Carson Ph., Mumford C. Hazardous Chemicals Handbook (Справочник по опасным химическим веществам)
Подождите немного. Документ загружается.


The brown nitrogen dioxide gas condenses to a yellow liquid which freezes to colourless
crystals of dinitrogen tetroxide. Below 150°C
the gas consists of molecules of dinitrogen tetroxide
and nitrogen dioxide in equilibrium and the proportion of dinitrogen tetroxide
increases as the
temperature falls. Above 150°C nitrogen dioxide dissociates into nitric oxide and oxygen.
Nitrogen dioxide is an oxidizing agent; it gives up all, or part, of its oxygen to reducing agents,
leaving a residue of nitrogen and nitric oxide. It reacts with potassium, hydrogen sulphide,
mercury, burning phosphorus or carbon, heated iron and copper. Explosions have been reported
between nitrogen dioxide and a host of materials including alcohols (to produce alkyl nitrates),
boron compounds, carbonyl metals, propyl nitrite, nitroaniline dust, sodium amide, triethylamine,
and vinyl chloride.
Nitrogen dioxide reacts with water, giving first a mixture of nitrous and nitric acids, and
ultimately nitric acid and nitric oxide:
H
2
O + 2NO
2
→ HNO
3
+ HNO
2
H
2
O + 3NO
2
→ 2HNO
3
+ NO
When dry the gas is not corrosive to mild steel at normal temperatures and pressures. Metals and
alloys such as carbon steel, stainless steel, aluminium, nickel and Inconel are satisfactory. For wet
usage stainless steels resistant to 60% nitric acid are suitable. Important uses include use as a
bleaching agent, an oxidation catalyst, polymerization inhibitor, a nitrating agent, oxidizing agent,
rocket fuel, and in explosives manufacture. Its physical properties are summarized in Table 9.23
and its vapour pressure/temperature relationship is shown in Figure 9.10.
Table 9.23 Physical properties of nitrogen dioxide
Molecular weight 46.005 (or 92.01 for the tetroxide)
Vapour pressure @ 21°C 14.7 psia
Specific volume @ 21°C, 1 atm 293.4 ml/g
Boiling point @ 1 atm 21.25°C
Freezing point @ 1 atm –9.3°C
Specific gravity (gas) @ 20°C, 1 atm 1.58
Density (gas) @ 21°C, 1 atm 3.3 g/l
Density (liquid) @ 20°C 1.448 g/ml
Critical temperature 158.0°C
Critical pressure 1470 psia (100 atm)
Critical density 0.56 g/ml
Latent heat of vaporization @ bp 99.0 cal/g
Specific heat (gas) @ 25°C, 1 atm
C
p
0.1986 cal/g °C
Viscosity (liquid) @ 20°C 4.275 millipoises
A key feature of its toxicity (page 154) at low concentrations is the delay between exposure and
onset of symptoms. The OES is 3 ppm (8 hr TWA) and 5 ppm (15 min STEL). Effects of exposure
are summarized in Table 5.33. Chronic exposures to low concentrations may cause chronic
irritation of the respiratory tract with cough, headache, loss of weight, loss of appetite, dyspepsia,
corrosion of the teeth and gradual loss of strength. Concentrations above 60 ppm produce immediate
irritation of the nose and throat with coughing, choking, headache, shortness of breath and
restlessness. Even brief exposures above 200 ppm may prove fatal. Liquid nitrogen dioxide
(dinitrogen tetroxide) is corrosive to skin.
First-aid measures include removal from the contaminated atmosphere, rest and administration
of pure oxygen. Skin or eyes in contact with liquid should be thoroughly irrigated. Medical
attention should be sought. Other special precautions include:
NITROGEN OXIDES 299

300 COMPRESSED GASES
1000
500
200
100
50
20
20015010070°F
93663721°C
Temperature
Vapour pressure (psia)
• Handle in well-ventilated area, preferably with hood equipped with forced extract ventilation.
• Ensure adequate number of emergency exits.
• Consider the need for personal protection such as skin, eye (goggles/face shield) and respiratory
protection including self-contained breathing apparatus.
• Provide instant-acting safety showers and eye-wash facilities in the location of the work
area.
• Train staff in appropriate first-aid measures including how to seek immediate medical assistance.
Figure 9.10
Nitrogen dioxide vapour pressure vs temperature

Oxygen
Oxygen occurs free in air in which it forms 21% by volume. It is also found combined with
hydrogen in water and constitutes 86% of the oceans, and with other elements such as minerals
constituting ca 50% of the earth’s crust. In the laboratory it is usually prepared by the thermal
decomposition of potassium chlorate in the presence of manganese dioxide catalyst:
2KClO
3
→ 2KCl + 3O
2
Industrially, it is manufactured either by fractional distillation of air, or by electrolysis of sodium
hydroxide and it is distributed as a non-liquefied gas in pressurized black cylinders at ca 2200 psig
at 21°C. Since it is non-corrosive no special materials of construction are required.
Selected physical properties of oxygen are included in Table 9.24. It is a colourless, odourless
and tasteless gas which is essential for life and considered to be non-toxic at atmospheric pressure.
It is somewhat soluble in water and is slightly heavier than air. Important uses are in the steel and
glass industries, oxyacetylene welding, as a chemical intermediate, waste-water treatment, fuel
cells, underwater operations and medical applications.
Table 9.24 Physical properties of oxygen
Molecular weight 32.00
Specific volume @ 21°C, 1 atm 755.4 ml/g
Boiling point @ 1 atm –183.0
o
C
Triple point –218.8°C
Density (gas) @ 0°C, 1 atm 1.4291 g/l
Density (liquid) @ bp 1.141 g/l
Critical temperature 1118.4°C
Critical pressure 737 psia (50.14 atm)
Critical density 0.427 g/l
Latent heat of vaporization @ bp 50.94 cal/g
Specific heat (gas) @ 15°C, 1 atm
C
p
0.2200 cal/g°C
C
v
0.1554 cal/g°C
ratio
C
p
/
C
v
1.42
Viscosity (gas) @ 25°C, 1 atm 0.02064 centipoise
Solubility in water @ 0°C, 1 atm 1 volume/21 volumes water
Explosive reactions can occur between oxygen and a wide range of chemicals including organic
compounds (such as acetone, acetylene, secondary alcohols, hydrocarbons), alkali and alkaline
earth metals, ammonia, biological specimens previously anaesthetized with ether, hydrogen and
foam rubber.
Oxygen supports combustion and the hazard is increased if the concentration in air exceeds
21% (page 199) or at pressures above atmospheric pressure. Substances ignite more readily, burn
at a faster rate, generate higher temperatures and may be extremely difficult to extinguish. Substances,
e.g. plastics, clothing or metals, which may not normally burn easily can burn vigorously in
oxygen-enriched air. Oxygen may become trapped within clothing; this can then be ignited and
cause serious burn injuries. Because some substances, e.g. oil and grease, may react explosively
with pressurized oxygen it is important never to use lubricants on oxygen equipment and to free
pipelines from deposits of them.
Enrichment of the atmosphere in any workplace to about 25% oxygen can be hazardous; this
is particularly so in a confined space. Inhalation of 100% oxygen at atmospheric pressure for
OXYGEN 301

302 COMPRESSED GASES
16 hr per day for several days poses no undue problems but longer periods of exposure to high
pressures can adversely affect neuromuscular coordination and the power of concentration.
In addition to the control measures given in Table 9.3, the following precautions are appropriate
when using industrial and medical oxygen, or mixtures of oxygen with other gases.
Never:
• Use oxygen to sweeten the air of a workplace.
• Use oxygen instead of fresh air to ventilate or cool a confined space.
• Use oxygen instead of compressed air or as a source of pressure, e.g. to clear blockages in
pipelines or to power air-driven tools.
• Use oxygen to blow down clothing, benches or machinery.
• Use oxygen to inflate vehicle tyres, rubber boats etc.
• Use oxygen to start a diesel engine.
• Use oxygen to cool the person.
• Use oil or grease on oxygen equipment.
• Use jointing compound or tape to cure leaks in oxygen equipment, e.g. at cylinder connections.
• Cut off the supply of oxygen by nipping or kinking flexible hose when changing equipment.
Always:
• Only use materials and equipment which are suitable for oxygen service and to a recognized
standard.
• Regularly check equipment for signs of leaks, e.g., at hose connections; replace damaged or
worn items. Usually the system is pre-tested with, e.g., nitrogen.
• Ensure that equipment, e.g. pipework for oxygen service, is kept clean and free from oil, grease
or dust.
• Ensure that the rated maximum inlet pressure of the regulator is not less than the cylinder
supply pressure. (For cylinder pressures up to 200 bar, pressure regulators should comply with
BS 5741. For higher cylinder pressures check with the manufacturer that the pressure regulator
has been shown to be suitable by appropriate testing.)
• Ensure that the pressure adjusting screw of a pressure regulator is fully unwound, so that the
regulator outlet valve is closed before opening the oxygen cylinder valve.
• Open cylinder valves slowly.
• Ensure that the cylinder valves are closed and piped supplies isolated whenever work is stopped.
• Ensure that flexible oxygen hose for welding etc. complies with BS 5120. The correct colour
for oxygen is blue.
• Ensure that proper fresh air ventilation is provided in oxy-fuel gas welding and cutting operations.
• Ensure that high-pressure oxygen systems are designed, constructed, installed and commissioned
by competent people with specialized knowledge of the subject.
• When working on ships, remove pipes or hoses when work stops, other than for short intervals.
• Store oxygen cylinders in a well-ventilated area or compound away from combustible materials
and separated from cylinders of flammable gases.
• Handle oxygen cylinders carefully, preferably using a purpose-built trolley and keep secured to
prevent cylinders from falling.
• Wear protective clothing appropriate to the work, e.g. leather gloves, fire-retardant overalls,
safety shoes or boots and eye protection.
• If applicable, locate oxygen cylinders outside any confined space and in an area of good
ventilation.

Ozone
Ozone is an allotrope of oxygen containing three oxygen atoms. It occurs naturally in the upper
atmosphere and is formed in small quantities during electrical discharges from electrical machines
or when white phosphorus smoulders in air. In the laboratory it is most conveniently obtained by
subjecting air to electrical discharges in ‘ozonizers’ when some of the oxygen molecules dissociate
into oxygen atoms which then combine with other oxygen molecules. However, the yield of
ozone is only 10% even when pure oxygen is used. It may also be prepared by electrolysis of ice-
cold dilute sulphuric acid using a high current density. Here the concentration of ozone liberated
at the platinum-in-glass anode is about 14%.
Pure ozone is made by fractional distillation of the blue liquid resulting from the cooling of
ozonized oxygen in liquid air. Commercially it is often supplied dissolved in chlorofluorocarbons
in stainless steel cylinders at ca 475 psig cylinder pressure at 20°C often transported chilled with
dry ice. These solutions can be handled safely at vapour concentrations of ca 20% by volume of
ozone.
The physical properties of ozone are summarized in Table 9.25.
Table 9.25 Physical properties of ozone
Molecular weight 47.998
Boiling point @ 1 atm –111.9°C
Freezing point @ 1 atm –192°C
Density (gas) @ 0°C, 1 atm 2.143 g/l
Density (liquid) @ –183°C 1.571 g/l
Critical temperature –12.1°C
Critical pressure 802.6 psia (54.6 atm)
Viscosity (liquid) @ –183°C 1.57 cP
Latent heat of vaporization @ bp 3410 cal/mole
Dielectric constant (liquid) @ –183°C 4.79
Dipole moment 0.55D
Solubility in water @ 0°C, 1 atm 0.494 volume/volume of water
Ozone is strongly exothermic in its reactions and neat solid or liquid phases are highly explosive.
Pure ozone is a toxic, slightly bluish, unstable, non-flammable but potentially explosive gas
with a smell akin to that of much-diluted chlorine. It is used mainly because of its extreme
oxidizing ability (second only to fluorine in oxidizing power) in chemical syntheses or because
of its powerful germicidal activity on many bacterial organisms, e.g. as a water purification agent
(e.g. swimming pools) although it may leave an unpleasant taste, as a bleach, in treatment of
industrial waste, sterilization of air (e.g. in the ventilation of premises of underground railways
with limited access to fresh air), deodorizing sewage and stack gases, and in food preservation.
Indeed, algae and certain fungi resistant to chlorine are highly susceptible to ozone. It decomposes
slowly at room temperature and rapidly at 200°C and is decomposed by many finely-divided
metals. It reacts readily with unsaturated organic moieties to form ozonides:
R
1
—CH
==
CH—R
2
+ O
3
→
R
1
—CH CH—R
2
O O
O
(Ozonide)
OZONE 303

304 COMPRESSED GASES
where R
1
and R
2
are univalent organic groups.
Violent reactions have occurred between ozone and many chemicals, a small selection being
acetylene, alkenes, dialkyl zincs, benzene/rubber solution, bromine, carbon monoxide and ethylene,
diethyl ether, hydrogen bromide, and nitrogen oxide.
Ozone is an irritant to eyes and mucous membranes. Inhalation can cause pulmonary oedema
and bleeding at ‘high’ concentrations, whilst at lower exposures symptoms include headache,
shortness of breath, or drowsiness. Long-term effects may include chronic pulmonary effects,
ageing, and possibly lung cancer. The ACGIH classify ozone as one of those ‘Agents which cause
concern that they could be carcinogenic for humans but which cannot be assessed conclusively
because of the lack of data. In vitro or animal studies do not provide indications of carcinogenicity
which are sufficient to classify the agent into one of the other categories.’ The TLV is set as a
sliding scale for exposures during different workloads, thus:
• 0.05 ppm for heavy work.
• 0.08 ppm for moderate work.
• 0.10 ppm for light work.
• 0.20 ppm for heavy, moderate, or light workloads for up to a maximum exposure of 2 hours.
Because of the odour threshold of ca 0.015 ppm, exposure to ozone below the TLV can usually
be detected by smell.
Precautions in addition to those in Table 9.3 include:
• Keep cylinders chilled (to help prevent decomposition rather than as a safety measure).
• Avoid copper and copper alloys since these can catalyse the decomposition, and rubber components
are unsuitable.
• Pre-test systems for leaks with inert gas.
• Prevent contact with grease, oil or other combustible material.
• Clean all equipment for oxygen service.
• Handle in a ventilated hood to protect surrounding atmosphere from leaks.
• Consider the need for appropriate personal protection including eye, skin and respiratory
equipment.
• Consider the need for leak detection systems.
• Where possible avoid explosions by working with dilute solutions at low temperatures in
suitable solvents.
Sulphur dioxide
Sulphur dioxide is used as a preservative for beer, wine and meats; in the production of sulphites
and hydrosulphites; in solvent extraction of lubricating oils; as a general bleaching agent for oils
and foods; in sulphite pulp manufacture; in the cellulose and paper industries; and for disinfection
and fumigation.
It is a non-flammable colourless gas which is twice as dense as air, and slightly soluble in water
forming sulphurous acid. It is readily liquefied as a gas under its own vapour pressure of about
35 psig (2.4 bar) at 21°C. Figure 9.11 depicts the effect of temperature on vapour pressure; Table
9.26 lists the physical properties. Cylinders tend to be protected against over–pressurization by
metal plugs melting at about 85°C.
Gaseous sulphur dioxide is highly irritant and practically irrespirable. Effects on the body are
summarized in Table 5.3. It can be detected at about 3.5 ppm and the irritating effects would
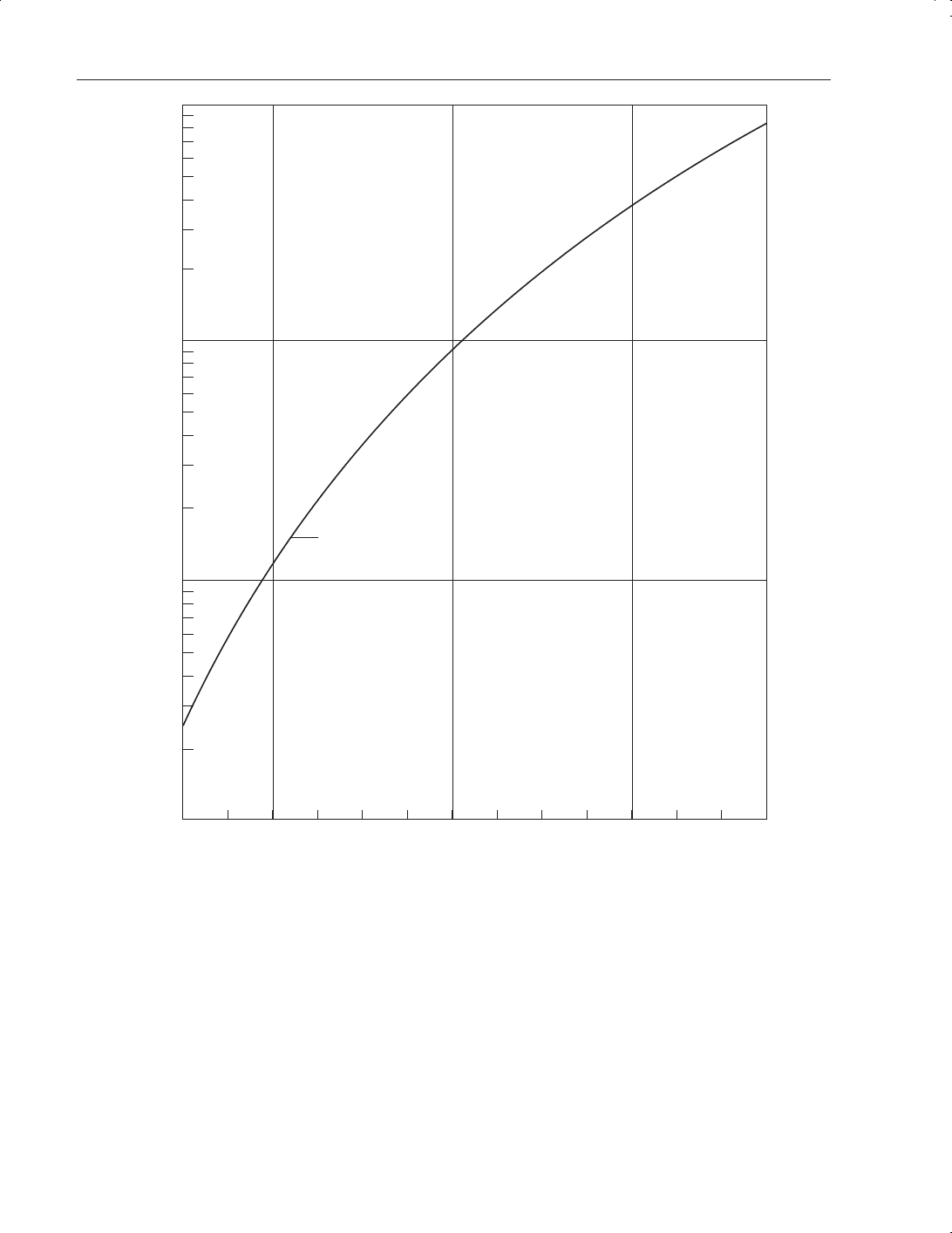
2752502252001751501251007550250–25–50°F
13512110793796652372510–4–18–32–46°C
Temperature
1000
900
800
700
600
500
400
300
200
100
90
70
60
50
40
30
20
10
9
8
7
6
5
4
3
2
80
1
Vapour pressure (psia)
Boiling point
Figure 9.11
Sulphur dioxide vapour pressure vs temperature
preclude anyone from suffering prolonged exposure at high concentrations unless unconscious, or
trapped.
Liquid sulphur dioxide may cause eye and skin burns resulting from the freezing effects upon
evaporation. Dry sulphur dioxide is non-corrosive to common materials of construction except
zinc. The presence of moisture renders the environment corrosive.
In addition to the precautions listed in Table 9.3, the following controls are appropriate:
• Use in well-ventilated areas.
SULPHUR DIOXIDE 305

306 COMPRESSED GASES
Table 9.26 Physical properties of sulphur dioxide
Molecular weight 64.063
Vapour pressure at 21°C 2.37 bar
Specific volume at 21°C, 1 atm 368.3 ml/g
Boiling point at 1 atm –10.0°C
Freezing point at 1 atm –75.5°C
Specific gravity, gas at 0°C, 1 atm (air = 1) 2.264
Density, gas at 0°C, 1 atm 2.927 g/l
Density, liquid at –10°C 1.46 g/ml
Critical temperature 157.5°C
Critical pressure 78.8 bar
Critical density 0.524 g/ml
Latent heat of vaporization at boiling point 92.8 cal/g
Latent heat of fusion at melting point 27.6 cal/g
Specific heat, liquid at 0°C 0.318 cal/g °C
Specific heat, gas at 25°C, 1 atm
C
p
0.1488 cal/g °C
C
v
0.1154 cal/g °C
ratio
C
p
/
C
v
1.29
Thermal conductivity at 0°C 2.06 × 10
–5
cal/s cm
2
°C/cm
Viscosity, gas at 18°C, 1 atm 124.2 mP
Solubility in water at 0°C, 1 atm 18.59% by weight
at 20°C, 1 atm 10.14% by weight
• Wear eye/face protection, approved footwear and rubber gloves.
• Showers and eye-wash facilities and respiratory protection should be conveniently located for
emergencies.
• Insert traps in the line to avoid liquid suck-back into the cylinder.
• Check for leaks with soap solution, aqueous ammonia or colour indicator tubes.
• First aid measures include those in Table 9.9.

Monitoring techniques
As mentioned in Chapters 4, 5 and 16 chemicals can be a nuisance or pose safety, health and
environmental risks, or become wasteful of expensive resources if allowed to escape excessively
and uncontrollably into the general or workplace environment. Escapes can result from inadequate
process control, errors in operation or maintenance, incomplete understanding of the process, etc.
Such problems can arise from both: periodic emissions of chemicals due to the need to open, or
enter, the ‘system’ occasionally (e.g. during sampling, cleaning, line-breaking) including both
planned and unplanned releases (e.g. due to accidents, human error) and, continuous low-level
fugitive emissions from normally-closed points, e.g. valve seals, flange gaskets, pump seals, drain
valves.
The need to monitor the impact of activities involving chemicals on the environment may stem
from sound management practice or to satisfy a host of specific legal requirements. Thus, in the
UK under the Environmental Protection (Prescribed Substances and Processes) Regulations 1991,
operators must apply BATNEEC to prevent or minimize the release of prescribed substances into
the environment, or to render harmless any emissions. The prescribed substances for release into
the air are given in Table 10.1. No prescribed process may be operated without an authorization
from the Environment Agency and air pollutants which must be measured and the frequency of
monitoring are set out in the authorization. Compliance with emission limits for municipal waste
incineration plants (Table 10.2) also requires monitoring.
Table 10.1 Prescribed substances for release into the air
Oxides of sulphur and other sulphur compounds
Oxides of nitrogen and other nitrogen compounds
Oxides of carbon
Organic compounds and partial oxidation compounds
Metals, metalloids and their compounds
Asbestos, glass fibres and mineral fibres
Halogens and their compounds
Phosphorus and its compounds
Particulate matter
In addition to pollution episodes, risks may arise due to atmospheric oxygen concentrations
fluctuating beyond its normal level of 21% posing health (page 72) or fire hazards. Fire and
explosion dangers may also arise from the presence of flammable gases, vapours, or dusts in the
atmosphere (Chapter 6).
Thus, as illustrated by Table 17.13 monitoring emissions of hazardous chemicals into the
environment may be required for a variety of reasons such as:
10

308 MONITORING TECHNIQUES
• Assessing fire or explosion risks from atmospheres containing flammable gas, vapour or dust.
• Determining oxygen content of the working atmosphere.
• Determining sources of leaks of toxic, flammable, or nuisance pollutants.
• Identifying unknown pollutants.
• Assessing process efficiency and control.
• Assessing environmental risk from effluent discharges or for formal environmental impact
assessment.
• Determining employee exposure to known toxic substances.
• Providing data for internal company environmental audits.
• Investigation of the causes of accidents.
• Investigation of the cause and nature of problems (e.g. local complaints of odour) or pollution
incidents.
Selected general analytical techniques for
monitoring environmental pollution
Stages in environmental monitoring include obtaining representative samples of the environment
in question and subsequent analysis of physical, chemical or microbiological attributes. Monitoring
techniques range from sophisticated in-line, continuous sampling and instantaneous analysis
linked to audible/visual alarms or features to control the pollution; samplers running continuously,
e.g. throughout a normal day for subsequent analysis; to grab samples (i.e. samples collected over
a short time span of, e.g., a few minutes). Continuous monitoring is common in personal dosimetry
studies where an appropriate collection device samples air wherever a worker is throughout a
specified period, e.g. 15 minutes or 8 hours (page 111). A selection of common analytical techniques
include the following.
Gases and vapours
Atomic absorption/emission spectrometry
Metal ions are most commonly measured using atomic absorption spectrometry. In this technique
Table 10.2 Selected emission limits for municipal waste incineration (units: mg/m
3
)
Country EU EU EU UK UK
Plant capacity 3 1–3 <1 >1 <1
(tonne per hour)
Particulates 30 100 200 30 200
CO 100 100 100 100 100
SO
2
300 300 – 300 300
Volatile organic 20 20 20 20 20
compounds
HF 2 4 – 2 –
NO
x
as NO
2
– – – 350 –
Cr, Cu, Mn, Pb 5 5 –
Ni, As 1 1 – 1 (incl Sn)
5
Cd, Hg 0.2 0.2 – 0.1 each
Dioxins – – – 1
}
