Ambaum M., Thermal Physics of the Atmosphere
Подождите немного. Документ загружается.


146 CH 7 CLOUD DROPS
where we have introduced a reduced vapour density
r
as
1
r
=
1
vs
+
1
K
. (7.76)
This density difference then leads to the total mass flux,
I F
m
= 4Dr
d
r
(RH − 1). (7.77)
As can be seen, Eq. 7.77 is the same as Eq. 7.69 with the far-field saturated
vapour density replaced by the reduced vapour density
r
.
It now becomes clear that latent heating slows the growth of the of the
drop because for all
K
we have
r
<
vs
. (7.78)
Physically this makes sense: a warmer drop reduces the supersaturation with
respect to the far field and thus reduces the vapour diffusion towards the
drop. There are two limiting cases of interest. If the heat conduction coeffi-
cient K is very large, then any excess heat is immediately diffused away from
the drop. We thus expect the heating effect to be small. Indeed, for K →∞
we have
K
→∞and we find
r
→
vs
. (7.79)
So the mass flux in Eq. 7.77 reduces to Eq. 7.69. The other limiting case is
for a small heat conduction coefficient. In this case the latent heat cannot
be transported away from the drop and we expect a strong reduction of the
supersaturation. Indeed, for K → 0 we have
K
→ 0 and we find
r
→
K
→ 0, (7.80)
so the mass flux to the drop vanishes. Realistic values of the diffusion and
conduction coefficients give a value of
K
of about 7 × 10
−3
kg m
−3
and it
is typically similar or somewhat larger than
vs
. The heating effect therefore
reduces the growth by a factor of two to four compared to the case without
heating.
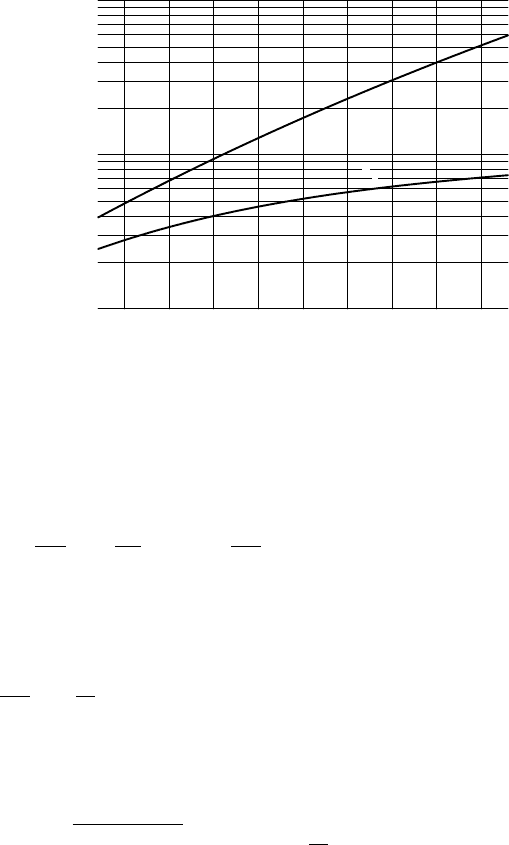
The reduction becomes larger for higher temperatures. In fact, the reduced
vapour density is a much weaker function of temperature than the saturated
vapour density, see Figure 7.9. As can be seen in this figure, the reduced
density is very nearly a linear function of the temperature. The approximation
r
= (2.80 + 0.11 T(
◦
C)) × 10
−3
kg m
−3
(7.81)
is accurate to within 4% between temperatures of –5
◦
C and 50
◦
C.
7.5 DROPLET GROWTH 147
1
10
100
0 10 20 30 40
T (ºC)
vapour density (gkg
−1
)
ρ
vs
ρ
r
FIGURE 7.9 Saturated vapour density
vs
and reduced vapour density
r
as a function of
temperature T.
Now we have an expression for the mass-flux, we can calculate the change
in droplet radius r
d
. We have
F
m
=
dM
dt
=
l
dV
dt
= 4
l
r
2
d
dr
d
dt
(7.82)
with
l
the density of the liquid and V the droplet volume, V = (4/3)r
3
d
. Sub-
stituting the expression for the mass flux, we find
r
d
dr
d
dt
= D
r
l
(RH − 1). (7.83)
The right-hand side of Eq. 7.83 is a constant at given temperature and relative
humidity, so we can integrate the equation to find:
I r
d
(t) =
r
2
d
(0) + 2D t with D = D
r
l
(RH − 1). (7.84)
The droplet radius grows as the square root of time. When the relative hu-
midity is smaller than 100%, this equation describes how the droplet radius
reduces in time through evaporation.
Square root change of scale with time is typical of phenomena that are
driven by diffusive processes. For example, the typical size of a cloud of
tracer released by a local source grows with the square root of time and heat
penetrates a conductor to a depth that grows with the square root of time.
An interesting property of Eq. 7.84 is that the radii of two droplets that
start growing from different initial radii become more and more similar. From
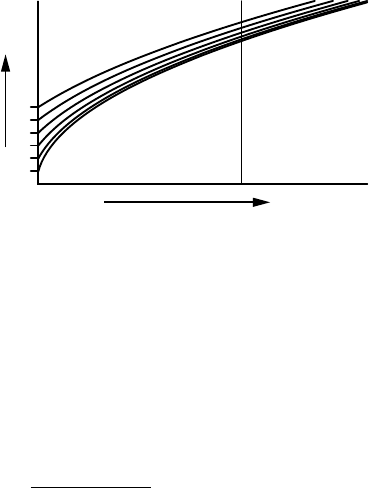
148 CH 7 CLOUD DROPS
t
r
d
FIGURE 7.10 Growth of droplet radius through vapour diffusion. The distribution of
droplet radii becomes ever narrower.
Eq. 7.84 we have
r
2
a
(t) − r
2
b
(t) = r
2
a
(0) − r
2
b
(0), (7.85)
with r
a
and r
b
the radii of two different droplets growing in the same back-
ground. This is equivalent to
r
a
(t) − r
b
(t) =
r
2
a
(0) − r
2
b
(0)
r
a
(t) + r
b
(t)
. (7.86)
Because both r
a
and r
b
will grow with time, their radii will get closer with
time. This effect is illustrated in Figure 7.10.
Note that with changing droplet radius, the mass flux F
m
is not constant
in time. We then need to ask ourselves whether the steady state assumption
leading to Eq. 7.61 is accurate. Any density fluctuations in the vapour field
around the drop will adjust to a steady state through a diffusion process with
diffusion coefficient D. The boundary condition change through the changing
droplet radius occurs with an effective diffusion coefficient D . Because
D D, (7.87)
the vapour field adjusts to a steady state on a much faster time scale than that
of the changing boundary (the relevant time scales are r
2
d
/D and r
2
d
/D). We
can therefore safely assume that the vapour flux is constant for any given
droplet radius during the growth process.
Let us now put some numbers in Eq. 7.84. A typical supersaturation at
activation is about 2% or less. At 10
◦
C, the reduced vapour density
r
is
about 4 × 10
−3
kg m
−3
. The time taken to grow from a small droplet to a
typical rain drop of radius about 1 mm is several days. Even for growth to
modest droplet size of several tens of micrometres we find times of several
hours. The typical time scale for rain to form in a convective cloud is very
much shorter, perhaps half an hour or less. Diffusive growth cannot be the
complete story to go from activation to raindrop.
The next stage in droplet growth is described by the process of collision and
coalescence. In its simplest form, drops at different radii will fall at different
7.5 DROPLET GROWTH 149
terminal velocities. This then allows the drops to collide and possibly coa-
lesce. This is a more efficient mechanism for droplet growth than diffusional
growth.
The terminal velocity of a drop increases with increasing size. Now consider
the following idealized case: A relatively large drop of radius r
d
is falling
through an environment with a number of smaller drops with a relative speed
of V . The large drop would sweep out a volume of V r
2
d
per unit time. The
smaller drops represent a total liquid water density,
b
. We assume that a
fraction E of the small drops in the volume swept out by the large drop would
collide and coalesce with the large drop. The mass increase of the large drop
would then be
dM
dt
= E V r
2
d
b
. (7.88)
The effective volume swept out by the drop is somewhat larger than this
because its effective radius has to be increased with the radius of the small
droplets. The fraction E is called the collection efficiency and it is a strong
function of the radius of the large drop and the radius of the smaller drops. The
collection efficiency is typically a few per cent if the small droplets are much
smaller than the larger droplets. This is because the air flow around the large
drop drags most small droplets around the large drop and thus prevents them
from colliding with the large drop. For small droplets larger than about 20 m
the collection efficiency is typically 90% or higher.
As in Eq. 7.82, this mass increase is related to a radius increase and we can
rewrite the above equation as
dr
d
dt
= E
V
b
4
l
. (7.89)
This is not a closed equation because the terminal velocity V is a function of
the radius of the drop. For small drops (typical cloud drops with radii smaller
than about 30 m), the terminal velocity is low and the Reynolds number of
the flow around the drop is small. This is the viscous Stokes regime, where
the terminal velocity is proportional to the square of the radius. For large
drops (larger than about a millimetre), the terminal velocity is large and the
Reynolds number of the flow is large. This is the turbulent regime where the
terminal velocity is proportional to the square root of the droplet radius.
43
In the intermediate regime, an empirical match is found for the terminal
43
In the Stokes regime, the terminal velocity is a function of the effective droplet buoy-
ancy g
, the viscosity , and the droplet radius r
d
. The only combination of these variables
with the dimension of speed is (g
) r
2
d
, which is, up to a constant factor, the Stokes
formula. For high Reynolds numbers, the terminal velocity cannot be a function of the
viscosity. The only possible combination of buoyancy and droplet radius with the dimen-
sion of speed is
√
g
r
d
. The effective buoyancy is g
= g (
l
− )/. Note that the effective
droplet inertia in the denominator is given by the density of the displaced air, .

150 CH 7 CLOUD DROPS
velocity. In its simplest form, a terminal velocity proportional to the radius is
chosen. Approximate formulae for droplet terminal velocity are
V = 1.2 × 10
8
r
2
d
ms
−1
if r
d
<
∼
30 m, (7.90a)
V = 250
√
r
d
ms
−1
if r
d
>
∼
1mm, (7.90b)
V = 8 × 10
3
r
d
ms
−1
otherwise. (7.90c)
These fall speed equations can be combined with Eq. 7.89 to find the growth
of drops by collisions and coalescence.
This theory has many approximations. The collection efficiency is not a
constant but a complex function of the sizes of the two colliding drops as
well as the relative speed of the colliding drops and of the turbulence in the
surrounding air. Also, to achieve realistic droplet growths from this model,
we need to assume a whole spectrum of initial droplet sizes. The largest of
the initial droplets will then grow at the expense of the smaller drops because
this process corresponds to the highest collection efficiencies.
We have only discussed the formation and growth of water droplets. So
we have been mainly looking at processes that occur in warm clouds, that is,
clouds below the freezing level. However, many clouds are above the freezing
level and are therefore likely to be at least partly made up of ice. Cold cloud
physics has a more empirical character as the ice processes are so complex.
Ice crystal activation is similar to droplet activation. Homogeneous nucle-
ation does not usually occur; we need a substrate for the ice crystal to nucleate
on. The most obvious choice would be for water droplets to freeze. It turns
out that for small drops this happens when the water droplet is severely su-
percooled. All drops would be frozen typically at around T<−40
◦
C. Droplets
can also freeze at higher temperatures, especially if they contain a solid nu-
cleus. However, many clouds contain substantial amounts of supercooled
water. Analogous to heterogeneous nucleation of water drops, ice crystals
can form on suitable substrates. Wind-blown clay-dust is a typical cause of nu-
cleation. Ice nuclei become more efficient at lower temperatures. Indeed, the
effective ice nucleus concentration increases exponentially with decreasing
temperatures.
When ice crystals are activated they can grow through diffusional growth,
just like water drops. In fact we find an analogue of Eq. 7.63 for ice crystals,
F
m
/C = 4D ((∞) − (r
d
)). (7.91)
Here the radius of the droplet in Eq. 7.63 is replaced by a so-called capacitance
C, which takes into account the shape of the ice crystal.
44
The capacitance
of a sphere is r. The word ‘capacitance’ has its origin in electrostatics: the
calculation for diffusional flux around a crystal turns out to be analogous to
44
See Westbrook, C. D. et al. (2008) J. Atmos. Sci. 65, 206–219.

7.5 DROPLET GROWTH 151
the calculation of the electric field around a capacitor of a certain shape with
capacitance C.
It is important to note that the growth rate of the crystal is now propor-
tional to the supersaturation over ice rather than over water. The saturated
vapour pressure over ice is lower than that over water, which means that the
supersaturation with respect to ice is higher than the supersaturation with re-
spect to water. It may then happen that in clouds where ice crystals and water
droplets are present, the ice crystals grow faster than the water droplets, per-
haps up to the point where the water droplets become subsaturated because
the ice crystals will have scavenged the water vapour. At this point the ice
crystals will grow at the expense of the water drops. This process is called the
Bergeron–Findeisen process. Hole clouds are thought to be spectacular mani-
festations of the Bergeron–Findeisen process: an aeroplane flying through a
supercooled water cloud can initiate ice crystal growth at the expense of the
water drops. The cloud is left with a hole filled with ice crystals.
The droplet can grow further by the collision of ice crystals with super-
cooled drops, a process usually called accrection, or the clumping together
of different ice crystals, a process usually called aggregation. The collection
efficiencies for these processes are hard to determine as they depend strongly
on crystal shape and temperature.
45
PROBLEMS
7.1. For a drop which grows by condensation, calculate a typical temperature
difference between the drop and the far field.
7.2. Wilson cloud chamber. Take a cylinder with some water and its satu-
rated vapour. Now expand the cylinder rapidly. The adiabatic expansion
is so fast the vapour does not immediately adjust to its new equilib-
rium. Using the Clausius–Clapeyron equation in its approximate form,
show that the relative humidity after rapid expansion from volume V
0
to volume V
1
satisfies
RH =
V
0
V
1
c
pv
/c
vv
exp
L
R
v
T
0
V
1
V
0
R
v
/c
vv
− 1
,
For water vapour, the specific gas constant is R
v
= 461Jkg
−1
K
−1
and
the specific heat capacity at constant volume is c
vv
= 1410 J kg
−1
K
−1
.
Hence show that an expansion by a factor of 1.3 is sufficient to achieve
a relative humidity of 400%. How does this result change when the
cylinder is filled with air at standard pressure and temperature, some
water, and its saturated vapour?
45
For further reading on cloud physics, see Mason, B. J. (1971) The physics of clouds, 2nd
edn. Oxford University Press, Oxford; Rogers R. R. & Yau, M. K. (1989) A short course in
cloud physics, 3rd edn. Butterworth–Heinemann; Pruppacher, H. R. & Klett, J. D. (1997)
Microphysics of clouds and precipitation, 2nd edn. Kluwer, Dordrecht.

152 CH 7 CLOUD DROPS
7.3. Supersaturation in updrafts. How far do we need to lift an initially sat-
urated air parcel to achieve a supersaturation of 1%? What is a typical
updraft velocity needed to prevent a cloud-drop from falling from the
air?
7.4. Evaporation of raindrops. Assume that raindrops have a terminal ve-
locity of V = Cr
d
with r
d
the drop radius and C = 8 × 10
3
s
−1
. Show
that, if we ignore the temperature dependence in the reduced density
r
, the radius of a raindrop falling through an environment with relative
humidity RH changes with height z as
r
3
d
(z) = r
3
d
(z
0
) +
3D
r
C
l
(RH − 1) (z
0
− z),
with z
0
the initial height. For a cloud base of 1 km, an environmental
temperature of 280 K, and relative humidity RH = 70%, how large does
the initial raindrop need to be to reach the ground? Estimate the error
made by assuming a constant
r
. The effective diffusion coefficient for
a falling drop will be increased because of ventilation. How will this
change the answer?

8
Mixtures and solutions
Variations in the composition of substances have thermodynamic implica-
tions. This contrasts with our previous emphasis on so-called simple sub-
stances, substances where the composition does not vary during the processes
considered. Here we will mainly concentrate on how the presence of solutes
in water affects the properties of the water. Although of considerable interest
to atmospheric science, we will not consider the thermodynamic aspects of
chemical reactions.
8.1 CHEMICAL POTENTIALS
Consider a substance which is made up of N different composites with the
i-th composite having mass M
i
. So the total mass M is
M =
i
M
i
. (8.1)
The Gibbs function G of such a composite substance will be a function of the
pressure p, temperature T and the composition,
G = G(p, T, M
1
,M
2
,M
3
,...). (8.2)
The differential of G can be written as
dG =
∂G
∂p
T,M
i
dp +
∂G
∂T
p,M
i
dT +
i
∂G
∂M
i
p,T
dM
i
= V dp − S dT +
i
g
i
dM
i
, (8.3)
where the g
i
are defined by the partial differentiation of the Gibbs function
with respect to M
i
,
I g
i
=
∂G
∂M
i
T,p
. (8.4)
153
Thermal Physics of the Atmosphere Maarten H. P. Ambaum
© 2010 John Wiley & Sons, Ltd. ISBN: 978-0-470-74515-1
154 CH 8 MIXTURES AND SOLUTIONS
The g
i
are called chemical potentials; the chemical potentials are intensive
quantities that are functions of the pressure, temperature, and the composi-
tion of the substance.
Note that the chemical potentials are not functions of the absolute values
of the M
i
but only of the ratios M
i
/M
j
: any uniform scaling of the masses M
i
by some factor does not change the definition of the chemical potentials, as
the Gibbs function G itself also scales with this factor. Such a uniform scaling
of the masses can be written as
dM
i
= M
i
d˛, (8.5)
where d˛ = dM
i
/M
i
is the same for all composites. So for constant composi-
tion we find that at fixed temperature and pressure
dG =
i
g
i
M
i
d˛. (8.6)
Because the g
i
do not change in this process, this equation can be integrated
to find
G =
i
M
i
g
i
. (8.7)
It now becomes clear that the chemical potentials g
i
are the multi-component
analogues of the specific Gibbs function for simple substances. The chemical
potentials g
i
are not the same as the specific Gibbs functions for the pure com-
ponents, as the chemical potentials in general will depend on the composition
of the substance.
The analogy between chemical potentials and specific Gibbs functions also
applies to the equilibrium between different phases. As shown in Chapter 5
this equilibrium follows from dG = 0 at constant pressure and temperature.
For simple substances this leads to the result that the specific Gibbs functions
of the substance in the two phases have to be the same, see Eq. 5.4. This can
be generalized to multi-component substances.
The phase equilibrium at constant pressure and temperature is still defined
by dG = 0, where G is the Gibbs function for the total system containing the
two phases. We can now consider the variation in the Gibbs function due
to the transfer of component i from one phase to another. For the sake of
argument, consider a system made up of a liquid and its vapour, and the
evaporation of a mass ıM
i
of component i from the liquid to the vapour. At
constant pressure and temperature the change in Gibbs function ıG is then
given by
ıG =
i
(g
i,v
− g
i,l
) ıM
i
. (8.8)
As ıG has to vanish for any variation in the system, we conclude that for all
components the chemical potentials have to be the same between the two

8.2 IDEAL GAS MIXTURES AND IDEAL SOLUTIONS 155
phases in equilibrium,
I g
i,v
= g
i,l
. (8.9)
This is the multi-component generalization of Eq. 5.4. Note that on adding a
component we also add another equilibrium relation for the chemical poten-
tial so the equilibrium state remains well defined.
The reliance on the use of the Gibbs function G to define chemical potentials
and phase equilibria is only apparent; the choice is one of convenience, not
necessity. The situation is analogous to that in the derivation of the Clausius–
Clapeyron equation in Section 5.1. For example, consider the enthalpy H of
a substance made up of several components. Because H = G +TS we can use
Eq. 8.3 to write for the differential of the enthalpy
dH = dG + T dS + S dT = V dp + T dS +
i
g
i
dM
i
. (8.10)
So we can also define the chemical potentials g
i
as partial derivatives of the
enthalpy g
i
= (∂H/∂M
i
)
S,p
. Analogous arguments work for the other thermo-
dynamic potentials and they lead to four equivalent definitions of the chem-
ical potentials:
g
i
=
∂U
∂M
i
S,V
=
∂H
∂M
i
S,p
=
∂F
∂M
i
T,V
=
∂G
∂M
i
T,p
. (8.11)
In contrast, the arguments leading to Eq. 8.7 cannot be straightforwardly
translated to the other thermodynamic potentials: only the Gibbs function
has two intensive variables as natural variables (T and p); the other potentials
have at least one extensive natural variable, which would change on a uniform
scaling of the masses of all constituents. So although we can use any of the
four standard thermodynamic potentials to define the chemical potential, the
chemical potential is the multi-component generalization of the specific Gibbs
function.
8.2 IDEAL GAS MIXTURES AND IDEAL SOLUTIONS
For ideal gas mixtures we can write down explicit expressions for the chem-
ical potentials. In order to calculate the chemical potentials in an ideal gas
mixture we will first calculate its total Gibbs function. We will take it that for
an ideal gas mixture the individual components contribute independently to
the extensive variables as each component, by definition of the ideal gas, is
not influenced by the presence of the other components.
For an ideal gas mixture the total internal energy U is given by
U =
i
M
i
u
i
. (8.12)
