Klipp E., Herwig R., Kowald A., Wierling C., Lehrach H. Systems Biology in Practice: Concepts, Implementation and Application
Подождите немного. Документ загружается.


by ultrasound treatment or intensive vortexing. However, this results in random
fragments, which are not useful for further processing.
This situation began to change when the first restriction endonucleases were iso-
lated from bacteria at the end of the 1960s. These enzymes recognize specific short
sequences of DNA and cut the molecule only at these positions. At first sight it
seems surprising that organisms contain enzymes that cut DNA into small pieces.
But it turns out that restriction enzymes are part of a bacterial defense system
against bacteriophages (prokaryote-specific viruses). This system consists of two
components. The first part, the restriction enzymes, cuts double-stranded DNA at
their recognition sequences. These sequences are typically between four and eight
base pairs long. The second component, the methylases, modifies DNA molecules at
specific sequences by adding methyl groups to the nucleotides of the target se-
quence. In order to work, a pair consisting of a methylase and a restriction enzyme
has to recognize the same sequence stretch. The DNA of the bacterium is methy-
lated by the methylase, and this protects it against the nuclease activity of the restric-
tion enzyme. However, the DNA of a phage that enters the cell is not methylated
and hence is degraded by the restriction enzyme.
Over the years several hundred different restriction enzymes have been isolated
from different bacterial strains with many different recognition sequences. Restric-
tion enzymes can cut the double helix in three different ways, as depicted by
Fig. 4.1. Some produce blunt ends, but others cut the DNA in a staggered way, re-
sulting in short stretches (here 4 bp) of single-stranded DNA. Depending on the
way the enzyme cuts the DNA, the single-stranded overhang can have either a 5'
end (if cut by Bam HI) or a 3' end (if cut by Kpn I). Because these overhanging sin-
gle strands can base pair with other complementary single strands, they are also
called sticky ends.
The following table provides an overview of some of the available restriction en-
zymes, the organism from which they have been isolated, and their recognition se-
quence. Restriction enzymes are normally named after the organism and strain in
which they were discovered. Eco RI, for instance, originates from Escherichia coli
strain RY13. The final Roman numeral indicates the order in which the enzymes
were isolated from single bacteria (i.e., Eco RI vs. Eco RV). If different enzymes re-
cognize the same sequence but produce different cuts, they are called isoschizomers.
110
4 Experimental Techniques in a Nutshell
Sma I
(blunt ends)
GGG
GGGCCC
CCC
5'
3'
3'
5'
Bam HI
(5' sticky ends)
CCT
CCTGGA
AGG
5'
3'
3'
5'
Kpn I
(3' sticky ends)
CCA
CCAGGT
TGG
5'
3'
3'
5'
Fig. 4.1 Restriction enzymes recognize short stretches of DNA that
often have a palindromic structure. The enzyme then cuts the DNA
in one of three ways, producing either blunt ends or sticky ends. This
behavior is shown here for the enzymes Sma I, Bam HI, and Kpn I.
The arrow indicates where the enzymes cut the double helix.

For instance, the enzymes Sma I and Xma I in the table are isoschizomers. Finally,
the “N” in the recognition sequence for Sau96 I indicates that it does not matter
which nucleotide is at this position; the enzyme will accept all of them.
Name Organism Recognition sequence
Alu I Arthrobacter luteus 5' AG: CT3'
Bam HI Bacillus amyloliquefaciens 5' G : GATCC3'
Bgl II Bacillus globigii 5' A : GATCT3'
Eco RI Escherichia coli 5' G : AATTC3'
Eco RV Escherichia coli 5' GAT: ATC3'
Hind III Haemophilus influenzae 5' A : AGCTT3'
Kpn I Klebsiella pneumonia 5' GGTAC: C3'
Pme I Pseudomonas mendocina 5' GTTT: AAAC3'
Sau96 I Staphylococcus aureus 5' G : GNCC3'
Sma I Serratia marcescens 5' CCC: GGG3'
Xma I Xanthomonas malvacearum 5' C : CCGGG3'
Why are restriction enzymes so useful? They generate reproducibly specific frag-
ments from large DNA molecules. This is a very important advantage over the ran-
dom fragments that can be generated by shear forces. If the restriction fragments
that result from an enzymatic digestion are separated according to size, they form a
specific pattern that represents a fingerprint of the digested DNA. Changes in this
fingerprint indicate that the number or position of the recognition sites of the used
restriction enzyme has changed. Restriction enzyme patterns can therefore be used
to characterize mutational changes or to compare orthologous genes from different
organisms.
The size separation of digested DNA is also a prerequisite to isolate and clone spe-
cific fragments. If the sequence of the DNA is known, the number and size of the re-
striction fragments for a given restriction enzyme can be predicted. By choosing the
right enzyme from the large number of available restriction enzymes, it is often pos-
sible to produce a fragment that contains the gene, or region of DNA, of interest.
This fragment can then be separated from the others and cloned into a vector (see
Section 4.1.2) for further investigation.
Electrophoresis is one of the most convenient and most often used methods of
molecular genetics to separate molecules that differ in size or charge. Many different
forms of electrophoresis exist, but they all work by applying an electrical field to the
charged molecules. Depending on the type of charge, the molecules will move to-
wards one of the two electrodes. Since each nucleotide of DNA (or RNA) carries a ne-
gative charge, nucleic acids move from the anode to the cathode. The separation is
carried out in a gel matrix to prevent convection currents and to present a barrier to
the moving molecules, which causes a sieving effect. The size, charge, and shape of
the molecules decide how fast they move through the gel. Generally it holds that the
smaller the molecule, the faster it moves. The pore size of the gel controls the size
range of the DNA fragments that can be separated successfully. For a typical restric-
111
4.1 Elementary Techniques
tion fragment, i.e., between 0.5 kb and 20 kb, agarose gels are used. Agarose is a lin-
ear polysaccharide that is extracted from seaweed. Agarose gels are not suited for
DNA fragments smaller than 500 bp. In this case, polyacrylamide gels that have
smaller pores are used. For such small molecules size differences of a single base
pair can be detected. Another problem are very large DNA molecules that are comple-
tely retarded by the gel matrix. Those fragments are not separated by the usual type
of electrophoresis. In this case, a special type of electrophoresis, the so-called pulse
field electrophoresis, can be used, which allows the separation of DNA molecules of
up to 10
7
bp. This technique varies the direction of the electric field periodically, so
that the molecules follow a zigzag path through the gel. Very long DNA fragments
move head-on through the gel, which results in a velocity that is independent of size.
Because of the oscillating field, the molecules have to re-orientate themselves. This
is easier for the smaller fragments, and the larger ones therefore lag behind. A typi-
cal application of this technique is the separation of whole chromosomes of microor-
ganisms (mammalian chromosomes are too large even for this technique).
Whatever type of electrophoresis or gel is used, the DNA is invisible unless it is
specially labeled or stained. A commonly used dye for staining is ethidium bromide,
which intercalates between DNA bases. In the intercalated state, ethidium exposed
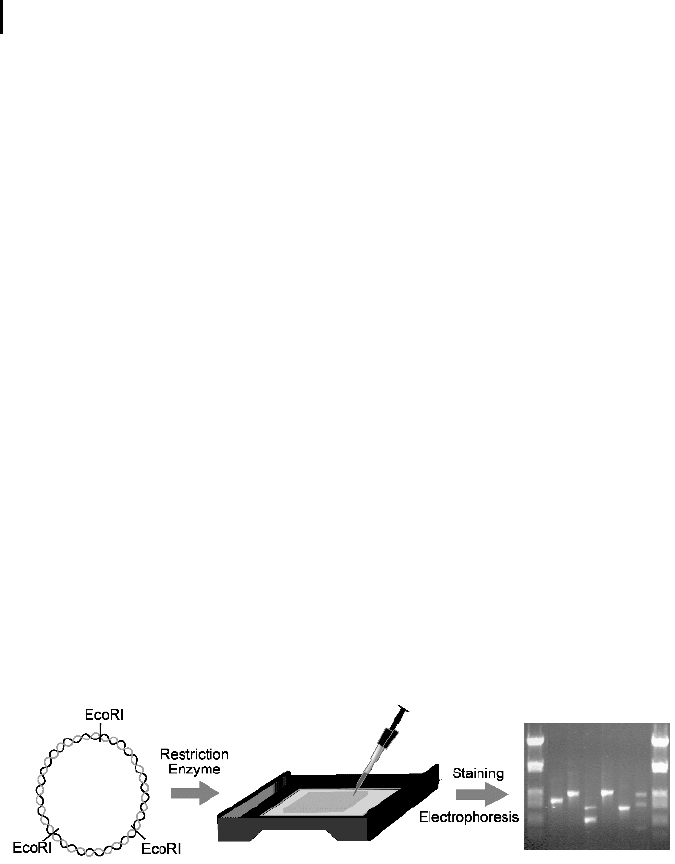
to UV light fluoresces a bright orange. Figure 4.2 sketches the different processing
steps from the DNA to the size-separated restriction fragments on an agarose gel.
On the gel (Fig. 4.2, right) different lanes can be seen, where different DNA probes
are separated in parallel. The concentrated solution of DNA fragments is filled in
the pockets at the top of the gel, and the fragments migrate during electrophoresis
to the bottom. The smallest fragments move fastest and appear at the bottom of the
gel. The lanes on the left and right sides contain fragments of known length that
serve as size markers.
112
4 Experimental Techniques in a Nutshell
Fig. 4.2 Agarose gel electrophoresis of DNA
restriction fragments. A plasmid containing
several recognition sites for a restriction enzyme
(here, Eco RI) is digested with the enzyme and
the resulting fragments are placed on an agarose
gel (middle). An applied electrical field moves the
charged molecules through the gel (here, from
top to bottom) and separates them according to
size. After staining, the individual fragments ap-
pear under UV light as bright bands (right).
(Courtesy of Dr. P. Weingarten, Protagen AG.)

4.1.2
Cloning Vectors and DNA Libraries
In the last section we discussed how restriction enzymes generate DNA fragments by
cutting the DNA at short, specific recognition sites. However, this is not the end of the
cloning procedure. In this section we will see how the generated fragments can be
used to generate billions of identical copies. But first we should clarify what is meant
by cloning, because, unfortunately, the terms clone and cloning have multiple mean-
ings. To create a clone of an organism means to generate a copy of an organism such
that both individuals are genetically identical. The first and most famous example of
such a clone generated from another adult organism is the sheep Dolly (Wilmut et al.
1997). Another example is of course monozygotic twins,which are natural clones. Sur-
prisingly, rough calculations show that currently approximately 60 million human
clones exist (Colletto et al. 2003)! However, most often the term cloning is used in a
different context, meaning to create an identical copy of a DNA molecule or to isolate
a specific DNA fragment from the total DNA content of a cell. In the remainder of this
chapter and the whole book, it is this second meaning that we are referring to.
For the actual cloning (amplification) step, a restriction fragment has to be in-
serted into a self-replicating genetic element. This can be, for instance, a virus or a
plasmid. Plasmids are small circular rings of DNA that occur naturally in many bac-
teria. They often carry a few genes for resistance to antibiotics or to enable the degra-
dation of unusual carbon sources and are normally only a few thousand base pairs
long (Fig. 4.3). Genetic elements that are used in the laboratory to amplify DNA frag-
ments of interest are called cloning vectors and the amplified DNA is said to be
cloned. In the following we will concentrate on the use of plasmids as vectors. The
actual insertion process requires that the DNA to be cloned and the vector be cut
with the same restriction enzyme and that the vector has only one recognition site
for this enzyme. This creates a linearized plasmid that has the same type of sticky
ends as the DNA to be cloned. If the linearized vector and the digested DNA are now
mixed at the right concentration and temperature, the complementary sticky ends
base pair and form a new recombinant DNA molecule. Initially, the resulting mole-
cule is held together only by hydrogen bonds. This is made permanent by adding
the enzyme DNA ligase that forms covalent bonds between the phosphodiester back-
bones of the DNA molecules. This procedure enables the combination of DNA from
arbitrary sources. It is in principle no problem to clone mammalian or plant DNA
into bacterial vectors, because the genetic code is universal (with a few exceptions in
mitochondrial DNA).
Finally, the vector is introduced into bacterial cells, which are then grown in cul-
ture. Every time the bacteria double (approximately every 30 min), the recombinant
plasmids also double. Each milliliter of the growth medium can eventually contain
up to 10
7
bacteria, an immense amplification! The actual process of introducing the
vector into the bacteria is called transformation. For this end the cells are specially
treated so that they are temporarily permeable for the DNA molecules.
But loss occurs at all steps of this genetic engineering. Not all vector molecules
will have received an insert, because it is possible that the sticky ends of some vectors
113
4.1 Elementary Techniques
114
4 Experimental Techniques in a Nutshell

self-ligate without insert. Furthermore, not all bacteria used in the transformation
step will have received a vector molecule. It can therefore be that there is only a small
proportion of cells that contain a vector with insert in the growing cell population.
There are different strategies to cope with this problem, but normally one tries to
make use of genes in the original vector that can be used as selection markers. Fig-
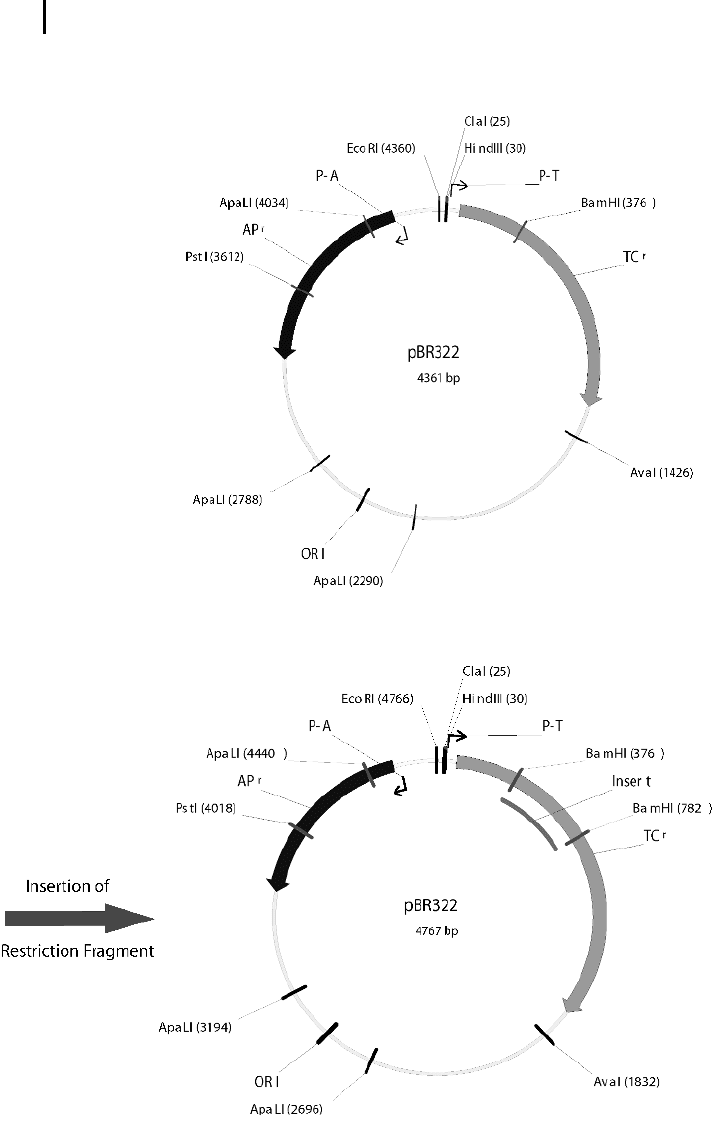
ure 4.3 shows pBR322, a typical cloning vector. Apart from a DNA sequence that en-
ables the cell machinery to replicate the plasmid (ori), it also contains two genes for
resistance against the antibiotics ampicillin and tetracycline. If the DNA fragment is
cloned into a restriction site that lies within one of the resistance genes, e.g., the
Bam HI site, simple selection steps can be used to end up with cells that contain the
desired construct. For this purpose the bacteria are grown in a medium that contains
ampicillin so that only cells that carry the plasmid can survive. The next step is more
complicated since we are interested in all those bacteria that contain a vector with a
nonfunctional tetracycline gene (caused by an insert). The cells are plated in high di-
lution on the surface of an agar plate, where each individual cell forms a colony. After
the colonies become visible, some bacteria of each colony are copied onto a second
agar plate by a stamping technique (which preserves the spatial arrangement of colo-
nies). This second plate contains tetracycline; therefore, only those cells with an in-
tact resistance gene can grow. By comparing the colony pattern of the two plates, it is
now possible to identify those colonies that exist on the first plate but not on the sec-
ond. These are the colonies that we are interested in.
For many years plasmid vectors have been used very successfully. However, there
is an upper size limit for the DNA one can clone into such a vector. Above 10 kb, the
cloning efficiency declines so much that other vectors are required. The following ta-
ble lists some of the types of vectors currently used. Lambda is a linear bacteriophage
of approximately 48 kb, and up to 20 kb of the original phage DNA can be replaced
by foreign DNA. Cosmids are artificial constructs that combine some features of the
phage lambda and of the classical plasmids. The advantage is that fragments up to
45 kb can be cloned. For really large fragments of up to one million base pairs, yeast
artificial chromosomes (YACs) have been developed (Burke et al. 1987), which are
now gradually being replaced by bacterial artificial chromosomes (BACs) (Shizuya
et al. 1992). BACs are based on the naturally occurring F-plasmid, which itself is
around 100 kb in length. While the copy number per cell of most smaller plasmids
is rather large, the F-plasmid and the derived BACs are maintained at only one to
two copies per cell. This reduces the risk of unwanted recombination events between
different copies and contributes to the stability of such large inserts.
115
4.1 Elementary Techniques
3 Fig. 4.3 pBR322 is a circular plasmid of 4.3 kb
that is often used as cloning vector. The diagram
on the top shows several important genetic ele-
ments of the plasmid. ORI marks a region of
DNA that controls the replication of the plas-
mid; it is the origin of replication. The boxes
represent genes that confer resistance to the
plasmid for the antibiotics ampicillin (APr) and
tetracycline (TCr). P-A and P-R are the promoters
of the resistance genes, and the lines and text
mark recognition sites for the corresponding
restriction enzyme. The lower part shows
pBR322 after a restriction fragment has been
inserted into the Bam HI restriction site.

Vector Maximum insert size Required number of clones in library
of Arabidopsis thaliana (115.4 Mb)
Plasmid 10 kb 53,141
Phage lambda 20 kb 26,570
Cosmid 45 kb 11,800
BAC Approx. 1 Mb 530
YAC Approx. 1 Mb 530
Because the average fragment size of restriction enzymes is much smaller than
1 Mb, the enzyme reaction is allowed to proceed for only a very short time. This time
is not long enough to cut all recognition sites, and therefore the resulting fragments
are much longer. This technique is called a partial digest.
So far we have discussed the situation when we want to clone a specific fragment
after a restriction digest. The DNA is separated on an agarose gel, and the desired
fragment is excised from the gel and cloned into an appropriate vector. However, of-
ten the situation is different insofar as we do not know in advance the fragment that
contains our gene of interest. In this case we can construct a so-called DNA library
by simply cloning all fragments that result from a digest into vectors. Such a library
is maintained in a population of bacteria that now contain vectors with many differ-
ent inserts. Bacteria with different inserts are either kept together or separated, so
that the library consists of thousands of clones (each kept in a separate plastic tube),
each carrying a specific fragment. This is, for instance, important for the construc-
tion of DNA chips (cf. Section 4.2.2). This strategy is also known as shotgun cloning.
To be sure that the library does contain the fragment of interest, it has to cover the
complete genome of the organism we are interested in. Because by chance some
fragments can occur more frequently than others in the library, more clones than ex-
pected are needed. The number of clones required to have a probability of P that a
particular fragment is represented by a clone in the library depends on the insert
size, I, and the genome size, GS, and is given by the following equation (Clarke and
Carbon 1976). The number of required clones in the above table has been calculated
for a probability of 99%.
N
ln 1 P
ln 1
I
GS
(4-1)
There are two basic types of DNA libraries that are extensively used in molecular
genetics. The first type is the genomic DNA library, which is exactly what we have de-
scribed so far. This type of library is directly created from the genetic material of an
organism. Restriction enzymes cut DNA regardless of the start and end points of
genes, and hence there is no guarantee that the gene of interest completely fits on a
single clone. Furthermore, in Chapter 2 we have seen that the genome of most
higher organisms contains large amounts of junk DNA. These sequences also end
up in the genomic DNA library and increase the number of required clones.
116
4 Experimental Techniques in a Nutshell

A different type of library, the cDNA library, circumvents these problems. This
technique does not use DNA as source material, but starts from the mRNA pool of
the cells or tissue of interest. The trick is that the mRNA molecules are a copy of ex-
actly those parts of the genome that are the most interesting. They represent the cod-
ing regions of the genes and contain neither introns nor inter-gene junk DNA.
Using the enzyme reverse transcriptase that exists in some viruses, mRNA can be
converted into cDNA. The resulting DNA is called complementary DNA (cDNA) be-
cause it is complementary to the mRNA. cDNA libraries differ from genomic
libraries in several important points: (1) they contain only coding regions; (2) they
are tissue-specific since they represent a snapshot of the current gene expression pat-
tern; and (3) because they are an image of the expression pattern, the frequency of
specific clones in the library is an indicator of the expression level of the correspond-
ing gene. cDNA libraries have many different applications. By sequencing cDNA
libraries, it is possible to experimentally determine the intron-exon boundaries of
eukaryotic genes. Constructing cDNA libraries from different tissues helps us to
understand which genes are expressed in which parts of the body. A derivative of the
cDNA library is the expression library. This type of library is constructed in such a
way that it contains a strong promoter in front of the cloned cDNAs. This makes it
possible not only to amplify the DNA of interest but also to synthesize the protein
that is encoded by this DNA insert. This technique is important especially for pro-
teins that are normally expressed only in very small amounts
4.1.3
1D and 2D Protein Gels
The basic principle of electrophoresis works for all charged molecules. This means
not only nucleic acids but also other kinds of cellular macromolecules, such as pro-
teins, can be separated by electrophoresis. But the distribution of charges in a typical
protein is quite different from the distribution in nucleic acids. DNA molecules carry
a negative charge that is proportional to the length of the DNA, since the overall
charge is controlled by the phosphodiester backbone. The net charge of proteins,
however, varies from protein to protein since it depends on the amount and type of
charged amino acids that are incorporated into the polypeptide chain. If proteins are
separated in this native form, their velocity is controlled by a difficult-to-predict func-
tion of charge, size, and shape.
It was a major improvement when Shapiro et al. (1967) introduced the strong de-
tergent sodium dodecyl sulfate (SDS) to protein electrophoresis in the 1960s. SDS
has a hydrophilic sulfate group and a hydrophobic part that binds to the hydrophobic
backbone of polypeptides. This has important consequences: (1) the negative charge
of the protein/detergent complex is now proportional to the protein size because the
number of SDS molecules that bind to a protein is proportional to the number of its
amino acids, (2) all proteins denature and adopt a linear conformation, and (3) even
very hydrophobic, normally insoluble proteins can be separated by gel electrophor-
esis. Under these conditions the separation of proteins is reduced to a function of
their size, as in the case of nucleic acids. Small proteins travel quickly, while large
117
4.1 Elementary Techniques
proteins are more strongly retained by the gel matrix. Images of protein gels are nor-
mally presented such that the large polypeptides are at the top and smaller ones at
the bottom.
For proteins, a different gel matrix is used than for nucleic acids. Acrylamide
monomers are polymerized to give a polyacrylamide gel. During the polymerization
step, the degree of cross-linking and thus the pore size of the network can be con-
trolled to be optimal for the size range of interest. Proteins often contain sulfide
bridges that connect either different parts of the same polypeptide or different pep-
tide chains of a multi-subunit protein complex. Therefore, in addition to SDS, a re-
ducing substance such as mercaptoethanol is often added, which reduces the sulfide
bridges to sulfhydryl groups. This linearizes single peptides and separates multi-
subunit complexes into the individual proteins. Over the years, SDS polyacrylamide
gel electrophoresis (SDS-PAGE) has become an easy-to-use standard technique for
separating proteins by size. As in the case of DNA, the gel has to be stained to make
the protein bands visible. The most frequently used dye is Coomassie blue, which
can detect quantities of proteins down to 100 ng. A more sensitive technique is so-
called silver staining, which can detect 5–10 ng of protein. However, Coomassie
blue staining is much simpler to perform and is therefore often the technique of
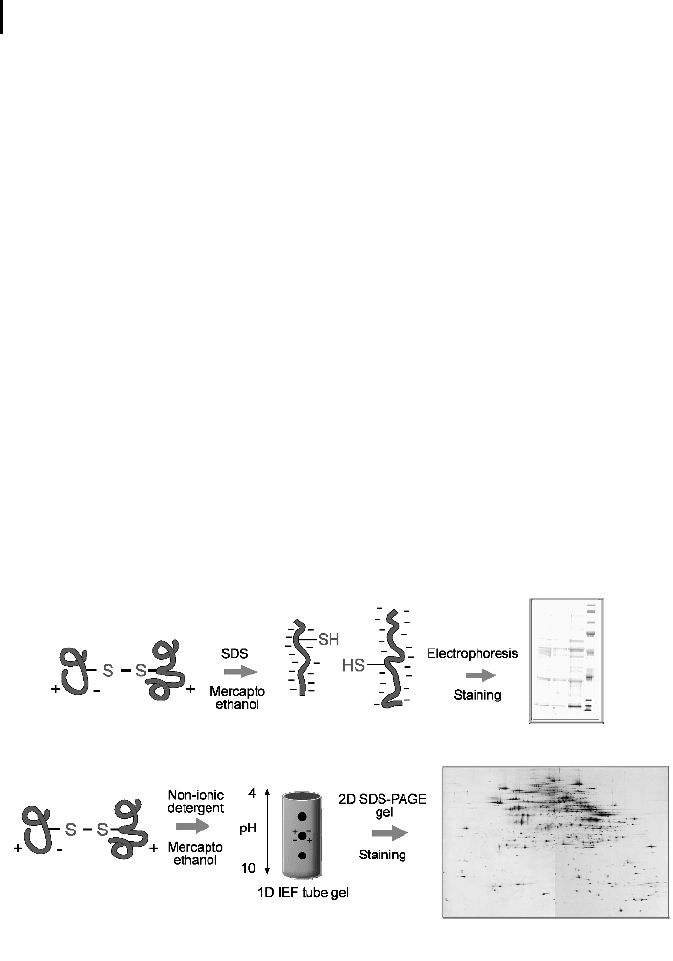
choice. Figure 4.4 top sketches the basic steps required for SDS-PAGE. In this ex-
ample, the outermost lanes contain protein size markers (large proteins at the top,
small ones at the bottom), and the middle lanes contain different samples of inter-
est.
118
4 Experimental Techniques in a Nutshell
Fig. 4.4 (Top) SDS-PAGE: Native proteins are
treated with the negatively charged detergent
SDS and the reducing agent mercaptoethanol to
break up disulfide bridges and unfold the pro-
tein. After this treatment even extremely hydro-
phobic proteins can be separated on a polyacry-
lamide gel according to their size. (Courtesy of
Dr. P. Weingarten, Protagen AG.) (Bottom) 2D
gel electrophoresis: For the first dimension, pro-
teins are separated in a tube gel according to
their isoelectric point. To preserve the native
charge of the proteins, a nonionic detergent is
used to unfold the polypeptides. For the second
dimension, the tube gel is placed on top of a
standard SDS-PAGE slab gel and the proteins
are now separated by size. Up to 2000 proteins
can be separated with this technique. (Courtesy
of Dr. L. Mao and Prof. J. Klose, Charité Berlin.)

On the gel shown in Fig. 4.4 top, only a few protein bands can be seen. This will
be the case after several protein purification steps. However, a cell or subcellular frac-
tion contains hundreds or thousands of different proteins. If such a mixture is used
for SDS-PAGE, individual bands overlap and proteins cannot be separated clearly.
The solution to this problem is the two-dimensional polyacrylamide gel electrophor-
esis (O’Farrell 1975). The idea is to separate the proteins in a second dimension ac-
cording to a property other than size.
Isoelectric focusing (IEF) is such a separation technique. The net charge of a pro-
tein depends on the number of charged amino acids, but also on the pH of the med-
ium. At a very low pH, the carboxy groups of aspartate and glutamate are uncharged
(–COOH), while the amino groups of lysine and arginine are fully ionized (–NH
3
+
),
conferring a positive net charge to the protein. At a very basic pH, by contrast, the
carboxy groups are charged (–COO
–
) and the amino groups are neutral (–NH
2
), re-
sulting in a negative net charge. Accordingly, for each protein a pH exists that results
in an equal amount of negative and positive charges. This is the isoelectric point of
the protein, at which it has no net charge. For isoelectric focusing, the proteins are
treated with a nonionic detergent so that the proteins unfold but retain their native
charge distribution (Fig. 4.4 bottom). Then they are placed onto a rod-like tube gel,
which has been prepared such that it has a pH gradient from one end to the other.
After a voltage is applied, the proteins travel until they reach the pH that corre-
sponds to their isoelectric point.
For the second dimension, the tube gel is soaked in SDS and then placed on top
of a normal SDS slab gel. A voltage is applied perpendicular to the direction of the
first dimension and the proteins are now separated according to size. The result is a
two dimensional (2D) distribution of proteins in the gel, as shown in Fig. 4.4 bottom.
This technique makes it possible to separate all proteins of a typical prokaryote in a
single experiment!
4.1.4
Hybridization and Blotting Techniques
Hybridization techniques are based on the specific recognition of a probe and target
molecule. The aim is to use such techniques to detect and visualize only those mole-
cules in a complex mixture that are of interest to the researcher. The base pairing of
complementary single-stranded nucleic acids is the source of specificity for Southern
blotting, Northern blotting, and in situ hybridization, which are described in the fol-
lowing sections. A short fragment of DNA, the probe, is labeled in such a way that it
can later easily be visualized. Originally, radioactive labeling was used, but in recent
years this has often been replaced by fluorescent labels. The probe is incubated with
the target sample, and after the recognition of probe and target molecules is com-
pleted, the location of the probe shows the location and existence of the sought-after
target molecule. In principle 16 nucleotides are sufficient to ensure that the se-
quence is unique in a typical mammalian genome (4
16
&4.29710
9
), but in practice
much longer probes are used. The Western blot is not a hybridization technique,
since it is not based on the formation of double-stranded DNA, RNA, or DNA/RNA
119
4.1 Elementary Techniques
