Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.

Amosorb
s
(Amoco Chemicals, currently a trademark of
Colormatrix, USA), ActiTUF
s
(M&G Polymers, Italy),
ZerO
2
s
(CSIRO, Australia), DarEval
s
(Darex Container
Products, USA), ValOR
s
(Valspar, USA), Oxbar
s
, Mon
Oxbar
s
, and DiamondClear
s
(Constar International,
USA).
SIZING AND CHEMISTRY SELECTION
Rapid removal of headspace oxygen and preventing oxy-
gen ingress through packaging for the longest possible
time constitute two different design targets in scavenger
applications. Simultaneous attainment of these goals
usually requires the use of more than one scavenging
solution and chemistry. Combination of rapidly absorbing
sachets or labels with high oxygen barrier passive or
reactive packaging often provides the best possible
performance.
For iron-based sachets, the major factors to consider are
the water activity (A
w
) of the food product or the equili-
brium relative humidity in the package headspace in
contact with the product, the total volume of the package,
the total weight of the food or other product in the package,
the temperatures that the package will experience,
whether carbon dioxide will be present in the headspace,
the percentage of oxygen initially present in the package,
the permeation rate of oxygen into the package, and the
total shelf life desired. Water activity, temperature, and
presence of carbon dioxide will determine the proper
chemistry while the rest determine the proper sizing (4).
The objective of the sizing calculations is to determine
actual volume of the oxygen that must be scavenged over
time (including both headspace and ingress oxygen). Once
the package volume and headspace volume are known,
then the percentage of oxygen in the initial headspace gas
mixture is used to calculate the actual volume of oxygen
that must be scavenged. If the product has a relatively
short shelf life, removing this initial oxygen may be all that
is required. However, for many packages, more oxygen
permeates through the package over its distribution life
than the oxygen initially present in the headspace. If this is
the case, then the surface area of the package should be
multiplied by the oxygen permeation rate (OTR) of the
barrier in use and by the expected shelf-life duration to
determine the amount of oxygen ingressing over the shelf
life. The initial oxygen amount and the oxygen ingress over
the shelf life are then added to determine the total capacity
necessary for the proper oxygen scavenger for that applica-
tion. Sachet sizing has been standardized such that most of
the chemistry designations are available in sachets of 20,
30, 50, 100, 200, 300, 500, 1000, and 2000 mL of oxygen
capacity (measured at STP conditions). Since oxygen is
approximately one-fifth of the atmosphere, these oxygen
capacities can be used to deoxygenate five times the stated
capacity of ambient air.
As better oxygen barrier packaging materials and
structures become available, the oxygen permeation rates
through packages can be reduced and smaller-sized sca-
vengers are needed to offset the ingress of oxygen. For
nearly absolute barriers like aluminum foil, only the
initial headspace oxygen has to be removed by enclosed
scavenger. Reactive barrier packaging solutions allow us
to eliminate loose sachets from the package. Design of
reactive barriers to reduce oxygen ingress rates is compli-
cated due to inapplicability of steady-state permeation
assumptions, nonuniform activation of scavenger capacity
across the barrier, and continuous inhomogeneous deple-
tion of this capacity in the course of scavenging reaction
(5). These effects are combined to produce transient non-
zero permeation rates (oxygen leakage) over the shelf-life
duration, unless the barrier reactivity with oxygen is high
enough to result in a zero measured permeation rate. As a
rule of thumb for uniformly reactive monolayer barriers,
the overall reactive capacity of the film should be twice
that of the enclosed scavenger to obtain the same time to
complete depletion of the scavenger capacity, provided
that the OTR of the passive barrier package and OTR of
the inactivated reactive barrier are the same. In both
cases of enclosed sachets and reactive barrier films, the
scavenger capacity needed to consume the headspace
oxygen has to be added to all sizing calculations.
TESTING
One common difficulty experienced by many first-time
users of individually packaged oxygen scavengers is the
difficulty of determining quantitatively just how well the
scavenger is working. The ability to accurately measure
1% residual oxygen in a package headspace is quite
common. The difficulty arises when there is only 0.01%
because the partial pressure of oxygen is far less inside the
package than outside, causing atmospheric oxygen to
rapidly equilibrate through any tiny aperture and con-
taminate many samples (4). Septa and sampling syringes
that are entirely adequate at 1% will prove inadequate at
0.01%, as many labs have learned. It is common to use a
gated or valved syringe with tight-fitting gaskets and
natural-rubber septa to overcome the propensity for leak-
age. One manufacturer recommends that the sample be
taken, the valve closed while the syringe is still in the
package, and thumb pressure be maintained on the syr-
inge plunger as it is withdrawn and until the syringe
needle is actually inserted into the oxygen meter, and only
then is the valve actually opened. By so doing, the sample
consistency can be improved. It is also difficult to obtain
accurate repeat samples from the same package unless
great care is exercised with the septa.
For testing reactive barrier films, the standard carrier
gas OTR test methods are suitable (29), provided that the
film sample is active or can be activated during the test.
Ox-Tran permeation analyzers by MOCON, Inc. (USA)
and 8000 series oxygen permeation analyzers by Illinois
Instruments (USA) are commonly used to measure OTR
with controlled relative humidity on both sides of the film.
Since no steady-state permeation can be observed in
reactive films until the scavenger capacity is depleted by
the reaction, waiting for the establishment of the steady-
state (passive) permeation pattern does not provide a
measure of the barrier performance. Commonly measured
performance properties include: the reactive lag time (or
848 OXYGEN SCAVENGERS
the time after which the steady-state passive permeation
pattern across the film is resumed), the initial barrier
improvement (or the initial rate of oxygen leakage
through the activated reactive film versus OTR of the
inactive film), and the induction period (or the duration of
full scavenger activation across the film during which the
OTR drops to its minimal measurable value). To prevent
inaccurate results, OTR measurements shoud be started
as soon as the film is loaded into the test chamber and the
downstream side is purged of oxygen. This way, no reac-
tive film capacity is wasted on equilibration and the
transient OTR profile will provide full information about
the film barrier performance. For films with a low oxygen
permeability of the polymer matrix material and a large
scavenging capacity, the OTR testing even with 100%
oxygen at the upstream film side can take a long time.
Even longer times will be needed when ambient air is
used. Thinner films and lower scavenging capacities in-
corporated into the film can be used to accelerate OTR
testing, quantify barrier properties, and scale-up the
barrier designs. The film reactive capacity to scavenge
oxygen can be tested separately by placing the activated
film sample in a sealed chamber and monitoring oxygen
levels in it until equilibration.
FOOD SAFETY AND REGULATORY CONCERNS
Ingestion of iron-containing sachets has been cited as a
concern, however, no evidence has been presented regard-
ing physical harm due to such ingestion. Mitsubishi Gas
Chemical Co. reports that they have tested the acute oral
toxicity (LD
50
) of their formulations in rats and find it to
exceed 16 g per kilogram of body weight (30). LD
50
values
of various food-grade iron powders generally exceed
5–15 g/kg of body weight. Multisorb Technologies, Inc.
reports that their oxygen scavenger components are either
food- or pharmaceutical-grade materials. Therefore iron-
based oxygen scavengers can be used with confidence. Iron
toxicity has been recognized as a problem among infants
in the United States as related to accidental ingestion of
dietary iron-supplement products. Reports of iron inges-
tion by persons of low body weight can be routinely
handled by poison control centers (31). Because of body
weight, it is highly unlikely that an adult could ingest
enough iron powder from commercially available sachets
to cause harm. Additionally, by the time a food package
reaches a consumer, the iron would no longer be in a very
reactive elemental form but partially oxidized. Use of
adhesive oxygen-scavenging labels and oxygen-scaven-
ging films, trays and lidstock eliminates the danger of
accidental ingestion.
There is also a necessity to prevent the use of oxygen
scavengers in applications where anaerobic bacteria may
grow. The food industry employs a number of precautions
to control anaerobes. Among these is to use oxygen scaven-
gers only when (a) the water activity of the product is below
the point at which Clostridium botulinum bacteria will
grow (32), (b) the food has been acidified to have sufficiently
low pH values, or (c) appropriate curing salts have been
used to prevent botulism. Temperature control is generally
discounted as a botulism prevention agent where retail
packages are concerned, but HAACP systems have been
employed for some products. Specific limits have not been
given because they will vary from product to product. There
is some evidence that the concern over oxygen scavengers
and botulism may be overstated. An examination of the
oxidative reductive potential in packages with and without
oxygen scavengers has been reported to show that the
scavenger does not increase the reductive potential (11),
and thus does not enhance the risk of outgrowth.
TROUBLESHOOTING
Because oxygen-scavenging technologies are not as pre-
valent as MAP and are somewhat complex, many users
have a need for a diagnostic tool to help evaluate the
oxygen scavenger performance. The following points
can be helpful for troubleshooting iron-based scavenging
sachet and label applications:
1. Is the package hermetically sealed? If not, a herme-
tically sealed package must be used.
2. Does the package provide sufficient oxygen barrier
(as a measured steady state OTR) at the conditions
of use? If not, the scavenger will be exhausted
prematurely and the product shelf-life expectations
will not be met.
3. Are the contents of the sachet or label rusty, or some
agglomeration is noticeable? If an evidence of reac-
tion is present, then examine the following factors.
a. Is the test method capable of measuring ppm
oxygen levels? Insufficiently accurate method
may indicate high oxygen concentrations when
the actual values may be significantly lower.
b. Is there carbon dioxide present in the headspace?
If so, use a scavenger chemistry compatible with
carbon dioxide that will scavenge oxygen instead.
c. Does the scavenger have enough capacity for the
product and package headspace? Recheck the
sizing calculations and use a larger format as
indicated.
d. If the scavenger is a self-acting type, was it left in
the atmosphere for too long before being placed in
the package? If so, it may have exhausted part or
all of its capacity. Limit the preapplication
exposure.
4. If the contents of the sachet or label are not rusty or
agglomerated:
a. Verify that the water activity range of the sca-
venger chosen matches the water activity range
of the food or other product being packaged. If
not, change scavenger specification to match the
product.
b. Check to make sure that the scavenger was not
blocked off from the headspace by being placed
under the food or a tray, or trapped tightly
between the product and the barrier film. If so,
alter the scavenger placement.
OXYGEN SCAVENGERS 849
BIBLIOGRAPHY
1. G. A. Cook and C. M. Lauer, ‘‘Oxygen’’ in C. A. Hampel, ed.,
The Encyclopedia of the Chemical Elements, Reinhold, New
York, 1968, pp. 499–512.
2. T. P. Labuza and W. M. Breene, J. Food Proc. Preserv. 13, 1–69
(1989).
3. J. D. Flores, Chem. Eng. Prog. June, 25–32 (1990).
4. R. C. Idol, ‘‘Oxygen Scavengers’’ in A. L. Brody and K. S.
Marsh, eds., The Wiley Encyclopedia of Packaging Technology,
2nd edition, Wiley-Interscience, New York, 1997.
5. S. E. Solovyov and A. Y. Goldman, Mass Transport and
Reactive Barriers in Packaging: Theory, Applications, and
Design, DEStech Publications, Lancaster, PA, 2007.
6. S. E. Solovyov, J. Phys. Chem. B 108, 15618–15630 (2004).
7. H. Nakamura and J. Hoshino, in Sanitation Control for
Food Sterilization Techniques, Sanyu Publishing, Tokyo,
1983, pp. 1–45.
8. V. W. Blanchard, U.S. Patent 96,871, 1869.
9. H. Tallgren, British Patent 496,935, 1938.
10. F. A. Isherwood, British Patent 553,991, 1943.
11. Y. Abe, Active Packaging—A Japanese Perspective, Mitsubishi
Gas Chemical Co., Ltd., Tokyo, Japan, 1990.
12. B. Zenner, J. Packag. Technol. January/February, 37–38
(1991).
13. R. Folland, ‘‘The Oxbar Super-Barrier System: A Total Oxy-
gen-Barrier System for PET Packaging,’’ in Proceedings of
Europak ’89, October 1989, pp. 483–494.
14. E. Powers and D. Berkowitz, J. Food Protect. 53(9), 767–771
(1990).
15. J. P. Smith, B. Ooraikul, W. J. Koersen, E. D. Jackson, and
R. A. Lawrence, Food Microbiol. 3, 315–320 (1986).
16. Y. Harima, ‘‘Free Oxygen Scavenging Packaging’’ in T. Ka-
doya, ed., Food Packaging, Academic Press, San Diego, 1990,
Chapter 13.
17. C. Y. Lee and J. R. Whitaker, Enzymatic Browning and Its
Prevention in Foods, ACS Symposium Series 600, American
Chemical Society, Washington, DC, 1995.
18. H. J. Andersen and M. A. Rasmussen, Int. J. Food Sci.
Technol. 27, 1–8 (1992).
19. J. Belcher, ‘‘Optimizing the Potential of Oxygen Absorber
Technology’’ in Proceedings of Pack Alimentaire ’93, June
1993.
20. Y. Abe and Y. Kondoh, in A. L. Brody, ed., Controlled/
Modified Atmosphere/Vacuum Packaging of Foods, Food &
Nutrition Press, Trumbull CT, 1989, pp. 149–158.
21. Y. Kondoh, ‘‘Oxygen Absorbers—Review of Current World-
wide Market Situation and Emerging New Technologies,’’
paper presented at Future Pak 1993, Atlanta, GA.
22. T. Klein and D. Knorr, J. Food Sci. 55(3), 869–870 (1990).
23. C. C. Loo and W. P. Jackson, U.S. Patent 2,825,651 (to
Carnation Co.), 1958.
24. W. R. Scholle, U.S. Patent 4,041,209 (to Scholle Corp.),
1977.
25. T. Y. Ching, G. Cai, D. Craig, M. S. Galland, J. L. Goodrich,
J. P. Leonard, A. Matthews, K. W. Russell, and H. Yang, World
Patent 1999/048963 (to Chevron Phillips Chemical Co. and
Sealed Air (NZ), Ltd.), 1999.
26. M. L. Rooney, J. Food Sci. 47, 291–294, 298 (1985).
27. B. L. Sarett and D. Scott, U.S. Patent 2,765,233 (to Sarett),
1956.
28.
A.
L.
Brody
, E. R. Strupinsky, and L. R. Kline, Active Packa-
ging for Food Applications, Technomic Publishing, Lancaster,
PA, 2001.
29. ASTM D3985-02e1, ‘‘Standard Test Method for Oxygen Gas
Transmission Rate Through Plastic Film and Sheeting Using
a Coulometric Sensor’’ in Annual Book of Standards 15.09;
ASTM, Philadelphia, PA, 2005.
30. Ageless—A New Age in Food Preservation, Mitsubishi Gas
Chemical Co. Ltd. brochure, Tokyo, Japan, 1991.
31. W. Klein-Schwartz, G. M. Oderda, R. L. Gorman, F. Favin,
and S. R. Rose, J. Clin. Pediatr. 29(6), 316–321 (1990).
32. K. Glass and M. Doyle, J. Food Protect. 54 (3), 162–165
(1991).
850 OXYGEN SCAVENGERS

P
PACKAGE-INTEGRITY IN STERILE DISPOSABLE
HEALTHCARE PRODUCTS
FRITZ YAMBRACH
Rochester Institute of
Technology, Rochester,
New York
The manufacturers of products used in healthcare
activities are referred to as the medical device industry
in the United States. Healthcare products include tongue
depressors, cardiac pacemakers, artificial joints, and hos-
pital beds and range from highly technological products
patented for unique design features, employing a variety
of technologies, to commodity products sold for a variety of
applications, healthcare being just one. The firms repre-
sented by the industry present an example of a business
that by its nature is an integration of technical activities
by various actors: biological sciences, mechanical engi-
neering, material science, and entrepreneur behavior, to
name a few. Unique to this industry is the high perfor-
mance requirement placed on the package and packaging
process. A failure in the package or process is viewed by
the Food and Drug Agency (FDA) as a critical defect equal
in gravity with a product design failure (1).
Sterile disposable medical products are a relatively
new product group, in existence only since the 1960s.
The arrival of this new product group created an increased
healthcare quality by utilizing single-use sterile medical
products. The development of these one-way products has
reduced the risk of contamination from previously used
products that were sterilized after each use in the hospital
where there existed a probability of error in the steriliz-
tion process. The advent of this product group, based on
the ‘‘Kleenex’’ principle, eliminated the cost and risk of in-
hospital sterilization. This product group has been grow-
ing, with more products being introduced in the health-
care industry each year. These products have a new set of
protective requirements that their packages have to meet.
Because the products are disposable after one use, the
packaging cost must not significantly increase the cost of
the product, so along with functional protective require-
ments the packaging is frequently restricted by cost
constraints. This is not true of all sterile disposable
products, because there are many that may cost several
hundred dollars and these can afford to have higher
package costs.
PACKAGE REQUIREMENTS IN HEALTHCARE PACKAGES
The basic requirements of a package are to protect the
product from the producer to the consumer. The definition
of ‘‘protection’’ is to prevent any activity from taking place
during the distribution channel that could render
the product useless or significantly reduce its usefulness.
It is beneficial to understand the basic requirements
and applications of a package prior to a more thorough
discussion of specific functional requirements. The main
requirements of a disposable device package include the
following:
1. Maintain Sterile Barrier around the Product. The
first and most important functional demand of a
device package is for the package to maintain a
sterile barrier around the product. This sterile bar-
rier is accomplished mostly by flexible packages or
blister packages. The simple, low-cost devices that
require little physical protection are usually pack-
aged in flexible bags or pouches. These simple
packages provide excellent barriers to microorgan-
isms when the correct materials are chosen, and
they are very material efficient. Complex devices
such as implants or multiple channeled IV (intrave-
nous) sets require more protection and often addi-
tional product-use functions. These products are
often packaged in blister trays that offer more
physical product protection and other features for
product use, such as separate areas in the package
for holding items before or after their specific use
(see also Skin packaging). If the sterile integrity is
compromised by a hole in the package or a ruptured
seal, the product is regarded as contaminated and
determined unusable. The potential contamination
of a patient from a nonsterile product due to package
failure is a very real concern with serious outcomes.
The maintenance of a sterile barrier is a result of
keeping anything in the external environment from
breaching the package and keeping the product from
breaching the package integrity. The breach does
not have to be more than a few micrometers in size
for a microorganism to enter the package. This
barrier function relies solely on material and pack-
age seal integrity.
2. Allow for Sterilization. The second major functional
demand that a package may have to meet is the
ability to allow for the product to be sterilized. This
demand translates into the package having to sur-
vive the stress of a sterilization process and allow
for complete sterilization of the product within that
package. This is a necessary demand on the package
system in most products because the products are
sterilized after they have been packaged. Steriliza-
tion occurs after the product has been sealed in the
primary package and the primary package has been
packaged into the corrugated shipper. Four main
sterilization processes are used to sterilize medical
devices, gamma radiation, ethylene oxide gas, elec-
tron-beam radiation, and steam. Gamma radiation
and ethylene oxide are responsible for about 85% of
the disposable market, with each currently having
851
approximately one-half of that group. The electron-
beam system is gaining market share because of
several new developments in that process that make
this sterilization technology particularly beneficial
to the medical-device industry. Other sterilization
systems are used to sterilize devices, including gas
plasma, vapor-phase hydrogen peroxide, peracetic
acid, and X-ray, but each has limited applications
due to certain factors and limitations in each system
(2). Certain products have unique needs that are
satisfied by these other sterilization systems, but
presently they represent minor applications for
device sterilization.
3. Aid in Aseptic Removal of Product. Another require-
ment of these packages is that they must allow for
an easy opening of the package by the medical
personnel. The opening of the package must not
compromise the sterility of the product. The package
must facilitate sterile, struggle-free opening without
the use of nonsterile opening devices. Low-aggres-
sion opening of the package allows for easy removal
of the sterile product and reduces the risk of
contaminating the product by struggling with the
package and touching sterile areas on the product or
dropping the product on a nonsterile area. This
requirement is particularly problematic in that the
package, to accomplish an easy opening, must have
a low resistance to force applied to the seal areas
while remaining intact. This low resistance to phy-
sical stress increases the difficulty of the package to
remain intact through the physical stresses occur-
ring during sterilization and distribution cycles.
The easy-opening feature is generally accomplished
through the use of a heat-seal material that has low
cohesive strength. Ethylene–vinyl acetate (EVA), a
copolymer of polyethylene and vinyl acetate, has a
low cohesive strength and allows for a thermoplastic
heat seal that gives the required microbial barrier
(see Aseptic packaging).
4. The Package Should Not Add to Airborne Contam-
ination. The package material should remain intact
and not tear and create airborne fiber that can settle
down and recontaminate the product during open-
ing. This translates to having packaging materials
that are higher in cohesive integrity than the
heat-seal materials used. When a package is opened,
the package material should not tear but the
adhesive will come apart. Additionally, heat-seal
adhesives should not be prone to particulate gen-
eration. The adhesive should separate and remain
on the package materials. This particulate concern
also includes dust that has settled on the exterior of
the package becoming airborne through a vigorous
opening action. The dust problem can be solved by
low-cohesive-strength heat-seal materials, design-
ing a flat package with few folds to allow for dust
collection, and the use of materials with smooth
surfaces.
5. Aid in Product Identification. The product should
be easily identifiable while still in the unopened
package. This eliminates the opening of a package,
with the consumer erroneously thinking that an-
other product or variation of the product was inside.
Ease in product identification is a particularly im-
portant feature where numerous products or pro-
duct variations need to be distinguished. In the case
of erroneously opening a package, the entire product
would be thrown out because the sterility was
compromised. Transferral of product-specific infor-
mation can occur in many ways in the unopened
package. Use of clear packaging materials can allow
for product inspection through the unopened pack-
age. The package can display an illustration of the
product with specific items highlighted on the ex-
terior of the package. The package can be color-
coded so that a line of similar products with different
specific features could be graphically identified by
the colors used for copy.
6. Evident Opening Features. The opening feature of
the package should be designed to clearly indicate
that the package has been opened. There should be
no reclosable feature on the package that would
allow for the package to be opened, contaminated,
and then resealed. The package seals should, when
opened, visually alert hospital personnel that the
package had been opened and sterility had been
compromised.
PACKAGE AND MATERIAL REQUIREMENTS FOR
STERILIZATION PROCEDURES
Generally, the maximum physical stress to which a pack-
age will be exposed is during transportation and distribu-
tion, including truck shock and vibration, the drops in
handling, and finally the twist and compression of hand-
ling the packages by hospital personnel. A current issue in
the device industry is the increased practice of using
flexible packages to contain products that are relatively
heavy. These heavier products will severely stress the
package materials and particularly the seals as they are
moved against the seals during transportation shock and
vibration stress. This is basically a package design issue
which must be addressed by utilizing more rigid packages
that ‘‘isolate’’ the product during distribution. When the
product is isolated in the package and not allowed to move,
it will not push against the seals and force the package to
open.
This is accomplished
by using blister packages with
design elements in the blister to hold the product imobile.
A medical package will face all these stresses, but
additionally the package will be exposed to stress during
product sterilization. The particular sterilization process
used exerts a unique set of stresses on the product and
also on the package. Ethylene oxide sterilization, for
example, will stress the package integrity more than it
stresses the materials. Both radiation and sterilization
seem to have the most stressful effect on the material
properties. The package and materials must withstand
these demands and after these processes remain function-
ally intact throughout distribution and storage.
852 PACKAGE-INTEGRITY IN STERILE DISPOSABLE HEALTHCARE PRODUCTS
Since sterilization occurs after the product has been
packaged, the package sees the same contact with radia-
tion, chemicals, moisture, and heat that the product is
exposed to during sterilization. The material properties
may change as a result of the sterilization exposure, and
some low level of change is acceptable. The package
properties may change as a result of the material changes,
but as a whole the package must maintain a high level
of microbial barrier. What is not allowable is any loss of
sterility. If the sterile integrity of the package is breached
to any degree by seal rupture or stress cracks in the
material, the product is considered contaminated and
unacceptable.
Each sterilization system used requires that the pack-
age perform in some way in order to facilitate sterilization.
Autoclave (steam) and ethylene oxide (EtO) sterilization
both require a vacuum process in a chamber as a mechan-
ism for the sterilant to pass through the package and
contact the product and sterilize it. Autoclave sterilization
requires that hot steam be allowed to pass through the
package and contact the product. The package must not
act as an insulator or be a barrier to the steam. An
additional requirement for steam sterilization is for the
package to tolerate high temperatures without losing
intended properties. Ethylene oxide (EtO) sterilization
requires a certain level of porosity in the package material
for the EtO gas to pass through the package material,
contact, and sterilize the product. Porosity is also neces-
sary for removal of the EtO gas from the package at the
end of the sterilization cycle. Gamma and electron-beam
sterilization both require a tolerance for low-level energy
without loss of mechanical properties. These packaging
materials must also not shelter the product from energies
required for an effective kill.
The stress a package must withstand in this industry
may be compounded in two ways:
1. It is possible for a package to see multiple steriliza-
tion cycles. This occurs when a batch of product is
sterilized and the batch fails to meet sterility
requirements. If this occurs, the batch is allowed to
be resterilized and retested. The sterilization effects
on a package are cumulative, and package design
must consider the worst-case challenge.
2. A single package design may be required to satisfy
the requirements of two different sterilization sys-
tems. This occurs when a company has multiple
manufacturing locations and different sterilization
facilities are used at each location. The company
does not want the product to be on the market in
two different packages, so one package must meet
the functional needs of two different sterilization
procedures.
Steam and (EtO) Sterilization
Steam and EtO sterilization systems are similar in that
they both use mechanical systems to deliver a sterilant to
the product. In the case of steam, the sterilant is hot moist
air; in the case of EtO, it is a warm gas. The mechanical
systems designed to deliver the sterilants rely on the
differences in pressure created by a vacuum in a vessel
to move the sterilants through the package walls and then
to contact the product. The packages are placed in a sealed
chamber, and then a vacuum is pulled on the entire
chamber. The sterilant is then released into the chamber;
and because of the pressure difference, the incoming gas
with the sterilant replaces the low-pressure areas, moving
through the package and contacting the product. This is
held for a period of time, and then another vacuum is
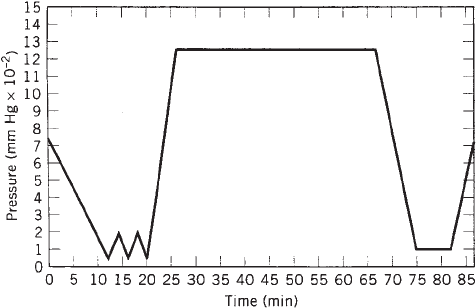
pulled on the chamber to remove the sterilant. Figure 1
shows the pressure changes in a typical steam-steriliza-
tion cycle.
The back-and-forth changes in pressure contribute to
package-seal rupture in these sterilization systems. The
decrease then increase in interior gas volume causes the
seals tbe stressed in a sort of ‘‘bellows’’ effect. This seal
stress is increased by the warm to hot temperatures used
during the cycle. The EVA heat seal is a low-melt-tem-
perature thermoplastic, and elevating the temperature
softens the material and weakens the seal strength. The
amount of vacuum drawn, quickness of the evacuation
and repressurization, package porosity, and temperature
are the key variables to control in order to maintain
package integrity. It would be possible for the pressure
cycles to be very slow and gradual, but that would increase
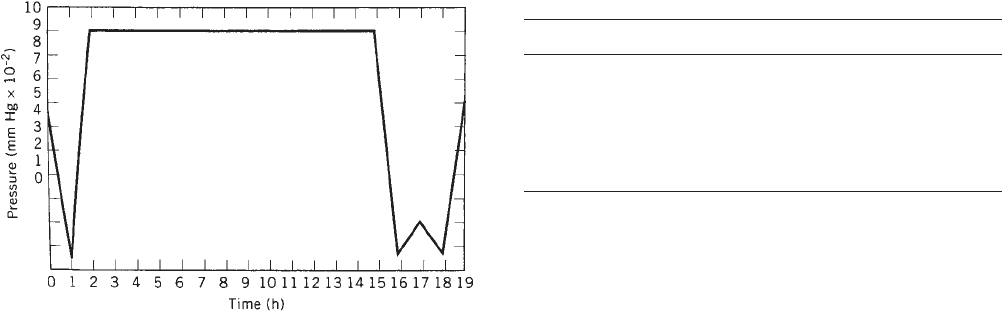
the processing times and add to the cost. In an EtO cycle,
the temperatures may be lowered to lessen the effect of
temperature softening the EVA heatseal, but to compen-
sate a longer exposure time may be required. Figure 2
shows the pressure changes that occur during a typical
EtO sterilization cycle. Steam sterilization requires a
minimum temperature that cannot be reduced. These
variables are balanced with the package’s ability to sur-
vive the sterilization system. If the need for improved
package integrity outweighs the cost of a longer cycle,
then the cycle could possibly be altered.
The mechanical similarities between the steam and
EtO processes allow for some common solutions to the
problems caused by the systems. The most serious com-
mon problem is the stressing and rupturing of the heat
seals during the pressure changes in the sterilization
cycles. There are package and package material variables
Figure 1. Changes in pressure in a typical steam sterilization
cycle.
PACKAGE-INTEGRITY IN STERILE DISPOSABLE HEALTHCARE PRODUCTS 853
that can be manipulated to alleviate the problem of
package seal stress during sterilization:
1. Porosity Rate of the Package. The porosity rate of
the package impacts on the stresses the seal face
because, if the porosity is high, the change in
relative pressures occur quickly and the package
does not bellow out and stress the seals. If the
package has a low porosity rate, the package tends
to bellow out and stress the seal by the pressure
exerted on the seal area. This property of the
material can be measured by a porosity tester.
Another solution is to increase the surface area of
porous material and, by doing so, increase the total
package porosity rate.
2. Seal Aggression. Another way to improve the seal’s
ability to survive stress is to increase the cohesive
strength of the seal material. The aggressiveness of
EVA seal material is designed so that the opening
force of the package is low and the health-
care professional does not have to struggle to open
the package. The drawback of an easy-opening
package is that the lower-aggression seals have a
hard time surviving sterilizer stresses. It is possible
to vary the vinyl acetate content of the copolymer
from 14% to 40%. The higher the vinyl acetate
content, the weaker the material’s cohesive strength
and the lower the seal strength. Another aspect of
this seal-strength issue is that if the seal is too
aggressive, it may cause the lidstock of the package
to tear when opening, increasing the amount of
airborne particle contaminants.
Steam Sterilization
The use of steam to sterilize devices is the oldest steriliza-
tion method and dates back to the time when devices were
reusable and were sterilized in the hospital after each use.
The process of using moist heat to kill microorganisms is a
very effective system with a great deal of data available
demonstrating its effectiveness. This process is very
difficult to monitor because there are many variables to
control, including temperature, moisture content, pres-
sure, and time of exposure. This process is very similar to
EtO in that the vacuum vessel is used and the differences
in pressure are created to move the sterilizing agent into
the packages. In this process the sterilizing agent is very
hot steam, with temperatures in excess of 1211C. This
process is common in most hospitals and in physicians’
and dentists’ offices. It is not used for many disposable
devices because the temperatures required will soften and
weaken most thermoplastic devices. Steam is most useful
with glass and metal devices that can tolerate the extreme
moist heat. Steam is not a major sterilization process in
the disposable-device industry but is more an in-house
system used by hospitals and medical offices on heat-
tolerant products. The use of steam sterilization as an
in-house system provides us with an example of packaging
medical devices on a relatively small scale. This is accom-
plished by the hospitals purchasing premanufactured
packages and loading the product into the three-sided
pouch and sealing the pouch. This package must then
withstand the rigors of a steam cycle and remain intact
and provide a sterile barrier until the product is to
be used. The advantages and disadvantages of steam
sterilization are summarized in Table 1.
Effects on Materials. The materials used in packaging
steam-sterilized items must be porous enough to allow for
sterilization and be able to withstand high-moist heat.
Steam has the ability to weaken papers because of the
combination of heat and moisture, so paper packaging
must be evaluated to determine the actual loss of material
integrity. Steam does not have the problem of toxic
residues found in packaging materials after sterilization
because the mechanism of lethality to microorganisms is
the high temperature rather than a toxic chemical. The
greatest concern with thermoplastic packaging materials
is their loss of physical properties on exposure to high
temperatures during the cycle. Polyethylene and polystyr-
ene have relatively low melt temperatures, so they soften
at too low a temperature for usefulness. Polypropylene
and polyesters have higher melt temperatures and may be
more suitable for steam cycles.
Effects of Package. The package stress stems from ex-
posure to pressure changes during the sterilization cycle.
These stresses manifest themselves in the same package-
seal problems occurring in EtO sterilization cycles. The
difference in temperatures used between steam and EtO
Figure 2. Changes in pressure in a typical EtO sterilization
cycle.
Table 1. Steam Sterilization
Advantages Disadvantages
Highly effective High heat exposure
Short product release time Many variables
In-house process High moisture
Low equipment cost Package seal stress
Package porosity requirements
Labor cost to process
854 PACKAGE-INTEGRITY IN STERILE DISPOSABLE HEALTHCARE PRODUCTS

accounts for the greater severity of package stress with
steam. Steam sterilization requires that the product reach
a temperature of 1211C to be considered sterile. This high
temperature weakens thermoplastic heat seals and packa-
ging films. The required porosity eliminates most plastic
films and makes paper a good choice. The one drawback
with paper is that it will weaken when saturated with
water and must be dried after sterilization.
Ethylene Oxide Sterilization
Ethylene oxide (EtO) is a chemical that, when in contact
with biological systems, disrupts certain cellular activities
interfering with the cell function, and in the case of small
organisms, the result is death. The use of EtO in steriliz-
ing medical devices is widespread, with approximately
45% of single-use devices using this method (3). This
sterilization system is favored over steam for disposables
because it employs a warm rather than hot environment
and is not as temperature-aggressive to thermoplastics.
The process involves, first, the prehumidification of
the devices and packages prior to the sterilization cycle.
This is done to activate any desiccated microorganisms
because the chemical has a more effective kill rate on
active microorganisms that are carrying out normal bio-
logical activities. This prehumidification process takes
at least 9 h at 401C and 65–80% relative humidity. After
prehumidification, the devices are moved into a vacuum
chamber. The products are still in their primary packages;
and in most high-volume applications, corrugated cases
of product are moved into the vacuum chamber still on
pallets. In the vacuum chamber a vacuum is drawn and
then held until the EtO gas is released into the evacuated
chamber. The difference in pressure is the mechanism
that draws the gas into the package and into the product
where all surfaces that come in contact with the gas are
sterilized. After a period of time, depending on the level of
bioburden and complexity of the device design, another
vacuum is drawn and the EtO gas is drawn out of the
chamber. A series of vacuums and air flushes are per-
formed to eliminate the EtO gas residuals from the
product and the package. These pallets of products are
then removed from the vacuum chamber and allowed to
vent while samples are evaluated for sterility before the
batch of product is released.
The effectiveness of this sterilization procedure relies
on the package having the properties of porosity to EtO
gas, heat and moisture resistance, and the ability to
maintain a sterile barrier. The maintenance of a sterile
barrier depends on the package material and heat seals
remaining intact without breaches throughout the re-
peated vacuum and pressurization cycles.
Effects on Materials. The effect of EtO gas on materials
is very well documented because this process has been
widely used for years. The main concern has been the
absorption of EtO and byproducts ethylene glycol and
ethylene chlorohydrin by packaging materials (4). Medical
papers and nonwoven materials such as Tyvek (manufac-
tured by DuPont) are porous and do not retain EtO
because they have ample surface area for the residuals
to evaporate and elute. EtO residuals can be absorbed into
plastics and exposure of the device manufacturer and
healthcare workers to residual amounts of the chemical
are a concern. There are differences between materials,
but a basic rule is that thicker materials will tend to
absorb more EtO than a thinner material and it is tougher
for a thicker piece of material to elute EtO residuals.
Polyvinyl chloride will absorb twice the amount of
EtO as polyethylene, polypropylene, and polystyrene (4).
The longer the sterilization cycle (the longer a material is
exposed to EtO), the greater the absorbency. The manu-
facturers who use EtO have designed aeration cycles
during and after sterilization that reduce the residual
problem. The residual issue continues to be monitored by
the U.S. government regulatory agencies, and this issue
has been instrumental in the increasing use of radiation
as a sterilization process (3). The advantages and disad-
vantages of EtO sterilization are summarized in Table 2.
Effects of Package. The effects of an EtO sterilization
cycle is more apparent on the package than on the
materials used in that package. The package effects center
on the ability of the package, as a combination of materials
assembled together, to maintain a sterile barrier:
1. Package Stress. The package stress comes from the
evacuation cycles during sterilization. The quick
changes in pressure have a bellows effect on the
heat seals. This effect is to blow the package up and
stress the package seal. This stress is compounded
by the fact that the plastic used for a heat-seal
material is chosen for its low melt temperature
and low cohesive force. The package must survive
stress on the seals at temperature conditions that
have already weakened the heat-seal material. The
most challenging part of packaging for EtO-steri-
lized packages is that the package has to encounter
stress when the heat seals are in a preweakened
warm condition. The packaging engineer is caught
between creating a strong, aggressive seal to with-
stand sterilization and creating a weak, easy-open
seal to facilitate a sterile removal of the product
from the package. The seal weakening from
Table 2. Ethylene Oxide Sterilization
Advantages Disadvantages
Relatively effective Toxic residuals
Relatively low heat (compared to
steam)
Package porosity
requirements
In-house process Many variables to monitor
Moderate heat
Regulatory issues
Moist process
Long process time
Long product release time
Package seal stress
Labor cost to process
PACKAGE-INTEGRITY IN STERILE DISPOSABLE HEALTHCARE PRODUCTS 855

evacuation and pressure cycles is referred to as
‘‘CREEP.’’ This is recognized by a scalloped area
along the seal that reduces the seal width and also
the seal strength at that point. The problem with a
seal weakened during sterilization is that during
normal distribution the seal may see additional
stress that may exceed the compromised seal
strength and cause it to open, compromising the
sterile integrity of the package. The scalloped void in
the seal can also actually be as wide as the seal,
breaching integrity and creating an opening for
microorganisms to enter.
2. Packaging Design to Aid in Penetration. The effec-
tiveness of EtO sterilization lies in the ability of the
cycle to drive the gas into the package and contact
the product. It is difficult for the gas to reach sealed
chambers in the product and other areas that do not
allow for airflow. Also, packages utilizing foam in-
serts are particularly difficult to sterilize because
the volume of air in the foam materials is difficult
to remove and impacts on sterilizer effectiveness (5).
The package can be designed with large areas of
porous material to improve the air-flow character-
istics of the package. This is necessary with blisters
that have only one side that is porous, and ‘‘Schus-
ter’’ or ‘‘header’’ pouches that reduce the surface
area of porous materials used in the package
because their design allows a minimal amountof
porous material (6).
Radiation Sterilization
Currently there are two popular methods of generating
radiation energy used to sterilize medical devices. Each
delivers the same effect, but they differ in mechanism of
delivering the energy. It is these differences that drive the
specific applications of each system now and in the future.
Gamma Sterilization. With gamma sterilization the pro-
ducts are sterilized by action of exposing the packaged
product to a decaying isotope, usually cobalt-60 (
60
Co).
The g-photon particles bombard the item and excite elec-
trons, causing them to break apart from their existing
positions. These ionized particles break other chemical
bonds, causing material and biological damage. This
damage is what kills microorganisms by disrupting cellu-
lar systems. Gamma particles are large, and because of
their size and mass, can travel relatively far distances.
This means that the source can be located yards away
from the product and still receive an effective dose.
Gamma sterilization requires a large technical facility
requiring a large capital investment. These facilities are
generally owned by companies whose business it is to
expose items to radiation through this process. The pro-
cess can achieve either (a) lethal doses as in the case of
medical devices or (b) sublethal doses as in the case of food
products (7). The sublethal process is exposure to much
lower doses. The process variables in this procedure are
few and microbial kill effectiveness is high, accounting
for high reliability in the procedure. The sterilization
takes place at ambient conditions, so the effects of high
temperature and humidity in steam and EtO sterilization
cycles are not factors affecting package integrity. Gamma
is characterized by very high penetration, which is one
reason for its high reliability of kill rate. The time for a
sterilization cycle is about 6–9 h at a normal dose rate of
4 kGy/h. The recognized average dose for sterilization
is 2.5 Mrad or 25 kGy. A gamma cycle will guarantee
exposure to at least 2.5 Mrad, but in order to do so, actual
doses will exceed that minimum. The advantages and
disadvantages of gamma sterilization are summarized in
Table 3.
Electron-Beam (E-Beam) Sterilization. E-beam steriliza-
tion is a lower energy system than gamma and has
particular advantages in particular applications. This
system excites electrons in an electron accelerator and
then bombards the package and product with these
excited electrons. The free electrons then break other
chemical bonds and cause biological damage. The similar-
ity between gamma and E-beam is the use of electrons
to effect sterilization; the difference is how the electrons
are excited. A gamma particle is much heavier than an
electron, so the gamma particle can travel farther and still
have the energy left to excite and ionize compounds.
Electrons, on the other hand, have very small masses
and lose energy quickly, so the effective use of excited
electrons occurs relatively close to the source. This dis-
tance is less than 12 in. for high-energy accelerators (8).
The advantage of E-beam sterilization is that a large
facility is not needed and consequently does not require
as large a capital expense as gamma. The electron accel-
erator can be placed in a production line with appropriate
shields employed.
The use of electron accelerators is not new in the device
industry. Johnson and Johnson was one of the first to
use this technique as far back as the 1960s (3). Currently
the technology is used for such varied applications as
(a) dosing polyester tennis strings to improve the tensile
strength and (b) curing inks and varnishes. The latter is
currently more common in Europe. There are currently
electron accelerators capable of producing 2–12 MeV of
energy. The lower energy is used mostly to cure inks and
varnishes, but this energy level is capable of producing a
sterile field within 1 in. of the source. This system has
potential for sterilizing very low-profile medical devices
(9). At these low energy levels, little shielding is required
Table 3. Gamma Sterilization
Advantages Disadvantages
Effective kill Costly facility
Few variables Degrades materials
High-volume cost-efficient Facility location not always
convenient
No residuals Effects are cumulative
No heat Variability in dose
Short product release time Long process time
856 PACKAGE-INTEGRITY IN STERILE DISPOSABLE HEALTHCARE PRODUCTS

because the energy is lost over small distances. This fact,
along with the short time required to dose the product,
would make this process a possible in-line operation. The
10 to 12–MeV accelerators are being used currently in the
industry to sterilize cases of products and have a penetra-
tion of roughly 1 MeV to 0.8 cm. These higher-energy
systems require shielding, but this is being accomplished
by subtle design changes to the production line so the
product can be sterilized in-line. Figure 3 shows an
E-beam accelerator being used in-line to sterilize medical
devices. Figure 4 shows a high-energy electron accelerator
sterilizing bottle caps. The advantages and disadvantages
of E-beam sterilization are summarized in Table 4.
Effects on Materials. The material changes in packages
exposed to radiation vary quite a bit. Some materials have
little change even when exposed to large doses of energy,
while other materials show profound effect with small
energy levels. The effects of radiation on materials has
been fairly well documented by research done by the U.S.
government and different private device companies (10).
The materials that are used in the majority of device
packages are generally low-cost common plastics and
papers, and consequently any method of limiting these
effects must be cost-effective.
Plastics are affected several ways when exposed to
doses of radiation. The polymer chains exhibit scission
and cross-linking, gas elution, and discoloration. The
dominant effect depends on the particular material. The
effect on materials has been shown to be dose-dependent
and cumulative. The effects are increased by repeated
exposure from multiple sterilization cycles. Most packa-
ging plastics are very resistant to radiation energies at
the levels used for sterilization, but two common materials
exhibit significant property changes. It is interesting
that two different materials show profound changes at
low-level energy exposure. Figure 5 shows the effects of
different doses of energy on the tensile strength of plastic
films commonly used as packaging materials. Polypropy-
lene (PP) and polyvinyl chloride (PVC) are two common
materials used to make devices and packages that exhibit
acute reactions to low levels of energy. At very low doses,
polypropylene will turn brittle, lose flexibility, and disco-
lor, turning yellow. Polypropylene shows significant loss in
mechanical strength at energy levels used in radiation
Figure 3. High-energy electron accelerator sterilizing cases of
medical devices. (Photograph provided by AECL Accelerators,
Ontario, Canada.)
Table 4. Electron-Beam Sterilization
Advantages Disadvantages
Low heat Degrades some materials
In-line process Low penetration
Highly effective High-energy systems require shielding
Few variables
Quick process time
Short release time
Low equipment cost
Controlled dose
Figure 4. High-energy electron accelerator steri-
lizing bottle caps. (Photograph provided by Ad-
vanced Electron Beams, Inc. Wilmington, MA.)
PACKAGE-INTEGRITY IN STERILE DISPOSABLE HEALTHCARE PRODUCTS 857
