers, Title 21, Code of Federal
Regulations, Part 113, U.S. Government Printing Office,
Washington, DC, 1992.
572 HAZARD ANALYSIS AND CRITICAL CONTROL POINTS

16. U.S. FDA/Center for Food Safety and Applied Nutrition,
Grade ‘‘A’’ Pasteurized Milk Ordinance, 2001 Revision, Ap-
pendix J, Part C (online), http://www.cfsan.fda.gov/~ear/
pmo01j-k.html.
HIGH-VOLTAGE LEAK DETECTION (HVLD)
TECHNIQUE FOR HERMETICALLY SEALED
PACKAGES
YOON S. SONG
United States Food and Drug
Administration, Office of Food
Safety (OFS), Process
Engineering Branch,
Summit-Argo, Illinois
INTRODUCTION
High-voltage leak detection (HVLD) is one of the emerging
technologies for inspecting package defects and tamper-
ing. HVLD operates by applying a high-voltage potential
to electrically conductive products inside non- or semicon-
ductive package materials. Pinholes are identified when
electrical discharges are detected between products and
device electrodes. The HVLD system with different fre-
quencies has been successfully validated and used for
years to test the integrity of glass ampoules and vials
in the pharmaceutical industry (1, 2) and recently was
introduced into the food industry within the United
States. It has been demonstrated that this detection
system is a powerful tool to detect pinholes in vacuum
pouches containing frankfurters. Therefore, the applic-
ability of this high-voltage technique to various containers
of different types and compositions is of great interest to
the industry.
PRINCIPLES
The HVLD system is composed of four unique elements:
high-voltage power generator, support electrode, inspec-
tion electrode, and a detector (Figure 1). The power
generator subjects the outside of a test package to a
certain predetermined high voltage (0.1–25 kV). In typical
HVLD systems, a package wall is nearly nonconductive
and thus can be regarded as the dielectricum of a con-
denser. When high voltage is applied to the support
electrode on which a test package is placed, it electrifies
product particles inside the package. While high voltages
are applied, the surface of a test package is scanned with
an inspection electrode as shown in Figure 2. In the
presence of a leak, electrons are discharged through the
hole. The detector can then measure current and/or
voltage discharges through the inspection electrode. A
data acquisition system in connection with an oscilloscope
is used to measure voltage or current profiles discharged
from a package. When voltage discharges are measured,
V
10
(kV) value, a high-voltage applied to the supporting
electrode when voltage discharge measured from the
inspection electrode exceeds the threshold value of 10 V,
Package
Products
Supporting
Electrode
Inspection
Electrode
High-Voltage
Power Supply
+
−
−
−
−
−
−
+
+
+
+
+
+
Data Acquisition
(Oscilloscope)
Discharge Current
Detector
Figure 1. Schematic diagram of HVLD system setup.
(a)
(b)
Figure 2. (a) The front and (b) top views of the Type-JH manual
HVLD unit (see text for explanation of operation).
HIGH-VOLTAGE LEAK DETECTION (HVLD) TECHNIQUE FOR HERMETICALLY SEALED PACKAGES 573
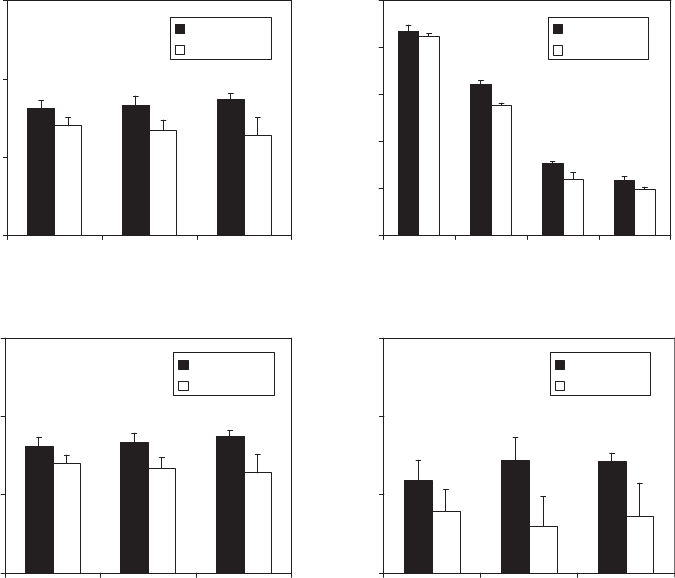
can be used as an indication of the detection. When
discharge current is monitored, the ability of the HVLD
to detect a leak is largely dependent on the gain setting.
With the gain control, a minimum current measured at a
constant high voltage applied can be adjusted and opti-
mized to within a measurable range to accommodate the
most important factors that could influence the detection
limit during a leak test.
FACTORS OF INFLUENCE
The ability of the HVLD system t o detect leaks can
be dependent upon various factors of product, package,
and defect. Jain and Song (3, 4) conducted a set of
experiments to evaluate the significance of various pro-
cessing var iables on detection of pinholes in flexible
pouches using a manual HVLD unit (Model DCC-1-AC,
Jovan Electronics Corp., Japan) (Figure 2). Sever al
critical variables, such as thi ckness (2.5, 4.5, 6.0 mil),
structure (plastic, paper and metallized laminates),
height (0.3, 0.7, 1.1 in . from a top of the support electrode
where a package is placed on), contact surface area (5, 11,
22, 33 in
2
), and dielectr ic constant (e) (1.63, 1.87, 2.01,
2.34) of pouches, volume (100, 200, 300 mL), electrical
condu ctivity (k) (0.7, 1.5, 8.5, 10.8 mS/cm), and type
(liquid, semisoli d (10% pectin), solid (0.85A
w
pecti n)) of
model products, an d location (0, 1.9, 3.7 in. from a
package center) and dia meter (10, 20, 30, 40, 50 mm) of
laser-drilled pinholes, were tested.
Effects of the critical variables on detection of 50-mm
pinholes in polypropylene (PP)/Nylon/PP laminated
pouches containing liquid model products are presented
in Figures 3 and 4 (3). In general, the greater the contact
surface area of packages and the higher the electrical
conductivity of products, the voltage required to detect
package defects is lower (po0.05). The statistical analysis
of data revealed that height and dielectric constant of
packages, product volume, and defect location were not
significant factors affecting detection by the HVLD unit
within the ranges tested (pW0.05), but HVLD responses
were significantly affected by thickness and surface area
of packages and by electrical conductivity of products
(po0.05). Results from a full factorial design experiment
with two levels and four factors (surface area, 5 and
33 in.
2
; film thickness, 2.5 and 4.5 mil; product conductiv-
ity, 1.5 and 10.8 mS/cm; pinhole size, 10 and 50 mm)
indicated that, among the independent variables, surface
area had the most significant effect followed by film
thickness, pinhole size, and product conductivity
0.45
0.55
0.65
0.75
0.27 0.66 1.08
Package height (in.)
V
10
(kV)
0.45
0.55
0.65
0.75
100 200 300
Food volume (ml)
V
10
(kV)
0.0
0.5
1.0
1.5
2.0
2.5
5112233
Contact surface area (in
2
)
V
10
(kV)
1.5 mS/cm
10.8 mS/cm
0.45
0.55
0.65
0.75
0.0 1.9 3.7
Distance from a geometrical center (in.)
V
10
(kV)
1.5 mS/cm
10.8 mS/cm
1.5 mS/cm
10.8 mS/cm
1.5 mS/cm
10.8 mS/cm
(b)
(a)
(
c
)
(
d
)
Figure 3. Effects of (a) package height, (b) package contact surface area, (c) product volume, and (d) defect location on V
10
values for
PP/nylon/PP laminated pouches (e = 2.01) with 50-mm pinholes, containing 100, 200, and 300 mL of liquid model products (k = 1.5 and
10.8 mS/cm).
574 HIGH-VOLTAGE LEAK DETECTION (HVLD) TECHNIQUE FOR HERMETICALLY SEALED PACKAGES
(po0.01) (4). Strong interactions between those variables
were also observed (po0.01). In particular, two evident
interactions—between film surface area and film thick-
ness and between product conductivity and film thick-
ness—contributed to the factorial model at a significant
level. HVLD responses to both paper/PP laminated
pouches and metallized pouches for different processing
variables were comparable with those from plastic lami-
nated pouches containing liquid model products. Similar
results were also reported for semisolid and solid model
products.
Moll et al. (5) validated the HVLD technique for 100%
inspection of low-dens ity polyethyle ne (LDPE) ampoules
by using a fully auto mated online unit (Model 920-004,
Rommelag, G ermany). Factors such as detector geometry,
electrical conductivity of semisolid model products
(7.8–8.5 mS/cm), and wall thickness of ampoules (20–
30 mil) were found to be critical in detecting defective
ampoules with 5- to 200-mm pinholes. They also suggested
that the temperatur e an d relative humidity at the site
of t esting, along with the speed of conveyor belt carry-
ing ampoules, may have an influence on equipment
sensi tivity.
LIMIT OF DETECTION
It has been reported that for plastic pouches, the HVLD
can detect pinholes as small as 10 mm, even under worst-
scenario conditions, such as 4.5-mil film thickness, contact
surface area of 5 in.
2
, and electrical conductivities of
0.7 and 1.5 mS/cm for semisolid and liquid products,
respectively (3). Limits of detections for liquid, semisolid,
and solid products were 10, 10, and 50 mm, respectively.
For metallized pouches, the HVLD can also detect
pinholes as small as 10 mm, even under worst-scenario
conditions, such as 6.0-mil film thickness, contact surface
area of 5 in.
2
, A
w
of 0.85 for solid products, and electrical
conductivities of 0.7 and 1.5 mS/cm for semisolid and
liquid products, respectively. The limit of detection was
10 mm for liquid, semisolid, and solid products. The HVLD
technique can also reliably detect 5- to 10-mm pinholes in
LDPE ampoules containing a medium-viscosity hydrogel
with conductivity of 7.8 mS/cm (5).
CONCLUSIONS
The HVLD is an innovative testing system for fully auto-
matic detection of package leaks and tampering. It requires
that products inside a package be in direct contact with
packaging material’s surface where the inspection elec-
trode is applied. The site on the package at which leaks
and/or tampering have occurred can be instantly and
effectively identified when electrical discharges are de-
tected between a product and device electrodes. Due to
high voltages applied, however, a defective package may be
further damaged, as evidenced by post detection results
where 5- to 10-mm pinholes were widened to 50–60 mm(5).
The HVLD technique is capable of detecting pinholes less
than 10 mm in diameter. Package surface area, thickness
and geometry, headspace, product type, and electrical con-
ductivity are parameters requiring testing to determine the
necessary system configuration. Parameters that do not
appear to be significant barriers to the use of HVLD were
height and dielectric constant of package, produce volume,
and location of pinhole. It can be concluded that the HVLD
technique is a promising nondestructive and online method
to detect pinhole defects, which may be applicable to a wide
range of hermetically sealed packages.
BIBLIOGRAPHY
1. P. Ruffieux, Pharm. Ind. 47, 976 (1985).
2. H. Barnickel, Pharm. Ind. 48, 388 (1986).
3. B. Jain, and Y. Song, Evaluation of a Non-destructive High
Voltage Technique for the Detection of Pinholes in Flexible
Pouches for Foods, Poster at IFT Annual Meeting, Chicago,
IL, 2007.
4. B. Jain, and Y. Song, Validation of a Non-Destructive High
Voltage Technique for the Detection of Pinhole Defects in F ood
Pouches , Poster at IFT Annual Meeting, New Orleans , LA, 2008.
5. M. Moll, D. L. Doyle, M. Haerer, and D. M., Guazzo, J. Pharm.
Sci. Tech. 52, 215–227 (1998).
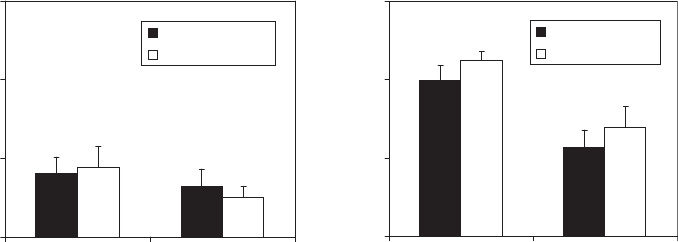
PET (ε = 1.87)
BOPA (ε = 1.63)
0.35
0.45
0.55
0.65
1.5 10.8
Electrical conductivity (mS/cm)
1.5 10.8
Electrical conductivity (mS/cm)
V
10
(kV)
0.35
0.45
0.55
0.65
V
10
(kV)
PET (ε = 1.87)
Nylon (ε = 2.01)
(b)(a)
Figure 4. Effects of dielectric constant (e) and electrical conductivity (k)onV
10
values for (a) 2.5- and (b) 4.5-mil-thick plastic pouches
(PET = PET/PE; BOPA = BOPA/PE; Nylon = PP/nylon/PP) with 50-mm pinholes, containing 100 mL of liquid model products.
HIGH-VOLTAGE LEAK DETECTION (HVLD) TECHNIQUE FOR HERMETICALLY SEALED PACKAGES 575
HOT-FILL TECHNOLOGY
RAYMOND A. BOURQUE
Ocean Spray Cranberries, Inc.,
Lakeville, Massachusetts
Food processors use a variety of processing, formulation,
and storage methods to assure that food products do not
spoil due to microbiological activity throughout their shelf
life. Products such as cereals, breads, and confectionaries
are formulated with sugars and/or salts to lower water
activity (a
w
). Water activity below B0.90 will not support
the growth of most food-spoilage bacteria (1). Chemical
preservatives, such as benzoate or sorbate, are added to
many products to inhibit microbiological growth. Many
dairy products, fruit juices, fruits, vegetables, meat, fish,
and prepared meals are distributed under refrigeration
or frozen conditions. Shelf-stable, low-acid foods—such as
vegetables, meat, and fish, with a pH Z 4.6—rely on a
thermal process. Thermal processing of low-acid foods is
achieved in a retort at pressures of 10–20 psi (7–14 kg/m
2
)
and temperatures of 240–2601F (116–1271C) (2,3). Food
products with a pHZ4.6 will support bacteria, yeast, and
mold growth. Commercial sterility is defined as the elim-
ination of all microorganisms that can grow and metabo-
lize in a product. To achieve commercial sterility in foods
with a pHZ4.6, bacteria must be destroyed as well as
yeast and mold. The destruction of many bacteria requires
temperatures in excess of 2121F (1001C). To achieve
temperatures above the boiling point, overpressurization
in a retort process is necessary. The high temperature
achieved not only destroys micro-organisms in the food
but also sterilizes the package. Packages historically used
for retorted low-acid foods include metal cans, glass jars,
and to a lesser extent, retortable pouches.
High-acid foods with a pHo4.6 support the growth of
yeast and mold but, unlike low-acid foods, only a limited
number of bacteria. Yeast, mold, and the types of bacteria
that can metabolize in high-acid foods are more tempera-
ture-sensitive than those that grow in low-acid foods.
Temperatures in the range of 170–2001F (77–901C) are
usually adequate to destroy these microorganisms. Com-
mercial sterility of high-acid food products and the ster-
ilization of the containers in which they are packed is
achieved with temperatures of o2121F (1001C). There-
fore, retorting with overpressure is not required. High-
acid foods are thermally processed in one of three different
ways: hot filling, postfill pasteurization, or pasteurization
with aseptic filling. This article focuses on hot filling.
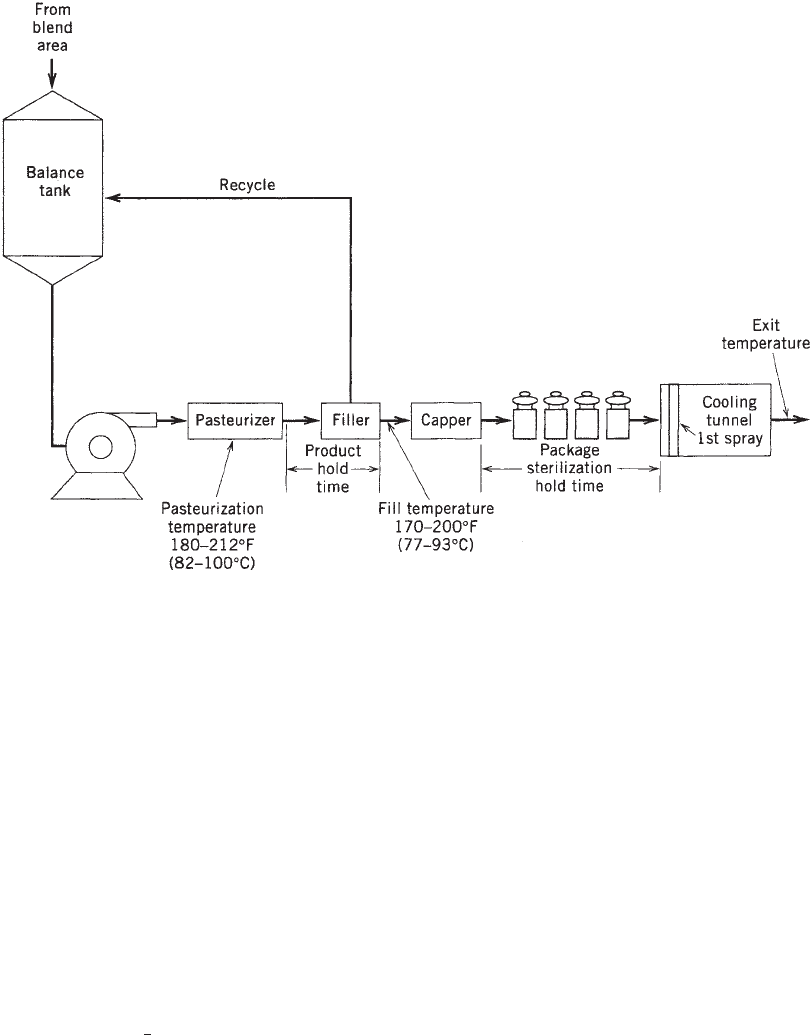
HOT-FILL PROCESSING
Foods such as fruit juices, fruit sauces, jams, jellies,
tomato sauces, ketchups, and barbeque sauces are often
hot-filled. The hot-fill process consists of heating the food
product to an adequate temperature to destroy the yeast,
mold, and limited types of bacteria that can grow in the
food, and then holding the food at that temperature
for an adequate time to destroy these microorganisms.
The temperatures required range from 170 to 2121F (77–
1001C), and the hold times vary from 30 to 60 s depending
on the food product. The hot product is then filled directly
into the container or package. The headspace in the
package may then be nitrogen- or steam-flushed to reduce
headspace oxygen. The package is then hermetically
sealed and inverted to sterilize the lid or closure. The
hot product sterilizes the package. To accomplish package
sterilization, the product must be at or above the mini-
mum temperatures [170–2001F (77–931C)] for an adequate
time (usually 1–3 min) (4). Thus, the control points for a
hot-fill process are the minimum fill temperature and the
minimum hold time (see Figure 1). Depending on the heat
sensitivity of the specific food, the products may or may
not be quickly cooled after the minimum hold time is
achieved. Cooling is usually accomplished in a water-
spray tunnel or immersion bath. Fruit juices and pre-
serves are examples of products that require cooling
directly after hot filling to protect product color, flavor,
and nutrient content (vitamin C). Barbecue sauce is an
example of a product that is not heat-sensitive and can be
allowed to cool under ambient conditions after being
packed in corrugated shipping containers.
Historically, high-acid products have been packaged
primarily in metal cans and glass jars. However, in the
1980s and 1990s, several rigid and flexible plastic
packages were developed for hot filling.
TINPLATE CANS
The traditional metal cans used for hot-filled foods are
made of tinplate. Some are enameled inside to protect the
food from the migration of iron or tin from the can into the
food, which may cause a loss of color or off-taste. Other
tinplate cans are not enameled inside; these are referred
to as plain tinplate cans. The lack of an inside enamel
allows the tin to migrate into the product. The color of
some canned products, such as white grapefruit juice, is
preserved by tin salts. Tinplate cans are also enameled to
protect
the can itself
from the corrosive effects of some
high-acid products. High-acid products, such as tomatoes
and cranberries, can corrode tinplate cans, which results
in perforations and leaking cans. The hot filling of tinplate
cans consists of hot-water washing the cans to remove
dust or other foreign contamination, hot filling the cans
with the high-acid food, flushing the headspace with
steam or nitrogen, double-steaming a metal lid onto the
can, inverting the can to sterilize the lid, and conveying
the hot can for an adequate distance to provide the
necessary hold time prior to entering a water-spray cooler.
The cans are cooled to approximately 90–1101F (32–431C).
The maximum cooler exit temperature is specified accord-
ing to the product’s heat sensitivity. The minimum cooler
exit temperature is established to ensure that the cans are
not too cool to prevent the evaporation of remaining water
beads. If the can is not warm, then rust will occur, staining
the label and distracting from the general appearance of
the can. If the can is too cool, then labeling may be difficult
as the hot melt used in labeling will set up too quickly,
which causes misapplied labels. A vacuum is generated in
576 HOT-FILL TECHNOLOGY
the can as it cools. The vacuum at the exit of the cooler
ranges from approximately 16 to 25 in. of mercury (41–
64 cm Hg) depending on the headspace, product density,
and size of the can. The vacuum is formed by a combina-
tion of the condensation of the steam in the headspace and
the contraction of the product as it cools. The vacuum is
important as it provides integrity to the can by pulling the
ends inward and not allowing them to flex during dis-
tribution. The concavity of the end caused by the vacuum
also provides visual evidence to the consumer that the can
is properly sealed.
LIGHTWEIGHT ALUMINUM CANS
In the 1980s, a new hot-filling process was developed that
allowed the hot filling of lightweight aluminum cans. This
technology is now used extensively for hot-filled fruit
juices and juice drinks in 11
1
2
¼ fl ¼ oz cans. Lightweight
aluminum cans could not be hot filled with traditional
processes because the aluminum cans would panel as a
result of the vacuum created when the product cooled. The
new hot-fill process includes dosing the filled cans with
liquid nitrogen immediately prior to applying and double-
seaming the can end. The cans consist of a drawn and
ironed aluminum body with an E-Z open aluminum end.
Directly following the dosing of the liquid nitrogen and the
application of the end, the nitrogen vaporizes and creates
internal pressure. In the case of hot fruit juices, this
nitrogen dosing creates an initial pressure of approxi-
mately 45–50 psi (31.6–35.1 kg/m
2
) prior to cooling and
25–35 psi (17.6–24.6 kg/m
2
) after cooling. Thus, the
nitrogen overpressure more than compensates for the
vacuum created by the product as it cools, and the final
package is overpressured to compensate for its light-
weight construction. The hot-fill nitrogen dosing process
for juice and juice drinks consists of cleaning the cans with
hot water or air, hot filling with juice, leaving a headspace
of approximately 0.3–0.4 in. (7–10 mm), dosing liquid ni-
trogen, applying the can end, double-seaming the can end
to the can body, and finally cooling the product to approxi-
mately 90–1101F (32–431C). This hot-fill process allows the
use of lightweight aluminum cans for fruit juice and offers
the potential for hot filling a variety of other food products.
(See also Cans, aluminum.)
GLASS CONTAINERS
Hot-filling technology has been used for ma ny years
for, not only tinplate cans, but also for vacuum-packed
glass jars and bottles. The hot-fill process used for glass
packages is very similar to that used for tinplate cans.
One main difference is that glass packages must be
preheated prior to hot filling to prevent breakage caused
by thermal shock. Th is problem is particularly trouble-
some in the w inter, when glass is either stored in cold
warehouses or filled dire ctly off delivery trucks. It is
gener ally belie ved that thermal-shoc k br eakage is not a
problem if the glass temperature after preheating is no
more than 751F(241C) less than the hot product. Thus, if
the prod uct fill temperature is 1851F (851C), then the
glass must be preheated to at least 1001F (431C) before
filling. The preheating process is usually accomplished
Figure 1. A typical hot-fill process for high-acid food or beverage products.
HOT-FILL TECHNOLOGY 577
