Hogg S. Essential microbiology
Подождите немного. Документ загружается.

JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
148 MICROBIAL METABOLISM
–
+
e
–
P
870
*
P
870
Red/infrared light
ADP
+ Pi
ATP
NADP
+
+ H
+
NADPH
External electron
donors e.g. H
2
S,
Fe
2+
, succinate
ADP + Pi
ATP
Cyt
b
/
c1
Cyt
c2
Q
Bph
Redox
Potential
(V)
Reverse
electron
flow
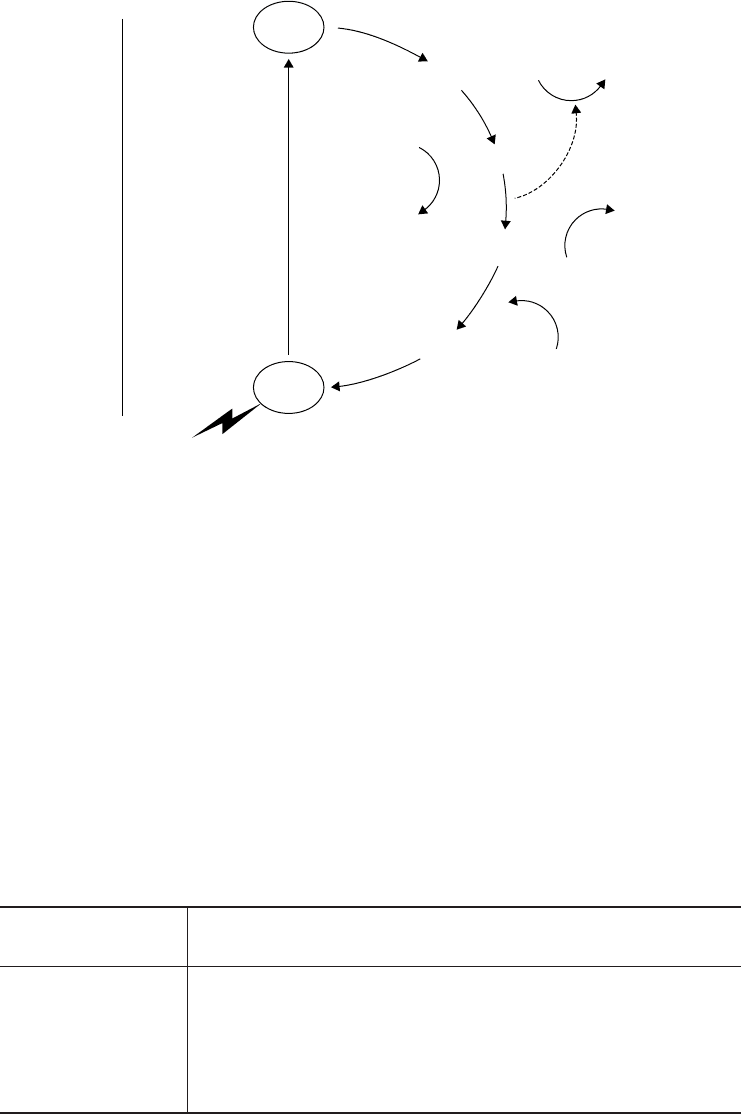
Figure 6.33 Electron flow in the anoxygenic photosynthesis of a purple bacterium. ATP is
generated by the passage of electrons down an electron transport chain back to the reaction
centre bacteriochlorophyll. Anoxygenic photosynthetic bacteria use molecules such as sul-
phur and hydrogen sulphide instead of water as external electron donors, hence no oxygen
is generated. NADPH for use in CO
2
fixation must be generated by reverse electron flow.
Bph = bacteriophaeophytin (bacteriochlorophyll a minus its magnesium atom)
Anabolic reactions
So far, in describing respiration and photosynthesis, we have considered those reactions
whereby a microorganism may generate cellular energy from its environment. As we
saw at the beginning of this chapter, one of the uses to which this may be put is the
synthesis of new cellular materials. In all of the pathways described in the following
Table 6.4 Comparison of anoxygenic photosynthetic bacteria
Green Green Purple Purple
sulphur non-sulphur sulphur non-sulphur
Bacteriochlorophyll c, d, ea, ca, ba, b
Electron donor H
2
S/reduced
S/H
2
Organic
compounds
H
2
S/reduced
S/H
2
Organic
compounds
CO
2
fixation Reverse
TCA cycle
Calvin cycle Calvin cycle Calvin cycle
Photoheterotrophy No Yes Some Mainly
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
ANABOLIC REACTIONS 149
section, the conversion of ATP to ADP is required at some point. The term biosynthesis
is used to describe those reactions by which nutrients are incorporated first into small
molecules such as amino acids and sugars and subsequently into biomacromolecules
such as proteins and polysaccharides.
Biosynthesis of carbohydrates
We have already seen in the preceding pages that autotrophic organisms (not necessarily
phototrophic ones) are able to incorporate inorganic carbon as CO
2
or HCO
3
−
into
hexose sugars, most commonly via the Calvin cycle. Heterotrophic organisms are unable
to do this, and must convert a range of organic compounds into glucose by a series of
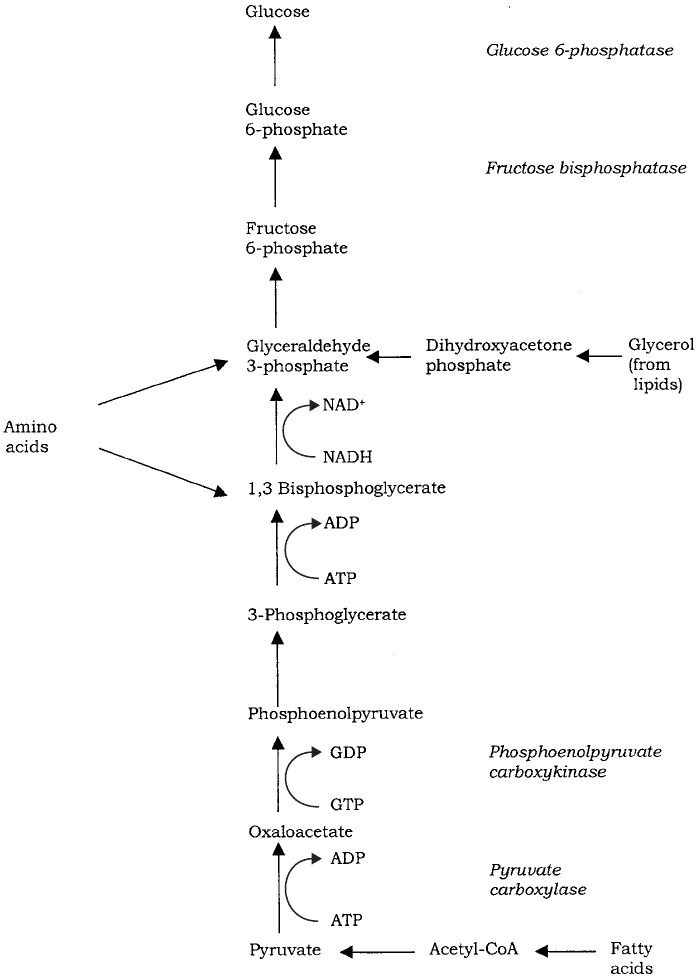
reactions called gluconeogenesis (Figure 6.34). Many compounds such as lactate or
certain amino acids can be converted to pyruvate directly or via other members of the
TCA cycle, and thence to glucose. To all intents and purposes, gluconeogenesis reverses
the steps of glycolysis (see above), although not all the enzymes involved are exactly
the same. This is because three of the reactions are essentially irreversible, so different
enzymes must be used to overcome this. These reactions are highlighted in Figure 6.34.
Once glucose or fructose has been produced, it can be converted to other hexose
sugars by simple rearrangement reactions. Building up these sugars into bigger carbo-
hydrates (polysaccharides) requires them to be in an energised form: this usually takes
the form of either an ADP- or UDP-sugar, and necessitates an input of energy. Pentose
sugars such as ribose are important in the synthesis of nucleotides for nucleic acids and
coenzymes (see below).
Biosynthesis of lipids
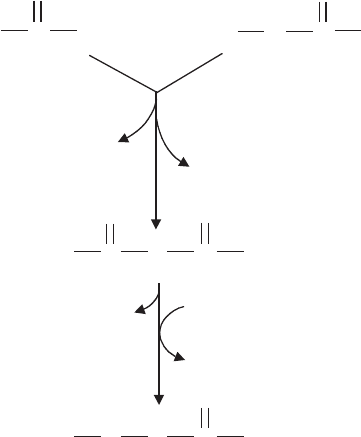
Fatty acids are synthesised by a stepwise process that involves the addition of two-
carbon units to form a chain, most commonly of 16–18 carbons. The starting point of
fatty acid metabolism is the two-carbon compound
The basic building blocks in the synthesis of fatty acids are acetyl-CoA (two-carbon)
and malonyl-CoA (three-carbon). We have encountered acetyl-CoA before, when dis-
cussing the TCA cycle; malonyl-CoA is formed by the carboxylation of acetyl-CoA.
Acetyl–CoA + CO
2
→ Malonyl–CoA
2 − carbon 3 − carbon
Carbon dioxide is essential for this step, but is not incorporated into the fatty acid as it is
removed in a subsequent decarboxylation step. In order to take part in the biosynthesis
of fatty acids, both molecules have their coenzyme A element replaced by an acyl carrier
protein (ACP). In a condensation reaction, one carbon is lost as CO
2
and one of the
ACPs is released. The resulting four-carbon molecule is reduced, with the involvement
of two NADPH molecules, to butyryl-ACP. This is then extended two carbon atoms at
a time by a series of further condensations with malonyl-ACP (Figure 6.35).
Thus, extending a fatty acid chain by two carbons involves the expenditure of one
ATP and two NADPH molecules. The overall equation for the synthesis of a 16-carbon
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
150 MICROBIAL METABOLISM
Figure 6.34 Gluconeogenesis. Non-carbohydrate precursors can feed into a pathway that
converts pyruvate to glucose in a series of reactions that are mostly the reverse of glycolysis.
Enzymes not found in glycolysis are shown in italics
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
ANABOLIC REACTIONS 151
Acetyl-ACP Malonyl-ACP
OO
H
3
C C ACP HOOC C C ACP
H
2
ACP
CO
2
O
O
H
3
C C C C ACP Acetoacetyl-ACP
H
2
2NADPH
H
2
O
2NADP
O
H
3
C C C C ACP Butyryl-ACP
H
2
H
2
Figure 6.35 Fatty acid biosynthesis. Acetyl- and malonyl-ACPs condense with the loss
of CO
2
to give a four-carbon molecule butyryl-ACP. The addition of further two-carbon
acetyl groups is achieved by re-entering the pathway and reacting with further molecules of
malonyl-ACP
fatty acid such as palmitic acid can be represented:
8 Acetyl-CoA + 7ATP+ 14 NADPH + 6H
+
−→ Palmitate + 14 NADP
+
+ 8 CoA +
6H
2
O + 7 ADP + 7Pi
Once formed, fatty acids may be incorporated into phospholipids, the major form of
lipid found in microbial cells. Recall from Chapter 2 that a phospholipid molecule has
three parts: fatty acid, glycerol and phosphate. These last two are provided in the form
of glycerol phosphate, which derives from the dihydroxyacetone phosphate of glycol-
ysis. Glycerol phosphate replaces the ACP of two fatty acid-ACP conjugates to yield
phosphatidic acid, an important precursor for a variety of membrane lipids. The energy
for this reaction is provided, unusually, not by ATP but by CTP (cytidine triphosphate).
Biosynthesis of nucleic acids
Most microorganisms are able to synthesise the purines and pyrimidines that comprise
the nitrogenous bases of DNA and RNA. These compounds are synthesised from a
number of sources in reactions that require an input of ATP. The contribution of different
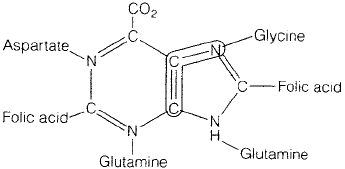
compounds towards the purine skeleton of guanine or adenine is shown in Figure 6.36.
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
152 MICROBIAL METABOLISM
Figure 6.36 Purine structure. Several precursors contribute towards the formation of the
purine base skeleton. The nitrogen atoms are donated by the amino acids glutamine, as-
partate and glycine. Note the important role played by folic acid. The antimicrobial agent
sulphonamide (Chapter 15) exerts its effect by inhibiting folic acid synthesis, which in turn
affects synthesis of purine nucleotides
The purines are not synthesised as free bases but are associated with ribose-5-phosphate
as complete nucleotides from the outset. Inosinic acid, which is formed initially, acts as
a precursor for the other purine nucleotides.
Pyrimidines have a similarly complex synthesis. The amino acids aspartate and glu-
tamine are involved in the synthesis of the precursor orotic acid. Note that unlike the
purines, the skeleton of pyrimidines is fully formed before association with the ribose-
5-phosphate moiety, which is itself derived from glucose (see biosynthesis of carbohy-
drates, above).
Ribonucleotides (as contained in RNA) are converted to deoxyribonucleotides (as
contained in DNA) by a reduction reaction that may involve vitamin B
12
acting as a
cofactor.
Biosynthesis of amino acids
A very limited number of microorganisms are able to utilise molecular nitrogen from
the atmosphere by incorporating it initially into ammonia and subsequently into or-
ganic compounds (see Chapter 7). Most organisms, however, need to have their ni-
trogen supplied as nitrate, nitrite, or ammonia itself. Ammonia can be incorporated
into organic nitrogen compounds in several ways, including glutamate formation from
α-ketoglutarate (see TCA cycle, above):
α-ketoglutarate + NH
4
+
+ NADPH* + H
+
−→ glutamate + NADP
+
+ H
2
O
(*Some species utilise NADH as their electron donor)
The amino group can subsequently be transferred from the glutamate to make other
amino acids by transamination reactions involving other keto acids:
e.g. glutamate + oxaloacetate −→ aspartate + α-ketoglutarate
Glutamate plays a central role in the biosynthesis of other amino acids, as it usually
donates the primary amino group of each:
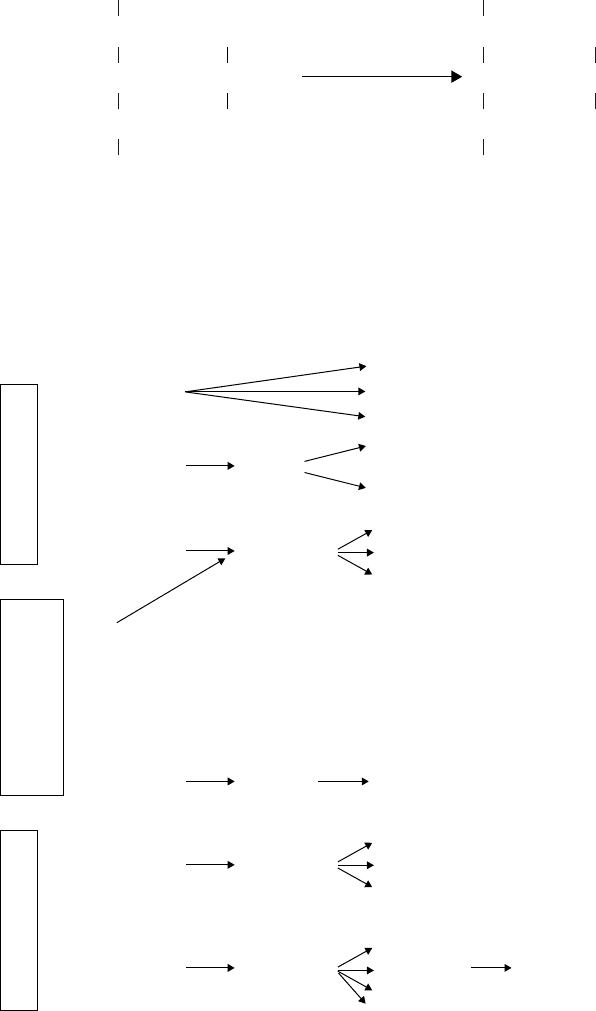
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
ANABOLIC REACTIONS 153
Glutamate
Pyruvic acid
CH
3
C=O
COOH
CH
2
COO
–
CH
2
H-C-NH
2
COOH
+
Alanine
H-C-NH
2
CH
3
COOH
+
α-Ketoglutarate
CH
2
COO
–
CH
2
C=O
COOH
According to the precursor molecule from which they derive, amino acids can be placed
into six ‘families’ (Figure 6.37). The precursors are intermediates in metabolic pathways
we have already encountered in this chapter, such as glycolysis or the TCA cycle. When
Pyruvate family
Pyruvate
3-Phospho-
glycerate
Alanine
Valine
Leucine
Serine Serine family
Aromatic family
Chorismate
Glycine
Cysteine
Phenylalanine
Tyrosine
Tryptophan
Phosphoenol-
pyruvate
Erythose 4-PO
4
Ribose 5-PO
4
GLYCOLYSISTCA CYCLE
Histidinol
Histidine
Lysine
Threonine
Isoleucine
Histidine family
Glutamate family
Glutamine
Arginine
Proline
Glutamate
Aspartate
Oxalo-
acetate
α-Keto-
glutarate
Aspartate
Aspartate family
Asparagine
PENTOSE PHOSPHATE
PATHWAY (see Fig.6.19)
Figure 6.37 Amino acid biosynthesis. The carbon skeleton of amino acids is obtained from
a limited number of precursor molecules, mainly intermediates in glycolysis or the TCA cycle.
The amino group originally derives from inorganic sources, but can then be transferred from
one organic molecule to another
JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
154 MICROBIAL METABOLISM
amino acids are broken down, they are likewise broken down into a handful of metabolic
intermediates, which then feed into the TCA cycle.
The regulation of metabolism
Microorganisms, like the rest of us, live in a changing world, and their needs do not
always remain the same. It would be highly inefficient and (frequently wasteful) if all
their metabolic reactions were going on with equal intensity all the time, regardless of
whether they were needed. Over evolutionary time, regulation systems have developed,
so that metabolism is tailored to the prevailing conditions.
Essentially, this regulation involves controlling the activity of enzymes which direct
the many biochemical reactions occurring in each cell. This can be done by:
r
directly affecting enzyme activity, or
r
indirectly, at the genetic level, by controlling the level at which enzymes are
synthesised.
Direct control of enzymatic activity occurs by the mechanism of feedback inhibition
(see Box 6.5), whereby the final product of a metabolic pathway acts as an inhibitor to
the enzyme that catalyses an early step (usually the first) in the pathway. It thus prevents
Box 6.5 Feedback inhibition
Biosynthetic pathways exist as a series of enzyme-mediated reactions, leading to
a final product required by the cell for structural or metabolic purposes. But what
happens if for some reason, the consumption of the final product slows down, or
even stops? Feedback inhibition, also known, perhaps more descriptively, as ‘end-
product inhibition’, ensures that excess amounts of the end product are not synthe-
sised. The pathways leading to the synthesis of many amino acids are regulated
in this way, for example isoleucine, which is synthesised from another amino acid,
threonine, via a series of intermediates:
Threonine Isoleucine
Here, the isoleucine itself acts as an inhibitor of threonine deaminase, the enzyme
which starts off the pathway. It does this by binding to an allosteric site on the enzyme,
distorting it and preventing its active site from binding to threonine. Note, that by
inhibiting the early part of the pathway, we not only prevent further production of
isoleucine but also unnecessary breakdown of threonine. When levels of isoleucine
starts to run low, less will be available to block the threonine deaminase, and thus
the pathway starts to function again.

JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
TEST YOURSELF 155
more of its own formation. When the concentration of the product subsequently falls
below a certain level, it is no longer inhibitory, and biosynthesis resumes.
Regulation of metabolic pathways can also be achieved by controlling whether or
not an enzyme is synthesised in the first place, and if so, the rate at which it is produced.
This is done at the DNA level, by one of two mechanisms, induction and repression,
which respectively ‘switch on’ and ‘switch off’ the machinery of protein synthesis dis-
cussed earlier in this chapter. These are discussed under the heading ‘Regulation of gene
expression’ in Chapter 11
Test yourself
1 Reactions which involve the breakdown of compounds are collectively
termed
and synthetic (building up) ones are termed .
2 Enzymes lower the
of a reaction.
3 The
of an enzyme is the part that is actually involved in
catalysis.
4 At pH values away from the optimum,
of ‘R’ groups causes denat-
uration of proteins.
5 When a molecule
an electron, it is said to be oxidised.
6 Compounds which are good electron donors have a strongly
redox
potential.
7 The energy ‘currency’ of cellular metabolism is
.
8 The first step in glucose metabolism is
, which can occur with or
without
.
9 ATP may be formed from ADP by
phosphorylation,
phosphorylation or phosphorylation.
10 Before entering the TCA cycle, pyruvate must be converted to the two-carbon
compound
. This type of reaction, in which a molecule of carbon
dioxide is removed, is called a
.
11 The main product of the TCA cycle is reducing power, in the form of
and .
12 The role of oxygen in aerobic respiration is to act as the
in the electron transport chain.

JWBK011-06 JWBK011-Hogg August 12, 2005 16:0 Char Count= 0
156 MICROBIAL METABOLISM
13 As envisaged by the chemiosmotic theory of Peter Mitchell, protons are
pumped out across a membrane, generating a
.
14
anaerobes are able to utilise anaerobic respiration if oxygen is
unavailable.
15 The two commonest products of fermentation reactions are
and
.
16 Fermentation yields far less ATP than aerobic respiration because it does not
involve an
.
17 Fatty acids are broken down
carbons at a time by the process of
.
18
are organisms that can derive their energy from the oxidation of
inorganic compounds such as ammonia or reduced iron.
19 Photosynthesis carried out by green and purple bacteria differs from that
carried out by algae and cyanobacteria in that no
is generated.
20 In the light reactions of photosynthesis, light energy and an electron donor
are used to generate
and .
21 In the dark reactions that make up the
cycle, carbon dioxide is
assimilated into glyceraldehyde-3-phosphate, which is then converted to glu-
cose in a series of steps that are the reverse of
.
22 Anoxygenic photosynthetic bacteria are unable to use
as an elec-
tron donor, and therefore use compounds such as hydrogen sulphide or suc-
cinate.
23 Organisms unable to synthesise glucose from inorganic carbon must derive
it from other organic compound by the process of
.
24 In amino acid synthesis, -NH
2
groups may be transferred between com-
pounds in
reactions.
25 The inhibition of a metabolic pathway by its final product is termed end-
product or
inhibition.
JWBK011-07 JWBK011-Hogg August 12, 2005 16:8 Char Count= 0
Part III
Microbial Diversity
157
