Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.

29. Nanotechwire.com. October 8, 2005. Available at http://
www.nanotechwire.com/news.asp?nid=2409&ntid=&pg=1.
Accessed Dec. 29, 2008.
30. nGimat, web information. Available at http://www.ngimat.-
com/barrier/anti_microb.html. Accessed December 22, 2008.
31. R. Lowry, Pira, Profit Through Innovation, web information.
Available at http://profitthroughinnovation.com/packaging/
antimicrobial-packaging.html. Accessed December 29, 2008.
32. R. Lowry, Pira, Profit Through Innovation, web information.
Available at http://profitthroughinnovation.com/company-
profiles/nano-materials-and-paper.html. Accessed December
29, 2008.
33. TopChim, web information. Available at http://www.topchim.-
com/?biopolymer. Accessed December 29, 2008.
34. A. ElAmin, Meatprocess.com, May 2, 2007. Available at http://
www.meatprocess.com/Financial/EU-funds-nano-packaging-
research. Accessed December 23, 2008.
35. Greenbox Systems, web information. Available at http://
www.greenboxsystems.com/index.htm. Accessed Dec. 29, 2008.
36. Nanotechnology Now, March 18, 2008. Available at http://
www.nanotech-now.com/news.cgi?story_id=28562. Accessed
December 29, 2008.
37. Small Times, June 14, 2004. Available at http://www.smallti-
mes.com/articles/stm_print_screen.cfm?ARTI-
CLE_ID=269497. Accessed December 29, 2008.
38. EcoSynthetix, web information. Available at http://www.eco-
synthetix.com/. Accessed December 30, 2008.
39. M. Taylor, Assuring the Safety of Nanomaterials in Food
Packaging: The Regulatory Process and Key Issues, Woodrow
Wilson International Center for Scholars, Washington, DC,
2008.
NETTING, PLASTIC
JIM OHLINGER
Tipper Tie-Net, West Chicago,
Illinois
INTRODUCTION
Today there are two popular manufacturing methods
to produce plastic netting: knitting and extrusion.
Both methods produce a tubular or sheeted netting pro-
duct in a wide variety of mesh sizes, diameters, widths,
and colors. Plastic netting is purchased as individual
pieces, in continuous rope form, or sheared onto cardboard
mandrels.
The primary materials used to produce plastic netting
are high-density polyethylene (HDPE) and polypropylene.
Ultraviolet inhibitors can be added to the resin to give the
netting extra durability in direct sunlight applications.
HDPE has a high elongation value and must undergo
the process of orientation to achieve its maximum tensile
strength. The process of orientation is when the material
is heated and then stretched to the point just before
breakage. This process also keeps the netting from droop-
ing when exposed to heat applications. The melt index of
HDPE is 2081F. Polypropylene has a high melt index
(3251F), and it is used when temperature requirements
exceed 2081F. Polypropylene tensile strength is almost
twice that of HDPE with the equivalent weight ratio.
With the heat and strength attributes, polypropylene
netting is widely used in the meat–poultry industry.
The process of extruding plastic netting was invented
in the mid-1950s. Extruded netting is produced through
counterrotating dies. As the inner and outer die rotate,
small strands of molten plastic overlap each other, bond-
ing themselves together where they overlap. The extruded
plastic is then cooled by water; this material is called a
cast. The next process is orientation. The cast is heated
and stretched to the point just before breakage. After the
cast is oriented and has become the final product, the net
is transverse-wound onto large spools (1).
MANUFACTURE
The manufacturing process of knitted plastic netting
offers the most versatile and variety of netting products.
Knitting machines offer simple and sophisticated stitch
patterns. The first knitting machine was invented by
Reverend William Lee, an Englishman, in 1589. The first
power knitting machine was introduced in 1832, in Cohoe,
New York by Egberts and American (2). The knitting
process is complex and varies by different types of ma-
chinery. To manufacture plastic netting via knitting, the
plastic resin must be manufactured into yarn or tape. The
yarn is then put onto a creel, with several individual
packages consisting of a single thread, or beam, and
several threads wound onto a large spool. The basic
concept of knitting is to take the yarn or plastic tape
from the creel or beam, and thread the set of needle guides
in the knitter. A bed of latch hooks move up and down
(vertically) as the needle guides move right and left
(horizontally). The needle guides are directed by a pattern
chain that controls the different mesh patterns; hexagon,
diamond, or countless other configurations. Knitting al-
lows the manufacturer to choose the style and the mate-
rial to be used, allowing a multitude of products.
APPLICATIONS
Plastic netting is used in an array of applications. As a
flexible material, it conforms to irregular products (toys,
houseware products, etc.). Netting provides an excellent
packaging forum as a decorative and protective overwrap.
It provides the necessary air circulation for products such
as plants (tropical and flowering) and various types of
fruits and vegetables. Because of plastic netting’s flexibil-
ity, it can be looped, making a convenient carrying handle
for the consumer (fresh or frozen whole turkey and ham
products). In the meat–poultry industry, netting is used as
a transportation vehicle. The meat and poultry items are
encased in the netting in the production area and then
hung on smoke racks ready to go into the smoke house.
The netting allows the meat and poultry products to
receive smoke for flavoring and coloring during the
818 NETTING, PLASTIC
cooking process. The netting leaves an attractive pattern
on the products, giving them the ‘‘old world’’ look. The
netting can be left on the product as a marketing tool for
product identity.
Netting has a wide variety of uses in sheet form. It
can be used to wrap pallets as an alternative to stretch
wrap. The netting can be put up the same way as stretch
wrap, to utilize modern equipment and hand applicators
that are available. This is an important tool where venti-
lation or heat dissipation is needed (produce, bagged
mulch, flowers, flour, etc). Because of its exceptional
strength, netting can be used to wrap heavy loads such
as bricks. In sheet form, netting has an abundance of uses.
It makes a great fence for construction or snow applica-
tions. It can be used as a safety net in construction to catch
debris and increase the safety of workers. It is widely used
in the horticulture industry as shade cloth protecting
plants from the harsh sun. It is also used as bird net,
protecting fruit trees, berry bushes, and fish ponds from
predator birds.
By using the knitted process to manufacture plastic
netting, different-color strings or threads can be combined
to form a multicolored netted product. An example is
Candy-Cane (trademark) red and white netting used to
package Christmas trees (3). This netted package allows
the tree to be easily transported from the retail lot to the
consumer’s tree stand at home. Netting is also used by
Christmas-tree growers to compress the tree. This gives
the grower the ability to ship more trees, saving in
transportation cost. The netting also protects the tree
from damage, and it makes the tree much easier to handle.
This packaging process also applies to other types of trees
(white oak, maple, bamboo, etc.).
Knitted netting is soft, durable, and strong, making it
an ideal packaging medium. The produce industry, which
uses extruded netting primarily for consumer-sized
packages, is seeing a trend moving toward knitted netting.
Both styles of netting offer adequate ventilation needed
for fruits and vegetables to reduce spoilage. The soft
texture of knitted netting does not damage the delicate
produce skin and makes an eye-pleasing package. A draw
string can be automatically inserted during the knitting
process, making a convenient closing mechanism as well as
a carrying handle. Potato sacks are a good example of this
process.
Plastic netting is a valuable packaging tool. It is
resilient, strong, and flexible. Netting can be frozen and
then heated, or vice versa, keeping its strength and
flexibility. Plastic netting is very cost-effective compared
to other forms of packaging and is recyclable. The applica-
tions and areas of usage keep growing as creative minds
keep developing new and innovative ways to use plastic
netting.
Some manufactures are
Tipper Tie-Net, 390 Wegner Drive, West Chicago, IL
60185
Polly Net Inc., P.O. Box 27, Three Rivers, MA 01080
C&K Manufacturing, 28025 Ranney Parkway, Wes-
tlake, OH 44140
BIBLIOGRAPHY
1. J. Scoba, Polly Net Inc., P.O. Box 27, Three Rivers, MA 01080.
2. The World Book Encyclopedia, Field Enterprises Educational
Corporation, 1970, pp. 279–280.
3. ‘‘Candy Cane,’’ manufactured by Tipper Tie-Net, 390 Wegner
Drive, West Chicago, IL 60185.
Cited Publications
J. Ohlinger, ‘‘Netting, Plastic’’, in A. J. Brody and K. S. Marsh,
eds., 2nd ed., The Wiley Encyclopedia of Packaging Technol-
ogy, 1997, pp. 666–667.
NITRILE POLYMERS
PAUL R. LUND
JOSEPH P. M CCAUL
The BP Chemical Company,
Cleveland, Ohio
INTRODUCTION
Nitrile polymers are generally those that contain the
cyano (C:N) functional group, also called the nitrile
group. The commercial development of these materials
was due in large part to the 1957 discovery by BP
Chemicals of a low-cost one-step process for acrylonitrile
(AN) production (1). The pure nitrile polymer, polyacrylo-
nitrile (PAN), is 49% nitrile. It is an amorphous, trans-
parent polymer with a relatively low glass-transition
temperature (T
g
=871C) (see Polymer properties) that
provides an outstanding barrier to gas permeation and
exceptional resistance to a wide range of chemical re-
agents. Unfortunately, its combination of properties is not
of commercial value in the packaging industry. Its pri-
mary deficiency is that it is not melt processable. It
degrades at 4281F (2201C), which is below that required
for melt processing. To overcome this handicap, nitrile
polymers are produced using acrylonitrile (CH
2
= CHCN)
as the monomer with other comonomers that impart melt
processability. Through this copolymerization process, the
desirable properties can be retained and the undesirable
properties can be suppressed.
COPOLYMERS
Styrene–Acrylonitrile (SAN)
Typical SAN polymers are made using a 3:1 ratio, by
weight, of styrene to acrylonitrile. The copolymer has a
combination of properties that reflect the processability of
the styrene component and the chemical resistance of the
AN. Gas-barrier properties are low because of the relatively
low AN concentration. SAN is of relatively minor signifi-
cance in packaging, used in applications where PS would
suffice with an added measure of chemical resistance.
NITRILE POLYMERS 819
Acrylonitrile–Butadiene–Styrene (ABS)
ABS is a graft copolymer of SAN onto a polybutadiene
backbone. The SAN forms a matrix phase, whereas the
polybutadiene forms a discrete (dispersed) phase. A tough
impact-resistance thermoplastic is produced by using the
grafting mechanism to compatibilize the two phases.
Although the polybutadiene is an excellent impact modi-
fier with low T
g
(D 851C), its refractive index is different
from the SAN matrix polymer. Therefore, in contrast to PS
(see Polystyrene), PAN, and SAN are not transparent.
ABS is a major commercial thermoplastic, but it is rarely
used in packaging because of its opacity, lack of gas-
barrier properties, and economics relative to other com-
modity resins. However, thermoformed ABS (see Thermo-
forming) has been used, for example, to produce
margarine tubs. This is an application in which PS would
suffice if not for its limited resistance to stress cracking. A
wide range of SAN ratios is used to achieve properties of
value in nonpackaging applications.
Acrylic Multipolymers
Some acrylic multipolymers are produced using AN as a
comonomer with methyl methacrylate (MMA). Refractive
index matched rubber modifiers are incorporated to com-
bine toughness with transparency. These materials are
used in healthcare packaging (2) (see Medical packaging)
and in some food packaging applications. Because the AN
concentration is low, these materials do not have excep-
tional gas-barrier properties [18.7 cm
3
mil/100 in.
2
d atm
at 731F (23 1 C)].
High-Nitrile Resins (HNR)
As noted above, SAN is a styrene–acrylonitrile copolymer.
Its gas-barrier properties are limited, however, by the low
nitrile content. High-barrier melt-processable copolymers
can be produced by raising the nitrile content above 25%.
Over this threshold, copolymer properties begin to resem-
ble those of PAN, particularly with respect to gas barrier
and chemical resistance.
Acrylonitrile–Styrene Copolymers (ANS). High-nitrile co-
polymers can be produced by combining acrylonitrile and
styrene in a 70:30 ratio. This was the approach taken by
Monsanto Company (Lopac) and Borg-Warner (Cycopac)
during the 1960s and early 1970s in their development of
resins for carbonated-beverage packaging. Lopac’s compo-
sition is 100% copolymer of 70% acrylonitrile and 30%
styrene. Cycopac’s composition is 90% copolymer of 74%
acrylonitrile and 26% methyl acrylate plus 10% butadiene
rubber graft (3). As serious commercial development
began, toxicological problems with AN surfaced (4). The
FDA banned the use of HNR in beverage packaging
because of concern for potential AN-extraction from the
bottle into the beverage. Since beverages are a major
component of the diet, their treatment by FDA was most
severe. The FDA continued to permit the use of HNR for
direct and continuous nonbeverage-food-contact applica-
tions (5) with filling and storage temperatures less than
1501F (65.61C); but because the principal commercial
significance of ANS polymers was in carbonated beverage
packaging, all commercial production of ANS polymers
was eventually discontinued.
In 1984, the FDA amended its position on HNR-bev-
erage applications (6). Monsanto Company had petitioned
on behalf of their ANS high nitrile resin for approval. As
described in their process patent (7), the bottle preform is
irradiated with an electron beam prior to blowing of the
bottle. Monsanto claimed that this process resulted in a
bottle that, because of the thermodynamics of the extrac-
tion process, would have essentially no extraction of AN by
the contained beverage. The FDA did not accept this
claim, but it did decide that the extraction would be below
the detection limit of 0.16 ppb. The agency ruled that AN
concentrations at or below this detection limit would be
considered acceptable, and it limited the residual AN
content of the finished container of 0.1 ppm. This concen-
tration is current (8).
Rubber-Modified, Acrylonitrile–Methacrylate Copolymers
(AN/MA). High barrier properties can also be achieved by
copolymerizing acrylonitrile and methacrylate in a 75:25
ratio onto a nitrile rubber backbone. This is the approach
taken by BP Chemicals in the production of Barex (regis-
tered trademark) resins. Barex’s composition is 90% copo-
lymer of 74% acrylonitrile and 26% sytrene plus 10%
butadiene rubber graft (3). This family of resins can be
processed on almost all conventional plastic processing
equipment including, but not limited to, injection, injec-
tion blow molding, injection stretch blow molding, extru-
sion, extrusion blow molding, extrusion stretch blow
molding, profile extrusion, pipe extrusion, tubing extru-
sion, and thermoforming (see individual processing sec-
tions). Transparency is retained through the use of a
refractive-index-matched rubber modifier.
The Barex family of resins are the only high-nitrile
resins in commercial production today and are in full
compliance with all applicable FDA regulations for direct
food contact and meets all the known requirements of a
USP Class VI plastic. Barex is now marketed by INEOS.
PROPERTIES
Among the commercial packaging polymers that have the
physical properties required for monolithic structures,
HNRs offer the greatest gas barrier [0.8 cm
3
mil/
100 in.
2
d atm or 3.0 cm
2
mm/m
2
d kPa] and chemical resis-
tance. Their gas-barrier properties (see Barrier polymers)
are surpassed only by EVOH (see Ethylene–vinyl alcohol)
and PVdC (see Vinylidene chloride copolymers), which are
used as components of multilayer structures (see Coex-
trusions for flexible packaging; Coextrusions for semirigid
packaging). Because of the polarity that the nitrile group
imparts to the molecule, HNRs show an affinity for water.
Water-vapor barrier is lower than that of the nonpolar
polyolefins (e.g., polyethylene, polypropylene), but that
same polarity imparts resistance to nonpolar solvents.
The relatively high flexural modulus (combined with the
lower specific gravity) means that for structures with
identical geometry, the HNR parts can be source-reduced
820 NITRILE POLYMERS

(down-gauged) and therefore designed with less material
for equivalent stiffness compared to the polyolefins, PVC,
PET, PETG, and many other plastic polymers. A summary
of the properties of high-nitrile resins is shown in Table 1.
APPLICATIONS
HNRs are used in packaging in a variety of physical forms.
These include: film, semirigid sheet, and injection-molded
(see Injection molding) and blow-molded containers. Blown
film (see Extrusion) is used in polyolefin-container struc-
tures to provide formability, chemical resistance, and gas
barrier. Spices , medical devices, and household chemical
products are example of such applications . Laminations
with polyolefins and aluminum foil are used in applications
ranging from food packaging to oil-drilling core wraps (9).
These structures have exceptional barrier properties, as
well as sealability and chemical resistance. Indeed, their
use in sachet packaging is growing rapidly as individual
dose and unit packaging become more popular.
The semirigid sheet market for HNR is primarily meat
and cheese packaging in thermoformed (see Thermoform-
ing) blister packages. With its excellent gas barrier,
clarity, and rigidity, the HNRs are the premium packaging
material. Of increasing importance in semirigid applica-
tions, however, is disposable medical device packaging
(see Medical packaging). Here HNR can be sterilized by
either ethylene oxide (ETO) or gamma radiation and is
unaffected by plasticizers present in many devices, mak-
ing it ideal for many medical packaging applications. As a
result of its unique combination of properties, including
source reduction and superior thermoforming, usage has
been steadily increasing in this market area.
Blow-molding applications for HNR are dominated by
chemical resistance requirements. Injection blow molding
is the most widely used method for manufacturing small
containers—for example, bottles for correction fluid, nail
enamel, and other cosmetics. Larger bottles are generally
extrusion blow-molded. Some important applications in-
clude: pesticides, herbicides and other agricultural chemi-
cals, fuel additives, and hard-to-hold household chemicals.
Extrusion stretch blow molding is gaining increasing
acceptance for bottles 16 oz (473 mL) or larger. The orienta-
tion achieved during stretching greatly increases the drop-
impact performance, and the walls can be relatively thin.
HNR has been coextruded with many different poly-
mers, but the polyolefins have been of greatest commercial
significance. Coextrusions are available in sheet, film, and
bottle form. They typically gain gas barrier or chemical
resistance from the HNR and water vapor barrier and
economics from the polyolefin. In structures with poly-
propylene, the heat-deflection temperature (HDT) of the
structure is increased by the higher HDT of the polypro-
pylene. This permits the use of HNR in high-temperature
environments such as microwave ovens.
The adhesive used to combine the layers in an HNR
coextrusion are typically styrene–isoprene or styrene–
butadiene block copolymers (see Multilayer flexible packa-
ging). Scrap is reusable in the polyolefin layer if the
nonolefin percentage in that layer is well-dispersed and
of lower concentration than about 15%.
Chemical-resistant coextruded bottles containing
HNR are now being commercialized. HNR is the inner
contact layer enclosed by adhesive and polyolefin, typi-
cally HDPE. Use of a three-layer structure limits the cost
and complexity of the machinery. It also places the sol-
vent-resistant polymer in contact with the chemicals. Five
layer or laminar structures (see Surface modification;
Nylon) using other barrier resins place the polyolefin in
direct contact with the aggressive contents of the con-
tainer. The three-layer structure also allows visual inspec-
tion of the barrier layer and maximizes the sealing area of
the barrier layer at the pinchoff of the bottle. HNR-
coextruded containers offer high performance with eco-
nomics superior to the other packaging alternatives (10).
Thermoplastic coextruded films of 5–7 layers have been
reported (11).
Alloying and blending also offer other unique proper-
ties that can be imparted to the base resins, typically
polyolefins. Here they can be used to reduce ESCR as well
as provide increased flexural modulus (stiffness) to poly-
olefinic resins (12).
The Barex family of resins offers a unique combination
of properties to the packaging industry. Barex resins
protect products from oxidation and are inert and resis-
tant to chemicals. They meet United States and European
regulations and, thus are available for a range of products,
They eliminate the need for a separate heat-seal layer and
are good against strong ingredients. As bottles, they keep
fragrances and flavors in place. The following are applica-
tions of Barex (13).
Table 1. Properties of High Nitrile Resins
Property ASTM Test Units Barex 210 Resin Barex 218 Resin
Specific gravity D1505 1.15 1.11
Yield in.
2
/lb at 1 mil 24,080 24,950
Brabender torque meter-grams 950 1050
Notched Izod D256 ft-lb/in. 5.0 9.0
Flexural modulus D790 psi 490,000 400,000
O
2
permeability (731F, 100% RH) D3985 cm
3
-mil/100 in.
2
-24 h-atm 0.8 1.6
CO
2
permeability (731F, 100% RH) D3985 cm
3
-mil/100 in.
2
-24 h-atm 1.2 1.6
N
2
permeability (731F, 100% RH) D3985 cm
3
-mil/100 in.
2
-24 h-atm 0.2 0.4
WVTR (1001F, 90% RH) F1249 g-mil/100 in.
2
-24 h-atm 5.0 7.5
Heat deflection temperature D648 1F 160 170
Heat seal temperature 1F 250–375 275–350
NITRILE POLYMERS 821
1. Medicinal Uses. Catheter trays, sachets, assay
trays, transdermic patches. These products are
sterilizable.
2. Personal Care. Cosmetic packs, perfume, mouth-
wash, nail enamel, and bath gel bottles.
3. Industrial Products. Among many nonpackaging
uses, criminal evidence bags and air fresheners.
4. Food and Beverages. Fruit juices, meats, and
confectionary.
BIBLIOGRAPHY
P. R. Lund and J. P. McCaul, ‘‘Nitrile Polymers’’ in A. J. Brody and
K. S. Marsh, eds., The Wiley Encyclopedia of Packaging
Technology, 2nd edition, Wiley, New York, 1997, pp. 669–672.
Cited Publications
1. J. D. Idol, U.S. Patent 2,904,580 (to BP Chemical, OH),
September 15, 1959.
2. J. M. Lasito, ‘‘Acrylic Multipolymers in Medical Packaging,’’
Proceedings of the TAPPI 71, 74, (1982).
3. P. Delassus, ‘‘Barrier Polymers’’ in Kirk-Othmer Encyclopedia
of Chemical Technology, Vol. 3, 5th edition, Wiley, Hoboken,
NJ, 2004, p. 386.
4. Report to the FDA by the Manufacturing Chemists Associa-
tion, January 14, 1977.
5. Federal Register 41 23,940. Title 21, Part 177.1480 (June 14,
1976).
6. Federal Register 49 36,637. Title 21, Part 177 (September 19,
1984).
7. M. Salome and S. Steingher, U.S. Patent 4,174,043 (to Mon-
santo), November 13, 1979.
8. 21CFR section 177.1041 (April 1, 2007).
9. P. K. Hunt and S. J. Waisala, U.S. Patent 4,505,161 (to BP
Chemical, OH), March 19, 1985.
10. J. P. McCaul, ‘‘The Economics of Coextrusion,’’ Proceedings of
the High Technology Plastic Container Conference, SPE,
November 11, 1985.
11. A. J. Lischefski, U.S. Patent 7,201,966 (to Curwood, Inc.),
April 10, 2007.
12. P. R. Lund and co-workers, ‘‘High Nitrile Polymer/Polyolefin
Blends: A Low Cost, High Performance Alternative,’’ Proceed-
ings of the 1995 Annual Technical Conference, SPE, May 7–
11, 1995.
13. Barex, brochure, INEOS, www.INEOS.com. accessed Febru-
ary 2008.
NONWOVENS
J. ROBERT WAGNER
Philadelphia College of Textiles
and Science, Philadelphia,
Pennsylvania
INTRODUCTION
‘‘Nonwovens are fabric-like materials consisting of a con-
glomeration of fibers that are bonded in some way or
other’’ (1). The total world wide sales of nonwovens in
2006 amounted to 19 billion dollars (5 109 t) (2). Non-
wovens may be sold by a yield or area basis, weight basis,
or product unit basis. Because of their unique properties
and high production rates, nonwovens offer a substan-
tially high performance/price ratio. However, in order to
appreciate the full range of products, properties, and end
uses of nonwovens, it is necessary to consider the fibers,
the web-formation method, and the bonding methods used
to make them.
FIBERS
The primary ingredient of nonwovens are the fibers used to
produce them. A fiber is substantially longer in length than
in diameter. In nonwovens, fibers may vary in length from
1 mm to a continuous length. Short (1-mm) fibers are used
in air-laid or wet-laid nonwovens. Spunbonded nonwovens
use continuous-length fibers in their fabrication. The
length of the fibers will affect both the uniformity of fiber
distribution and the strength of the nonwoven. Short fibers
tend to give good fiber distribution, but longer fibers tend to
produce greater strength. However, the fiber length actu-
ally utilized is dependent on the nonwoven-web-formation
process and the type of product being manufactured.
The diameter of the fiber affects the properties of the
nonwoven considerably. If it was desired to produce a soft
nonwoven facial wipe, baby wipe, or polishing cloth, a fine
fiber should be used. If, on the other hand, it was desired
to produce a scrubbing pad of the required stiffness for a
frying pan, large-diameter fibers should be used. The
latter would also give high incompressibility or resilience.
While many fibers are perfectly round, some are trian-
gular, square, hollow, and of other shapes. In addition,
they may consist of one or more generic type of materials
in their makeup.
One major variable in nonwovens is the type of fiber
used in its construction. Virtually any fiber can be utilized
to make nonwovens. The principal natural fibers found in
nonwovens are cotton, jute, and especially wood pulp. The
majority of spunbonded fabrics are based on either isotactic
polypropylene or polyester. Small quantities are made from
nylon 6,6 and an increasing tonnage from flash spun high
density polyethylene (3). Because of their far-reaching
properties, the range of fiber types available offer great
latitude in the performance characteristics of the final
nonwoven. Because of their high hydrophobic properties,
moisture-barrier properties are provided by polypropy-
lene, polyethylene, polyester, and polytetrafluoroethylene
(PTFE). High tensile strength may be obtained using
Kevlar aramid, Spectra olefin, nylon, polyester, silk, and
glass. High-temperature resistance or low flammability can
be obtained with carbon, glass, Kynol novoloid, Kevlar and
Nomex aramid, modacrylic, PBI, PTFE, ceramic, and me-
tallic fibers. For composites, glass, carbon, Kevlar, and
Nomex aramid may be used. Chemical resistance may be
obtained with PTFE, glass, carbon, acrylic, olefin, and some
metallic fibers. It should be apparent, therefore, that proper
fiber selection is essential in order to ensure desirable end-
use performance characteristics of the nonwoven.
822 NONWOVENS

NONWOVEN WEB-MANUFACTURING METHODS
Nonwoven webs can be produced in four primary ways:
carding, air laying, wet laying, or spunbonding. Within
each one of these, the process and resulting product will
often vary considerably.
A carding machine feeds a mass of crimped fibers,
opens, disentangles, and drafts the fibers and then delivers
a thin wide web of uniformly distributed fibers to some
form of delivery apron. The fibers used to produce carded
nonwovens must have crimp and will usually have a fiber
length ranging between 34 and 152 mm.The fibers exiting
the card tend to be aligned in the machine direction, and, if
used in this manner, the product is referred to as a
parallel-laid nonwoven. Often it is necessary to combine
the web from several carding machines to achieve the
desired nonwoven weight. A parallel-laid nonwoven
may be up to 11 times stronger in the machine direction
or fiber-oriented direction than it is in the cross-machine
direction. Also, the elongation in the machine direction is
substantially less than that found in the cross-machine
direction.
The web exiting the card may be laid back and forth
across a moving apron to produce what is known as a
cross-laid nonwoven. Because systems of fibers cross each
other, the resulting nonwoven has more uniform strength
and elongation properties than found in parallel-laid
nonwovens.
Sometimes a parallel-laid web is combined with a cross-
laid web to produce what is termed a composite carded
nonwoven. The latter has the greatest uniformity in
strength and elongation properties of carded nonwovens.
In randomized carded nonwovens the card is similar to
that used for parallel laying, but the doffer roll is run in a
direction counter to that of the main cylinder at the point
of near-wire contact. Such a configuration deparallelizes
the web, permitting more loft and greater uniformity of
strength and elongation in the machine direction and
cross direction.
Because of the great latitude in weight, thickness, fiber
orientation, strength, elongation, and fibers utilized,
carded nonwovens have a vast number of product end-
use applications.
Air-laid nonwovens utilize fibers having a length ran-
ging between 1 and 76 mm.The fibers are first opened,
then conveyed with an air stream against either a con-
densing cylinder or a screen to form the web. The product
produced tends to have an x, y, z-fiber orientation, and as a
result the webs are soft and lofty. Such a web is especially
useful in padding, insulation, filtration, and wipes.
Wet-laid nonwovens utilize papermaking equipment
and technology. The fiber length ranges between 1 and
38 mm.The longer fibers are either glass for roofing shin-
gles and felt or carbon for composites. Shorter natural and
synthetic fibers are used in tea bags, automotive air and
oil filters, coffee filters, bunting, hang tags, and compo-
sites. While flat-tabled fourdrinier machines have been
used to make some nonwovens, the best machine for
producing wet-laid nonwovens is the inclined-wire ma-
chine since long fibers are held in suspension in water at
the time of formation on the moving wire. Often multiple
headboxes are used to produce layered or stratified high-
efficiency air filters and other products.
Spunbonded and melt-blown nonwovens (see Figure 1)
are produced at the time the fiber is extruded from the
spinneretts. Spunbonded nonwovens are produced with
continuous filament fibers that are drafted with either
multiple drafting rollers or high-velocity air used in
combination with a venture to achieve molecular fiber
orientation. Most spunbonded nonwovens are produced
with polypropylene or PET. However, polyethylene, polye-
ster, nylon, acrylic, and rayon are also used.
Melt-blown nonwovens are similar to spunbonded non-
wovens but instead use a spinnerette in which both the
polymer and hot air exit the spinnerette from different
orifices simultaneously. The velocity of the air is sufficient
to draft the polymer into very fine discontinuous fibers.
Melt-blown nonwovens are used for thermal insulation,
filters, padding, personal products, synthetic leather, and
other products.
Flash spinning is a radical departure from conven-
tional melt spinning. It begins with a 10–15% polymer
solution prepared by dissolving a solid polymer, such as
high-density polyethylene, with a suitable solvent. The
solution is pressurized and then permitted to expand
rapidly through a hole in a spinnerette. The solvent is
instantaneously flashed off, leaving behind a three-dimen-
sional film-fibril network called a plexifilament. These
plexifilaments are substantially oriented and possess re-
latively high tenacities. Flash spinning is the most com-
plex and sophisticated method for manufacturing
spunbonded fabrics (3).
BONDING METHODS
There are four principal methods of bonding nonwovens:
chemical, mechanical, thermal, and inherent or self-bond-
ing. Chemical bonding may rely on binders, solvents, or
hydrophilic fibers. Of these, binders are by far the most
important. Binders act like adhesive and include acrylic,
phenol, urea and melamine formaldehyde, poly(vinyl
Figure 1. Illustration of a polypropylene melt blown nonwoven
that is bonded using a heated pattern engraved calender roll.
Such a product is manufactured using a fine oil-loving fiber and is
excellent for use as a shop towel or wipe.
NONWOVENS 823
acetate), styrene–butadiene rubber, vinyl acetate–ethylene,
poly(vinyl chloride), nitrile rubber, poly(vinyl alcohol), and
other lesser binders (4). Such binders vary considerably in
cost, properties, and performance characteristics.
Mechanical bonding includes needle punching, stitch-
through, and hydroentangling. Needle punching may use
either barbed or forked needles. Barbed needles interlock
the fibers to provide mechanical strength to produce such
products as papermaker’s felts, blankets, geotextiles, syn-
thetic shoe leather, thermal lining, and automotive trunk
liners. The forked needles can produce velour and pat-
terned products for use as institutional and automotive
carpeting, and throwrugs.
Stitch through technology is a knitting-through process
that may be accomplished either with or without yarns to
stitch the nonwoven web together. The Mali and Arachne
machines are most common and produce products such as
carpets, upholstery, blankets, interlining, geotextiles, and
automotive products.
The hydrogenating process was invented by E. I.
DuPont and was first referred to as the spunlace process.
It utilizes very fine water jets to mechanically entangle
fibrous webs.
Thermal bonding may include the use of thermoplastic
fibers, yarns, powders, or films. Often bicomponent fibers
having differential melting temperatures in either a core-
and-sheath or a side-by-side configuration may be utilized.
Bonding is facilitated by through-air heaters, and smooth
or patterned calender rolls.
Inherent or self-bonded nonwovens are bonded at the
time they are produced. This might include film splitting,
blow extrusion, or static-charge extrusion. However, be-
cause of their limited properties and aesthetics, nonwo-
vens produced in this manner have limited uses.
Because of the fiber types available, the differences in
web-manufacturing processes possible, and the various
bonding methods, a cost-effective nonwoven product
can be produced with properties that will perform in
almost any application. However, adequate performance
characteristics are ensured only if a nonwoven incorpo-
rates the proper fibers, web manufacturing process,
bonding method, and proper finishing and fabrication to
produce the final product for the end-use application
desired.
PACKAGING APPLICATIONS
Packaging applications for spunbonded fabrics are for the
most part a specialty area in which paper products or
plastic films do not adequately perform. One of the largest
packaging applications is high-performance envelopes.
The major use for spunlace fabrics is in medical pro-
ducts. In medical applications, great progress has been
made in the substitution of traditional reuseable woven
materials with higher-performing spunbondeds. Histori-
cally, flash spunbonded polyethylene was the first 100%
spunbonded to find use in sterilzable packaging. Recently,
structures of spubonded–meltblown–spunbonded polypro-
pylene has gained acceptance in sterilizable wrap. Spun-
bonded fabrics with elastomeric properties are now
commercial. An economical approach using polypropylene
has recently become commercial and is used in medical
and hygienic applications (5).
Medical devices or trays of devices are often sterilized
after the nonsterile device is sealed in a package. A part of
the package, such as the lid, is made from flashspun or
spunbonded–meltblown fabric because it possesses the
unique property of permitting the sterilizing gas of ethy-
lene oxide to pass through while remaining impenetrable
to bacteria (3).
BIBLIOGRAPHY
J. Robert Wagner, ‘‘Nonwovens,’’ in The Wiley Encyclopedia of
Packaging, 2nd edition, John Wiley & Sons, New York, 1997,
pp. 672–674.
Cited Publications
1. J. R. Wagner, Nonwoven Fabrics, Norristown, PA, 1982.
2. F. Hajduk, M. Blogoer, and T. Sasano, ‘‘Nonwovens,’’ Chemical
Economics Handbook, SRI Consulting, Menlo Park, CA, 2006.
3. R. L. Smorada, ‘‘Nonwoven Fabrics, Spunbonded’ in Kirk–
Othmer Encyclopedia of Chemical Technology, Vol. 20, John
Wiley & Sons, Hoboken, NJ, 2006.
4. J. R. Wagner, Nonwoven Notes, Norristown, PA, 1989.
5. J. Austin, (to BBA Nonwovens), U.S. Patent 6,225,243 May 1,
2001.
NUTRITION LABELING
JEAN STORLIE
Nutrition Labeling Solutions,
Ithaca, New York
KAREN HARE
Nutrition Services, Inc., Fort
Collins, Colorado
The regulatory issues associated with nutrition labeling of
food products are complex and extensive. Specific nutri-
tion information must be included on most food products
following particular design requirements. The regulations
also delineate what nonmandatory nutrition information
may be included and how it can be presented. Profes-
sionals involved in food processing, manufacturing, mar-
keting, and packaging need to be aware of nutrition
labeling regulations and understand how they affect food
packaging and marketing.
The purpose of this article is to provide a broad under-
standing of the nutrition-labeling regulations, explain
why they were developed, and describe some of the
approaches that may be used for developing nutrition
labels. A heavier emphasis will be placed on the issues
related to design and format, rather than those pertaining
to the content of the nutrition label. Because the regula-
tions are extensive and detailed, it is not possible in the
824 NUTRITION LABELING
scope of this article to cover all the issues necessary to
make this a ‘‘how to’’ reference. In order to apply the
regulatory requirements to specific packaging issues, it is
necessary to consult with regulatory source documents,
regulatory guidebooks, and/or legal or regulatory advisors.
HISTORY OF NUTRITION LABELING
Although the history of food labeling laws reach back as
far as 1906 with the Pure Food and Drug Act, it was not
until after World War II that consumer interest in and
need for nutrition information became evident (1). During
the war, food preparation shifted from the household to
large-scale food processing. The extended shipping times
for delivery of goods needed in war encouraged the devel-
opment of additives to extend shelf life. Wartime health
problems led to the recognition of the relationship be-
tween nutrition and diseases, spawning interest in nutri-
tion research and food labeling. Scientists recognized the
need for vitamins and minerals to prevent certain diseases
such as beriberi and pellagra.
By 1969, there was significant public interest in the
relationship between diet and health. As a response to this
interest, President Nixon convened the White House
Conference on Food, Nutrition, and Health, addressing
malnutrition in America (2). He told Congress of the need
to ensure that the private food industry serves all Amer-
icans well and that people are educated in choosing proper
foods. As an outcome of this conference, along with Con-
gress’ effort to mandate nutrition labeling, the Food and
Drug Administration adopted a voluntary nutrition label-
ing program in 1973 that required the labeling of nutrition
information whenever a nutrient claim was made or a
product was fortified (3). At this time, the voluntary
nutrition label focused on key vitamins or minerals known
to cause deficiency disease.
Proponents of mandatory nutrition labeling were not
satisfied with the limitations of the voluntary program—
typically, only products with a good nutrition story
included nutrition labeling. Throughout the 1970s, man-
datory nutrition labeling continued to be addressed by
legislators—in particular, Senator George McGovern, who
chaired the Senate Select Committee on Nutrition and
Human Needs. This Committee investigated a number of
nutrition issues affecting Americans and was responsible
for issuing the U.S. Dietary Goals, the first government
report setting prudent dietary guidelines for Americans
(4). In 1978, McGovern initiated a series of hearings to
explore nutrition labeling and information, where he is
quoted: ‘‘It appears from all I have heard and read to date
that the present labeling system is not useful or appro-
priate. Therefore we must determine what nutrition in-
formation the public wants and needs, and how best to
convey that information’’ (5). Although the hearing dis-
played the bipartisan nature of the issue, it took Congress
another 13 years before the Nutrition Labeling and Edu-
cation Act of 1990 (NLEA) was passed.
With the election of President Reagan in 1980, the
thrust to adopt mandatory nutrition-labeling legislation
came to an abrupt halt (1). Deregulation limited available
resources, causing FDA to focus on food-safety issues
and to deemphasize economic issues such as food labeling.
Despite this shift in legislative priorities, there were
major advances in science that documented the link
between nutrition and health. Several reports issued by
the federal government (6–8), in addition to reports re-
leased by nonprofit agencies, such as the American Cancer
Society (9) and the American Heart Association (10),
clearly showed the growing consensus about the relation-
ship between dietary imbalances and chronic diseases,
specifically the overconsumption of calories, fat, saturated
fat, cholesterol, and sodium and the underconsumption
of dietary fiber. As a result of extensive media coverage
of these findings, consumers began to demand healthier
food products and more information about the fat, choles-
terol, and fiber content of foods. Food manufacturers
responded by introducing nutrient-focused products—pro-
ducts lower in fat, cholesterol, and sodium and high in
fiber proliferated. Unfortunately, there were no guidelines
for defining ‘‘low’’ or ‘‘high’’ or for labeling nutrients such
as dietary fiber, saturated fat, or cholesterol. As a result,
manufacturers’ efforts to produce and market such pro-
ducts without consistent rules led to incidence of abuse,
and consumer groups complained of food-labeling decep-
tions. By the end of the 1980s, the food industry, as well as
regulators and consumers, were frustrated by the contra-
dictions that prevailed on grocery-store shelves.
It was not until President Bush took office in the late
1980s that legislative and regulatory efforts again focused
on nutrition labeling. Senators Metzenbaum, Kennedy,
and Hatch and Representatives Waxman and Madigan
renewed the spirit of the 1970s with nutrition labeling
regulations that ultimately resulted in mandatory label-
ing, nutrient content and health claim definitions, and
federal preemption, now known as NLEA. While these
legislators were lobbying for mandatory nutrition labeling
laws, the National Academy of Sciences’ Institute of
Medicine (IOM) issued a report entitled Nutrition Label-
ing: Issues and Directions for the 1990s (11). The IOM
report recommended changes in food labeling to assist
consumers in implementing the recommendations of the
Surgeon General (7) and the National Research Council
(8). The FDA responded by working with all concerned
parties, including industry, consumer groups, and the
states, to define regulations that would meet the goals
set out by the IOM report.
On November 8, 1990, President George Bush signed
into law the Nutrition Labeling and Education Act
(NLEA) of 1990 (12), which dramatically changed the
way that food products were labeled in the United States.
The
NLEA represents a
comprehensive mandatory nutri-
tion-labeling system, designed to help consumers meet the
U.S. Dietary Guidelines (6) and reduce their risk of chronic
diseases. The intent of these regulations was to the
provide guidance to food manufacturers and package
designers needed to ensure consistent presentation of
nutrition information and to prevent erroneous nutrient
content and health claims.
The only significant change that has been made to the
Nutrition Facts panel since the implementation of NLEA
in the early 1990s was on July 9, 2003, when FDA issued a
NUTRITION LABELING 825
regulation requiring manufacturers to also list trans fat
on the Nutrition Facts panel of food products. This regula-
tion was the result of growing scientific reports confirming
the relationship between trans fat and an increased risk of
coronary heart disease.
REGULATORY AGENCIES
Regulation of food labeling falls primarily under the jur-
isdiction of two federal agencies: the Food and Drug Admin-
istration (FDA) and the United States Department of
Agriculture (USDA) (13). The USDA’s Food Safety and
Inspection Service (FSIS) oversees food labeling of products
containing meat or poultry (Z2% or more cooked, Z3%
raw). All other food products fall under the jurisdiction of
the Center for Food Safety and Applied Nutrition (CFSAN)
of FDA. FDA regulations are governed by the F ederal F ood,
Drug, and Cosmetic Act (FDCA) and the F air Packaging and
Labeling Act (FPLA). [The Nutrition Labeling and Educa-
tion Act of 1990 (NLEA) is an amendment to the FDCA.]
The USDA’s regulatory role and responsibilities are defined
by the Federal Meat Inspection and P oultry Products
Inspection Acts . Because the two agencies are governed by
separate laws, they have different missions, philosophies,
and approaches to food labeling. This results in subtle
differences throughout the regulations.
The USDA’s primary regulatory role has been to pre-
vent public health hazards resulting from improper hand-
ling of meat and poultry during production and packaging.
The USDA’s network of field offices are responsible for
conducting frequent plant inspections, and the national
office administers a label preapproval program. Through
these mechanisms, the USDA tightly controls food label-
ing during the production process. Unlike the USDA, the
FDA has not been funded to function as an inspection
service; therefore, the agency does not have the staff
resources to play a hands-on role in label development
and approval. The FDA relies on postmarket surveys to
enforce the regulations.
As discussed above, Congress passed the NLEA in 1990,
mandating the FDA to initiate extensive changes in the
content and format of the nutrition label. FDA responded
by issuing final rules in January 1993 (14). Nutrition
labeling of USDA products was not included in the Con-
gressional mandate; however, the USDA decided to follow
suit in order to prevent consumer confusion arising from
two different nutrition labels (15). In most of the signifi-
cant aspects of nutrition labeling, the USDA regulations
mimic the FDA’s; however, the regulations diverge in a few
areas. For example, the USDA has different exemption
criteria, more relaxed rules for use of the ‘‘Simplified’’
format, and less extensive requirements for statements
that accompany nutrient content claims.
NUTRITION FACTS PANEL
The Nutrition Facts panel presents the nutrient profile of
a food product on a per serving basis. Since a goal of NLEA
was to make nutrition information easy for consumers to
locate, read, and understand, both the content and layout
of the Nutrition Facts panel were studied extensively
before the regulations were finalized. The Nutrition Facts
panel focuses on the nutrients important to the health
of Americans (e.g., calories, fat, saturated fat, trans fat,
cholesterol, sodium, fiber). The nutrient content of the
food is presented in absolute terms (e.g., grams, milli-
grams) and as a percentage of the Daily Value (% Daily
Value) for those nutrients where FDA has establised a
Daily Value. Percentages provide a method for comparing
the nutrient profile of the food to recommended nutrient
intakes. Larger food packages are also required to include
a list of the Daily Values at two calorie levels (i.e., 2000
and 2500) in a footnote at the bottom of the Nutrition
Facts Panel. The serving size information has been stan-
dardized to make it easier to compare similar products.
Serving sizes are presented in common household mea-
sures (e.g., cups, tablespoons) to help consumers visualize
portion sizes, as well as metric units (e.g., grams), to
encourage familiarity with the International System of
Measures (13).
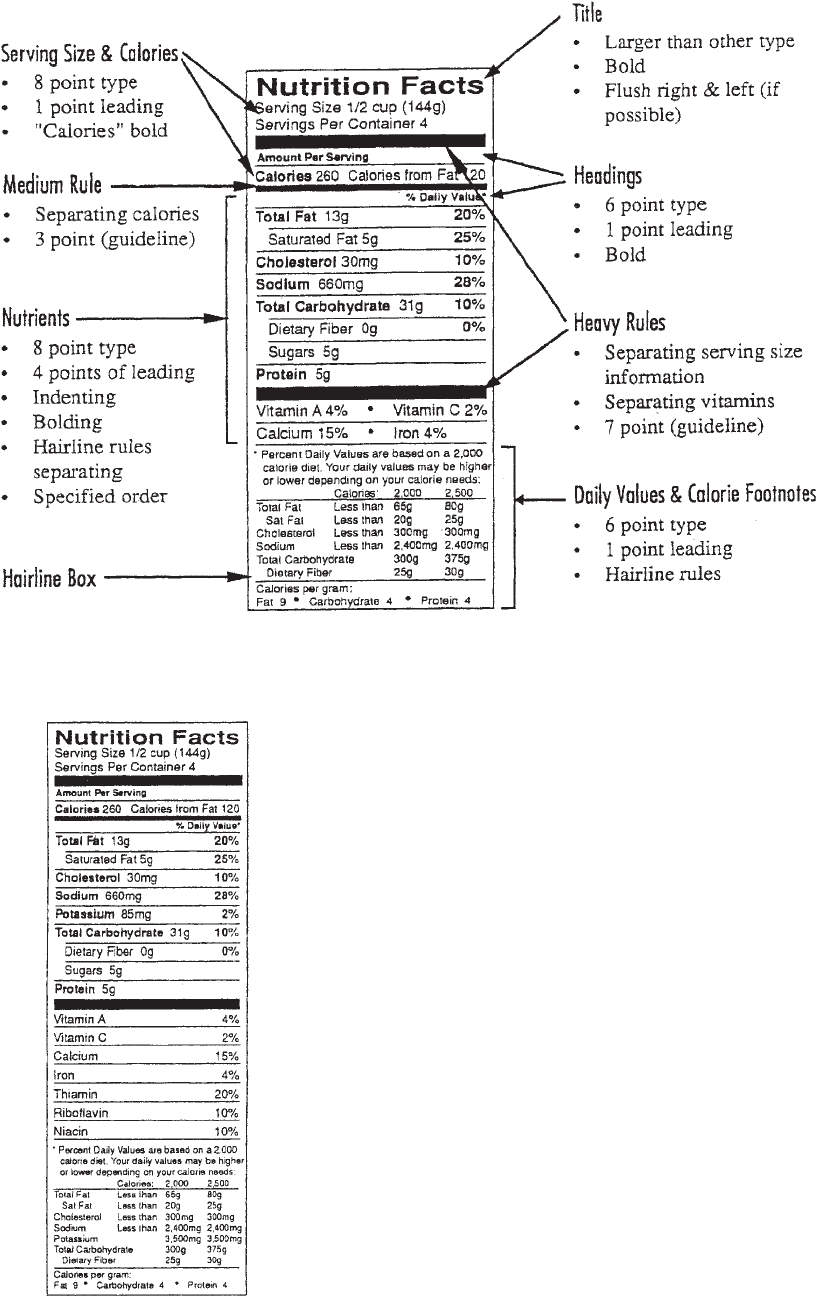
To maintain a consistent look from package to package,
both FDA and USDA regulations explicitly define
the layout of the Nutrition Facts panel (Figure 1). A
hairline box surrounds the nutrition information, which
is presented in a required order. The type size and leading
(spacing between lines of type) of each line is specified.
Boldface type is used to enhance certain nutrients,
and other nutrients are indented. Heavy-, medium-,
and lightweight rules are incorporated in specified
places.
To provide some flexibility in tailoring the Nutrition
Facts panel to various food packages, the regulations allow
for several variations of the Nutrition Facts layout pic-
tured in Figure 1. These variations are not a random
choice; they each have explicit usage criteria (13). Format
options are based on the nutrient profile of the food.
Display options are available to accommodate different
package shapes and sizes, as well as special situations
(e.g., variety packages, foods for children, bilingual labels).
In addition, the regulations allow for modification of the
Nutrition Facts on packages that have limited space for
labeling.
Format Options
The format of the Nutrition Facts panel refers to the
nutrients listed on the panel (13). The regulations define
14 mandatory nutrients for inclusion in the Nutrition Facts
pa
nel; th
e ‘‘Full’’ format includes these 14 nutrients (Figure
1). In addition, the regulations allow for inclusion of certain
nonmandatory nutrients, on either a required or voluntary
basis. The Extended format accommodates the inclusion of
nonmandatory nutrients in a specified order. An example of
the Extended format is presented in Figure 2.
When a food product has one or more mandatory
nutrients at an insignificant level (e.g., nutrient decla-
ration of 0), an abbreviated format may be used.
The ‘‘Shortened’’ format is available for FDA products
that have between one and six insignificant nutrients.
With this format, insignificant nutrients may be listed in
a footnote stating, ‘‘Not a significant source of [list of
826 NUTRITION LABELING
insignificant nutrients].’’ FDA products with seven or
more insignificant nutrients may bear the Simplified
format that allows insignificant nutrients and the Daily
Values table in the footnote to be omitted. However, if
nonmandatory nutrients are declared, then a variation,
called the ‘‘Simplified Extended’’ format, is used. USDA
employs a different approach to the Simplified format. The
Simplified format is available for products that have one
or more noncore, mandatory nutrients at insignificant
levels. With all these abbreviated formats, five core
nutrients (viz., calories, total fat, sodium, total carbohy-
drate, protein) must be listed, even if they are insignif-
icant. Figure 3 presents examples of the abbreviated
formats.
Display Options
The ‘‘Standard’’ display (vertical column) pictured in
Figure 1 is the ‘‘model’’ Nutrition Facts layout from which
all others are derived (13). On packages with limited
vertical space, the ‘‘Modified’’ (footnote to the side) display
may be used. If neither the Standard nor Modified dis-
plays will fit, then the ‘‘Tabular’’ (horizontal) display may
be used. Figure 4 presents the Modified and Tabular
displays.
To accommodate special packaging and labeling situa-
tions, other displays are allowed. The ‘‘Dual Column’’
display provides for listing nutrients on an ‘‘as prepared’’
as well as an ‘‘as packaged’’ basis. The ‘‘Aggregate’’ display
Figure 2. Extended Nutrition Facts format.
Figure 1. Design features of the Nutrition Facts panel.
NUTRITION LABELING 827
