Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.


24. See http://www.thecoca-colacompany.com/presscenter/
presskit_contour_bottle_time_line.html.
25. 15 U.S.C. y 1127.
26. See In re Clarke, 17 U.S.P.Q. 2d 1238 (T.T.A.B. 1990).
27. See, Qualitex Co. v. Jacobson Products Co., Inc. 514 U.S. 159,
1303 (1995).
28. J. T. McCarthy, McCarthy on Trademarks and Unfair Com-
petition Fourth Edition Vol. 2, Thomson West, 2008, 11-1 to
11-210, 12-1 to 12-212.
29. Grace & Co. v. Union Carbide Corp., 581 F.Supp. 148
(S.D.N.Y. 1984).
30. In re Serv-A-Portion, Inc., 1 U.S.P.Q.2d 1915 (T.T.A.B. 1986).
31. In re Polytop, 167 U.S.P.Q 383 (T.T.A.B. 1970).
32. 15 U.S.C. yy 1058, 1059.
33. 17 U.S.C. y 102; A. R. Miller and M. H. Davis, Intellectual
Property: Patents, Trademarks, and Copyright, 3rd edition,
West Group, St. Paul, MN 2000, pp. 288–290; P. Goldstein and
R. A.Reese, Copyright, Patent Trademark and Related State
Doctrines: Cases and Materials on the Law of Intellectual
Property, 6th edition, Foundation Press, New York, 1999,
pp. 661–664.
34. Kieselstein-Cord v. Accessories By Pearl, Inc., 208 U.S.P.Q. 1
(2d Cir. 1980).
35. General information on copyrights is available at http://
www.copyright.gov/. See also Copyright Basics at http://
www.copyright.gov/circs/circ1.html.
36. Oscar Mayer Foods Corp. v. Sara Lee Corp., 15 U.S.P.Q.2d
1204 (W.D. Wisc. 1990).
37. Del Monte Fresh Produce Co. v. Dole Food Co., 59 U.S.P.Q. 2d
(S.D. Fla. 2001).
38. See uspto.gov.
39. See, for example., Crown Packaging Technology, Inc. v. Rexam
Beverage Can Co., 05-608MPT (D. Del. Jan. 22, 2008).
40. The USPTO assignment database can be accessed at http://
assignments.uspto.gov/assignments/?db=pat.
41. The USPTO classification index can be accessed at http://
www.uspto.gov/go/classification/uspcindex/indextouspc.htm
42. See for example, R. C. Farber, Landis on Mechanics of Patent
Claim Drafting, 3rd edition, Practicing Law Institute, New
York, 1990.
43. See, for example, North American Container Inc. v. Plastipak
Packaging Inc., 415 F3d 1335 (Fed. Cir. 2005).
PERMEATION OF AROMAS AND SOLVENTS
THROUGH POLYMERIC PACKAGING
MATERIALS
DONGHWA CHUNG
Department of Marine Food
Science and Technology,
Kangnung-Wonju National
University, Gangneung,
Gangwong,
Republic of Korea
Mass transport in food and pharmaceutical package sys-
tems encompasses a number of phenomena referred to as
either permeation, adsorption,ormigration. Permeation
includes the transfer of molecules from the product to the
external environment through the package, or from the
storage environment through the package to the product.
Adsorption involves the takeup of molecules contained by
the product into, but not through, the package, while
migration is the passage of molecules originally contained
by the package itself into the product. These mass transfer
processes also provide the basis of further physiochemical
activities within the package system. Such activities may
induce physiochemical changes in the product, as well as
physical damage of the package, or both (1). In a package/
product system, the mass-transfer processes between
product, package, and storage environment as well as
the physiochemical activities associated with them are
referred to as product/package interactions (2) (see Phar-
maceutical packaging; Packaging, food).
The permeation of water vapor and gases, such as oxygen
and carbon dioxide, has been extensively investigated for
food and pharmaceutical package systems due to its sig-
nificant effects on product shelf life. In the last decade, the
overall concern on the mass transport in package systems
has shifted a little more toward the transfer of volatile
organic compounds of low molecular mass, such as aromas
and solvents, due both to shelf-life reasons and to an
incresing interest in safe and healthy food packaging .
Aromas or flavors are one of the key attributes affecting
the consumer acceptance of a packaged food. Their permea-
tion through the package as well as their adsorption on the
package, called flavor scalping, causes the loss of original
sensorial characteristics of the product during storage,
resulting in the reduction of product shelf life. Solvents,
such as toluene, ethyl acetate, hexane, and abietic acid, are
often contained in package materials, printing inks, or
adhesive systems. Their migration into the packaged pro-
duct not only changes the original sensorial properties of the
product but also in some cases causes the generation of off-
flavors that negatively alterate the consumer perception. In
addition, the migrating solvents raise significant concerns
about the toxicological safety of the product. Obviously, all
these concerns are related to the product/package interac-
tions. In this chapter, the permeation, rather than sorption
or migration, of aromas and solvents has been discussed
because the theory and experimental data on permeation
can be expanded to understant the other processes.
The permeation process occurs in five consecutive
steps, as schematically illustrated in Figure 1: (1) mass
transfer from the bulk of phase 1 to the adjacent surface
of package film, (2) sorption from the adjacent surface into
package film, (3) molecular diffusion, (4) desorption from
package film into the adjacent surface in contact with
phase 2, and (5) mass transfer from the adjacent surface to
the bulk of phase 2.
Since the molecular diffusion within package film is the
rate-limiting step of the whole process, the rate of permea-
tion can be described by Fick’s law of diffusion. Under
steady-state conditions, the flux (J) of permeant through
the film is given by the following Fick’s first law:
J ¼D
dc
dx
ð1Þ
938 PERMEATION OF AROMAS AND SOLVENTS THROUGH POLYMERIC PACKAGING MATERIALS

where D is the diffusion coefficient of permenat in the film,
c is the concentration of permeant in the film, and x is the
distance in the film. Integration of equation (1) yields the
following expression of J:
J ¼
D
l
Dc ð2Þ
where l(=x
2
x
1
) is the thickness of the film and
Dc(=c
1
c
2
) is the difference in permeant concentration
between the two surfaces of the film. Experimental deter-
mination of these surface concentrations is very difficult.
Therefore, the surface concentration is necessary to be
converted to the partial pressure (p) in bulk phase, which
is easily measurable, using Henry’s law of solubility:
c ¼ Sp ð3Þ
where S is the solubility coefficient of permeant in the
film. By substituting equation (3), equation (2) is rewrit-
ten as
J ¼
DS
l
Dp ð4Þ
where Dp(=p
1
p
2
) is the drop of partial pressure across
the film. The product of D and S is often called perme-
ability P, which is the most widely used coefficient for
expressing the rate of packaging permeation:
P ¼ DS ð5Þ
Another coefficient that has been frequently used is
permeance R, which has a physical meaning similar to
that of mass transfer coefficient:
R ¼
DS
l
ð6Þ
Substitution of equation (5) into equation (4) gives the
following well-known packaging permeation equation:
J ¼
P
l
Dp ð7Þ
The gas flux J is often called the permeant transmission
rate; and in the case of water vapor, the water-vapor
transmission rate (WVTR) is commonly used.
The above equations are applicable only for situations
where D is independent of permeant concentration and S
follows Henry’s law. However, when the permeation pro-
cess involves highly interactive organic permeants such as
aroma, flavor, or solvent molecules, the diffusion process is
more complex than the diffusion of simple gases, and the
diffusion coefficient may vary as a function of permeant
concentration and time (3–6). Where D varies with time,
the diffusion is often called non-Fickian. Mears (7) pro-
posed the following expressions for cases where the diffu-
sion coefficient is concentration-dependent:
D ¼ D
0
1 þacðÞ ð8Þ
or
D ¼ D
0
exp bcðÞ ð9Þ
Equation (9) is more suitable in cases where the diffusion
coefficient is strongly concentration-dependent. Here D is
the differential diffusion coefficient, D
0
is the limiting
diffusion coefficient, and a and b are constants.
The variables affecting permeation and diffusion pro-
cesses in a package can be grouped as follows:
1. Compositional variables:
a. Chemical composition of the polymer and
permeant
b. Morphology of the polymer
c. Permeant characterstics
d. Concentration of the permeant
e. Presence of copermeant
2. Environmental and geometric factors
a. Temperature
b. Relative humidity
c. Packaging geometry
While an in-depth treatment of each of the above factors is
beyond the scope of this article, selected examples are
discussed to illustrate their role in the transport of organic
permeants in barrier polymers. For a more in-depth
treatment, the reader is referred to the references listed
in the Bibliography and references cited therein.
CHEMICAL COMPOSITION OF THE PACKAGING
MATERIAL
The relationship between permeant transfer characteris-
tics and the basic molecular structure and chemical
composition of a polymer is rather complex, and a number
of factors contribute to the permeation and diffusion
processes; the most important are as follows:
. Cohesive-energy density, which produces strong in-
termolecular bonds, van der Waals or hydrogen
Package film
J J
p
1
p
2
Phase 1 Phase 2
Sorption
Desorption
Diffusion
Surface
mass transfer
Surface
mass transfe
r
c
1
c
2
x
1
x
2
Figure 1. Schematic diagram of gas or vapor permeation
through a package film.
PERMEATION OF AROMAS AND SOLVENTS THROUGH POLYMERIC PACKAGING MATERIALS 939

bonds, and regular, periodic arrangement of such
bonding groups.
. The glass-transition temperature (T
g
) of the polymer,
above which free vibrational motion and rotational
motion of polymer chains occur, so that different
chain conformations can be assumed.
With respect to the glass-transition temperature of
barrier polymer structures, DeLassus (8) reported that
glassy polymers have very low diffusion coefficients for
flavor, aroma, and solvent molecules at low concentra-
tions. Typically, these values are too low to measure by
standard analytical procedures. The diffusion coefficient
determines the dynamics of the permeation process and
thus the time to reach steady state, which accounts for
glassy polymers exhibiting high-barrier characteristics to
organic permeants. Polyolefins, being well above their
glass-transition temperature, are non-glassy polymers
and have high diffusion coefficients for organic permeants,
and steady-state permeation is established quickly in such
structures.
Polymer-free volume is also a function of structural
regularity, orientation, and cohesive-energy density. The
aforementioned structure–property relationships all con-
tribute to a decrease in solubility and diffusivity and thus
permeability.
Salame (2) has proposed a relationship between poly-
mer molecular structure and permeability based on an
empirical constant (p), or ‘‘Permachor’’ constant, which,
when substituted into the Permachor equation, predicts
the gas permeability of polymer structures. The correla-
tion parameter or the ‘‘Permachor’’ constant is based on
the cohesive-energy density and free volume of the poly-
mer, two major properties of the polymer. Agreement
between the Permachor constant and film permeability
has been shown to be quite good for oxygen, CO
2
, and
nitrogen, but not for water vapor and organic vapors.
The equation for relating gas permeability to the
Permachor constant is as follows:
P ¼ A expðspÞð10Þ
where A and s are constants for any given gas at tem-
perature T and p is the Permachor constant of the
polymer.
POLYMER MORPHOLOGY
Solid-state polymer chains can be found in a random
arrangement to yield an amorphous structure or a highly
ordered crystalline phase. Most polymers used in packa-
ging are semicrystalline or amorphous materials (see
Polymer properties).
Morphology refers to the physical state by which
amorphous and semicrystalline regions coexist and relate
to each other in a polymer, and it depends not only on its
stereochemistry but also on whether the polymer has been
oriented, on the conditions of temperature, strain rate,
and cooling temperature, and on the melt cooling rate.
Fundamental properties that are associated with poly-
mer morphology and will therefore influence the perme-
ability and diffusivity characteristics of the polymer
include:
. Structural regularity or chain symmetry, which can
readily lead to a three-dimensional order or crystal-
linity. This is determined by the type of monomer(s)
and the conditions of the polymerization reaction.
. Chain alignment or orientation, which allows later-
ally bonding groups to approach each other to the
distance of the best interaction, enhancing the ten-
dency to form crystalline materials.
Morphology is thus important in determining the bar-
rier properties of semicrystalline polymers. This is illu-
strated by the results of permeation studies carried out on
biaxially oriented polyethylene terephthalate (PET) films
of varying thermomechanical history (9).
Film samples were biaxially stretched at a strain of
350%/s, based on the initial dimensions of 4 in. 4 in.,
which corresponded to an orientation rate of 14 in./s
biaxially.
The degree of orientation was 400% based on the initial
dimensions. The orientation temperature was 901C,
1001C, and 1151C, respectively.
Table 1 summarizes the results of permeation studies
carried out with ethyl acetate in PET film biaxially
oriented at 901C and 1151C, respectively, and serves to
illustrate the effect of thermomechanical history (stretch-
ing-temperature values) on the relative barrier properties
of PET, for the permeation of ethyl acetate. The percent
crystallinity of PET film oriented at 901C was 22%, while
the percent crystallinity of the film sample oriented at
Table 1. Permeability of Ethyl Acetate through PET Film Biaxially Oriented at 901C and 1151C
Orientation
Temperature (1C) Vapor Activity (a
v
) Run Temperature (1C) Permeability
a
, P 10
20
Lag Diffusion
Coefficient
b
, D 10
12
90 0.59 30 2.6 1.8
0.43 37 4.8 2.9
0.21 54 15.4 11.0
115 0.59 30 0.014
c
—
0.21 54 3.6 5.3
a
Permeability units are (kg m) (m
2
s
1
Pa
1
).
b
Diffusion coefficient units are cm
2
s
1
.
c
No permeation after 550 h. Value of P reported represents an upper bound.
940 PERMEATION OF AROMAS AND SOLVENTS THROUGH POLYMERIC PACKAGING MATERIALS
1151C was 31%. As shown, ethyl acetate permeability
values decreased by approximately four times by increas-
ing the film orientation temperature from 901Cto1151C.
PERMEANT CHARACTERISTICS
The most important characteristics of the permeant af-
fecting its transfer in and through a film include:
. Carbon chain length or boiling point
. Functional groups and polarity
With respect to the carbon chain length, different
studies have been carried out to evaluate the influence
of this characteristic on permeation phenomenon, parti-
cularly influencing the aforementioned second step of this
process, that is, sorption. In particular, homologous series
of hydrocarbons, esters, aldehydes, alcohols, and benzo-
ates in the vapor phase were taken into consideration (10,
11). The obtained results showed that, in compounds
below 10 carbon atoms, both the solubility and the perme-
ability coefficients (S and P, respectively) of the volatiles
increased two- to five-fold with each additional methylene
group, depending on the types of packaging films. Ob-
viously, diffusivity (D) decreased due to the steric hin-
drance. On the contrary, for molecules with 11 or more
carbon atoms, the increments were less or negative, as in
the case of aldehydes. Different experiments on the per-
meation of alkyl esters (C
5
–C
10
), methyl ketones (C
7
–C
10
),
aldehydes (C
7
–C
10
), and sulfur compounds through differ-
ent plastic films (PP, PE, PVDC, EVOH) led to similar
results (12–16). Sorption into plastic films can take place
also in the case of longer molecules as, for instance, fatty
acids. A specific study (17) showed that these kinds
of compounds with 16–18 carbon atoms moved through
the LDPE film, with the final result of delaminating the
composite (multilayer) flexible packaging film. Finally,
carbon chain length and boiling point are strictly linked
each other. For instance, within sorbates, there is a
proportional relationship between boiling point and solu-
bility. Moreover, a further relationship exists between the
logarithm of the solubility coefficient and the boiling point
of linear esters, alkanes, and ketones (e.g., hexyl butyrate,
n-decane, or 3-octanone) through PVDC polymers (12–16).
The effect of functional groups on the transfer proper-
ties of aroma compounds through a film is much larger
than the boiling point in the liquid phase rather than in
the vapor phase, due to close interactions between mole-
cules (10). Hydrocarbons and oxygenated terpenes, for
example, did not over PP nor LDPE by permeation when
put in aqueous model solutions (11). The polarity of the
solutes can be effectively considered as the most impor-
tant controlling factor to explain the tendency to permea-
tion. Researchers pointed out that carvone (C
10
H
14
O) and
limonene (C
10
H
16
O), even if similar terpenes, show differ-
ent sorption and diffusion properties. In general, permea-
tion of limonene (less polar than carvone), through plastic
films, takes place quickly, because of lesser cohesive forces
(11). In another study on the permeation of citrus oil
compounds through polyolefins, it was found that terpenes
(C
10
carbon length) exhibited the highest affinity for the
polymers, followed by sesquiterpenes (C
15
) (18). In addi-
tion, the permeation of esters and aldehydes was more
intense than alcohols due to polarity. Within aldehydes,
those saturated showed a greater permeation compared
to the unsaturated ones. For orange juice aromas, the
following ranking was established in terms of affinity
to LDPE: hydrocarbonsWchetonesWestersWaldehydesW
alcohols (15, 16, 19). The same ranking can be extended
to apple juice aromas associated with LDPE, LLDPE,
PET, PA, and PP films (20). Another important factor
influencing the permeation of flavor components into
plastic films (in particular concerning the sorption step)
is the pH of the system, which can modify the polarity of
the permeant. The sorption of 2-hexanal into PE was
found to be increased by a factor of 40 between pH 3.0
and pH 5.0 (21). Analogously, sorption of alcohols into
LDPE was increased by lowering the pH up to 3.0 (19).
Finally, sorption of limonene into LDPE at 221C was
higher at pH 5.2 than at pH 2.6 (1.3 times more; results
significant at 95% level) (22).
CONCENTRATION DEPENDENCE OF THE TRANSPORT
PROCESS
Permeance of limonene vapor through (1) oriented poly-
propylene, (2) saran-coated oriented polypropylene, (3)
two-sided acrylic (heat-seal)-coated biaxially oriented
polypropylene, and (4) one-side saran-coated, one-side
acrylic-coated polypropylene film samples, as a function
of permeant concentration, is presented graphically in
Figure 2, where permeance (R) is plotted as a function of
permeant concentration (23). The observed concentration
dependency of the permeance values may be attributed to
the permeant/polymer interaction, resulting in configura-
tional changes and alteration of polymer-chain conforma-
tional mobility. Zobel (24) reported similar findings for the
transport of the permeant benzyl acetate through coex-
truded oriented polypropylene and saran-coated oriented
polypropylene, at various permeant concentrations.
PRESENCE OF COPERMEANT
As shown above, organic vapors are capable of exhibiting
concentration-dependent mass transport processes.
Therefore, the type and/or mixture of organic vapors
permeating will determine the magnitude of sorption
and permeation, as well as the effect of a copermeant on
permeant permeability. The synergistic effect of a coper-
meant is illustrated by the results of permeation studies
carried out on a biaxially oriented polypropylene film. The
degree of film orientation was 430% (machine direction)
and 800% (cross-machine direction), based on the initial
dimensions. Binary mixtures of ethyl acetate and limo-
nene of varying concentration were evaluated as the
organic permeants (25).
Results of permeation studies for selected ethyl acetate/
limonene binary vapor mixtures are presented in
Figures 3 and 4, respectively. As shown in Figure 3 (ethyl
PERMEATION OF AROMAS AND SOLVENTS THROUGH POLYMERIC PACKAGING MATERIALS 941
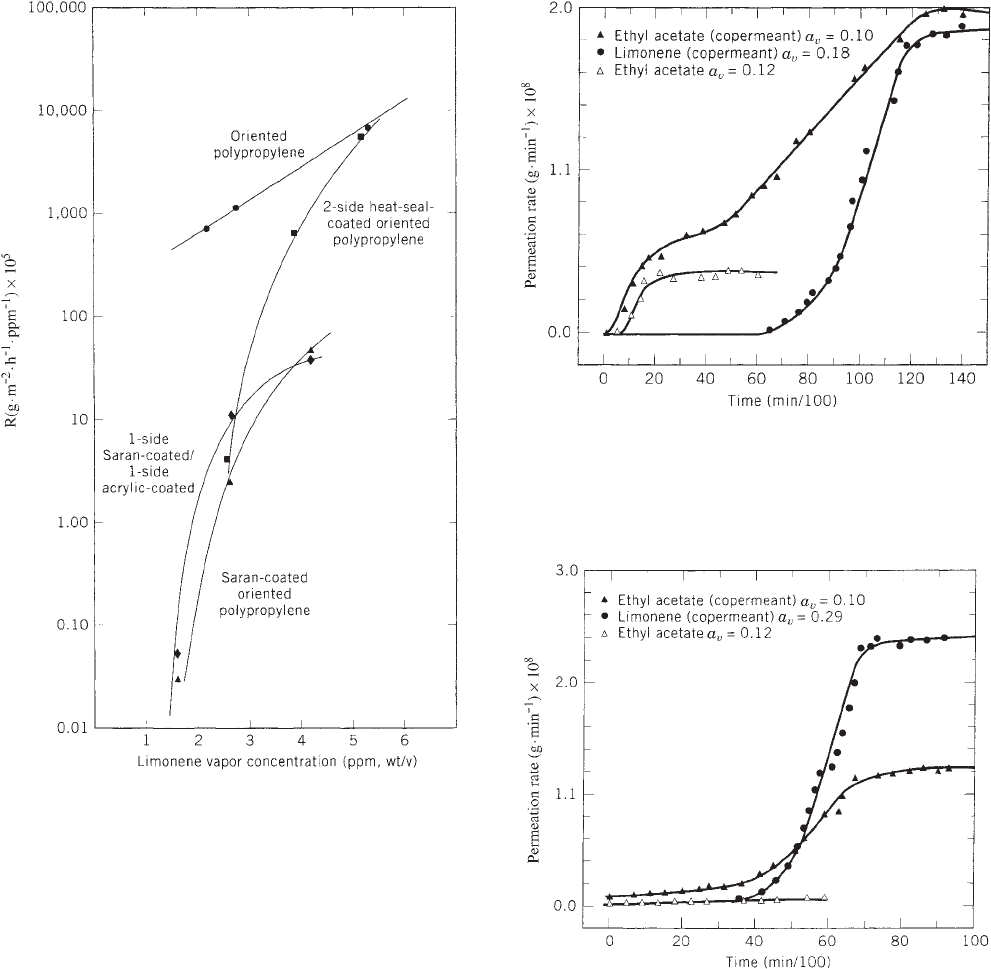
acetate a
v
= 0.10 and limonene a
v
= 0.18), limonene vapor
had a significant effect on the transport properties of the
copermeant. A 500% increase in the permeability constant
of ethyl acetate was obtained, when compared to ethyl
acetate vapor permeability alone, at similar test condi-
tions. However, at this concentration level, ethyl acetate
did not appear to influence the permeation of the limonene
vapor. The transmission-rate profile curve for limonene
vapor in the binary mixture is superimposed in Figure 3,
to provide a complete description of the transmission
characteristics of the mixed-vapor system.
For the ethyl acetate (a
v
= 0.1)/limonene (a
v
= 0.29) bin-
ary mixture, a permeation rate 40 times greater than the
transmission rate of pure ethyl acetate vapor, of an equiva-
lent concentration, was obtained. This is illustrated in
Figure 4, where the transmission–profile plot of the binary
mixture is presented, and compared to the transmission-
rate profile curve for ethyl acetate vapor alone. Again, at
this concentration level, ethyl acetate did not appear to
affect the permeation characteristics of limonene vapor.
For studies carried out with the binary mixture of ethyl
acetate a
v
= 0.48 and limonene a
v
= 0.18, the individual
components of the mixture were found to have a signifi-
cant effect on the permeation rates of the copermeant.
Since the permeability is drastically affected in the
sorbate/polymer systems studied, the diffusion coefficient
and/or the solubility coefficient must also vary in the
presence of a copermeant to account for the observed
increases in the transmission rates for the components
of ethyl acetate/limonene binary mixtures. By direct mea-
surement of the equilibrium solubility, Nielsen and Giacin
Figure 3. Comparison of the transmission profile of the binary
mixture, ethyl acetate (a
v
= 0.1)/limonene (a
v
= 0.18), with the
transmission profile of ethyl acetate (a
v
= 0.12) through oriented
polypropylene.
Figure 2. The effect of limonene-vapor concentration on log R for
oriented polypropylene and coated oriented polypropylene struc-
tures (221711C).
Figure 4. Comparison of the transmission profile of the binary
mixture, ethyl acetate (a
v
= 0.1)/limonene (a
v
= 0.29), with the
transmission profile of ethyl acetate (a
v
= 0.12).
942 PERMEATION OF AROMAS AND SOLVENTS THROUGH POLYMERIC PACKAGING MATERIALS

(26) found that the solubility coefficient values were
independent of sorbate vapor activity, over the range of
activity levels studied, and were not affected by the
presence of a copermeant.
For the ethylacetate (a
v
= 0.10)/limonene (a
v
= 0.21) bin-
ary mixture, Hensley (25) reported a permeation rate of
ethyl acetate 40 times greater than the transmission rate
for pure ethyl acetate vapor of an equivalent concentra-
tion, while sorption studies showed the solubility coeffi-
cient of ethyl acetate to be constant and did not deviate
from Henry’s law in the presence of a copermeant. A
possible explanation for the dramatic increase in the
permeability coefficients for ethyl acetate in the presence
of limonene as a copermeant lies with the high coper-
meant dependence of the diffusion coefficient.
For ethyl acetate/limonene binary mixtures, limonene
as a copermeant appears to have little or no effect on the
solubility of ethyl acetate in oriented polypropylene film,
while significantly changing the inherent mobility of ethyl
acetate within the polymer bulk phase. This accounts for
the observed increase in the transmission rates for ethyl
acetate through the OPP (oriented polypropylene) film in
the presence of limonene.
While not fully understood, the proposed copermeant
dependence of the diffusion coefficient may be due in part
to copermeant-induced relaxation effects occurring within
the polymer matrix. The absorption of organic vapors can
result in polymer swelling and thus change the conforma-
tion of the polymer chains. These conformational changes
are controlled by the retardation times of polymer chains.
If these times are long, stresses may be set up that relax
slowly. Thus, the absorption and diffusion of organic
vapors can be accompanied by concentration as well as
time-dependent processes within the polymer bulk phase,
which are slower than the micro-Brownian motion of
polymer chain segments that promote diffusion (7).
There is precedence in the literature in support of such
long-time-period relaxation effects occurring in polymer
films above their glass-transition temperature (27, 28).
Thus, there may be copermeant-induced relaxation effects
occurring during the diffusion of ethyl acetate/limonene
binary mixtures in the oriented polypropylene film inves-
tigated. Such relaxation processes, which occur over a
longer time scale than diffusion, may be related to a
structural reordering of the free-volume elements in the
polymer, thus providing additional sites of appropriate
size and frequency of formation, which promote diffusion
and account for the observed increase in the permeation
rate of ethyl acetate in the presence of limonene as a
copermeant.
EFFECT OF RELATIVE HUMIDITY
The permeation of acetone vapor through amorphous
polyamide (nylon 6I/6 T) under dry conditions and in the
presence of a humid environment serves to illustrate the
effect of water activity or moisture content on the barrier
properties of hydrophilic polymer films. Studies were
conducted at 601C, 751C, 851C, and 951C, at a constant
permeant partial pressure value of 92 mm Hg (0.29 g/L).
Water activity (a
w
) of the permeant steam was maintained
at 0.7 (70% RH), when measured at 231C. Different film
samples were used for dry- and humid-condition experi-
ments. For an experiment, each run was maintained for a
period of 8–14 days after attaining steady state, to ensure
that the system was at equilibrium (29).
The respective permeability values are summarized in
Table 2. As shown, sorption of water vapor resulted in an
increase in permeability, as compared to dry conditions,
with an increase of approximately 1.5 times being
observed.
A further illustration of the effect of water activity on
the barrier properties of polymer films is presented in
Figure 5, where the total quantity of ethyl acetate per-
meated is plotted as a function of time, for the permeation
of ethyl acetate through SiOx PET and EVAL-F films (30).
The test conditions were as follows: temperature 221C,
190 ppm (wt/v) concentration of ethyl acetate vapor, at
56% and 87% RH, respectively. Fluctuation of relative
humidity was 72%, and fluctuation of organic vapor
concentration was 75%.
For time, W500 h of continuous testing, there was no
measurable permeation at 56% RH. The results indicate
that both test films were excellent ethyl acetate vapor
barriers at 56% RH and ambient temperature. However,
Table 2. Summary of Permeability Values of Acetone
through Nylon 6I/6 T
a
Temperature (1C)
P 10
19
(Dry
Condition)
a
P 10
19
(Humidified
Conditions)
a
60 3.7 4.9
75 6.5 11.2
85 9.8 17.6
95 11.8 —
a
Permeability units are expressed in (kg m) (m
2
s
1
Pa
1
).
Figure 5. Transmission-rate profile curves of ethyl acetate vapor
through SiO
x
PET and EVAL-F films at 221C, 190 ppm (wt/v), and
56% and 87% RH.
PERMEATION OF AROMAS AND SOLVENTS THROUGH POLYMERIC PACKAGING MATERIALS 943

as shown, at 87% RH the EVAL-F film had a significant
increase in the permeation rate of ethyl acetate vapor,
while the SiO
x
PET film still showed no measurable
increase in the rate of permeation. Table 3 summarizes
the permeance values. Also presented in Table 3 are
upper-limit-value estimations for film permeance.
Liu et al. (31) studied the effect of both water activity
and permeant vapor concentration on the permeation of
toluene vapor through a saran (PVDC)-coated OPP film.
Studies were conducted at 22711C. Since the film inves-
tigated in the present study is a multilayer structure,
permeance values are presented to describe the barrier
properties of the total structure.
The permeability parameters determined are summar-
ized in Table 4.
A comparison of the permeance values for toluene,
determined as a function of toluene vapor concentration
and water activity, showed that the permeance values are
highly dependent on both water-activity levels and to-
luene-vapor concentration. The findings suggested that
the sorption of water vapor and/or toluene vapor leads to
relaxation in the molecular structure of the saran barrier
coating, resulting in a high concentration dependency of
the permeance values. Toluene appears to be much more
effective than water vapor in promoting relaxation of the
saran coatings, since at the highest toluene-vapor concen-
tration level studied (81 ppm), the permeance rate is
independent of water-activity or sorbed-water levels.
However, at the lower permeant concentration levels
studied, the effect of sorbed water is quite significant,
making this polymer structure very vulnerable to the
permeation of low concentrations of toluene in the pre-
sence of sorbed water.
The results of this study also serve to illustrate the
complex interrelationship of the barrier characteristics of
the polymer film, permeant vapor activity, and sorbed-
water concentration.
EFFECT OF TEMPERATURE ON PERMEATION
PARAMETERS
Permeability, diffusion, and solubility coefficients follow a
van’t Hoff–Arrhenius relationship as given in equations
(11–13):
P ¼ P
0
exp
E
p
R
c
T
ð11Þ
D ¼ D
0
exp
E
D
R
c
T
ð12Þ
S ¼ S
0
exp
DH
s
R
c
T
ð13Þ
where E
p
= activation energy for permeation
E
D
= activation energy for diffusion
DH
s
= molar enthalpy of sorption
P
0
, D
0
, S
0
= constants
R
c
= universal gas constant
T = absolute temperature
Lin (32) determined the permeability of ethyl acetate
and toluene for the following commodity films: (a) oriented
polypropylene, (b) high-density polyethylene, (c) glassine,
(d) saran-coated oriented polypropylene, (e) acrylic-coated
oriented polypropylene, and (f) metallized polyethylene
terephthalate/OPP laminate.
Permeation studies were carried out at three tempera-
tures to allow evaluation of the activation energies.
For each temperature, three vapor activity levels were
Table 3. The Effect of Relative Humidity on the
Permeation of Ethyl Acetate Vapor through SiO
x
PET
and EVAL-F Films
Relative Humidity (%) Sample Permeance
a
R 10
17
56 SiO
x
PET o1.1
EVAL-F o2.2
87 SiO
x
PET o2.2
EVAL-F 840740
a
Permeance units are expressed in kg m
2
s
1
Pa
1
.
Table 4. The Effect of Water Activity on the Permeation of Toluene Vapor through a Two-Side-PVDC Coated OPP Film
Toluene-Vapor
Concentration
(ppm) (Mass/
Volume)
Toluene-Vapor
Pressure (Pa) Water Activity (a
w
)
Transmission
Rate
a,b
(J) Permeance
a,c
(R) Lag Time (h)
40 1203.8 0.50 0.73 10
13
6.0 10
17
94.0
0.86 0.72 10
10
6.0 10
14
55.0
60 1778.2 0 0.4 10
11
2.2 10
15
82.0
0.50 0.32 10
10
1.8 10
14
38.0
0.86 1.6 10
10
9 10
14
29.0
81 2407.6 0 1.1 10
7
1.6 10
11
2.1
0.50 1.1 10
9
4.6 10
11
2.2
0.86 1.2 10
7
5.0 10
11
1.9
a
Average of replicate runs, with a 2s confidence limit within 710%.
b
Transmission rate units are expressed in kg m
2
s
1
.
c
Permeance units are expressed in kg m
2
s
1
Pa
1
.
944 PERMEATION OF AROMAS AND SOLVENTS THROUGH POLYMERIC PACKAGING MATERIALS

evaluated. Since several films investigated are multilayer
or barrier-coated structures, permeance values are pre-
sented to describe the barrier properties of the total
structure (see also Barrier polymers).
Permeance values determined at ethyl acetate vapor
activity levels of a
v
= 0.095, 0.21, and 0.41 for the respec-
tive barrier structures are summarized in Tables 5–9. For
the metallized polyethylene terephthalate/OPP laminate,
no measurable rate of diffusion was detected, following
continuous testing for 44 h at 701C and a vapor activity of
a = 0.41.
Determined permeance values for toluene activity le-
vels of a
v
= 0.067, 0.22, and 0.44 for the respective barrier
structures are summarized in Tables 10–14. There was
no measurable rate of diffusion for the polyethylene
Table 5. Permeance of Ethyl Acetate through Oriented
Polypropylene (OPP) as a Function of Vapor Activity and
Temperature
a
Vapor Activity
b
Temperature (1C) Permeance
c
R 10
12
0.095 30 0.6
40 1.7
50 4.4
0.21 30 0.7
40 1.9
50 4.4
0.41 30 0.9
40 2.1
50 4.8
a
The results reported are the average of duplicate analyses.
b
Vapor-activity values were deter mined at ambient temperature (241C).
c
Permeance units are expressed in kg m
2
s
1
Pa
1
.
Table 6. Permeance of Ethyl Acetate through
High-Density Polyethylene (HDPE) as a Function of
Vapor Activity and Temperature
a
Vapor Activity
b
Temperature (1C) Permeance
c
R 10
11
0.095 30 1.0
40 1.7
50 2.7
0.21 30 2.4
40 2.8
50 3.4
0.41 30 2.9
40 3.8
50 4.8
a
The results reported are the average of duplicate analyses.
b
Vapor-activity values were deter mined at ambient temperature (241C).
c
Permeance units are expressed in kg m
2
s
1
Pa
1
.
Table 7. Permeance of Ethyl Acetate through Glassine as
a Function of Vapor Activity and Temperature
a
Vapor Activity
b
Temperature (1C) Permeance
c
R 10
12
0.095 23 1.1
30 2.0
40 4.2
0.21 23 2.3
30 2.9
40 5.2
a
The results reported are the average of duplicate analyses.
b
Vapor-activity values were deter mined at ambient temperature (241C).
c
Permeance units are expressed in kg m
2
s
1
Pa
1
.
Table 8. Permeance of Ethyl Acetate through
Saran-Coated OPP as a Function of Vapor Activity
and Temperature
a
Vapor Activity
b
Temperature (1C) Permeance
c
R 10
13
0.095 40 1.0
50 5.0
60 15.1
0.21 40 1.6
50 5.2
60 14.9
0.41 40 2.8
50 7.7
60 26.5
a
The results reported are the average of duplicate analyses.
b
Vapor-activity values were determined at ambient temperature (241C).
c
Permeance units are expressed in kg m
2
s
1
Pa
1
.
Table 9. Permeance of Ethyl Acetate through
Acrylic-Coated OPP as a Function of Vapor Activity
and Temperature
a
Vapor Activity
b
Temperature (1C) Permeance
c
R 10
12
0.095 50 0.25
60 1.0
70 2.6
0.21 50 0.3
60 1.2
70 3.6
0.41 50 0.3
60 1.2
70 3.3
a
The results reported are the average of duplicate analyses.
b
Vapor-activity values were determined at ambient temperature (241C).
c
Permeance units are expressed in kg m
2
s
1
Pa
1
.
Table 10. Permeance of Toluene through OPP as a
Function of Vapor Activity and Temperature
a
Vapor Activity
b
Temperature (1C) Permeance
c
R 10
11
0.067 30 0.5
40 1.15
50 1.8
0.22 30 0.75
40 1.4
50 2.7
0.44 30 1.4
40 2.0
50 3.1
a
The results reported are the average of duplicate analyses.
b
Vapor-activity values were determined at ambient temperature (241C).
c
Permeance units are expressed in kg m
2
s
1
Pa
1
.
PERMEATION OF AROMAS AND SOLVENTS THROUGH POLYMERIC PACKAGING MATERIALS 945

terephthalate/OPP laminate structure following continu-
ous testing for 44 h at 701C and a vapor activity of 0.44.
From equation (11) the temperature dependency of the
transport process associated with the respective barrier
membranes, over the temperature range studied, was
found to follow the Arrhenius relationship. From the
slopes of the Arrhenius plots, the activation energy for
the permeation process (E
p
) was determined for the
respective film samples, as a function of vapor activity.
The determined activation energy values for both ethyl
acetate and toluene are summarized in Tables 15, 16,
respectively.
NUMERICAL CONSISTENCY OF PERMEATION DATA
In addition to determining the permeability values of
organic permeants, it is also important to determine the
diffusion coefficients and to evaluate the consistency of the
experimental data obtained. The numerical consistency of
Table 11. Permeance of Toluene through HDPE as a
Function of Vapor Activity and Temperature
a
Vapor Activity
b
Temperature (1C) Permeance
c
R 10
11
0.067 30 2.1
40 3.4
50 5.6
0.22 30 4.0
40 5.5
50 8.0
0.44 30 7.7
40 8.6
50 9.2
a
The results reported are the average of duplicate analyses.
b
Vapor-activity values were deter mined at ambient temperature (241C).
c
Permeance units are expressed in kg m
2
s
1
Pa
1
.
Table 12. Permeance of Toluene through Glassine as a
Function of Vapor Activity and Temperature
a
Vapor Activity
b
Temperature (1C) Permeance
c
R 10
12
0.067 30 2.8
40 8.9
50 5.1
0.22 30 3.5
40 5.3
50 6.6
0.44 30 4.1
40 5.5
50 6.8
a
The results reported are the average of duplicate analyses.
b
Vapor-activity values were deter mined at ambient temperature (241C).
c
Permeance units are expressed in kg m
2
s
1
Pa
1
.
Table 13. Permeance of Toluene through Saran-Coated
Oriented Polyethylene (Saran OPP) as a Function of
Vapor Activity and Temperature
a
Vapor Activity
b
Temperature (1C) Permeance
c
R 10
13
0.067 40 1.0
50 5.8
60 21.9
0.22 40 2.5
50 9.7
60 25.0
0.44 40 3.1
50 12.8
60 28.9
a
The results reported are the average of duplicate analyses.
b
Vapor-activity values were deter mined at ambient temperature (241C).
c
Permeance units are expressed in kg m
2
s
1
Pa
1
.
Table 14. Permeance of Toluene through Acrylic-Coated
Polypropylene as a Function of Vapor Activity and
Temperature
a
Vapor Activity
b
Temperature (1C) Permeance
c
R 10
13
0.067 50 ND
d
60 ND
d
70 ND
d
0.22 50 ND
d
60 ND
d
70 ND
d
0.44 50 1.4
60 2.0
70 3.1
a
The results reported are the average of duplicate analyses.
b
Vapor-activity values were determined at ambient temperature (241C).
c
Permeance units are expressed in kg m
2
s
1
Pa
1
.
d
Without detectable response after 44-h test.
Table 15. Activation-Energy Values for the Permeation of
Ethyl Acetate through Polymer Membranes
E
p
(kcal mol
1
)
Polymer Membranes a
v
= 0.095 a
v
= 0.21 a
v
= 0.41
OPP 17.7 14.6 14.5
HDPE 9.3 4.4 4.9
Glassine 14.6 9.6 N/A
(a)
Saran OPP 24.9 23.0 24.5
Acrylic OPP 25.9 27.9 25.0
a
N/A, data not available.
Table 16. Activation Energy Values for the Permeation of
Toluene through Polymer Membranes
E
p
(kcal mol
1
)
Polymer Membranes a
v
= 0.067 a
v
= 0.22 a
v
= 0.44
OPP 12.63 12.53 7.79
HDPE 9.45 6.90 1.90
Glassine 5.78 6.21 5.02
Saran OPP 34.00 34.10 35.41
Acrylic OPP N/A
a
N/A
a
30.25
a
N/A, data not available.
946 PERMEATION OF AROMAS AND SOLVENTS THROUGH POLYMERIC PACKAGING MATERIALS
the permeation data will affect the values of both the
diffusion coefficient and the permeability and would in-
dicate any variations of the system parameters, such as
temperature, or permeant concentration changes during
the course of the permeation experiment. Gavara and
Hernandez (33) have described a simple procedure for
determining the diffusion coefficient and for performing a
consistency analysis on a set of experimental permeation
data from a continuous-flow permeation study. This pro-
cedure was applied to the continuous-flow permeation
data obtained, to provide a better understanding of the
mechanism of the diffusion and sorption processes asso-
ciated with the permeation process. The consistency test
for continuous-flow permeation experimental data has
been described in detail by Gavara and Hernandez (33)
and is summarized briefly below.
The value of the permeation rate at any time J
t
, during
the unsteady-state portion of the permeation experiment
varies from zero, at time equal to zero, up to the transmis-
sion rate value (J
N
) reached at the steady state. This is
described by the following expression (34):
J
t
J
N
¼
4
p
1=2
‘
2
4Dt
1=2
X
N
n¼1;3;5
exp
n
2
‘
2
4Dt
ð14Þ
Equation (14) is simplified to the following form:
g ¼
4
p
1=2
ðXÞ
1=2
expðXÞð15Þ
where g is equal to the transmission rate ratio J
t
/J
N
and
X = ‘
2
/4Dt. In equation (14), D is assumed to be indepen-
dent of permeant concentration and time. For each value
of g, a value of X can be calculated, and from a plot of 1/X
versus t, the diffusion coefficient (D) can be determined.
The authors further described two dimensionless con-
stants, k
1
and k
2
;
k
1
¼
t
1=4
t
3=4
¼
X
1=4
X
3=4
¼ 0:4405 ð16Þ
k
2
¼
t
1=4
t
1=2
¼
X
1=4
X
1=2
¼ 0:6681 ð17Þ
where X
1/4
, X
1/2
, and X
3/4
denote the numerical values of X
when the permeation experiment has reached values of
0.25, 0.5 and 0.75, respectively, for g, the transmission rate
ratio.
The numerical values of the constants k
1
and k
2
as
given in Equations (16) and (17), together with the linear
relationship of 1/X versus t, will provide values of the
diffusion coefficient and a criteria to evaluate the consis-
tency of the experimental data.
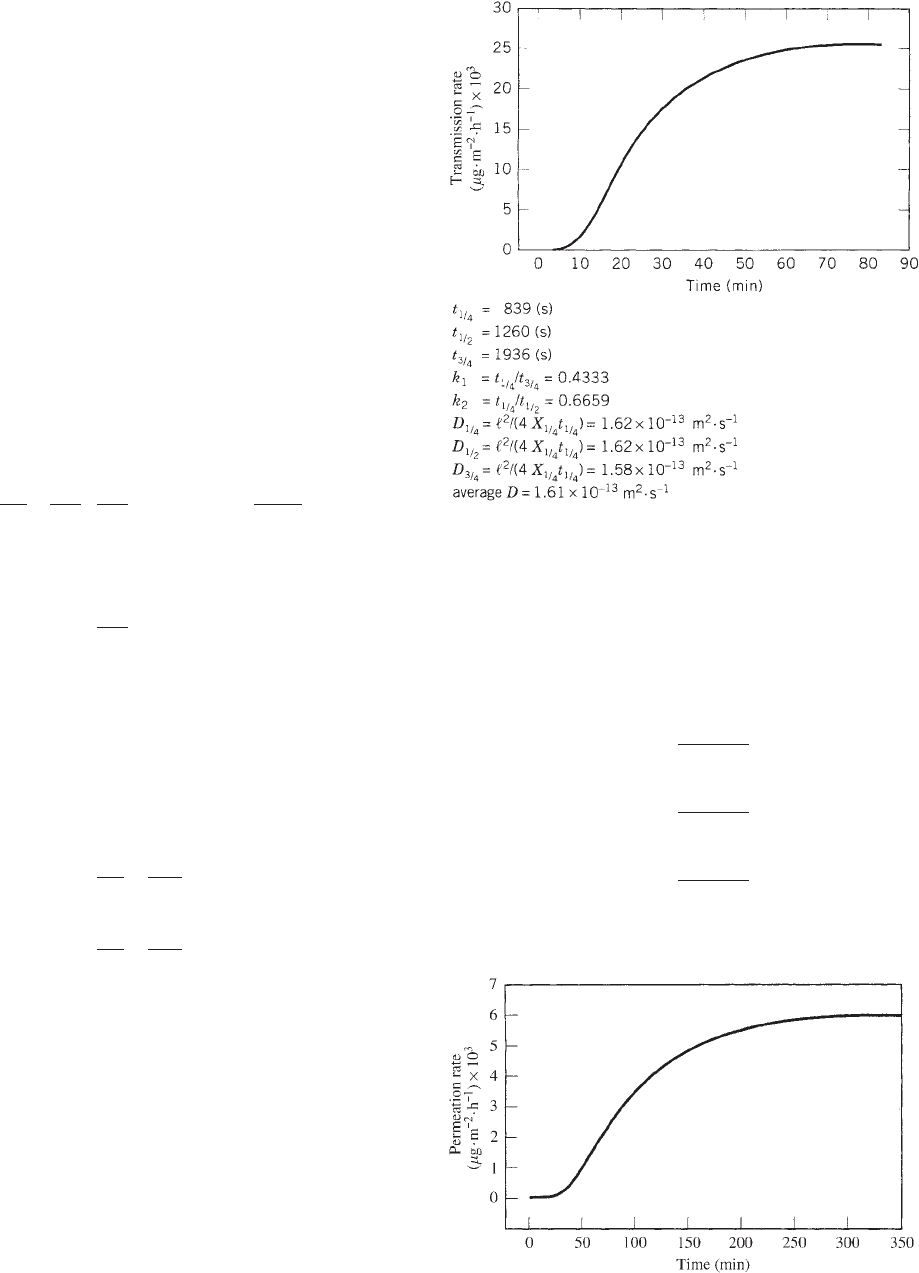
The results of a series of permeation studies carried out
with limonene vapor are presented graphically in Figures
6, 7, where the transmission rate is plotted as a function
of time, and serves to illustrate the applicability of the
consistency test to permeation data obtained for polyethy-
lene and polypropylene (35). The values of k
1
and k
2
calculated from the experimental data and the associated
diffusion coefficients, D
1/4
, D
1/2
, and D
3/4
, obtained by
substitution into the expressions
D
1=4
¼
‘
2
4X
1=4
t
1=4
ð18Þ
D
1=2
¼
‘
2
4X
1=2
t
1=2
ð19Þ
D
3=4
¼
‘
2
4X
3=4
t
3=4
ð20Þ
Figure 6. Transmission-rate profile and consistency test for the
permeation of limonene (a
v
= 0.4) through high-density polyethy-
lene at 501C.
Figure 7. Transmission-rate profile and consistency test for the
permeation of limonene (a
v
= 0.2) through oriented polypropylene.
PERMEATION OF AROMAS AND SOLVENTS THROUGH POLYMERIC PACKAGING MATERIALS 947
