Yao N. Focused Ion Beam Systems: Basics and Applications
Подождите немного. Документ загружается.

hard samples are also applicable to biological samples. In-situ sample
manipulation allows for site-specific morphological and structural analysis
through quasi-real time viewing of the sample during processing. The ability
to quickly mill cross sections in a variety of directions provides a strong
understanding of the relationship between cell structure and morphology.
Imaging and sectioning of biological specimens from single yeast cells to
small arthropods have provided high-resolution images of biological struc-
tures from the subcellular level to the microstructural level of tissue and
organs [3,6]. Preliminary results indicate the sample preparation must be of
primary concern during the imaging and manipulation of biological samples
[3,5,9]. Care must be taken to ensure the fidelity of the biological structures
on all size scales and to minimize artefacts due to milling or sample charging.
Novel techniques for the study of ultrastructure and morphology in bio-
logical samples include FIB tomography with subsequent three-dimensional
reconstruction as well as the proposal of nano-biomachining [5,8,11]. The
incorporation of a cooled sample chamber holds the promise for minimizing
sample damage while helping to retain integrity of biological structures
without artefacts introduced by chemical fixation. This chapter covers the
topics important to successful FIB milling and imaging of biological samples.
13.2 Sample preparation
Existing procedures for sample preparation have been developed and opti-
mized for effective TEM and SEM imaging. These techniques exist to
strengthen biological structures from bulk tissue down to subcellular
nanostructures. Only with extensive processing will these samples be able to
retain native configurations with minimal loss of structural components. The
general procedure for sample preparation consists of the following steps:
chemical fixation, dehydration, embedment, and staining or coating. Other
alternatives to chemical fixation are cryotechniques in which structural pre-
servation is accomplished through the ‘‘freezing’’ of cellular components.
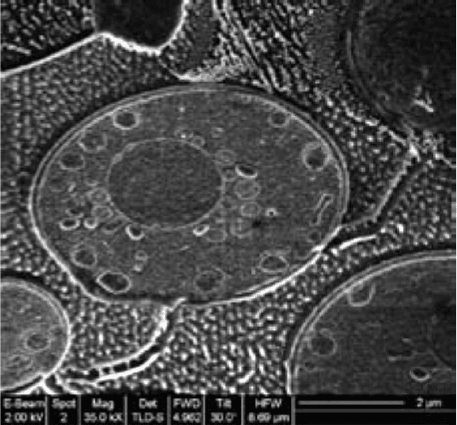
Recent developments have shown that cryo-dual stage SEM/FIB systems are
adequate for imaging unprepared biological samples (Figure 13.1)[31,32].
However, chemical methods are more widely employed as samples prepared
through cryotechniques require the availability of a cold stage, and ‘‘frozen’’
cells are thermodynamically unstable at low temperatures. FIB imaging and
sectioning of biological samples is a relatively unexplored field and use of
varying degrees of sample preservation have been employed in existing
literature [1–10,22]. Although there exist some instances in which no pre-
paration was required, chemical fixation and cryotechniques remain popular.
Focused ion beam systems338
For an in-depth discussion of sample preparation the reader is referred to
Introduction to Biological Electron Microscopy: Theory and Techniques by
Clinton Dawes [12].
13.2.1 Chemical methods for sample preparation
Chemical fixation
By beginning with chemical fixation, cellular structure is preserved in a near
in vivo state for the subsequent processing. The success of these procedures
has been supported by comparison with samples preserved by freezing
techniques. Chemicals used for fixation function by forming inter- and intra-
molecular cross linkages in proteins to form a gel, preserving cellular support
structures and the membrane system of various organelles with little struc-
tural change. The quality of a fixation is indicated by the appearance of lipid
membranes making up the endoplasmic reticulum and golgi systems. The
choice of fixing agent is determined by what cellular constituents are to be
preserved with the highest integrity. Common chemicals for fixation include
osmium tetroxide, aldehydes, and permanganates. Osmium tetroxide reacts
with nearly all cellular components, making it the most popular choice for
chemical fixation. The considerations important to successful staining with
osmium tetroxide are discussed next.
Figure 13.1 Cryo-FIB milled yeast sample originally published in an
application note from FEI company [31].
Applications for biological materials 339
Osmium tetroxide (OsO
4
) is a nonpolar compound, allowing unhindered
diffusion through cellular membranes. OsO
4
also has the ability to penetrate
hydrophilic lipids as well as aqueous areas of the cell. Osmium has five stable
oxidation states, allowing for reactions with most cellular constituents [12].
However, OsO
4
reacts most strongly with carbon–carbon double bonds. As a
result, osmium tetroxide reacts most strongly with unsaturated fats and
phospholipids. OsO
4
also partially fixes proteins by reaction with phenols
and SH groups. This also allows some fixation of nucleic acids by reacting
with histone proteins which are involved in DNA packaging. However, OsO
4
is a strong oxidizing agent and may result in the complete oxidation of
double bonds to form diols if concentration and fixation duration are not
closely monitored [13].
During this procedure, temperature, pH, and osmolarity of the fixing
solution are of utmost importance. Buffer solutions are required to maintain
a constant pH. As OsO
4
infiltrates the cell, the pH drops drastically,
resulting in the denaturation of proteins, and the degradation of protein
structures such as microtubules, microfilaments, and intermediate filaments.
For animal tissue, the buffer should maintain a pH of 7, while for plant
matter a slightly acidic environment of pH 5 to 6 is required. The most
common buffers are cacodylate and phosphate due to low reactivity with
fixing agents. The fixation solution should also be nearly isotonic with the
cellular environment to prevent shrinking and swelling. Also, a hypotonic
solution will result in ion leaching, which could destabilize the membrane
structures of organelles [12].
As the rate of fixation is directly related to diffusion, temperature begins to
play a large role in fixation duration and quality. Room temperature allows
for shorter durations, which is less disruptive to the cell; however, leaching of
cellular components is highly likely and autolysis of cell membranes will also
occur. Temperatures as low as 4
C prevent these deleterious effects, but require
a longer fixation time. If the fixation duration with OsO
4
is too long, however,
cellular components may be oxidized too far, resulting in the degradation of
cellular structures [12]. Due to the difficulties associated with OsO
4
fixation,
aldehydes are often employed for primary fixation of proteins, followed by
secondary fixation with OsO
4
to provide electron density and stabilize lipid
membranes. By the end of such fixation procedures, electron micrographs will
show a granular cytoplasm due to stabilization of cytoplasmic lipo-proteins
and dark staining of unsaturated fatty acids. Cell walls will appear darkened
along the edges due to low penetration into the dense cell wall. Loss of up
to 70% of carbohydrates, 21–50% of lipids, and 12–50% of proteins is com-
mon, but these numbers can be reduced by lower temperatures [12].
Focused ion beam systems340
Post-fixation procedures
For preparation for vacuum, water must be removed to avoid cellular
changes upon water evaporation from biological samples. When embedding
the sample, dehydration is also required as most embedding media are not
miscible with water. Polymerization of plastics will also be hindered by the
presence of water. During dehydration, water is slowly replaced by another
solvent, usually ethanol or acetone, by slowly increasing the concentration
of dehydrant in the infiltrating solution. Acetone is the most popular choice
due to miscibility with many plastics, and a lack of reaction with osmium
tetroxide. It also will not cause removal of phospholipids. Ethanol is
used more sparingly due to cell shrinkage. Special dehydrating agents such as
2,2-dimethoxypropane or 2,2-diethoxypropane can be used to remove water
by reaction to produce methanol and acetone or ethanol and acetone [12].
Nonpolar dehydrants can give improved results due to increased retention of
lipids and proteins. However, cell compression and membrane expansion will
occur during freeze-drying or freeze-substitution.
Although embedding the sample in a plastic or resin is most essential when
sectioning in preparation for TEM imaging, sample embedding is also
important for the imaging of animal cells without cell walls. Embedment
requires the use of a low viscosity plastic to infiltrate the sample. Such plastics
can not have volume change upon polymerization, and have low electron
scattering effects. Most importantly, the plastic must be stable under ion
beam bombardment. To provide the most uniform sectioning, the polymer
embedment must be similar to the sample in both density and chemical
composition as both parameters affect etching rate. Common embedments
include vinyl plastics, methacrylates, epoxy resins, and polyester resins [14].
Drobne et al. have used paraplast for the embedding of a crustacean digestive
system [8]. Paraffin is another popular choice, although maintaining hot
paraffin during preparation is difficult. If the sample is to be imaged in a
dual-beam system, stability under electron beam bombardment must also be
considered. Epoxies are most stable, while methacrylates are least stable [14].
Staining or coating is required for biological samples in which a large content
of carbon, oxygen, and nitrogen prevent high contrast due to low electron
density. As a result, coating with heavy metals or post-fixation heavy metal
staining is required. Staining refers to either chemical or physical incorporation
of heavy atoms into the sample. Common stains include metals such as tung-
sten, ruthenium, osmium, and lead. These metal ions form coordinate com-
plexes with active groups within the sample, adhering to certain structures to
increase contrast. For surface morphology, thin films of gold or palladium are
Applications for biological materials 341
deposited on sample surfaces by sputter coating or high vacuum evaporation.
Thin film coatings have the advantage of being an in-situ process that can be
performed within the FIB as new sections are generated.
13.2.2 Cryotechniques for sample preparation
Crytotechniques can be used with or without prior chemical fixation and can
be performed on living or fixed samples. These techniques are often com-
bined to prepare samples for vacuum by solvent removal. For bulk samples
such as arthropods with strong exoskeletons or even yeast cells with thick cell
walls, sample embedment is not required; however, removal of liquid within
the sample is important [3,9,10]. During ordinary drying, surface tension
causes specimen distortion as the liquid volume within the sample decreases
[14]. Cryotechniques minimize these effects through increased evaporation or
elimination of surface tension effects.
The favored techniques for this task are freeze-drying and critical point
drying. Freeze-drying employs vacuum induced sublimation of the solvent to
accomplish solvent removal. The largest drawback of freeze-drying is sample
shrinkage during freezing. As a result, critical point drying is the more
popular choice for solvent removal. During critical point drying, the sample
is heated until the solvent reaches its critical point, allowing the solvent to be
bled off without surface tension effects or other interactions that could
destabilize cellular structures. The critical points of liquid solvents occur at
temperatures and pressures too high for practical application, therefore,
carbon dioxide or Freon are the preferred solvents. Nevertheless, surface
tension damage is not completely avoidable, and a minimal amount of
shrinkage is still observed.
Yonehara et al.[1] have imaged the small intestine of Wister mice which
had been prepared using critical point drying. These samples were first ion
beam etched to reveal the small intestine where microvilli were imaged by
SEM to obtain high-resolution images after coating with 10 nm of heavy
metal [1] (Figures 13.2 and 13.3).
13.2.3 FIB imaging without sample preparation
Ballerini et al.[6] have directly imaged and sectioned yeast cells without prior
sample preparation (Figure 13.4). Yeast cells on filter paper were directly
transferred into the sample chamber. The semipermeable membrane allows
for the evaporation of water from within the cell, while the thick cellular wall
provided the structure to prevent sample shrinkage and collapse. Despite a
Focused ion beam systems342

Figure 13.2 Apical view of microvilli of mouse small intestine taken with
SEM at 3 kV after ion beam etching to remove surface mucosa [1].
Figure 13.3 Side view of microvilli of mouse small intestine taken with SEM
at 3 kV after ion beam etching to remove surface mucosa [1].
Applications for biological materials 343

lack of chemical fixation, ultracellular support structures were clearly visible.
They have suggested that imaging of cells such as lymphocytes or chon-
drocytes will also be possible without much sample preparation (Figure 13.5).
Yeast cells may be a special case, however, as they are known to survive
extreme conditions including complete dehydration.
13.2.4 Sample mounting
Due to sample embedding, the capsule must be trimmed to reveal the
specimen before imaging. Special considerations must be taken to avoid
build up of charge on the sample during ion beam or electron beam
bombardment depending on the system. Copper tape connecting the sample
to the sample holder is an option that is simple and easy to apply. Con-
ductive paints such as silver or carbon have also proven successful in
maintaining conduction between nonconducting polymer samples and the
sample holder [15]. Complete coatings of iridium, platinum, or gold have
been successful for SEM imaging; however, due to ion beam bombardment
in the FIB, these methods are less successful as they will be removed. In-
situ coatings are possible using ion assisted deposition for small cross
Figure 13.4 FIB microsection of yeast cells taken by Ballerini et al. with no
sample preparation [6].
Focused ion beam systems344
sections, which can increase the effectiveness of dual-beam systems in which
SEM imaging is possible.
If serial ultra-thin sectioning is to be performed in a dual-beam system, the
angle of the sample face with regard to the ion and electron beams is
extremely important. The ion beam must have a grazing angle of incidence to
decrease damage to the sample such as amorphous surface layers and ion
incorporation into the sample. The configuration of the ion and electron
beams must be considered when serial cross sections are examined. Dual-
beam machines currently on the market have an FIB at between 48 and 52
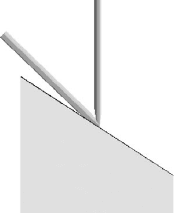
from the normal electron beam. Due to the limited tilt of the stage, to obtain
a grazing incidence angle of the ion beam, the sample face must be tilted as
illustrated in Figure 13.6.
13.3 Ion–sample interactions
Understanding the reactions that occur when energetic ions interact with
sample surfaces is essential to preparing high-quality images and uniform cross
sections. As with traditional FIB applications within the field of semi-
conductors and metals, interactions ranging from ion implantation to sample
sputtering and secondary electron emission involve the transfer of energy from
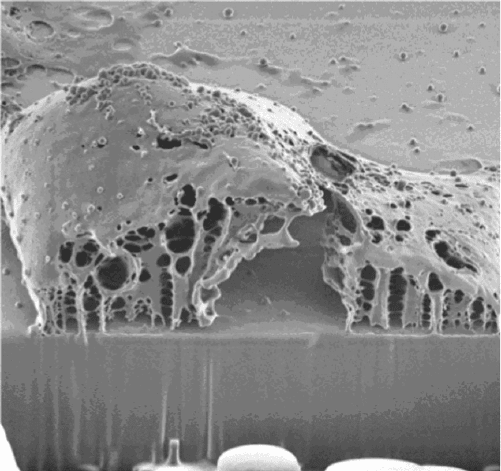
Figure 13.5 Chondrocyte imaged after FIB sectioning by Milani et al.[7].
Applications for biological materials 345
the incident ion to the solid surface. Knowledge of these interactions, specifi-
cally molecular sputtering theory, enhances the ability to utilize the focused ion
beam as an effective milling device. Unfortunately the FIB’s imaging cap-
abilities are compromised by artefacts resulting from ion bombardment. Due
to the nature of organic materials, artefacts specific to imaging of biological
samples become a matter of importance. Careful study of reactions resulting
from an incident beam of ions provides a foundation for the analysis and
minimization of artefacts generated during FIB milling and sectioning.
With both hard (semiconductors, ceramics, metals, etc.) and soft (organic)
materials transfer of momentum from incident ions to the sample surface is
the result of elastic collisions and inelastic interactions. The former leads to
physical sputtering of surface molecules if the kinetic energy of the
impinging ion beam is greater than the bonding energy of those molecules.
Complex inelastic interactions with soft materials give similar results to
those noted in hard materials such as the generation of secondary electrons
and phonons. All ion interactions with the sample result in a decrease in
kinetic energy of the impinging ions. If the ions are not backscattered, they
will remain implanted within the material. Artefacts such as defects, sample
heating, and amorphization typical of hard materials may have even more
complex effects in organic materials. Denaturation of proteins, instability of
lipid proteins and preferential sputtering may result. Detailed analysis of
ion–solid interactions in traditional hard materials are provided in the work
by Nastasi et al.[16].
13.3.1 Molecular sputtering theory
Physical ejection of atoms from the surface of a target depends on the kinetic
energy of the incident electron beam and the energy with which atoms are
Electron beam
Ion beam
Embedment
Figure 13.6 Proposed ample embedment angle to achieve grazing surface
angle to mini mize damage during cross-sectioning with eucentric stage.
Focused ion beam systems346

bound to the surface (sublimation energy). The energy transfer depends on
the size, mass, and charge of the ions and atoms. The power potential law
proposed by Lindhard et al.[17] suggests that the power potential between
ions and atoms varies as
VðrÞ¼
Z
1
Z
2
e
2
a
n1
nr
n
; ð13:1Þ
where Z
1
and Z
2
are the atomic numbers of the incident ion and target atom,
respectively, r is the distance between the two interacting atoms, and e is the
charge of an electron. n depends on the type of collision: n =1 for Rutherford
type, n ¼2 for weakly screened collisions, and n ¼5 for collisions of hard
spheres. a is the effective screen radius of the atoms as given by
a ¼ 0:8853 · a
H
· ðZ
1
2=3
þ Z
2
2=3
Þ
1=2
; ð13:2Þ
where a
H
is the Bohr radius for the hydrogen atom.
Kanaya et al. (1988) enhance this theory to develop atomic sputtering
theory by accounting for the weak-screening effects and hard sphere colli-
sions [2]. Molecular sputtering theory as proposed by Kanaya et al. (1992)
builds upon atomic sputtering theory to account for bonding energies of
molecules [18]. For samples of a pure solid, the sputtering yield is dependent
on the nuclear stopping cross section, the number of atoms per unit volume,
and the number of primary knock-on atoms per incident particle, as well as
the number of atoms that recoil as a result of collision cascade effects.
The proposed semi-empirical relationship for the sputtering yield (ejected
atoms per incident ion) as a function of the energy of the incident beam (E)
derived by Kanaya et al.[2,18] takes the form
Y
A
¼ Y
m
E
E
m
1=21=n
: ð13:3Þ
The maximum theoretical yield Y
m
is given by
Y
m
¼
0:45 · Z
1
Z
2
ð1 þ M
1
=M
2
Þ
M
2
E
s
ðZ
1
2=3
þ Z
2
2=3
Þ
1=2
; ð13:4Þ
and E
m
is given by
E
m
¼
0:4Z
1
Z
2
e
2
ðM
1
þ M
2
Þ
aM
2
; ð13:5Þ
where the coefficients 0.45 and 0.4 have been determined experimentally, M
1
and M
2
are the atomic masses of the incident ion and target atom, respectively,
Applications for biological materials 347
