Hogg S. Essential microbiology
Подождите немного. Документ загружается.

JWBK011-12 JWBK011-Hogg August 12, 2005 20:21 Char Count= 0
318 MICROORGANISMS IN GENETIC ENGINEERING
V
ector
Donor DNA
RE digestion
Recombinant vector
Host bacterial cell V
ector replication
Bacterial replication
Clone of identical cells
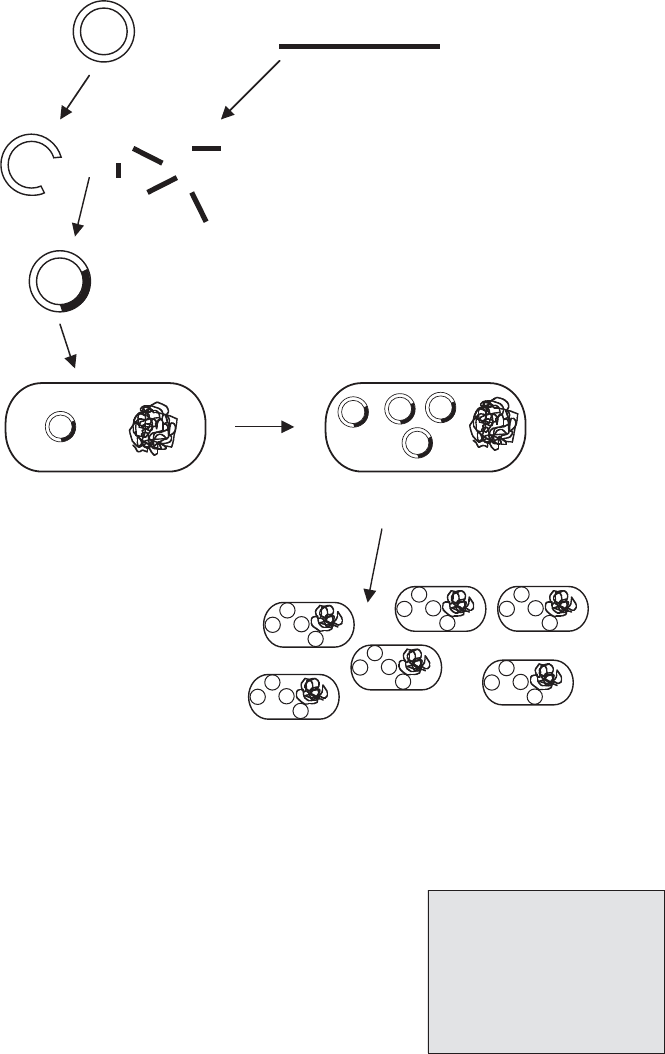
Figure 12.3 Gene cloning. DNA fragments obtained by restriction digestion can be spliced
into a similarly digested vector molecule and transformed into a host bacterial cell. See the
text for details
A selectable marker is
a gene that allows cells
containing it to be identi-
fied by the expression of
a recognisable charac-
teristic.
r
it must carry a selectable marker – since only a pro-
portion of host cells will take up the vector, there must
be a means of differentiating them from those that do
not. A common way to do this is to use a vector that
carries a gene that confers resistance to an antibiotic
such as ampicillin. When bacterial cells are plated out
on a medium containing the antibiotic, only those that
have taken up the vector will be able to form colonies.
(The host strain must, of course, normally be suscep-
tible to the antibiotic.)
JWBK011-12 JWBK011-Hogg August 12, 2005 20:21 Char Count= 0
PLASMID CLONING VECTORS 319
r
it must carry a single copy of RE restriction sites – in order to accommodate a piece
of donor DNA, a vector must be cut by a restriction endonuclease in one place only
(Figure 12.3).
Plasmid cloning vectors
A multiple cloning site
or polylinker is a region
of a cloning vector de-
signed to contain recog-
nition sequences for sev-
eral REs.
Two main types of vector system are used in cloning,
those that use plasmids and those that use bacterio-
phages (revisit Chapters 3 and 10, respectively, for a
reminder of the main features of these). Naturally occur-
ring examples of these are manipulated so that they pos-
sess the above properties. A popular vector in the early
days of gene cloning was the plasmid pBR322; Figure
12.4 shows how it contains the features described above.
The lacZ
gene actu-
ally only encodes a part
of the β-galactosidase
enzyme, called the α-
peptide. The strain of E.
coli used as host makes
an incomplete version of
the enzyme, which lacks
this portion. Only if the
cells contain the plasmid
with the lac Z’ gene can
they produce functional
β-galactosidase, by α-
complementation.
Let us consider now what happens, at a molecular
level, when donor DNA is ligated into a plasmid vector
(Figure 12.5). Sticky ends of the donor fragment form hy-
drogen bonds with the exposed compatible ends of the
opened up plasmid by complementary base pairing, and
DNA ligase consolidates the join. Many plasmids con-
tain engineered sequences called multiple cloning sites
(MCS); these provide additional flexibility with respect
to the restriction fragments that may be accommodated.
The ligation of insert DNA into the cloning site of
the plasmid is not the only possible outcome of the pro-
cedure described above, however. Unless experimental
conditions are carefully controlled (there are ways of
doing this), a more likely outcome is that the two com-
patible ends of the plasmid will simply ‘find’ each other
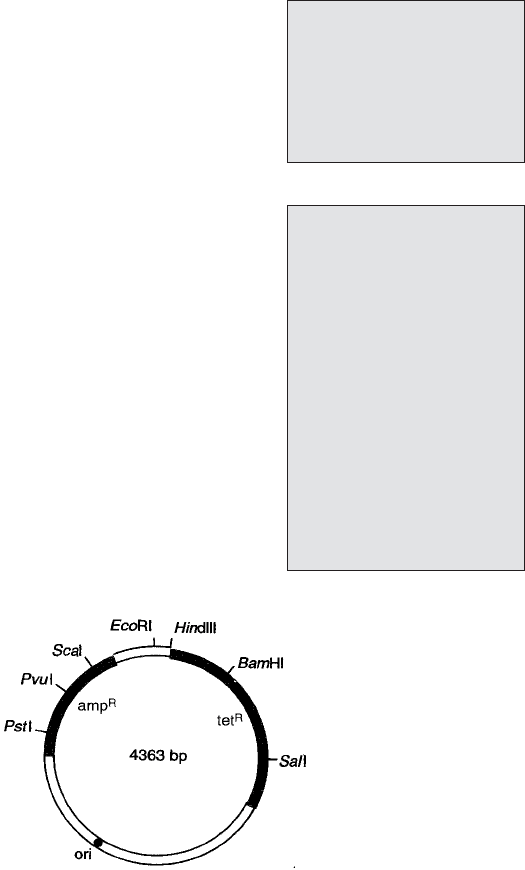
Figure 12.4 Plasmid pBR322. One of the earliest plasmid vectors, pBR322 illustrates the
major features required for use in gene cloning: an origin of replication (ori), selectable
markers (genes for resistance to ampicillin and tetracycline), and single recognition sites for
a number of REs
JWBK011-12 JWBK011-Hogg August 12, 2005 20:21 Char Count= 0
320 MICROORGANISMS IN GENETIC ENGINEERING
Recognition sites
for restriction
endonuclease (RE)
Digestion with
RE fragments
donor DNA and
opens up
vector
Donor
DNA
Formation of
recombinant vector
molecule. DNA
ligase consolidates
the join by repairing
the phosphate-sugar
b
ackbone.
..…G
..…CCTAG
GATCC…..
G…..
Compatible ‘sticky ends’
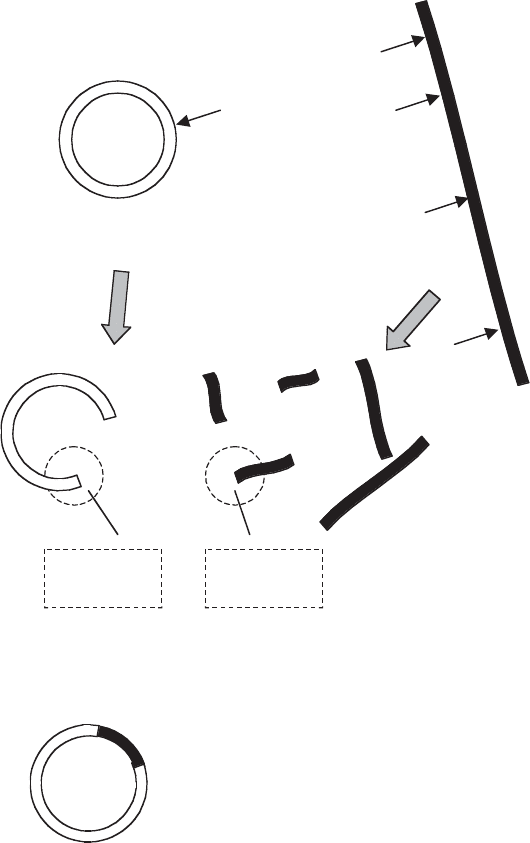
Figure 12.5 Formation of a recombinant plasmid. A recombinant plasmid is formed when
a fragment of foreign DNA is taken up and ligated into the plasmid. By cleaving both plasmid
and foreign DNA with the same RE, compatible ‘sticky’ ends are created, facilitating the
join. Treatment with the enzyme alkaline phosphatase prevents the cut plasmid ends rejoining
together, thereby favouring the formation of recombinant molecules
JWBK011-12 JWBK011-Hogg August 12, 2005 20:21 Char Count= 0
PLASMID CLONING VECTORS 321
pUC19
Fragment with
BamHI sticky
ends
ori
lacZ’amp
R
pUC19
BamH1
recognition
site
Inserted
fragment
Transform into E. coli cells
and plate onto medium
containing ampicillin and
Xgal (artificial substrate
for β-galactosidase)
Recombinant colony
(white)
Non-recombinant
colony (blue)
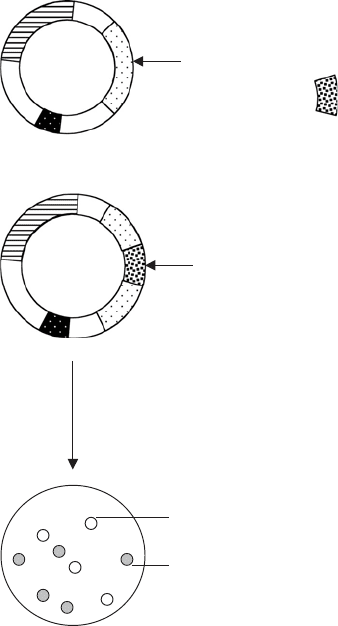
Figure 12.6 Recombinant plasmids can be detected by insertional inactivation. Insertion
of foreign DNA is carried out at a site within one of the selectable markers, thus interrupting
its gene sequence. Here, a fragment has been inserted into the BamHI site situated within
the gene that codes for β-galactosidase. Bacteria transformed with such a plasmid will not
produce the functional enzyme, and so can be distinguished from those carrying plasmids
with no inserted DNA
again, and rejoin. Since a certain amount of this is inevitable, how are we able to
tell the difference between those bacteria that contain a recombinant plasmid (one
containing a piece of donor DNA) and those that have taken up a recircularised ‘native’
plasmid? Since both types will contain the gene for ampicillin resistance, we cannot
distinguish them by this means. A strategy commonly used to get around the problem
is insertional inactivation (Figure 12.6). This clever ploy exploits the fact that we can
manipulate DNA, and, for example, insert RE recognition sequences at desired points.
If a recognition site occurs in the middle of a gene sequence, and a piece of foreign
DNA is inserted at this position, the gene will be interrupted, and unable to produce
a functional gene product. In the example shown, the gene is lacZ
, necessary for the
successful expression of the enzyme β-galactosidase. This will only be expressed in
those bacteria that contain plasmids in which the gene has remained uninterrupted,
JWBK011-12 JWBK011-Hogg August 12, 2005 20:21 Char Count= 0
322 MICROORGANISMS IN GENETIC ENGINEERING
i.e. those that have not taken up an insert. Expression of the β-galactosidase can be
detected by growing the bacteria on an artificial substrate, which is converted to a
coloured (usually blue) product when acted on by the enzyme. Those cells that contain
recombinant plasmids are easily identified because disruption of the lacZ
gene means
that no β-galactosidase is produced, resulting in non-pigmented (white) colonies.
One problem remains. Remember that our inserted DNA was derived from the di-
gestion of total (genomic) DNA from the donor organism; this means that our DNA
library will contain recombinant plasmids with a whole range of fragments from that
digestion, and not just the specific one that interests us. How are we able to distinguish
this fragment from the others?
A technique called nucleic acid hybridisation is used to solve the problem. This once
again depends on complementary base pairing, and involves the creation of a probe,
a short length of single-stranded DNA that is complementary to part of the desired
‘target’ sequence, and therefore able to seek it out. If searching for the right clone can
be likened to looking for a needle in a haystack, then the probe is a powerful ‘magnet’
that makes the task much easier. The probe carries a tag or label, so that its location
can be identified (Figure 12.7).
Once we have identified the clone of bacteria containing plasmids with the insert that
interests us, we can grow a pure culture of it and then isolate plasmid DNA. Using the
same RE as before, the inserted donor DNA can be removed and purified. We now have
enough of this specific DNA sequence (a tiny proportion of the donor organism’s total
genome) to analyse and manipulate.
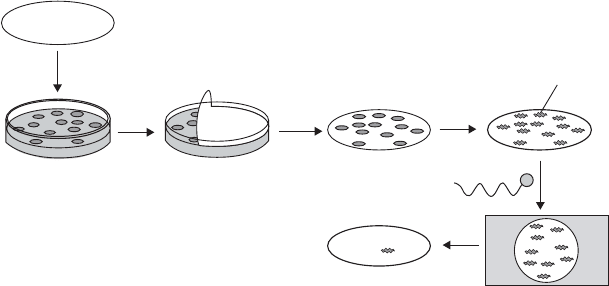
Nitrocellulose
Petri dish with colonies
of bacteria containing
recombinant plasmids
Peel sheet to
produce replica
of colonies
Lyse bacteria
and denature
DNA
DNA bound
Incubate
with probe
and wash
Expose
to film
a) b)
c)
d)
Figure 12.7 Colony probing. (a) A replica of the bacterial colonies is made using a nitro-
cellulose membrane. (b) Alkali treatment lyses the cells and denatures the DNA, making it
single stranded. (c) Following a period of incubation with a radiolabelled probe to allow
hybridisation to take place, the membrane is washed to remove any unbound radioactivity.
(d) Position of bound radioactivity revealed by autoradiography. Comparison with original
master plate reveals the location of colonies carrying specific target sequence. Alternative
detection systems such as biotin–streptavidin that avoid the use of radioactivity have be-
come much more commonly used in recent years. From Reece, RJ: Analysis of Genes and
Genomes, John Wiley & Sons, 2003. Reproduced by permission of the publishers

JWBK011-12 JWBK011-Hogg August 12, 2005 20:21 Char Count= 0
BACTERIOPHAGES AS CLONING VECTORS 323
Although plasmids are easily isolated and manipulated, their use as cloning vectors
is limited by the fact that they tend to become unstable if we attempt to insert much
more than about 5kb of foreign DNA. For inserts larger than this, we must turn to other
vector systems.
Bacteriophages as cloning vectors
The most commonly used bacteriophage vectors are those based on phage Lambda (λ)
(Figure 12.8). The genome of λ is 48.5kb in length; it was the first genome to have its
entire sequence determined (1982). As drawn conventionally, the genome is linear, and
contains 46 genes. At each 5
end of the double-stranded structure is a 12 base single-
stranded sequence known as a cos site; these have a complementary base sequence (i.e.
they are cohesive, or ‘sticky’ ends). They can join together to form a double-stranded,
circular λ molecule, a conformation that is essential for insertion and integration into
the host genome (see Chapter 10).
The naturally occurring phage is unsuitable as a vector, because being relatively
large, it contains multiple copies of recognition sites for a number of REs. By using
genetic manipulation techniques, however, unwanted sites can be removed, allowing
the DNA to be ‘opened up’ at a single location, and fragments of foreign DNA with
complementary sticky ends to be ligated.
A further limitation that must be overcome is that in this form, phage λ can only
accommodate about another 3kb of DNA. This is because if the genome exceeds 52kb,
it cannot be packaged properly into the protein head to produce viable phage particles.
The arrangement of genes on the phage λ genome offers a solution to this problem.
It is known that genes encoding specific functions are clustered together, with genes
necessary for the lytic replication cycle being found at the ends of the map as it is
conventionally drawn (Figure 12.8). It is possible, therefore, to remove much of the
central part of the λ genome without affecting its ability to infect and lyse its bacterial
host.
Insertion vectors have had some of this non-essential material removed, reducing their
genome size to around 42kb, and thus allowing them to carry an insert of up to 10kb. A
single-copy restriction site is opened up, and a fragment with complementary sticky ends
inserted with the aid of DNA ligase. Vectors such as λZAPII have a multiple cloning site
or polylinker, positioned so that it falls within the lacZ
gene. This allows insertional
Tail Head Recombination cIII N cI cro cII DNA replication Q Lysis
Non-essential genes
Figure 12.8 Bacteriophage λ. The central region of the λ genome contains genes involved
in its lysogenic cycle. This region can therefore be excised, without affecting the ability of
the phage to infect E. coli via the lytic cycle. The genes at the right-hand end of the genome
as shown have a regulatory function, whilst the structural genes are situated at the left
JWBK011-12 JWBK011-Hogg August 12, 2005 20:21 Char Count= 0
324 MICROORGANISMS IN GENETIC ENGINEERING
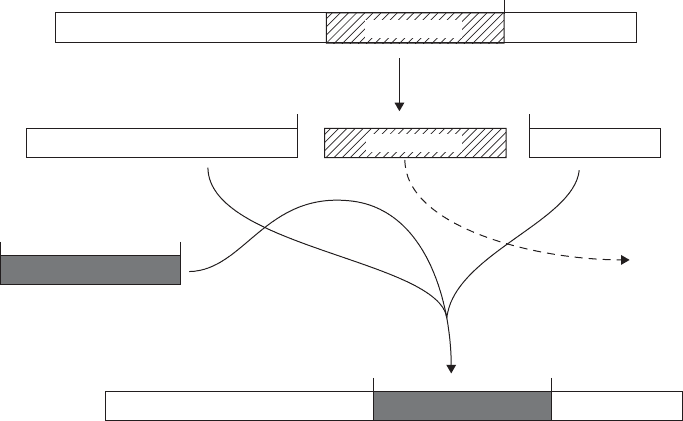
cos cos
BamH1
Cut with
BamH1
BamH1
BamH1 BamH1
BamH1 BamH1
BamH1
BamH1
Left arm 20 kb
Left arm 20 kb Right arm 9 kb
Left arm 20 kb
Insert
Recombinant
phage DNA
Ligate
Discarded
EMBL4Right arm 9 kb
Right arm 9 kb
Stuffer 14 kb
Stuffer 14 kb
Figure 12.9 Cloning in bacteriophage λ. The removal of non-essential genes in the centre
of the genome allows the incorporation of large fragments of foreign DNA. From Dale,
JW and von Schantz, M: From Genes to Genomes: Concepts and Applications of DNA
Technology, John Wiley & Sons, 2002. Reproduced by permission of the publishers
inactivation with blue/white selection to be used for the detection of recombinants as
described above for pUC plasmids.
Replacement vectors (Figure 12.9) are able to accommodate larger inserts, because
instead of a single copy of a particular RE site, they have two, one situated either side
of a disposable central section known as the ‘stuffer’ fragment. This can be removed by
digestion with the appropriate RE and replaced by insert DNA. The vector λEMBL3,
which can accommodate inserts of between 9 and 23kb, has two polylinker sequences
containing sites for SalI, BamHI and EcoRI flanking the stuffer fragment, allowing a
variety of restriction fragments to be inserted.
Recombinants in replacement vector systems can be detected by a method that ex-
ploits the limitations in the phage’s ability to package DNA. Just as too big a genome
cannot be packaged (see above), neither can one that is too small (<37kb); consequently,
constructs lacking an insert will not result in the formation of plaques.
Another naturally occurring bacteriophage of E. coli that has been adapted for use
as a cloning vector is phage M13. This is a single-stranded phage that for part of
its replication cycle inside the host cell exists as a double-stranded replicative form,
which can be isolated and manipulated like a plasmid. Systems based on M13 have
proved popular as a means of obtaining DNA in a single-stranded form for applications
such as DNA sequencing using the dideoxy (Sanger) method. M13 vectors contain a
multiple cloning site situated within the lacZ
gene, allowing blue/white selection of
recombinants. The cloning capacity is generally limited to fragments of less than 1.5kb,

JWBK011-12 JWBK011-Hogg August 12, 2005 20:21 Char Count= 0
BACTERIOPHAGES AS CLONING VECTORS 325
Box 12.1 A large number of clones are required to cover
an entire genome
The factors that determine the number of clones necessary to make a genomic
library are the size of the genome and the average size of each insert. In addition
the probability that a given fragment will be present in the library must be set: the
higher the probability, the more clones will be necessary.
N =
log(1 − P)
log(1 − a/b)
Where: N = number of clones required; a = average insert size; b = total genome
size; P = probability that a given fragment will be present.
e.g. Number of clones required for a 95% probability of a given fragment to be
present in a genomic library of E. coli (genome size 4.8 × 10
6
) with an average insert
size of 20kb:
N = log(1 − 0.95)/ log(1 − 4.167 × 10
−3
)
= log 0.05/ log(0.9958)
= 712 clones
This may seem a manageable number, but remember that the genome of E. coli is
very small compared to that of higher organisms (Homo sapiens:3× 10
9
bp).
but hybrid M13/plasmid vectors (phagemids) have been developed that are able to take
inserts of as much as 10kb.
As we have seen, a large collection of cloned DNA fragments is called a DNA library. If
the original donor DNA comprised the entire genome of an organism, then the collection
of clones, which between them contain an entire donor genome, is known as a genomic
library. We could create a library of the entire E. coli genome (total size 4.8 × 10
6
bp)
for example, by having just over 700 clones with an average insert size of 20kb (see
Box 12.1 for calculation). For a more complex genome, however, we would either need
to have very many more clones, or to increase the average fragment size.
For this reason, further types of cloning vectors have been developed, to allow the
cloning of larger fragments. Cosmids are entirely man-made creations, and have no
naturally occurring counterpart. They incorporate beneficial features of both plasmids
and phage vectors, and may be capable of accommodating insert fragments of more than
45kb. Cosmids are essentially plasmids that contain the cos sites from λ phage, with
the plasmid supplying the necessary origin of replication, restriction sites and selectable
marker. As we’ve seen, λ DNA will be packaged into phage heads if the cos sites are
37–52kb apart; however the only parts of the DNA recognised by the packaging enzymes
are the cos sites, so any DNA can be used to fill the intervening sequence.
The recombinant DNA is packaged into phage particles by a process called in vitro
packaging, and introduced into the host E. coli. Lacking phage genes, cosmids do not
lead to lysis of the host cells and plaque production, but are instead replicated as if
they were plasmids. Growth of host cells on a selective medium allows the detection of

JWBK011-12 JWBK011-Hogg August 12, 2005 20:21 Char Count= 0
326 MICROORGANISMS IN GENETIC ENGINEERING
transformants, i.e. those that have taken up the cosmid. There is no need to select for
recombinants, because non-recombinants are too small to be packaged into the phage
heads.
Expression vectors
Sometimes, the aim of a cloning experiment is not just to obtain large amounts of a
specific gene, but for the gene to be expressed. This involves using the host cell as a
sort of ‘factory’, to manufacture the specific protein encoded by the cloned gene. One
of the earliest applications of genetic engineering was the production of human insulin
in E. coli (Figure 12.10). Insulin is needed in considerable quantities for the treat-
ment of diabetics; for years it was obtained from the pancreas of pigs and cattle, but
this had several disadvantages including immunological complications and the risk of
viral contamination. Insulin generated by recombinant means is free from these prob-
lems. Many proteins can now be produced in this way by microorganisms at a rate
several times that of the normal host cell. In order for a gene to be expressed, it must
have specific nucleotide sequences around it that act as signals for the host cell’s tran-
scription/translation machinery (promoter, ribosomal binding site and terminator – see
Chapter 11). Since these sequences differ between, say, humans and E. coli, the bacte-
rial RNA polymerase will not recognise the human sequences, and therefore, although
a human gene may be cloned in E. coli using a simple vector, it will not be expressed. If,
however, the human gene could be inserted so that it was under the control of the E. coli
expression signals, then transcription should take place. Specially designed vectors that
provide these signals are called expression vectors. The choice of promoter sequence is
particularly important; often, a strong (i.e. very efficient) promoter is selected, so as to
maximise the amount of protein product obtained. Genes whose protein products are
naturally synthesised in abundance are likely to have such promoters. It is often helpful
to be able to regulate gene expression; inducible promoters can be switched on and
off by the presence of certain substances. The lac promoter (which controls the lacZ
gene) is an example of this. Cassette vectors have promoter, ribosomal binding site and
terminator sequences clustered together as a discrete unit, with a single recognition site
for one or more REs being situated downstream of the promoter (Figure 12.11).
The small size of the insulin molecule (and gene) and the size of the potential market
made it a prime early candidate for production by recombinant DNA technology. Most
insulin used in the treatment of diabetes nowadays is produced in this way. Systems
based on E. coli have also been used to synthesise other small human proteins with
The polypeptide pro-
ducts of translation in
eucaryotes may require
post-translational modifi-
cation before functional
protein is produced. Ex-
amples include phos-
phorylation, acetylation
and glycosylation.
therapeutic potential such as human growth hormone,
γ -interferon and tumour necrosis factor (TNF).
Bacteria, however, are not suitable host cells for the
production of many other human proteins such as tis-
sue plasminogen activator (TPA) or blood clotting Fac-
tor VIII, due to the size and complexity of their genes.
This is because many proteins of complex eucaryotes
are subject to post-translational modifications; this does
not occur in procaryotes, so bacteria such as E. coli are
not equipped with the cellular machinery to make the
necessary modifications to human proteins.
JWBK011-12 JWBK011-Hogg August 12, 2005 20:21 Char Count= 0
EXPRESSION VECTORS 327
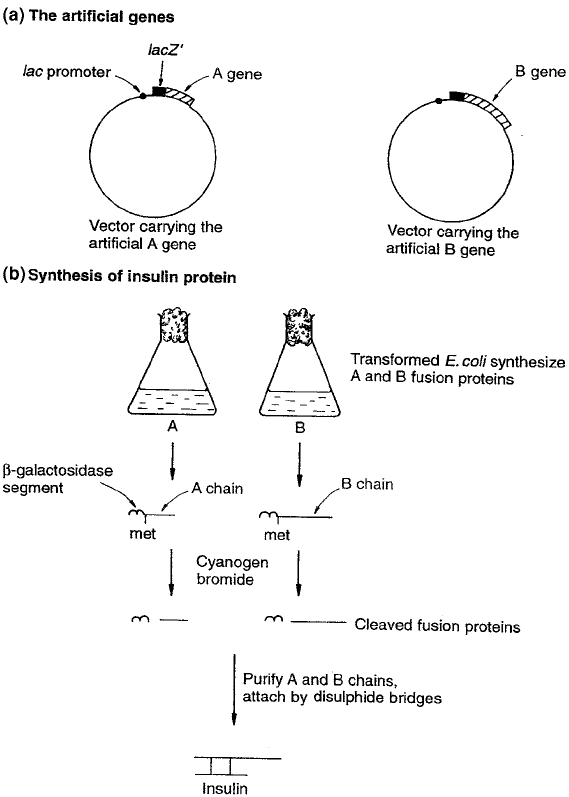
Figure 12.10 Production of recombinant human insulin. Human insulin comprises two
short peptide chains, 21 and 30 amino acid residues in length. Because they are so short,
the nucleotide sequence of their genes can be predicted from their amino acid sequence, and
synthetic genes produced. Each is cloned under the influence of the lac promoter and down-
stream of part of the lacZ
gene. By being produced as fusion proteins with β-galactosidase,
the insulin chains are protected against being degraded by the E. coli cell. From Brown, TA:
Gene Cloning, 3rd edn, Chapman & Hall, 1995. Reproduced by permission of Blackwell
Publishing Ltd.
