Brewster H.D. Solid State Physics
Подождите немного. Документ загружается.


262
Then,
is replaced by
where
~
Ie
-lq.RO(q)
V R
~
"
/,
e-
iq
.
R
vL..
q
R
Broken Translational Invariance
/q
=
IUb(x)el-iq.x
b
As a result
of
the structure factor,
fq'
the scattering amplitude need not
have a peak at every reciprocallattic vector,
ij.
Of
course, the probability that the detector is set up at precisely the right
angle to receive
k
f
= k
i
+
{;
is very low. Hence, these experiments are usually
done with a
powder
so
that
there will be Bragg scattering
whenever
2k sin % =
IGI
. By varying
e,
a series
of
peaks are seen at, e.g.
1t16,
1t/4,
etc.,
from which the reciprocal lattice
vectors are reconstructed.
Since
Ikl-IGI-(IA r
1
,
the energy
of
the incoming photons is
-nck-
I04eV which is definitely in the X-ray range.
Thus far, we have not looked closely at the factor:
I(
mle-iq.ii(O)1
nt
This factor results from the vibration
of
the lattice due to phonons. In
elastic scattering, the amplitude
of
the peak will be reduced by this factor
since the probability
of
the ions forming a perfect lattice is less than I. The
inelastic amplitude will contain contributions from processes in which the
incoming photon or neutron creates a phonon, thereby losing some energy.
By measuring inelastic neutron scattering (for which the energy resolution
is
better than for X -rays), we can learn a great deal about the phonon spectrum.

Chapter 12
Electronic
Bands
Thus far, we have ignored the dynamics
of
the elctrons and focussed on
the ionic vibrations. However, the electrons are important for many properties
of
solids. In metals, the specific heat is actually C v = yT +
aT3.
The linear
term is due to the electrons. Electrical conduction is almost always due to the
electrons, so we will need to understand the dynamics
of
electrons in solids in
order to compute, for instance, the conductivity
cr(T
,co).
In
order to do this, we will need to understand the quantum mechanics
of
electrons
in
the periodic potential due to the ions. Such an analysis will
enable us to understand some broad features
of
the electronic properties
of
crystalline solids, such as the distinction between metals and insulators.
BLOCH'S
THEOREM
Let us first neglect all interactions between the electrons and focus on
the interactions between each electron and the ions. This may seem crazy since
the inter-electron interaction isn't small, but let us make this approximation
and proceed. At some level, we can say that we will include the electronic
contribution to the potential in some average sense so that the electrons move
in the potential created by the ions and by the average electron density
(of
course, we should actually do this self consistently). Later, we will see why
this
is
sensible.
When the electrons do not interact with each other, the many-electron
wavefunction can be constructed as a Slater determinant
of
single-electron
wavefunctions.
Hence, we have reduced the problem to that
of
a single electron moving
in
a lattice
of
ions. The Hamiltonian for such a problem
is:
li
2
H =
__
V
2
+
IV(x
-i?-u(i?))
2m R
expanding
in
powers
of
u
(i?)
,
2
H
=_~V2
+
IV(x
-i?)-IVV(x-i?).u(i?)+
...
2m R R

264
Electronic Bands
The third term and the ... are electron-phonon interaction terms. They
can
be
treated
as
a
perturoation.
W e w
ill
fOcus
on
the1t
first two terms, which
describe an electron moving
in
a periodic potential. This highly simplified
problem already contains much
of
the qualitative physics
of
a solid.
Let us begin by proving
an
important theorem due to Bloch.
Bloch's
Theorem:
If
V
(F
+
R)
= V
(F)
for all lattice vectors R
of
some given lattice, then for any solution
of
the Schrodinger equation in this
potential,
2
-~
V
2
\!f(F) + V(F)\!f(F) = E\!f(F)
2m
there exists a k such that
\!f(F
+ R) =
i~oR\!f(F)
Proof
Consider the lattice translation operator
TR
which acts according
to
Then
TRHX
(F) =
HTRX(F
+ R)
i.e.
[TR'
H]
=
O.
Hence, we can take our energy eigenstates to be eigenstates
of
T
R
·
Hence, for any energy eigenstate
\!fCF)
,
TRIjf(F)
= c(R)\!f(F)
The additivity
of
the translation group implies that,
e(R)c(R) = e(R +
R')
Hence, there
is
some k such that
c(R)=/foR
Since e
100R
= 1
if
G
is
a reciprocal lattice vector, we can always take
~k
to be
in
the first Brillouin zone.
TIGHT-BINDING
MODELS
Let's
consider a very simple model
of
a ID solid in which we imagine
that the atomic nuclei lie along a chain
of
spacing
a.
Consider a single ion and
focus on two
of
its electronic energy levels.
In
real systems, we will probably
consider
sand
d orbitals, but this
is
not important here;
in
our toy model,
these are simply two electronic states which are localized about the atomic
nucleus.
We'll call them
11)
and
12),
with energies
E?
and E
g.
Let's further imagine
that the splitting
E g -
E?
between these levels
is
large. Now, when we put

Electronic Bands
265
this atom in the linear chain, there will be some overlap between these levels
and the corresponding energy levels on neighboring atoms. We can model
such a system by the Hamiltonian:
H=
L(E~
IR,I)
(R,Jl+E6JR,2)(R,21)-
~
(tIIR,l)
(R',ll
+t
2
I
R
,2)(R',21)
R R,R
n.n
We have assumed that
tl
is the amplitude for an electron at
IR,
1)
to hop
to
IR',
I), and similarly
for~.
For simplicity, we have ignored the possibility
of
hopping from
IR,
I) to
IR',
2), which
is
unimportant anyway when
EO
is
large. The eigenstates
of
this Hamiltonian are:
Ik,l) = IeikRIR,l)
R
with energy
and
Ik,2)
=
Ie
,kR
IR,2)
R
with energy
E 2 (k) = E g -
2t2
cos ka
Note, first, that
k lives
in
the first Brillouin zone since
Ik,j)
==
Ik
+
2:n
,j)
Now, observe that the two atomic energy levels have broadened into two
energy bands. There is a band gap between these bands
<?f
magnitude E
~
- E
~
-
21
1
21
2
,
This is a characteristic feature
of
electronic states in a periodic
potential: the states break up into bands with energy gaps separating the bands.
How many states are there in each band? As we discussed in the context
of
phonons, there are as many allowed
k's
in
the Brillouin zone as there are
ions in the crystal.
Let's
repeat the argument. The Brillouin zone has k-space
extent
2rc/a.
In a finite-size system
of
length L with periodic boundary
conditions, allowed
k's
are
of
the form
2rcn/L
where n is an integer. Hence,
there are
Lla = N
ions
allowed
k's
in the Brillouin zone. (This argument
generalizes to arbitrary lattices
in
arbitrary dimension.)
Hence, there are as many states as lattice sites. Each state can
be
filled by
one up-spin electon and one down-spin electron.
Hence,
if
the atom
is
monovalent - i.e.
if
there
is
one electron per
site-
then,
in
the ground state, the lower band,
Ik,
I)
is
half-filled and the upper
band is empty. The Fermi energy
is
at
E~.
The Fermi momentum (or, more
properly, Fermi crystal momentum)
is
at
±rrJ2a.
At low temperature, the fact

266
Electronic Bands
that there is a gap far away from the Fermi momentum is unimportant, and the
Fermi sea will behave just like the Fermi sea
of
a free Fermi gas. In particular,
there is
no
energy gap
in
the many-electron spectrum since we can always
excite an electron from a filled state
just
below the Fermi surface to one
of
the unfilled states
just
above the gap.
For instance, the electronic contribution to the specific heat will be
C
v
-
T.
The difference is that the density
of
states will be different from that
of
a
free Fermi gas. In situations such as this, when a band is partially filled, the
crystal
is
(almost always) a metal.
If
there are two electrons per lattice site, then the lower band
is<J>illed
and
the upper band is empty in the ground state. In such a case, there
is
an energy
gap
E g = E g -E
~
-
2t
1 -
2t2
between the ground state and the lowest excited
state which necessarily involves exciting an electron from the lower band to
the upper band. Crystals
ofthis
type, which have no
partially<J>illed
bands, are
insulators. The electronic contribution to the specific heat will be suppressed
by a factor
of
e-
Egf[.
Note that the above tight-binding model can be generalized to arbitrary
dimension oflattice. For instance, a cubic lattice with one orbital per site has
tight-binding spectrum:
E (k) = - 2t(cos
kxa
+ cos k a + cos
k=a)
Again,
if
there is one electron per site, the band will be half-filled (and
metallic);
if
there are two electrons per site the band will be filled (and
insulating).
The model which we have
just
examined is grossly oversimplified but
can, never-the less, be justified, to an extent. Let us reconsider our lattice
of
atoms.
Electronic orbitals
of
an isolated atom:
with energies
En:
<Pn(F)
-~
V
2
<pn(r)
+ V(r)<Pn(F) =
En
<Pn(r)
2m
We now want to solve:
2
-~V2't)JnCr)+
LV(r
+R)'t)Jn(r)=E(k)'lPn(r)
2m R
Let's
try the ansatz:
tPkCr)
=
LCneif.R<pnCr
+
R)
R,n
which satisfies Bloch's theorem. Substituting into
Schrodinge~s
equation and
taking the matrix element with
<Pm'
we get:
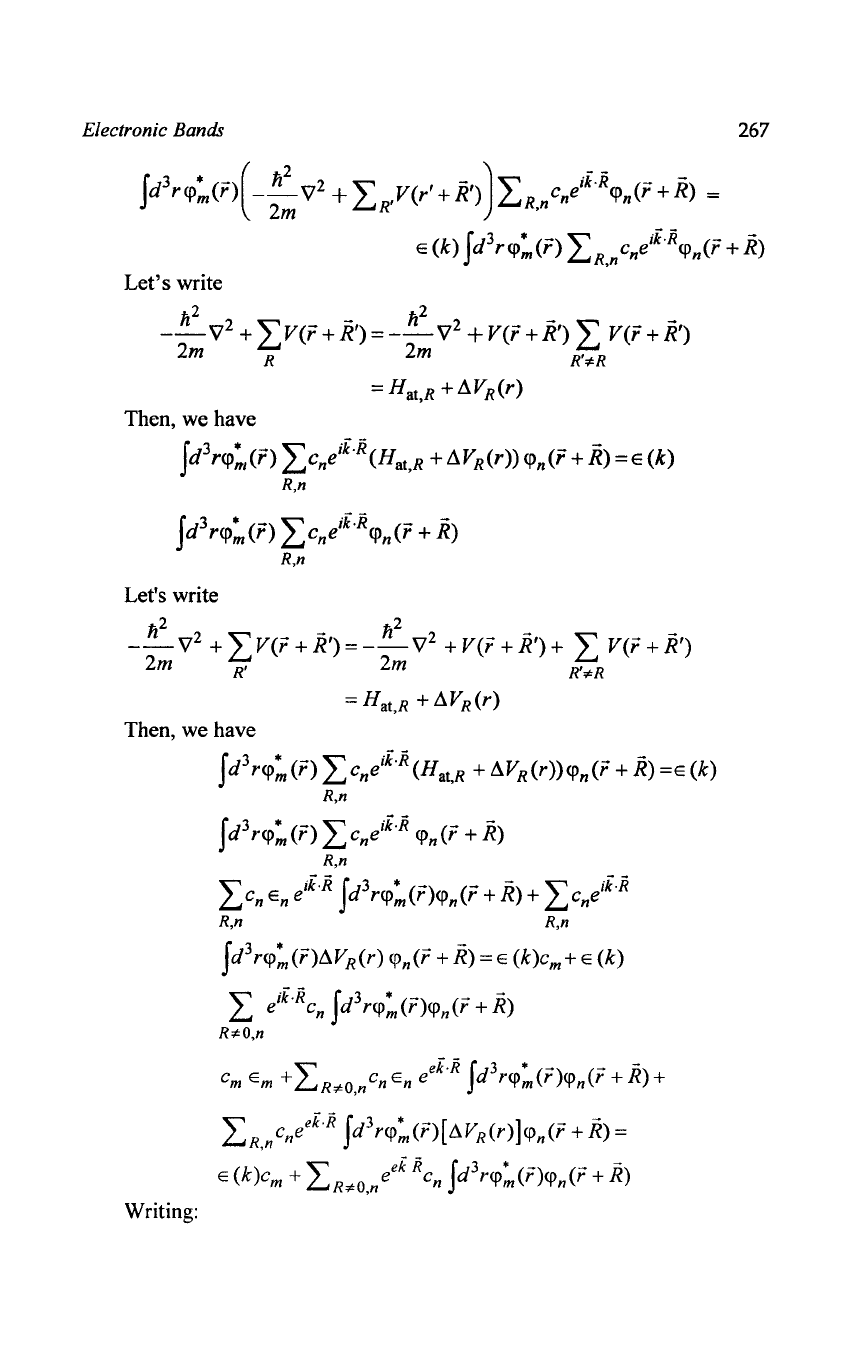
Electronic Bands
267
Jd
3
r<p:O:{
-
;:
\7
2
+
LRY(r'
+R'»)LR,nCneiioR<Pn(r
+R)
=
E (k)
Jd
3
r<p:(r) LR,nCneifoR<pn(r
+ R)
Let's
write
h
2
2"
- h
2
2
-"
-
--\7
+
L.JV(r+R')
=--\7
+
V(r+R')
L.J
V(r
+ R')
2m R 2m
R'*R
=
Hat,R
+ LlVR(r)
Then, we have
Jd
3
r<p:/(r) Lcneik-R(Hat,R
+ LlVR(r»<Pn(r
+R)=E(k)
R,n
Jd
3
r<p:(r) LCneifoR<pn(r
+
R)
R,n
Let's write
h
2
2"
- h
2
2
-"
-
--\7
+
L.J
VCr
+ R') =
--\7
+
VCr
+ R') +
L.J
VCr
+ R')
2m
R'
2m
R'*R
=
Hat,R
+ LlVR(r)
Then, we have
Writing:
Jd
3
r<p:(r) LCneifoR(Hat,R
+ LlVR(r»<Pn(r + R)
=E
(k)
R,n
Jd
3
r<p:(r) LCneifoR
<Pn(r
+ R)
R,n
LCn
En
e
ifoR
Jd
3
r<p:(r)<Pn(r
+
R)
+ Lcneik-R
R,n R,n
Jd
3
r<p:
(r)LlV
R
(r)
<Pn(r
+ R) = E (k)c
m
+ E (k)
L
eik-Rc
n
Jd
3
r<p:(r)<Pn(r
+ R)
R*O,n
LR,ncneei-R
Jd3r<p~(r)[LlVR(r)]<Pn(r
+R)=
E (k)c
m
+ L °
eek
RC
n
fd3r<p~(r)<Pn(r
+
R)
R*
,n

268 Electronic Bands
amn(R) = fd3r<p:(r)<pn(r +
R)
Y mn(R) = -
fd3r<p:
(r)[LlVR (r)]
<Pn(r
+
R)
We have:
Cm(Em
-E(k»+
I
Cn(En
-E(k»i~·Ramn(R)=
IC
n
i;·Rymn(R)
R~O,n
R,n
Both
arnn(
R ) and
mn(
R)
are exponentially small, _ e
-RI
Go
• In particular,
(lmn(
R)
and Y
mn
(
R)
are much larger for nearest neighbors than for any other
sites, so let's neglect the other matrix elements and write
(lmn
= (lmn(Rn.n)'
Y
mn
=
YmnC
R
n
.
n
),
vrnn
=
Ymn(O).
In problem 2
of
problem set 7, so may make
these approximations. Suppose that we make the approximation that the
lth
orbital
is
well separated
in
energy from the others. Then we can neglect
(l[n(
R)
and Y[n(R) for n
*-
I.
We write B =
vll'
Focusing on the m = 1 equation, we
have:
(E[-E(k)+all(E[-E(k»
Ieik-R
=P+Yll
Ie
ik
.
R
R.,n R.,n
Hence,
P +
Yll
IR
eik-R
E (k) = E (k) =E[
-1
"
n.n.
ik.R
+ all
L..J
R.,
e
If
we neglect the
1t'
S and retain only the y's, then we r;cover the result
of
our phenomenological model. For instance, for the cubic lattice, we have:
E (k) =
[E
[-
B]
-
2Yll
[cos
kxa
+ cos
kp
+ cos
kp]
Tight-binding models give electronic wavefunctions which are a coherent
superposition oflocalized atomic orbitals. Such wavefunctions have very small
amplitude
in
the interstitial regions between the ions.
Such models are valid, as we have seen, when there is very little overlap
between atomic wavefunctions on neighboring atoms.
In other words, a tight-
binding model will
be
valid when the size
of
an atomic orbital
is
smaller than
the interatomic distance, i.e.
a
o
«R.
In the case
of
core electrons, e.g. Is, 2s,
2p, this
is
the case. However, this
is
often not the case for valence electrons,
e.g. 3s electrons. Nevertheless, the tight-binding
method is a simple method
which gives many qualitative features
of
electronic bands. In the study
of
high-
Tc
superconductivity, it has proven useful for this reason.
THE 0-
Let
us
now consider another simple toy-model
of
a solid,
aID
array
of
o-functions:

Electronic Bands 269
(
li2
d2
00
)
- 2m
dx2
+ V
n~oo
o(x - na) \jJ(x) = E\jJ(x)
Between the peaks
of
the 0 functions,'I'(x) must be a superposition
of
the
plane waves e
iqx
and
e-
1qx
with energy E(q) =
li2q2/2m.
Between x = 0 and
x=a,
'I'(x) =
e,qx+ia.
+ e
-iqx-11t
with a complex. According to Bloch's theorem,
'I'(x + a) = e
ika
'I'(x)
Hence, in the region between x = a and x = 2a,
\jJ(x) = e
ika
(eiq(x-a)+w
+ e
-iq(x-a)-Ia.
)
Note that
k which determines the transformation property under a
translation
x
~
x + a is not the same
as
q,
which
is
the 'local' momentum
of
the electron, which determines the energy. Continuity at x = a implies that
cos(qa
+
(,.)
= e
ika
cos a
or,
or,
cosqa-e
,ka
tan
a=
smqa
Integrating Schrodinge's equation from x = a - E to X = a + E , we have,
sin( qa
+ a.) -
~ika
sin a =
2~
V
e,ka
cos
a.
Ii q
2mV
lka
.
--e
-smqa
li
2
tan a = _-'qO--
___
_
cosqa
_e
ika
Combining these equations,
e
21ka
_
2(cosqa
+ m V sinqa)e1ka + 1 = 0
li
2
q
The sum
of
the two roots is cos
ka:
k
cos(qa-o)
cos a = -"":":'---'-
coso
where
mV
tan 8
=-2-
Ii q
For each k E [
-~,~],
there are infinitely many roots q
of
this equation,
qn(k). The energy spectrum
of
the
nth
band
is:

270
Electronic Bands
2
En(k)
=~
[qn(k)]2
2m
±Ie
have the same root qn(k) =
qn(-k).
Not all
q's
are allowed. For instance, the
values
qa
- B =
me
are not allowed. These regions are the energy gaps between
bands. Consider, for instance,
k = 1t/a.
cos(qa
-1t)
= cos B
This has the solutions
qa
= 1t, 1t +
2B
. 2m
Va
For V small, the latter solution occurs at qa =
1t
+
1th
2
. The energy gap
is:
r:::!2V/a
NEARLY
FREE
ELECTRON
APPROXIMATION
as:
According to Bloch's theorem, electronic wavetUnctions can be expanded
\jI(X) = LCk_Gei(k-G)-x
G
In the nearly free electron approximation, we assume that electronic
wavefunctions are given by the superposition
of
a small number
of
plane waves.
This approximation is valid, for instance, when the periodic potential is weak
and contains a limited number
of
reciprocal lattice vectors.
Let's see how this works. Schrodinge's equation
in
momentum space reads:
---E(k)
+ck
+
LCk-G
VG
=0
(
h
2
k
2
_ )
2m G
Second-order perturbation theory tells us that (let's assume that V
k
= 0)
where,
_
-"
IV
G
I
2
E(k)=EO
(k)+
~
-
-_
_ h
2
k
2
EO
(k)=--
2m
G#OEO
(k)-
EO
(k
-G)
Perturbation theory will be valid so long as the second term is small, i.e.
so long as
IVGI«
EO
(k)-
EO
(k -
G)
For generic k, this will be valid
if
VG
is
small. The correction to the

Electronic Bands
271
energy will be
O(lv
G
I
2
).
However, no matter how small V G is, perturbation
theory fails for degenerate states,
/i
2
k
2
/i
2
(k
_ G)2
--=-
2m 2m
or, when the Bragg condition is satisfied,
G
2
=2k·a
In other words, perturbation theory fails when k is near a Brillouin zone
boundary. Suppose that
V G
is
very small so that we can neglect it away from
the Brillouin zone boundaries. Near a zone boundary, we can focus on the
reciprocal lattice vector which it bisects,
a and ignore V
G
'
for
a"*
a'.
We
keep only
c
k
and ck-G' where E o(k)
:::::
E o(k - G). We can thereby reduce
Schrodinge's equation to a 2 x 2 equation:
(EO
(k)
- E (k))Ck
+ck-G
VG
=0
(EO
(k-a)-E(k-a))Ck_G
+Ck
V~
=0
V
G
'
for a
"*
fj' can be handled by perturbation theory and, therefore, neglected
in the small
V
G'
limit. In this approximation, the eigenvalues are:
E±
(k)
=~[
EO
(k)
+
EO
(k
-G)±~(EO
(k)
-
EO
(k
_G))2
+41VG12
]
At the zone boundary, the bands have been split by
E+
(f)
-
E_
(f)
=
21VGI
The effects
of
V
G
'
for
G"*
G'
are now handled perturbatively.
To summarize, the nearly free electron approximation gives energy bands
which are essentially free electron bands away from the Brillouin zone
boundaries; near the Brillouin zone boundaries, where the electronic crystal
momenta satisfy the Bragg condition, gaps are opened.
Though intuitively appealing, the nearly free electron approximation is
not very reasonable for real solids. Since
41tZe
2
VG
~--2--13.6eV
G
IVGI-Eo
(f)-EO
(f
-G)
and the nearly free electron approximation
is
not valid.
SOME
GENERAL PROPERTIES
OF
ELECTRONIC
BAND
STRUCTURE
Much, much more can be said about electronic band structure. There are
many approximate methods
of
obtaining energy spectra for more realistic
