Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.


O
OFF-ODORS PACKAGING
GREGORY V. PACE
Sun Chemical Corporation, and
Department of Food Science
Graduate Program,
Rutgers University
THOMAS G. HARTMAN
School of Environmental and
Biological Sciences
Department of Food Science,
Rutgers University
INTRODUCTION
Off-odor and off-flavor compounds that are responsible for
the sensory response are detected by the taste and olfac-
tory systems. The challenge for analytical instrumenta-
tion is to detect and identify those compounds most
responsible for the off-odor or off-flavor. This task is
further complicated by the fact that the compounds with
the highest sensory response have very low odor thresh-
olds. That is, they can be present at part per million (ppm)
or part per billion (ppb) levels. The analytical methodology
must be able to separate, identify, and quantitate these
off-odor compounds at very low levels in the package.
Since the 1970s, there have been excellent advances in
the instrumentation, experimentation, interpretation,
and application of measurement techniques, that have
increased the separation and lowered the detection limit
of compounds that have low odor thresholds and high
human sensory response. Much of the off-odor references
for investigation are derived from the flavor and aroma
chemistry field, in which the analytical methodology has
been centered on isolation and identification of the volatile
compounds known to give the organoleptic human re-
sponse. Because of their volatility, these compounds are
predominantly analyzed by some form of gas chromato-
graphy (GC). Capillary GC can be tailored to give the
separation and sensitivity needed for the analysis of off-
odor compounds. It is easily amenable to various sample
introduction techniques, and specific, universal, or con-
firmatory detectors.
ANALYTICAL INSTRUMENTATION FOR DETERMINING
THE CHEMICAL COMPOSTION OFF-ODOR
COMPOUNDS
Gas chromatography (GC) is the separation of a mixture of
compounds that are put into the vapor state, carried
through a chemically coated column, selectively separated
based on a physical or chemical property, and identified
with a specific detector. GC was developed using a packed
metal column of about 2 m, with a stationary support that
was coated with a nonvolatile liquid phase. It is now
predominately performed with high-resolution GC
(HRGC), using capillary columns ranging from 15 to
100 m long. The type of GC detectors most often employed
in aroma and flavor analyses are; flame ionization detector
(FID); mass spectrometry—full scan (or total ion) mode
MS-TIC; mass spectrometry in selective ion monitoring
mode (MS-SIM); GC—olfactometry (GC-O). The selection
of the detector depends on the complexity of the volatile
mixture being separated, the properties of the compounds
in the mixture, and the levels at which those analytes of
sensory response interest exist.
The methodology chosen for off-odor and off-flavor com-
pound isolation is a critical step in ensuring that the entire
mixtures that cause the sensory response are analyzed.
Solid-phase extractions (SPE) and SP-micro-extraction
(SPME), headspace (HS) extractions, thermal desorption
(TD), and purge and trap (P&T) are common techniques
employed to facilitate the extraction of the compounds and
then analyzed by the GC instrumental system.
Gas Chromatography—Olfactory (GC-O)
Gas chromatography—olfactometry (GC-O) is a true hy-
phenated analytical and sensory technique. It is widely
applied to confirm the actual olfactometric property of a
suspected off-odor, off-flavor, or off-aroma compound. The
isolated mixture is injected into a GC, the compounds
in the mixtures are separated, and the effluent vapors
from the column are usually split to an instrumental
detector and a sniff port (Figure 1). The sniff port is
conditioned with humidified air to protect the nasal
passages of the analyst (or panelist) and the vapor
compounds separated from the mixture by the column
elute out of the sniff port and are detected by the human
nose. An electronic signal is generated by the analyst
Figure 1. Gas Chromatograph equipped with a sniff port.
839
upon detection of the odor. A descriptor is also recorded
for each compound. The final olfactogram is recon-
structed by averaging the individual olfactograms of the
replicate samples, identifying the odor-active compounds
in the odorant areas (1). GC-O was successfully used in
conjunction with triangle tests to determine the effects of
oxidation of critical flavor active compounds during wine
processing (4).
Gas Chromatography–Flame Ionization Detector (FID)
GC-FID is the most widely used GC system in many
different environments from production quality control
to fragrance research. There is an abundance of published
literature and application notes using this technique in a
wide variety of fields where the analysis of volatile com-
pounds is important (e.g., environmental, petroleum, in-
dustrial solvents, food packaging, flavors and fragrances).
The identification of a compound is based on the retention
time on a particular column, compared to a known stan-
dard. They can also be compared to a series of n-paraffin
hydrocarbons, which determines their retention index
(RI). This comparative retention index of a compound
allows comparison with different instrument configura-
tions to published data, as was done in the wine research
of Le Fur et al. (1). In the wine industry, GC-FID is used in
conjunction with GC-MS to quantitate the chemical com-
position of the volatile aroma compounds in foods (2, 3).
The GC-FID is also commonly used in-line concurrently
with GC-O, where the column effluent is split to the FID
and sniff port at the same time (5). These techniques used
in the food and wine industry have been adapted to off-
odor analysis by the authors of this article.
Gas Chromatography–Mass Spectrometry (GC-MS)
The improvement of the mass spectrometer since the 1980s
has made it readily available to a wide variety of indus-
tries. It is the most appropriate analytical tool to enable
the detection, identification, and quantitation of flavor
aroma active compounds with odor thresholds in the very
low ppb range. The utility of the MS is in its generation of a
third dimension of the mass spectral identification to the
chromatographic separation of column retention time in
the column and response at the detector. There are many
examples in the literature search for the utilization of GC-
MS in the confirmation and identification of sensory sig-
nificant compounds (1–8). The ability to generate a mass
spectrum that is consistent and predictable to the chemical
structure has led to large (W500,000 compounds) commer-
cial databases of compounds that can be used to search and
identify unknowns. The specific GC–mass spectrum of the
individual off-odor and off-flavor compounds allows for
their detection, identification, and quantitation at ppm or
very low ppb levels (2).
Static Headspace Gas Chromatography–Mass Spectrometry
(HS-GC-MS)
Static HS-GC-MS is used to obtain an overview of the
volatile aroma compound mixtures. It is the simplest of
the volatilization techniques, reproducing the natural
volatilization of the aroma and flavor compounds in the
sample. A liquid sample is heated in a closed container,
and the volatile mixture of compounds present in the
headspace above the liquid is sampled and analyzed
by GC-MS. This technique is effectively used in food
packaging and wine profiling to compare acceptable and
unacceptable samples (i.e., off-odor and off-flavor). Its
limitation is that the concentration of some off-odor or
off-flavor compounds with very low odor thresholds are at
levels below the detection limit of this method (i.e., ppb).
The instrumentation available on the market allows for
automated sampling of up to 100 samples, facilitating
analysis of replicate samples to generate statistical sig-
nificance of the results.
Purge and Trap Gas Chromatography–Mass Spectrometry
(P&T-GC-MS)
Purge and trap (P&T) is a dynamic headspace volatile
concentration technique. The volatiles are purged from
the sample and trapped on an absorbent material (i.e.,
Tenaxt). P&T is also used to prepare samples for con-
centrated GC-O (1) analysis. The trapped volatile com-
pounds are then desorbed into the GC-MS, and the
mixture is separated by the GC column and identified
using the MS. P&T has been mostly done off-line, where
the volatiles are trapped onto an absorbent material
(i.e., Tenax). The trap is then installed onto a desorption
apparatus, which sits over the injection port of the
GC-MS, and the volatiles are desorbed at high heat
(e.g., 3001C) and analyzed. P&T was used as a sample
preparation on many of the experiments performed in the
author’s laboratories.
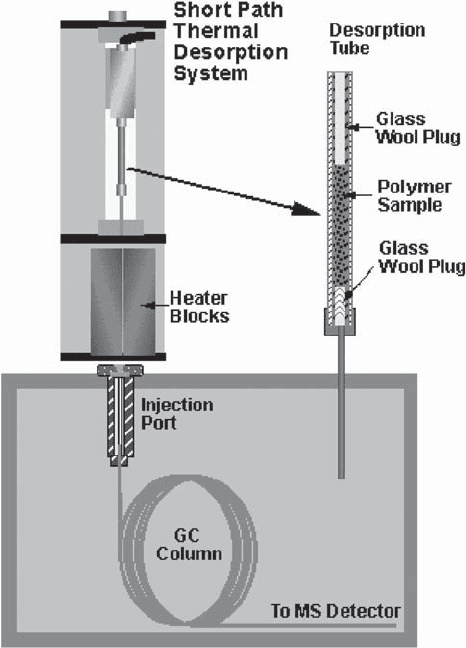
Thermal Desorption–Gas Chromatography–Mass
Spectrometry (TD-GC-MS)
Thermal desorption (Figure 2) can be used to extract
trapped volatiles that have been captured onto a chemical
trap, as stated above in P&T-GC-MS. It has been used for
the identification of off-flavors from food packaging mate-
rials (9). One way it is used is by trapping the outgassed
volatiles P&T, which are then desorbed into the injection
port of the GC-MS or GC-O (1). Another approach used is
by direct thermal analysis (TA-GC-MS) of the plastic
polymer film used for the food packaging, to release
trapped off-odor compounds from the polymer matrix
(11, 12). In that work by one of the authors, the cause of
an off-odor in a polypropylene food packaging made from
recycled resins was determined. Two compounds out of
175 chromatographic peaks were identified as the most
organoleptic significant.
Electronic Gas Sensor Array (Electronic Nose)
Th
e el
ectronic nose (e-nose) is a measurement device that
combines chemical sensing with electronic processing. Two
of the most popular designs of e-nose sensors used in flavors
and aromas are based on a conductive organic polymer
sensor array and piezoelectric gas sensor. In the conductive
organic polymer sensor array, the polymers swell to differ-
ent degrees upon response to a volatile compound, changing
their resistance. The resistance patterns of the array of
840 OFF-ODORS PACKAGING
polymers are unique for each type of compound. The
pattern display resembles the cross fiber pattern theory of
taste, although the response mechanism is much different.
The signal pattern generated by the e-nose sensor are
downloaded to a chip (for handheld models), and also
analyzed by computer. To be successfully applied, it is
critical that the e-nose is ‘‘trained’’ with the compounds
and volatiles of interest. This sensor was used in food
quality control (6). It was shown that once the
e-nose is trained with the standard, it is successful in
detecting deviations and changes. The e-nose’s portability
make it an excellent in-line production screening tool,
capable of identifying foods that fall outside of the cali-
brated, acceptable range. Another type of e-nose made from
a piezoelectric gas sensor was applied to record and repro-
duce the odor of citrus to manufacture artificial citrus
flavors (10). The array output pattern from the sensor
was compared to the ten essential citrus oil standards.
The multi-component mixtures were treated as single
components, allowing successful duplication of typical
orange flavor. The e-nose can be trained to identify
if a food or package is different that the standard, but is
less capable to identify and quantitate an off-odor or off-
flavor compound as well as GC-MS.
REFERENCES
1. Y. Le Fur, V. Mercurio, L. Moio, J. Blanquet, and J. M.
Meunier, A New Approach to Examine the Relationships
Between Sensory and Gas Chromatography–Olfactometry
Data Using Generalized Procrustes Analysis Applied to Six
French Chardonnay Wines. J. Agric. Food Chem. 51(2), 443–
452 (2003).
2. M. Aznar, R. Lopez, J. Cacho, and V Ferreira, Prediction of
Aged Red Wine Aroma Properties from Aroma Chemical
Composition. Partial Least Squares Regression Models. J.
Agric. Food Chem. 51(9), 2700–2707 (2003).
3. M. Bucking and H. Steinhart, Headspace GC and Sensory
Analysis Characterization of the Influence of Different
Milk Additives on the Flavor Release of Coffee Beverages.
J. Agric. Food Chem. 50(6), 1529–1534 (2002).
4. A. Escudero and P. Etievant, Effect of Antioxidants on the
Flavor Characteristics and the Gas Chromatography/Olfacto-
metry Profiles of Champagne Extracts. J. Agric. Food Chem.
47(8), 3303–3308 (1999).
5. C. Berger, N. Martin, S. Collin, L. Gijs, J. A. Khan, G.
Piraprez, H. E. Spinnler, and E. N. Vulfson, Combinatorial
Approach to Flavor Analysis. 2. Olfactory Investigation of a
Library of S-methyl thioesters and sensory evaluation of
Selected Components. J. Agric. Food Chem. 47(8), 3274–
3279 (1999).
6. R. Desrochers, P. Keane, S. Ellis, and K. Dowell, ‘‘Expanding
the Sensitivity of Conventional Analytical Techniques in
Quality Control Using Sensory Technology,’’ Food Qual.
Pref. 13, 397–407 (2002).
7. T. Aishima, Correlating Sensory Attributes to Gas Chromato-
graphy–Mass Spectrometry Profiles and e-Nose Responses
Using Partial Least Squares Regression Analysis. J. Chroma-
togr. A 1054(1–2), 39–46 (2004).
8. S. J. Lee and A. C. Noble, ‘‘Characterization of Odor–Active
Compounds in Californian Chardonnay Wines Using GC–
Olfactometry and GC–Mass Spectrometry,’’ J. Agric. Food
Chem. 51(27), 8036–8044 (2003).
9. M. Huber, J. Ruiz, and F. Chastellain, ‘‘Off-Flavour Release
from Packaging Materials and Its Prevention: A Foods Com-
pany’s Approach,’’ Food Addit. Contam. 19(Suppl), 221–228
(2002).
10. M. Hirschfelder, A. Forster, S. Kuhne, J. Langbehn, W.
Junghanns, F. G. Pank, and D. Hanrieder, Using Multivariate
Statistics to Predict Sensory Quality of Marjoram from
Instrumental Data. Sensors and Actuators B: Chemical
69(3), 404–409 (2000).
11. T. Hartman, Determination of Off-Odors and Other Volatile
Organics in Food Packaging Films by Direct Thermal Analy-
sis-GC-MS, Scientific Instrument Services Application Note
1a, updated 1999, CAFT/New Jersey Agricultural Experi-
mental Station Publication # E-10570-1-90.
12. J. Manura, ‘‘Detection and Identification of Volatile and Semi-
Volatile Organics in Synthetic Polymers Used in Food and
Pharmaceutical Packaging,’’ LC/GC 11(2), 140–146 (1993).
OXYGEN SCAVENGERS
STANISLAV E. SOLOVYOV
Multisorb Technologies, Inc.,
Buffalo, New York
Oxygen scavengers comprise the most commercially im-
portant subcategory in active packaging applications and
Figure 2. Thermal Desorption/Analysis System.
OXYGEN SCAVENGERS 841
are a part of a much broader category of absorbers or
sorbent materials for gases, vapors, liquids, and volatile
organic compounds. Depending on their physicochemical
nature, such absorbers can either reversibly or irreversi-
bly remove an undesirable gaseous or liquid species from
environments contained within a package. Common re-
versible absorption processes include physical adsorption
of gas molecules to solid surfaces, bulk dissolution, and
molecular diffusion of mobile species in solids and liquids.
Mobile species absorbers, whose action is irreversible at
the conditions of use, are generally referred to as scaven-
gers of such species. Irreversible absorption (scavenging)
generally proceeds via chemical reactions of the mobile
species with the components of a scavenger system to form
stable compounds. Oxygen scavenging processes include,
for example: oxygen chemisorption, catalyzed oxidation of
hydrogen gas to form water: catalyzed oxidation of ethy-
lenically unsaturated hydrocarbons and organic polymers
to aldehydes, ketones, and carboxylic acids; and oxidation
of reduced transition metals to form stable oxides.
Molecular oxygen (O
2
) is a highly reactive diatomic gas
that forms about 20.9% of the earth atmopshere (1) and is
necessary for sustaining organic lifeforms, but at the same
time it causes many types of food and beverage spoilage,
degradation of pharmaceutical formulations, and oxida-
tion and corrosion of metals, electronic components, and
various other products. Internal volume of any package is
necessarily limited and contains a limited amount of
oxygen; therefore it is possible to reduce or essentially
eliminate the oxygen present inside the package by using
oxygen scavengers. In food packaging, removal of oxygen
trapped inside the package allows us to control aerobic
microorganisms such as mold and many bacteria, slow
down enzymatic activity, and reduce detrimental color and
flavor changes due to oxidative deterioration (2, 3). The
use of individually packaged oxygen scavenger formats,
which are inserted in a package but do not form a part of
food formulations, also allows food processors to reduce or
eliminate less desirable direct food additives such as
antioxidants and preservatives.
Individually packaged oxygen scavengers are produced
in the form of packets or sachets containing a sacrificial
compound that is formulated to rapidly oxidize and
thereby eliminate free oxygen by chemically binding
with it before it reacts with the packaged product (4). In
addition to sachets, oxygen-scavenging internal labels
applied with pressure-sensitive adhesive backings and
oxygen-scavenging liners in bottle caps and crowns are
marketed. Oxygen-scavenging polymeric films that ap-
peared in the early 1990s allow us to eliminate loose
sachets from the package and add an oxygen-scavenging
functionality to the entire packaging structure or its part.
The resulting hermetically sealed packages have a dual
function, simultaneously providing for oxygen removal
from the package interior and preventing ingress of atmo-
spheric oxygen through otherwise permeable container
walls by forming a reactive barrier to oxygen permeation.
Applications of reactive barrier technology include oxygen
scavenging plastic bottles for beer, juices, ketchup, and so
on, pharmaceutical blister packs, plastic lidstocks, and
thermoformed food trays.
The majority of all oxygen scavengers in commercial use
are based on the oxidation of iron powders, although
systems based on other oxidizable transition metals such
as copper, zinc, magnesium, manganese, aluminum, tita-
nium, and so on, have been proposed and patented. In
recent years, multiple patents have been issued, describing
organic oxygen scavenging chemistries with and without
use of metal-based oxidation catalysts, specifically for the
use as active barriers in plastic packaging applications. In
this case the low oxygen permeability of polymer matrix is
combined with high reactivity and substantial reactive
capacity of oxidizable substrate to create an efficient and
long-lasting active barrier to oxygen permeation (5). Or-
ganic oxygen scavengers are often preferable due to their
transparency and compatibility with polymeric matrices
that make it possible to produce optically clear oxygen
barrier films and structures. Several formulations have
been claimed as having zero effective oxygen transmission
rates through the reactive barrier films for periods ranging
from 1 to 3 months up to 2 years, thus outperforming glass
packaging with essentially permeable seals and closures.
In such cases the essentially zero oxygen transmission
rates are achieved for reactive barrier systems character-
ized by a high degree of diffusion control of the overall
forward rate of oxygen scavenging reaction. Diffusion-
controlled reactions refer to the reactions of dissolved
oxygen with the scavenging agent immobilized in a solid
matrix, where the rate of oxygen removal by the reaction is
much faster than the rate of oxygen diffusion through the
solid matrix of the barrier layer (6).
Performance and packaging requirements to enclosed
oxygen savengers differ substantially from those for re-
active barrier applications. Enclosed scavenger formats
are designed for a controlled mode and rate of activation,
rate of oxygen absorption, and specific overall reactive
capacity to absorb oxygen. Scavenging chemistries that
are initially stable and inactive but can be activated on
demand are preferable because they reduce the possibility
of premature reaction and loss of scavenger activity before
application. Scavenging systems are generally designed to
be activated by external fields and penetrating agents
such as UV radiation (photoreducible organic compounds)
or moisture diffusion (oxidation of transition metals).
Scavengers that are immediately active upon their man-
ufacture are less desirable since they generally require
high levels of protection from environmental oxygen be-
fore use. To rapidly deoxygenate a package, an oxygen-
scavenging chemistry has to be readily accessible to both
activating agent and oxygen. Therefore the scavenger
packets are often made from microperforated or otherwise
highly permeable film substrates. Numerous sizes and
formats of packaged oxygen scavengers with varying
oxygen absorption capacity and activated at different RH
levels have been developed for specific applications.
Oxygen-scavenging capacity of the packaged scavenger
unit as well as the mode and rate of its activation are
critical for packaging system design. The rate of oxygen
absorption by the scavenger, determined by its format and
chemistry, and overall oxygen transmission rate (OTR) of
the package wall determine the minimal attainable oxy-
gen concentration in the package headspace. OTR of the
842 OXYGEN SCAVENGERS
package, together with the overall reactive capacity of
the scavenger, control the overall duration of protection
from ingress of ambient oxygen. In well-designed oxygen-
scavenger/package systems, attaining and maintaining
100 ppm or less of residual headspace oxygen is consi-
dered to be the norm. At 100 ppm, effective control
of the detrimental effects of oxygen on foods has been
demonstrated (7). Oxygen scavengers are often used in
conjunction with modified atmosphere packaging (MAP)
techniques, such as vacuum packing and flushing head-
space with inert gas before sealing, to reduce residual
oxygen amounts. The gas flush reduces the scavenger
reactive capacity requirement per package and is a more
economic means of reducing the oxygen down to as little as
0.5%, but flushing alone is not capable of reaching the
oxygen levels possible with the use of the scavenger (4).
The oxygen-scavenging technologies constitute a major
part of active packaging techniques and methods, and they
have been extensively patented worldwide, especially in
Japan. Commercial use of oxygen scavengers started in
1976 in Japan and in 1984 in the United States. UK-based
consulting and media concern Pira International Ltd.
estimated the global value of the total active packaging
market in 2005 to be worth more than US$1.5 billion and
has forecasted this market to be worth more than US$2.6
billion in 2010. The global market for packaged oxygen
scavengers was estimated to be 12 billion units in Japan,
500 million in the United States, and 300 million in
Western Europe in 2001. This market was forecast to
grow to 14.4 billion units in Japan, 4.5 billion units in
the United States, and 5.7 billion units in Western Europe
in 2007.
HISTORY
In 1869 Virgil Blanchard of Vermont obtained a U.S.
patent for ‘‘improvement in preserving fruits, meats, and
other substances’’ (8) by placing an insert with an alkaline
solution of pyrogallic acid into an air-tight vessel. Removal
of both headspace oxygen and oxygen leaked through the
seals was claimed via reaction of pyrogallic acid with
oxygen. In 1938 Henrik Tallgren of Finland was granted
a British patent for ‘‘keeping food in closed containers with
water carrier and oxidizable agents such as zinc dust, iron
powder, manganese dust, etc.’’ (9). F. Isherwood of the Low
Temperature Research Station at University of Cam-
bridge obtained a patent for ‘‘removing oxygen from a
container containing vacuum or gas packed food in which
a metal absorbs oxygen to form an oxide’’ in 1943 (10). This
early activity was followed by attempts to use powdered
metals to purify industrial gases and the development of a
system to scavenge oxygen by use of palladium catalysts
in a hydrogen-flushed package by American National Can
in the 1960s. In 1969, Mr. Fujishima attempted to market
a hydrosulfite-based oxygen scavenger that reacted very
quickly, causing problems with lost capacity, and also had
a propensity to react with food volatiles to form noxious
sulfur-containing gases. None of these early developments
resulted in any significant commercial activity. Mitsubishi
Gas Chemical Co. (MGC) of Tokyo, Japan has been issued
a patent for a ferrous-iron-based scavenger formulation in
1977 and was the first company to start marketing of their
Ageless
s
brand sachets in 1976.
The early success of oxygen scavengers in Japan ap-
pears to be tied to the tradition of gift-giving and the
importance of flawless presentation and quality (11),
especially in the hot and humid summer climate of Japan.
When the gift was a food item, the quality assurance
provided by an oxygen scavenger was a reasonable pre-
caution. Several confectionery items given as traditional
gifts also suffered from short shelf life due to mold growth.
For foods such as Japanese rice cake, the shelf life and
quality improvements afforded by a combination of a
barrier package and an oxygen scavenger allowed for a
longer distribution cycle and made Ageless
s
a commercial
success. Competitors followed in Japan and other Asian
countries, and today there are more than a dozen,
although MGC is still leading the industry. Metal-based
oxygen scavengers are not the only option, because many
organic scavenging systems have been proposed with the
goal to alleviate the possibility of food contamination by
metallic compounds, prevent setting off metal detectors,
and allow making microwaveable containers. Toppan
Printing Co. has marketed an ascorbic-acid-based oxygen
absorber since 1979. A catechol-based scavenger has been
marketed by Oji Kako Co. under the Tamotsut name.
Since early 1990s, many scavenger-based oxygen barrier
films and structures have been developed with the goal of
preventing oxygen ingress into the sealed package.
In the United States, oxygen scavengers were first used
in 1984 when General Foods began to ship ground roast
coffee with their ‘‘Fresh Lock,’’ an oxygen scavenger for-
mulation supplied by MGC. Multiform Desiccants Inc.
(currently Multisorb Technologies, Inc.) of Buffalo, New
York entered the U.S. market with their Freshpax
s
brand
oxygen scavengers in 1988 and an initial application for
‘‘Meal, Ready-to-Eat’’ (MRE) rations as developed by the
U.S. Army Natick Research, Development and Engineering
Command. The Aquanautics Corp. introduced ‘‘SmartMix
and SmartCup’’ technology for compounding into packa-
ging such as bottle-cap liners in the late 1980s (12). In
addition, CMB Technology (France) introduced their Ox-
bar
s
oxygen barrier system, which used a cobalt salt
catalyst to accelerate oxidation of MXD-6 nylon blended
with PET (13). The original Oxbar
s
did not become a
commercial technology; however, its later reincarnations
as MonOxbar
s
and optically clear DiamondClear
s
tech-
nology, currently a part of Constar International (USA)
portfolio, have gained significant publicity and resulted in
several commercial applications.
A wide acceptance of oxygen scavengers by the Japa-
nese food industry has not yet been repeated in other
markets. Food is more expensive in Japan than in the
United States; thus the cost of an oxygen scavenger versus
the value of preservation afforded a greater incremental
packaging expense in the Japanese market. The U.S. food
industry has had other reservations, especially regarding
the liability of loose scavenging sachets and potential
food contamination by their contents. The acceptance is
growing in the United States and Europe as the technol-
ogy has matured and adapted to specific applications that
OXYGEN SCAVENGERS 843
make economic sense in the U.S. and EU markets. Nota-
bly, food stores at Walmart supercenters in the United
States started switching from high-oxygen packaging for
case-ready meats to low-oxygen master packs with en-
closed oxygen scavengers in late 2007 to improve preser-
vation and shelf life of red meats such as beef steaks and
ground beef.
SPOILAGE CONTROL WITH SCAVENGERS
Preservation of foods and other products with the use of
an oxygen scavenger will be improved only when an
oxygen-related spoilage is the primary pathway of qual-
ity deterioration. The economic significance of the var-
ious spoilage types attributable to oxygen also varies and
should be factored into any decision to employ a scaven-
ger. In situations where control of oxygen is warranted,
the observable advantages include greatly reduced losses
and returns due to spoilage, improvement in consumer
confidence and relations, increased distribution radius
due to a longer shelf life with correspondingly increased
sales expectations, and improved plant scheduling due
to longer product runs taking advantage of the longer
shelf life.
Primary use of an oxygen scavenger is to control
aerobic microorganisms. While both MAP and vacuum
packaging have the capability to aid in control of aerobic
bacteria and mold, it has been reported that mold colonies
may grow in even 0.4% residual oxygen in as little as 10
days (7). Both vacuum and MAP techniques may not be
able to effectively deliver or maintain such low initial
residual oxygen concentrations, although an appropriate
concentration of carbon dioxide may also effectively sup-
press mold growth. However, Powers and Berkowitz
(14) noted that an oxygen absorber prevented mold
growth in specially formulated and packaged bread for
up to 3 years, and oxygen scavengers are also reported to
control mold growth on crusty rolls for at least 60 days
(15). In commercial practice, use of an oxygen scavenger to
control mold is a major use in Japan but has been of minor
importance elsewhere in the world. Use of barrier packa-
ging for baked goods appears to be more prevalent in
Japan than in the United States, and oxygen scavenger
use makes sense only where a suitable barrier to oxygen
permeation is employed. In the United States, the MRE,
beef jerky, and fresh pasta industries have adopted pack-
aged oxygen scavengers to control mold growth on their
products. Permeation of oxygen into the package over
time, which leads to mold growth, is the major concern
that can be addressed with either enclosed scavengers or
reactive barrier packaging.
Another oxygen related mode of food spoilage is
through oxidation of various pigments in the food itself
(16). This generally takes the form of a darkening
of color—for example, due to enzymatic browning reac-
tions (17). While color change does not in and of itself
make the food product inedible, consumers appear to
perceive that such darkening is an effective indicator
of other spoilage problems that may have occurred simul-
taneously. Therefore, such pigment darkening may
effectively end the shelf life of the packaged product.
The smoked/processed meat industry is especially aware
of the problems of discoloration and consumer reluctance
to purchase packages exhibiting a color darker than
other packages in the same retail case. Oxygen scaven-
gers, more than any other packaging method, excel at
reducing headspace oxygen to such low levels that pig-
ment oxidation does not occur for extended periods of time
(16, 18). Color preservation is often cited as a reason for
using oxygen scavengers in the United States and
Europe.
Another mode of oxygen related deterioration of foods is
through oxidation of fats and oils in the product (7, 16, 19).
This process is commonly referred to as the rancidity of
lipids and yields organoleptic changes in them that are
usually considered undesirable even though rancidity is
commonly accepted in some food products such as fried fish
and nutmeats. The byproducts of this type of oxidation may
include peroxides, aldehydes, ketones, and carboxylic acids.
The presence and undesirable odor of many aldehydes and
ketones is detected by humans at levels well below 1 ppm,
and this fact presents a major problem for food processors.
Use of an oxygen scavenger has been shown to positively
control this type of oxidation (20) because peroxide values
have remained essentially unchanged over time in
packages protected by the scavenger. Conversely, reduction
of headspace oxygen to the low ppm range tends to preserve
vitamin and nutrient content of foods.
Another example of a type of food spoilage, related to
oxygen in the package headspace, is insect infestation.
Many grains and flour products suffer losses due to weevils
and the like, and it has been shown that these can be
controlled by use of an oxygen scavenger (21). While an
infestation may have been suppressed during the storage
of the bulk grain, either a later hatch or a reinfestation
may occur during packaging or distribution. Insect eggs,
larvae, or pupae may be present, but all need some amount
of oxygen to survive. Elimination of the headspace oxygen
will eventually kill all forms of the infestation and prevent
further damage, although it may take as long as 12 days to
kill the eggs of some of the more resilient insects. There
have been limited uses of this concept in the United States
for bird seed and preservation of museum artifacts, but it is
an accepted practice in Japan.
When the product to be protected is not a food product,
oxygen scavengers can also provide significant benefits.
Some pharmaceutical formulations are easily degraded by
oxygen: In particular, vitamin C, vitamin E, and b-car-
otene employed as antioxidants are commonly packaged
with an oxygen scavenger. Many newer hormone replace-
ment therapies and similar drugs are unstable in the the
presence of oxygen and will not meet common pharma-
ceutical shelf-life requirements without reactive oxygen
barrier packaging. Closely related to the food industry,
pet food and pet snacks are often protected with an oxygen
scavenger
as rancidity and
mold appear to be a problem
for pets. Protection of metals from corrosion in demanding
applications such as electronics often requires the use of
oxygen scavengers. In the meantime, the food industry
remains the dominant worldwide consumer of oxygen
scavengers.
844 OXYGEN SCAVENGERS
CHEMISTRY
Applications of scavenging technology are complex pri-
marily because of different chemistries involved, their
activation mechanisms, the endpoints of oxidation, the
reaction byproducts, and the interactions between the
scavenger and the product. The iron-based oxygen sca-
vengers are the most prevalent and they are examined
first.
The iron technology is based on rapid oxidation of iron
powders to prevent headspace oxygen from reacting with
foodstuff components or to reduce oxygen permeation
rates through container walls. Reduced iron powders
(zero-valent iron Fe
0
) are highly reactive and possess a
higher capacity to react with oxygen than partially oxi-
dized ferrous iron (Fe
2+
) compounds. Large exposed sur-
face area of iron powders per unit weight, which increases
with reduction in mean particle size and especially when
using highly porous ‘‘sponge’’ iron morphologies, promotes
simultaneous oxidation at multiple reactive sites, result-
ing in faster rates of oxygen scavenging. Presence of
electrolytes and an aqueous interface to facilitate electron
transfer during oxidation is known to accelerate oxidation
reactions of transition metals. This is exemplified by rapid
rusting of ferrous metals and steels exposed to road salt
and salted seawater, in contrast with slow rates of corro-
sion in the dry climates. Ferrous and ferric iron oxides and
hydroxides produced by oxidative reactions are basic. As a
result, acidic environments promote oxygen scavenging by
shifting equilibrium to iron oxides. At low pH values,
oxidation of reduced iron proceeds in humid environments
even in the absence of oxygen, by deoxygenizing water
molecules and resulting in hydrogen gas formation.
Since low pH values and aqueous interfaces require the
presence of water, water vapor diffusion and variation in
relative humidity of the environment can be used to
control activation of oxygen scavenging capacity of iron
powders. The specific formulations are designed to trigger
the activation at desired RH levels, control the reaction
rate, achieve the full oxidative potential of the iron
powder, maintain compatibility with various products,
and ensure food safety. The reaction formula for complete
oxidation of reduced iron to eventually form ferric oxide
trihydrate complex (commonly known as rust) is
4Fe
0
þ 3O
2
þ 6H
2
O ! 4FeðOHÞ
3
! 2ðFe
2
O
3
3H
2
OÞ
Many transition metals are recognized as being suffi-
ciently oxidative to become candidates for an oxygen-
scavenger formulation; however, iron does offer unique
advantages that have driven the industry to general use
of this medium. Iron has a relatively high affinity to
combine with oxygen on a per unit weight basis com-
pared to most alternatives. Complete oxidation of 1 g of
reduced iron removes 300 cm
3
of oxygen at STP condi-
tions. Elemental candidates that exceed it have draw-
backs such as the odor problems with the use of sulfur
and the propensity of aluminum to form an almost
impermeable oxidized skin layer that limits further
oxidation. Iron rust, on the other hand, flakes off as it
forms and it is highly permeable to further duffusion of
water and oxygen. Of great importance is the food safety
of iron powders, which are recognized as nutrients
important for healthy blood and commonly used as food
enrichments in milled flour, breads, breakfast cereals,
and baby foods. The same cannot be said about copper,
zinc, aluminum, and many other transition metals. The
abundance and relatively low cost of producing elemental
iron are also important, especially in comparison to
choices such as palladium and platinum metals and
catalysts based on them. Then there is the ready ability
to manipulate reactive capability of iron powder formula-
tions to adapt to a wide variety of applications for both
the rate of oxygen scavenging and the activation by the
moisture in a package headspace. One of the few draw-
backs is that the oxidation of iron is a temperature-
dependent reaction and normally slows dramatically as
the temperature approaches freezing.
Electrolytes and acidifying agents in a scavenging
formulation are commonly supplied by metallic salts
(22). The practical considerations of food safety have
dictated that common table salt (NaCl) is often the best
choice for the electrolyte, while various metal and organic
salts of strong acids are often used to increase the acidity
of the formulation. Another important factor in oxygen
scavenging chemistry is the desired control of activation
or triggering the scavenging reaction on demand in every
application. When the packaged product does not have a
required water activity to fully activate an iron-based
scavenger and provide the desired reaction rate, premois-
turized silicas, zeolites, and activated carbons are used as
self-contained water carriers in various proprietary for-
mulations to provide a reliable activation.
There are two broad categories of iron-based scavenger
formats, commonly described as ‘‘moisture-dependent’’
type and ‘‘self-reaction’’ type. For the moisture-dependent
chemistries, either no moisture or insufficient moisture is
present in the sachet or label formulation and the chem-
istry must have the capability to absorb moisture from the
headspace of the package. A significant advantage of these
formulations is that they are stable in ambient environ-
ments until sufficient moisture has been absorbed to allow
reaction to occur. The package into which they are inserted
must provide a moisture source, which limits the use of
these formulations to food products with relatively high
water activity. For the self-reaction type, the necessary
moisture is added right into the sachet on a suitable
carrier. These carriers must carry a high percentage by
weight of moisture and must bind the moisture strongly
enough so that the formulation powder remains free-
flowing, yet they must release the moisture as needed for
the reaction. This type of formulation has to be protected
from environmental exposure because it will start scaven-
ging oxygen immediately and can exhaust its capacity if
exposed for extended periods. An additional disadvantage
is that the preloaded moisture is subject to desorption and
equilibration in the package over time, which means that
with extremely dry products, the moisture will leave the
sachet. The loss of preloaded moisture means that the
oxygen scavenger will eventually stop working even
though it may still contain unoxidized iron.
OXYGEN SCAVENGERS 845
It should be noted that since oxygen scavengers are
routinely used with MAP, there will be situations in which
the application dictates gas flushing of the package head-
space with gas mixtures containing carbon dioxide. This is
an area for caution because many iron-based formulations
are unsuitable for use with carbon dioxide because they will
absorb it preferentially over oxygen. Many of these formu-
lations have a capacity for about five times as much CO
2
as
O
2
and will thus fail to remove enough oxygen to prevent
spoilage. Furthermore, these formulations can also reduce
the beneficial effects of carbon dioxide by reducing its
intended concentration in the headspace. However, formu-
lations are available that both scavenge oxygen and release
carbon dioxide (CO
2
emitters), and one of these should be
used when gas flushing with CO
2
is used.
Sulfites, bisulfites, and their analogues have been
suggested and used as oxygen scavengers for a long time
(23). Sulfites are commonly added to bottled wines to
preserve their quality. Desirable oxygen scavenging action
is produced by oxidation of sulfites to sulfates. The reac-
tive capacity of such scavengers is rather small to be used
as a reactive filler in barrier structures, therefore, their
use was limited to sachets and liquid sulfite layers (24).
Use of iron-based oxygen scavengers can create pro-
blems with metal detection because there is enough iron in
most sachets and labels to set off a metal detector. This is
generally overcome by using metal detectors just before
the scavenger insertion to assert that no extraneous metal
is present and then another just after to assert that the
scavenger has been added. In situations where this has
not been possible, organic oxygen scavenging chemistries
can be successfully used. Organic scavengers are also
beneficial for making polymeric barrier films and struc-
tures due to often improved compatibility with matrix
resins and higher achieveable levels of dispersion com-
pared to dispersion of inorganic powders, generally inso-
luble in plastics.
Ascorbic acid and other ascorbates are known organic
antioxidants because they oxidize to dehydroascorbic acid
in the presence of transition metal catalysts, preferably
copper. Pillsbury Co. (now a part of General Mills, Inc.)
holds a patent to use ascorbic acid as a reducing agent. A
combination of ascorbates and sulfites was used as an
oxygen scavenger embedded in plastic closure liners by
the Darex Container Products division of W. R. Grace &
Co. The technology has only achieved minor acceptance in
the United States, but it does have unique advantages and
disadvantages. Due to a lower capacity to scavenge oxy-
gen, it generally requires a larger sachet and weight of
reactant to accomplish the same oxygen removal as for the
iron-based technology. Carbon dioxide is also released as a
byproduct of oxidation.
Other proposed organic oxygen scavengers included
gallic acid, n-propyl gallate, butylated hydroxyanisoles
(BHA), butylated hydroxytoluene (BHT), and dihydro-
guaretic acid, which can serve as reactive barriers when
added to plastic packaging structures rather than being
used as food additives. Migration of these low-molecular-
weight chemicals as well as byproducts of their oxidation
into the product cannot be completely prevented. To over-
come this problem and increase the available activated
capacity of scavengers, more recent developments focused
on oxidizable polymers that can be used as barrier resins
or blended with common packaging polymers to create
reactive barriers to oxygen permeation. Ethylenic unsa-
turation in linear hydrocarbon polymer backbones and
pendant groups as well as in polyunsaturated fatty acids
is subject to auto-oxidation, especially in the presence of
transition-metal-based catalysts:
2CH ¼ CH2 þ O
2
! 2CH ¼ CðOOHÞ2
The resulting hydroperoxides can decompose to produce
free radicals, which can in turn combine by crosslinking,
can initiate free radical polymerization of other double
bonds if enough of them are present, or can cleave the
double bonds in the course of successive oxidation reac-
tions. Such reactions can be catalyzed by salts of transi-
tion metals (e.g., cobalt is most common). Rearrangement
and cleavage of the hydroperoxides to aldehydes and
ketones result in low-molecular-weight byproducts that
adversely affect odor and flavor of packaged foods in very
low concentrations. These effects limited the use of un-
saturated hydrocarbons in food packaging. Oxygen
Scavenging Polymer (OSP) developed by Chevron-Phillips
Chemical Co. gets around this problem by including the
double bond in a pendant cyclohexene ring (25). This way
pendant carboxylic acid groups are formed on the polymer
backbone as a result of oxidation rather than free low-
molecular-weight byproducts.
Rapid rates of oxidation of certain polyamides in the
presence of transition metal catalysts have been exploited
to produce reactive oxygen-scavenging barriers. MXD-6
nylon with a cobalt salt catalyst has been the primary
system patented by MGC and is targeted for incorporation
into the inner layers of polymeric packaging structures.
Having a large reactive capacity and high reaction rates,
MXD-6 nylon-based scavengers can provide nearly zero
oxygen transmission rates through nylon barrier layers.
However, their use is limited by layer delamination issues,
the potential migration of low-molecular-weight reaction
byproducts into the packaged product, relatively high cost,
and a lack of regulatory approval for direct food contact.
Oxygen-scavenging barrier systems based on MXD-6
nylon and cobalt salts have experienced some limited
success when multiple producers started blending them
as minor additives to reduce migration and improve
barrier properties of PET and other packaging resins.
CSIRO Australia has introduced photosensitive dyes as
oxygen scavengers independent of transition metal cata-
lysts (26). Functionalized anthraquinone-based chemis-
tries can act as singlet oxygen acceptors after being
excited by UV light. Excited dye molecules sensitize
ground-state oxygen molecules that diffuse through a
polymer in a singlet state. Singlet O
2
molecules react
with the dye and are consumed. ZerO
2
s
is a CSIRO line
of UV-activated oxygen scavengers incorporated into re-
active barrier films. Despite lower capacity than that
provided by metal-based scavengers, ZerO
2
s
scavengers
have advantages such as efficient low-energy activation on
demand and their reactivity being independent of moist-
ure diffusion and transition metal based catalysts.
846 OXYGEN SCAVENGERS
Biochemical reactions in living cells are accelerated by
biologically specific organic catalysts called enzymes. Cer-
tain enzymes can function as oxygen scavengers within
barrier structures. To act as scavengers, enzymes must be
first immobilized within the polymer matrix. Although
many enzyme candidates can be thought of as oxygen
absorbers, few systems have been attempted in practice.
Oxygen removal by a combination of glucose oxidase and
catalase is a primary example (27). It should be noted that
oxygen-scavenging enzymes and coupled enzyme systems
cannot act without water and are very sensitive to
changes in pH, water activity, temperature, salt content,
and other factors. On the other hand, common thermo-
plastics such as polyethylene and polypropylene are good
substrates for enzyme immobilization. Stability of immo-
bilized enzymes in the film is not fully analyzed. Other
food-grade enzymes can potentially be used for oxygen
scavenging: One example is ethanol oxidase, which oxi-
dizes ethanol to acetaldehyde. Bioka Ltd. of Finland
commercially produces enzyme-based scavenging sachets.
Some organometallic complexes known as ligands are
capable of trapping free oxygen in a manner similar to
that of blood hemoglobin (containing Fe
2+
ligand in heme)
and other peptides with metal-containing prosthetic
groups. When immobilized on a solid support such as
silica, these ligands have been demonstrated to form a
highly reactive oxygen-scavenging medium (28). Such
systems anchored to an insoluble support can be dispersed
in polymer matrices as powders to form reactive barriers.
Advanced Oxygen Technologies, Inc. (New York, NY) has
developed a series of oxygen scavengers based on synthe-
tically produced ligands. Incorporation of ligands into
plastic film structures has also been claimed.
APPLICATIONS
Individually packaged oxygen scavenger formats are now
found in multiple applications with packaged foods being
the primary use. Fresh refrigerated pasta is a major user
of oxygen scavengers to maintain quality during its dis-
tribution cycle. The smoked/processed-meat industry has
several segments that rely on oxygen scavengers to main-
tain color or prevent mold during distribution. Both pre-
sliced pepperoni in thermoformed packages and bacon bits
in glass and plastic jars use an oxygen scavenger for color
retention. In Great Britain and the United States, sliced
deli meats are often produced with very low levels of
preservatives and are thus susceptible to rapid discolora-
tion under retail lighting. These are now increasingly
packaged with oxygen scavengers to prevent discoloration
for up to one month. Beef jerky is commonly packaged
with an oxygen scavenger for reasons of both color reten-
tion and prevention of mold growth. Case-ready meat
master packs often include oxygen scavengers to extend
distribution time through deoxygenation. The individual
cases are then exposed to ambient oxygen before placing
them on the shelves to reoxygenate the meat and restore
its original red color. Roasted nuts are often protected
from rancidity by scavengers, especially where the pro-
duct will be used for export or for upscale packages. The
MRE military rations remain a major U.S. application that
is being adopted by other military organizations around
the world. Freeze-dried products often have an elevated
oxygen sensitivity, and items such as backpackers’ trail
foods are now packaged with scavengers. Pizza crusts are
also being packaged for some markets with a scavenger to
prevent mold during fresh distribution. Baked goods and
confections are often packaged with scavengers to prevent
mold. Oxygen-scavenging liners in juice and wine bottle
caps have been introduced with limited success in some
markets. Oxygen scavengers with simultaneous humidity
control (e.g., StabilOx
s
from Multisorb Technologies) are
used for protection of pharmaceuticals, dietary supple-
ments, diagnostic tests, and other medical supplies. Other
uses include prevention of metal corrosion in archeology,
art storage, and electronic components, as well as con-
servation of oxygen sensitive products such as rubber,
plastics, and greases.
Reactive oxygen barrier packaging is another rapidly
growing market. Beer, wine, and fruit juices in PET
bottles, ketchup in plastic containers, and oxygen-sensi-
tive pharmaceuticals and nutraceuticals in oxygen-
scavenging blister packs are some of the current and
emerging applications. Sulfur-containing components in
beer originating from hops are particularly subject to
oxidation and, when oxidized, adversely change beer taste
profile. Reaction with 1–2 ppm of oxygen is often enough to
cause a beer taste unacceptable to many consumers.
Protection of beer in plastic packaging is a major applica-
tion of oxygen-scavenging barrier technology.
PRODUCTS
Sorted by application format, the following oxygen-
scavenging products have been offered by major
manufacturers:
Sachets and Labels. Ageless
s
sachets and labels (Mit-
subishi Gas Chemical Co., Japan), Freshmax
s
labels,
Freshpax
s
and StabilOx
s
sachets (Multisorb Technolo-
gies, USA), ATCO
s
sachets and labels (Standa Industrie,
France), O-Buster
s
sachets (Desiccare, USA), Bioka sa-
chets (Bioka Ltd., Finland), Freshilizer
s
sachets (Toppan
Printing Co., Japan), Keplont sachets (Keplon Co., Ltd.
Japan), Tamotsut sachets (Oji Kako, Japan), Oxyeater
s
sachets (Ueno Seiyaku, Japan), Vitalon
s
sachets (Toago-
sei Chemical Industry Co., Japan), Seculet (Oxysorbt in
the U.S.) sachets (Nippon Soda Co., Japan), Sansocutt
sachets (Finetec Co., Japan), and several other Japanese,
Korean, and Chinese sachet producers.
Closure Liners. Pureseal
s
(W. R. Grace and Co., USA),
DarExtend
s
(Darex Container Products, USA), Tri-
SO2RB
s
(Tri-Seal International, USA), Oxycap
s
bottle
crowns (Standa Industrie, France).
Oxygen Scavenging Plastic Packaging Materials. OS
2000
s
(Cryovac, USA), OSP
s
(Chevron-Phillips, USA),
Shelfplus
s
(Ciba Specialty Chemicals Corp., Switzer-
land), Oxyguard
s
(Toyo Seikan Kaisha Ltd., Japan),
OXYGEN SCAVENGERS 847
