Hogg S. Essential microbiology
Подождите немного. Документ загружается.

JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
128 MICROBIAL METABOLISM
Aerobic respiration
We shall now examine the fate of the pyruvate produced as the end-product of glycol-
ysis. As we have seen, this depends on whether the organism in question is aerobic or
anaerobic.
You will recall that during glycolysis, NAD
+
was reduced to NADH. In order for glu-
cose metabolism to continue, this supply of NAD
+
must be replenished; this is achieved
either by respiration or fermentation. Respiration is the term used to describe those ATP-
generating processes, aerobic or anaerobic, by which oxidation of a substrate occurs,
with an inorganic substance acting as the final electron acceptor.Inaerobic respira-
tion, that substance is oxygen; in anaerobic respiration, a substance such as nitrate or
sulphate can fulfil the role.
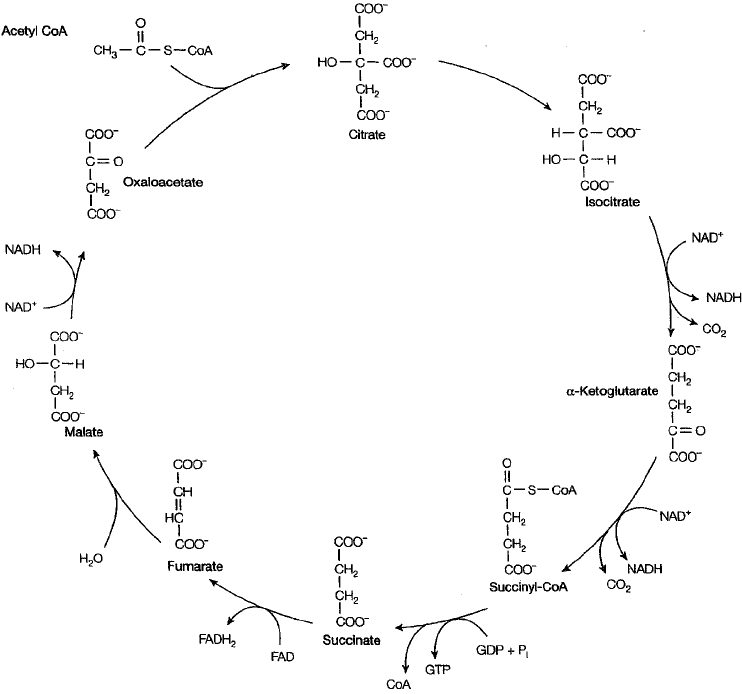
The TCA cycle is a series
of reactions that oxidize
acetate to CO
2
, gener-
ating reducing power in
the form of NADH and
FADH
2
for use in the elec-
tron transport chain.
In most aerobic organisms, the pyruvate is completely
oxidised to CO
2
and water by entering the tricarboxylic
acid (TCA) cycle, also known as the Krebs cycle or sim-
ply the citric acid cycle (Figure 6.20). During this cycle,
a series of redox reactions result in the gradual transfer
of the energy contained in the pyruvate to coenzymes
(mostly NADH). This energy is finally conserved in the
form of ATP by a process of oxidative phosphorylation.
We shall turn our attention to these important reactions
in due course, but first let us examine the role of the TCA cycle in a little more detail.
Pyruvate does not itself directly participate in the TCA cycle, but must first be con-
verted into the two-carbon compound acetyl-Coenzyme A:
Pyruvate Acetyl-CoA + CO
2
(2C)
NAD
+
NADH
+ H
+
(3C)
Coenzyme A
This is an important intermediate, as lipids and amino acids can also be metabolised
into this form, and thereby feed into the TCA cycle. The main features of the cycle are
as follows:
r
each reaction is catalysed by a separate enzyme
r
four of the reactions involve substrate oxidation, with energy, in the form of
electrons, passing to form NADH (mainly) and FADH
2
r
the two carbons present in acetyl-CoA are removed as CO
2
r
one reaction involves the generation of ATP by substrate-level phosphorylation.
For each ‘turn’ of the citric acid cycle, one molecule of ATP, three molecules of NADH
and one molecule of FADH
2
are produced (FADH
2
is the reduced form of another
coenzyme, FAD). Since these derive from oxidation of a single acetyl-CoA molecule,
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
PRINCIPLES OF ENERGY GENERATION 129
Figure 6.20 The TCA cycle. Acetyl-CoA may derive from the pyruvate of glycolysis or
from lipid or amino acid metabolism. It joins with the four-carbon oxaloacetate to form the
six-carbon citric acid. Two decarboxylation steps reduce the carbon number back to four and
oxaloacetate re-enters the cycle once more. Although no ATP results directly from the cycle,
the third phosphate on GTP can be easily transferred to ADP (GTP + ADP = GDP + ATP),
thus generating one molecule of ATP per cycle. In addition, substantial reducing power is
generated in the form of NADH and FADH
2
. These carry electrons to the electron transport
chain, where further ATPs are generated
we need to double these values per molecule of glucose originally entering glycolysis.
Several of the intermediate molecules in the TCA cycle also act as precursors in other
pathways, such as the synthesis of amino acids, fatty acids or purines and pyrimidines
(see Anabolic metabolism, below). Other pathways regenerate such intermediates for
continued use in the TCA cycle (see Box 6.3).
So far, we are a long way short of the 38 molecules of ATP per molecule of glucose
mentioned earlier; we have only managed two ATPs from glycolysis and a further two
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
130 MICROBIAL METABOLISM
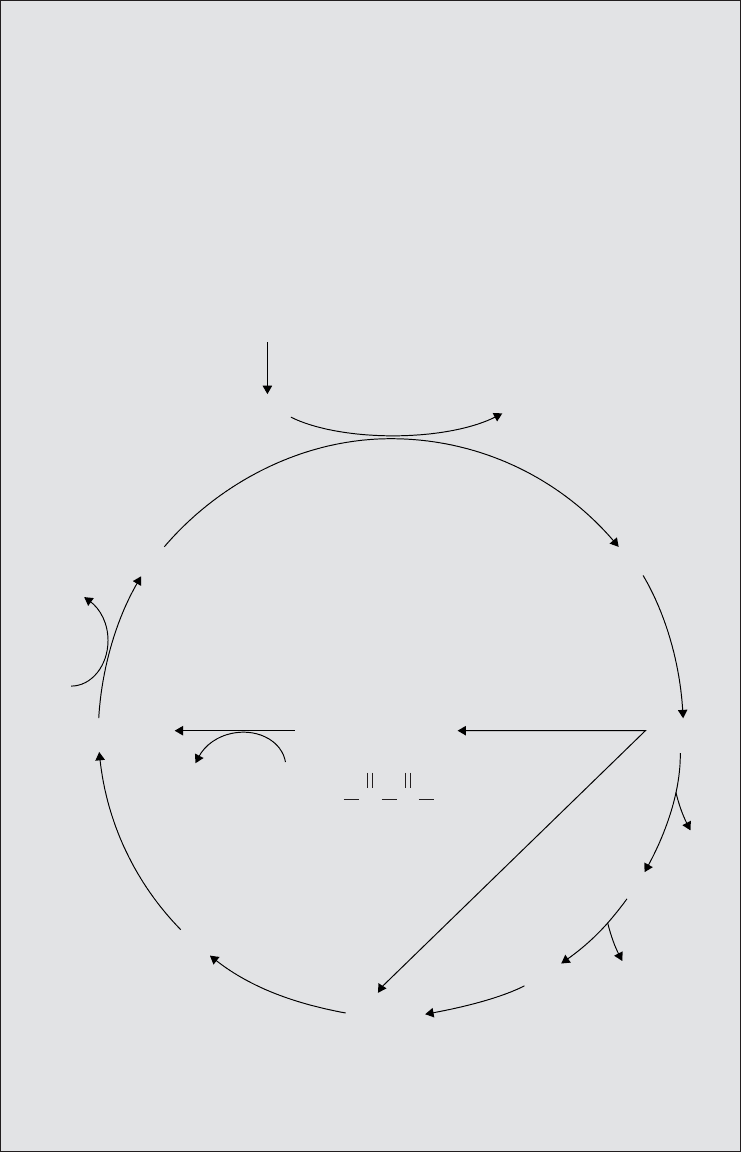
Box 6.3 The glyoxylate cycle
The components of the TCA cycle may act as precursors for the biosynthesis of other
molecules (e.g., both α-ketoglutarate and oxaloacetate can be used for the synthe-
sis of amino acids). For the TCA cycle to continue, it must replace these compounds.
Many microorganisms are able to do this by converting pyruvate to oxaloacetate
via a carboxylation reaction. A pathway that replenishes intermediate compounds
of another in this way is termed anaplerotic. Organisms that use acetate (or molecules
that give rise to it e.g. fatty acids) as sole carbon source regenerate TCA interme-
diates by means of the glyoxylate cycle (sometimes known as the glyoxylate shunt or
bypass). This resembles the TCA cycle, but the two decarboxylation reactions (i.e.
those where CO
2
is removed) are missed out (compare with Figure 6.20).
Succinate
α-Ketaglutarate
Isocitrate
Citrate
Fumarate
Malate
Oxaloacetate
SuccinylCoA
CO
2
CO
2
AcetylCoA CoA
Fatty Acids
CoA Acetyl
CoA
Malate
synthase
Isocitrate
Iyase
Glyoxylate
OO
CHCO
–
NADH
+H
+
NAD
+
Thus, isocitrate is converted directly to succinate and glyoxylate, and in another
unique reaction, the glyoxylate is joined by acetyl-coA to form malate. The result of
this is that succinate can be removed to participate in a biosynthetic pathway, but
oxaloacetate is still renewed via glyoxylate and malate.
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
PRINCIPLES OF ENERGY GENERATION 131
NADH
NAD
+
FADH
2
FM N FAD
Q
Cyt b
Energy Cyt c
1
Cyt c
Cyt a
Cyt a
3
O
2
2
1
2H
+
H
2
O
Figure 6.21 The electron transport chain. Electrons from NADH and FADH
2
pass from
one electron carrier to another, with a gradual release of energy as ATP by chemiosmosis (see
Figure 6.22). The electron carriers are arranged in order of their reduction potential (tendency
to gain electrons) and oscillate between the oxidised and the reduced state. FMN = flavin
mononucleotide, Q = coenzyne Q.
from the TCA cycle. Where do all the rest come from? Most of the energy originally
stored in the glucose molecule is now held in the form of the reduced coenzymes (NADH
and FADH
2
) produced during glycolysis and the TCA cycle. This is now converted to
no less than 34 molecules of ATP per glucose molecule by oxidative phosphorylation
in the remaining steps in aerobic respiration (three from each molecule of NADH and
two from each of FADH
2
).
The electron transport
chain is a series of donor/
acceptor molecules
that transfer electrons
from donors (e.g. NADH)
to a terminal electron
acceptor (e.g. O2).
In the final phase of aerobic respiration, electrons
are transferred from NADH and FADH
2
, via a series
of carrier molecules known collectively as the electron
transport (or respiratory) chain to oxygen, the terminal
electron acceptor (Figure 6.21). This in turn is reduced
to the molecules of water you will remember from our
overall equation on page 122. In procaryotes, this elec-
tron transfer occurs at the plasma membrane, while in
eucaryotes it takes place on the inner membrane of mi-
tochondria. Table 6.2 summarises the locations of the reactions in the different phases
of carbohydrate metabolism.

JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
132 MICROBIAL METABOLISM
Table 6.2 Location of respiratory enzymes
Reaction Procaryotes Eucaryotes
Glycolysis Cytoplasm Cytoplasm
TCA cycle Cytoplasm Mitochondrial matrix
Electron Transport Plasma membrane Mitochondrial inner
membrane
Oxidative phosphorylation and the electron transport chain
The components of the electron transport chain differ between procaryotes and eu-
caryotes, and even among bacterial systems, thus details may differ from the example
outlined below. The purpose of the electron transport is the same for all systems, how-
ever, that is, the transfer of electrons from NADH/FADH
2
via a series of carriers to,
ultimately, oxygen. Around half of the energy released during this process is conserved
as ATP.
The carrier molecules, which act alternately as acceptors and donors of electrons,
are mostly complex modified proteins such as flavoproteins and cytochromes, together
with a class of lipid-soluble molecules called ubiquinones (also called coenzyme Q). The
carriers are arranged in the chain such that each one has a more positive redox potential
than the previous one. In the first step in the chain, NADH passes electrons to flavin
mononucleotide (FMN), and in so doing becomes converted back to NAD
+
, thereby
ensuring a ready supply of the latter for the continuation of glycolysis (Figure 6.21).
From FMN, the electrons are transferred to coenzyme Q, and thence to a series of
cytochromes; at each transfer of electrons the donor reverts back to its oxidised form,
ready to pick up more electrons. You may recall that FADH
2
yields only two, rather than
three molecules of ATP per molecule; this is because it enters the electron transport chain
at a later point than NADH, thereby missing one of the points where export of protons
occurs. The final cytochrome in the chain transfers its electrons to molecular oxygen,
which, as we have seen, acts as the terminal oxygen acceptor. The negatively charged
oxygen combines with protons from its surroundings to form water. Four electrons and
protons are required for the formation of each water molecule:
O
2
+ 4e
−
+ 4H
+
−→ 2H
2
O
Since two electrons are released by the oxidation of each NADH, it follows that two
NADH are needed for the oxidation of each oxygen.
How does this transfer of electrons lead to the formation of ATP? The chemiosmotic
theory proposed by Peter Mitchell in 1961 offers an explanation. Although it was not
immediately accepted, the validity of the chemiosmotic model is now widely recognised,
and in 1978 Mitchell received a Nobel Prize for his work. As envisaged by Mitchell,
sufficient energy is released at three points in the electron transport chain for the transfer
of protons to the outside of the membrane, resulting in a gradient of both concentration
and charge (proton motive force). The protons are able to return across the membrane
and achieve an equilibrium through specific protein channels within the enzyme ATP
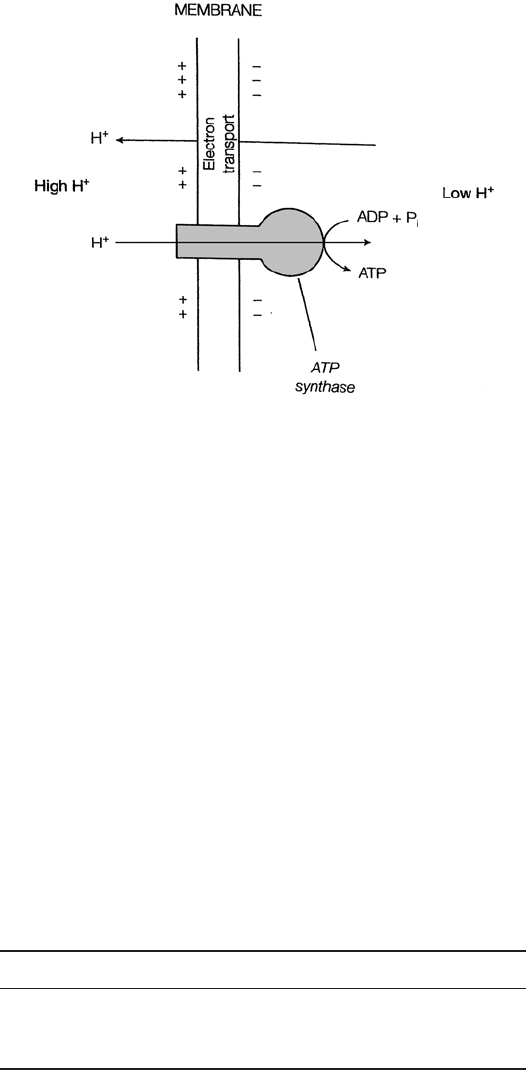
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
PRINCIPLES OF ENERGY GENERATION 133
Figure 6.22 Chemiosmosis. The active transport of protons across the membrane creates
a gradient of charge and concentration (proton motive force). Special channels containing
ATP synthase allow the return of the protons; the energy released is captured as ATP. From
Hames, BD, Hooper, NM & Houghton, JD: Instant Notes in Biochemistry, Bios Scientific
Publishers, 1997. Reproduced by permission of Thomson Publishing Services
synthase. The energy released by the protons as they return through these channels
enables the ATP synthase to convert ADP to ATP (Figure 6.22).
Aerobic respiration in eucaryotes is slightly less efficient than in procaryotes due to
the fact that the three stages take place at separate locations (see Table 6.2). Thus the
total number of ATPs generated is 36 rather than the 38 in procaryotes (Table 6.3).
Fermentation
We turn now to the fate of pyruvate when oxygen is not available for aerobic respira-
tion to take place. Microorganisms are able, by means of fermentation, to oxidise the
pyruvate incompletely to a variety of end products.
Table 6.3 Yield of ATP by aerobic respiration in procaryotes
Process ATP Yield (per glucose molecule)
Glycolysis 2
TCA Cycle 2
Electron transport chain 34
∗
∗
Derived from the oxidative phosphorylation of 2 × NADH from glycolysis,
8 × NADH and 2 × FADH
2
from TCA cycle.
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
134 MICROBIAL METABOLISM
It is worth spending a moment defining the word fermentation. The term can cause
some confusion to students, as it has come to have different meanings in different
contexts. In everyday parlance, it is understood to mean simply alcohol production,
while in an industrial context it generally means any large scale microbial process such as
beer or antibiotic production, which may be aerobic or anaerobic. To the microbiologist,
the meaning is more precise:
a microbial process by which an organic substrate (usually a carbohydrate) is broken down
without the involvement of oxygen or an electron transport chain, generating energy by substrate-
level phosphorylation
Two common fermentation pathways result in the production of respectively lactic
acid and ethanol. Both are extremely important in the food and drink industries, and
are discussed in more detail in Chapter 17. Alcoholic fermentation, which is more
common in yeasts than in bacteria, results in pyruvate being oxidised via the intermediate
compound acetaldehyde to ethanol.
CH
3
COCOO
–
CH
3
CHO CH
3
CH
2
OH
CO
2
Pyruvate Acetaldehyde Ethanol
pyruvate decarboxylase alcohol dehydrogenase
NAD
+
NADH
Electrons pass from the reduced coenzyme NADH to acetaldehyde, which acts as an
electron acceptor, and NAD
+
is thereby regenerated for use in the glycolytic pathway.
No further ATP is generated during these reactions, so the only ATP generated in the
fermentation of a molecule such as glucose is that produced by the glycolysis steps. Thus,
in contrast to aerobic respiration, which generates 38 molecules of ATP per molecule
of glucose, fermentation is a very inefficient process, producing just two. Note that all
the ATP produced by any fermentation is due to substrate-level phosphorylation, and
does not involve an electron transport chain.
A variety of microorganisms carry out lactic acid fermentation. Some, such as Strep-
tococcus and Lactobacillus, produce lactic acid as the only end product; this is referred
to as homolactic fermentation.
CH
3
COCOO
–
CH
3
CHOHCOO
–
Pyruvate Lactate dehydrogenase
NAD
+
NADH
Lactate
Certain other microorganisms, such as Leuconostoc, generate additional products such
as alcohols and acids in a process called heterolactic fermentation.
In both alcohol and lactic acid fermentation, the two NADH molecules produced
per molecule of glucose have been reoxidised to NAD
+
, ready to re-enter the glycolytic
pathway.
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
PRINCIPLES OF ENERGY GENERATION 135
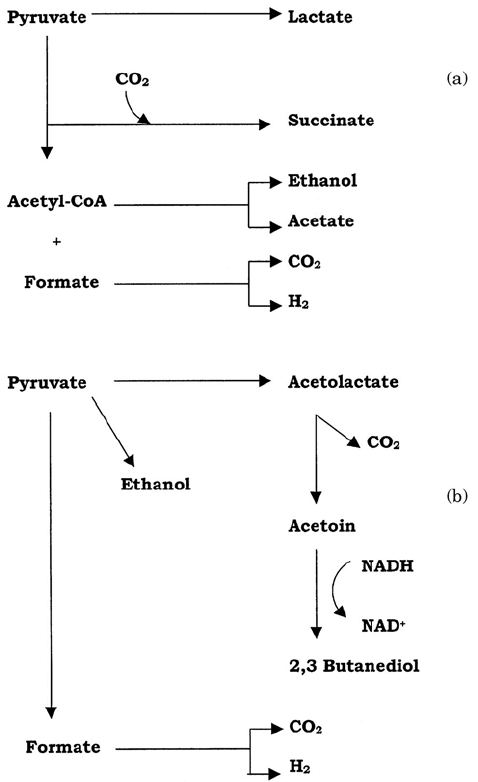
Figure 6.23 Fermentation patterns in enteric proteobacteria. All members of the group
produce pyruvate via the Embden–Meyerhof pathway (glycolysis), but subsequent reactions
fall into one of two main types. (a) Mixed acid fermentation results in ethanol and a mixture
of acids, mainly acetic, lactic, succinic and formic, e.g. Escherichia, Salmonella. (b) Butane-
diol fermentation involves the conversion of pyruvate to acetoin, then to 2,3-butanediol, e.g.
Enterobacter, Klebsiella. The ratio of CO
2
to H
2
production is much greater in butanediol
fermentation
Other types of fermentation
Members of the enteric bacteria metabolise pyruvate to a variety of organic compounds;
two principal pathways are involved, both of them involving formic acid. In mixed acid
fermentation, pyruvate is reduced by the NADH to give succinic, formic and acetic acids,
together with ethanol (Figure 6.23a). Escherichia, Shigella and Salmonella belong to

JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
136 MICROBIAL METABOLISM
this group (see Chapter 7). Other enteric bacteria, such as Klebsiella and Enterobacter,
carry out 2,3-butanediol fermentation. In this, the products are not acidic, and include
an intermediate called acetoin. Much more CO
2
is produced per molecule of glucose
than in mixed acid fermentation (Figure 6.23b). The end products of the two types of
fermentation provide a useful diagnostic test for the identification of unknown enteric
bacteria.
Metabolism of lipids and proteins
We have concentrated so far on the metabolism of carbohydrates, but both lipids and
proteins may also act as energy sources. Both are converted by a series of reactions to
an intermediate compound that can then enter the pathways of metabolism we have
discussed above.
Lipids often form an important energy source for microorganisms; they are plentiful
in nature, as they form the major component of cell membranes, and may also exist as
cellular storage structures. Lipids are hydrolysed by a class of enzymes called lipases to
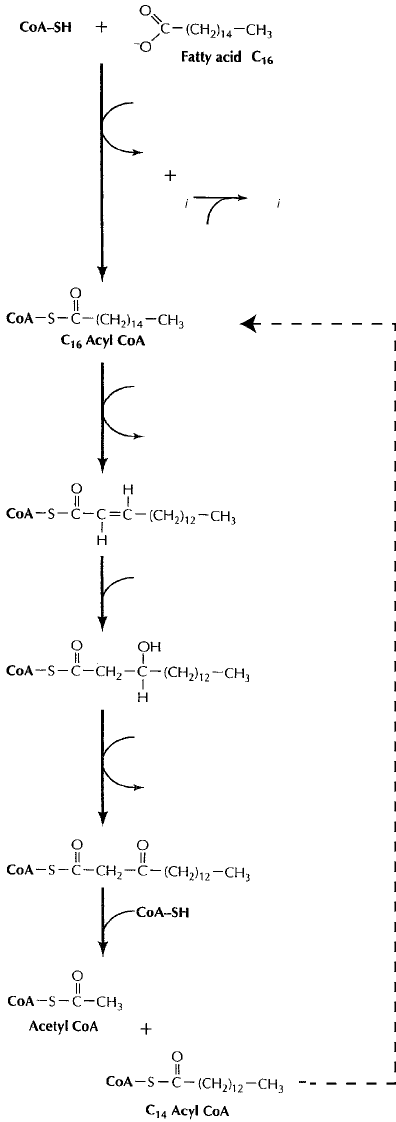
their constituent parts; the fatty acids so produced enter the cyclic β-oxidation pathway.
In this, fatty acids are joined to coenzyme A to form an acyl-CoA, and shortened by two
carbons in a series of reactions (Figure 6.24). Molecules of NADH and FADH
2
derived
from β-oxidation can enter the electron transport chain to produce ATP. Acetyl-CoA,
you will recall from earlier in this chapter, serves as the entry point into the TCA cycle.
When you consider that a single turn of the TCA cycle gives rise to the production of 14
molecules of ATP, you can appreciate what a rich source of energy a 16- or 18-carbon
fatty acid represents. The glycerol component of a lipid requires only slight modification
in order to enter the glycolytic pathway as dihydroxyacetone phosphate (see ‘Glycolysis’
above).
Proteins are a less useful source of energy than lipids or carbohydrates, but may be
utilised when these are in short supply. Like lipids, they are initially hydrolysed to their
constituent ‘building blocks’, in this case, amino acids. These then undergo the loss of
an amino group (deamination), resulting in a compound that is able to enter, either
directly or indirectly, the TCA cycle.
CH
3
CH
3
CCH COOH COO
–
+ NH
4
+
Alanine Pyruvate
NH
2
O
NH
2
O
−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−−→
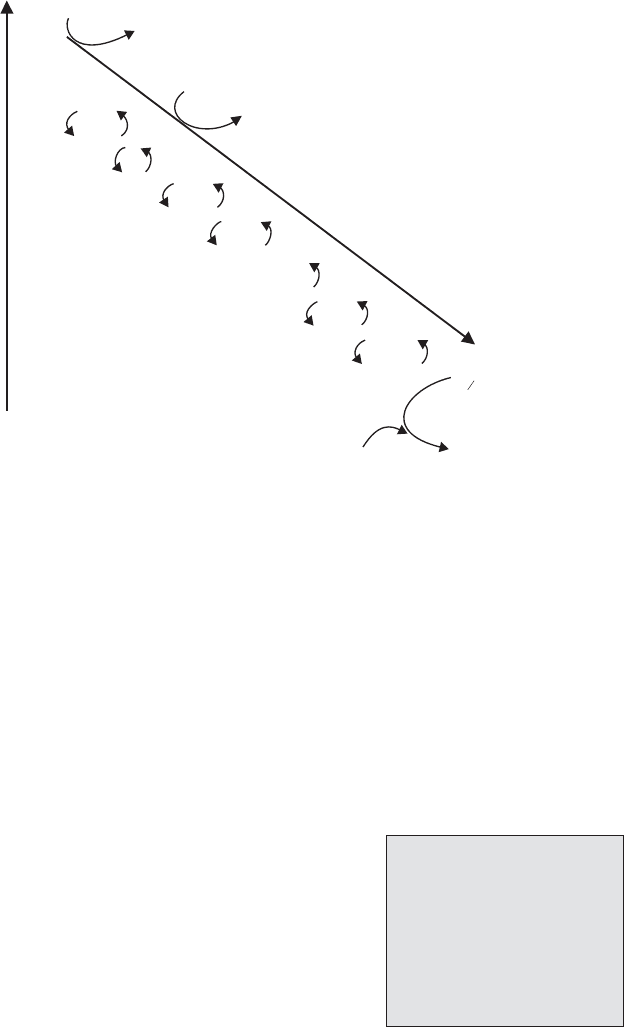
Figure 6.24 β-oxidation of lipids. β–Oxidation comprises a series of four reactions,
repeated for the removal of each two-carbon unit. Formation of the acyl CoA ester
requires the expenditure of ATP, but there is a net gain in reducing power (NADH + FADH
2
),
which can feed into the electron transport chain. The shortened acyl chain at the end of the
process can re-enter the cycle and become further shortened, while acetyl CoA can enter
directly into the TCA cycle (CoA-SH = coenzyme A)
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
PRINCIPLES OF ENERGY GENERATION 137
Oxidation
Oxidation
Cleavage
(thiolysis)
Hydration
ATP
AMP
P P 2 P
H
2
O
H
2
O
NAD
+
NADH + H
+
FAD
FADH
2
