Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.


Using Eq. 7.1.3, the needed reactor volume is
V
R
¼ tv
0
t
cr
¼ 100:8L (f)
c. If the variation in the volumetric flow rate along the reactor is not considered
(D ¼ 0), the design equation is
dZ
dt
¼ 1 Z (g)
We solve (g) by separating the variables and integrating. The solution is
Z(t) ¼ 1 e
t
For Z
out
¼ 0.8
t ¼
ð
0:8
0
1
1 Z
dZ ¼ 1:61 (h)
Hence, the calculated reactor volume is
V
R
¼ tv
0
t
cr
¼ 67:18 L (i)
Note that by ignoring the effect of the volume expansion, we specify a reactor
volume that is only 68% of the required volume.
d. For a reactor with a volume of 67.1 L, t ¼ 1.61. Using the proper design
equation, (e), the outlet extent, Z
out
, is determined by
ð
Z
out
0
1 þ Z
1 Z
dZ ¼ 1:61 ( j)
The solution is Z
out
¼ f
A
out
¼ 0:680. Hence, if the wrongly specified reactor
volume is used, only 68% conversion is obtained.
e. To attain 80% conversion on the 67.1-L reactor, the feed flow rate should be
reduced. From the design equation (e), t ¼ 2.42. Using Eq. 7.1.3 to deter-
mine the feed rate,
v
0
¼
V
R
tt
cr
¼ 166:3L=min (k)
(F
tot
)
0
¼ v
0
C
0
¼ 6:65 mol=min
Hence, when the wrongly specified reactor volume is used, the feed flow
rate should be lowered by about 34% to maintain the specified conversion
level of 80%.
250 PLUG-FLOW REACTOR
Example 7.2 The second-order, gas-phase chemical reaction
2A ! B
is carried out in a 1000 L isothermal plug-flow reactor. A feed stream consisting
of 80% A and 20% inert (% mole) is fed to the reactor at a molar flow rate of
125 mol/min. The concentration of reactant A in the feed stream is
C
A
in
¼ 0:05 mol=L. The molar fraction of reactant A at the outlet of the reactor
is 0.16. We want to modify the reactor such that it provides 90% conversion of
A. What is the required additional volume?
Solution The stoichiometric coefficients of the chemical reaction are
s
A
¼2 s
B
¼ 1 s
I
¼ 0 D ¼1
In this case, the reaction rate constant is not provided, and we have to determine
it from the operating data. First, we derive the design equation for this case. We
select the inlet stream to the system as the reference stream. Hence,
(F
tot
)
0
¼ (F
tot
)
in
, y
A
0
¼ 0:8, y
I
0
¼ 0:2. The reference concentration is
C
0
¼
C
A
0
y
A
0
¼ 0:0625 mol=L
and the volumetric flow rate of the reference stream is
v
0
¼
(F
tot
)
0
C
0
¼ 2000 L=min
For a gas-phase reaction, the species concentration is given by Eq. 7.2.6, and the
rate expression is
r ¼ kC
0
2
y
A
0
þ s
A
Z
1 þ D Z
2
(a)
Using Eq. 3.5.4, for a second-order reaction, the characteristic reaction time, t
cr
,is
t
cr
¼
1
kC
0
(b)
Substituting (a) and (b) into Eq. 7.2.2, the design equation is
dZ
dt
¼
0:8 2Z
1 Z
2
(c)
7.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 251

Separating the variables, the design equation for the reactor is
t ¼
ð
Z
out
0
1 Z
0:8 2Z
2
dZ (d)
where t is the dimensionless space time of the reactor, defined by Eq. 7.1.3. To
determine Z
out
, we use Eq. 2.7.8 and Eq. 2.7.10 to express the molar fraction of
reactant A at the reactor outlet:
y
A
out
;
F
A
out
F
tot
out
¼
0:8 2Z
out
1 Z
out
¼ 0:16 (e)
Solving (e), Z
out
¼ 0.348. Substituting this value into (d), we obtain t ¼ 2.2.
Using Eq. 7.1.3, the characteristic reaction time is
t
cr
¼
V
R
v
0
t
¼ 0:2278 min ( f)
Using (b), the reaction rate constant is
k ¼
1
C
0
t
cr
¼ 70:39 L =mol min
Now we design a reactor to provide 90% conversion. Using Eq. 2.6.5,
Z
out
¼
y
A
in
s
A
f
A
out
¼
0:8
2
(0:9) ¼ 0:36
Substituting in (d),
t
new
¼
ð
0:36
0
1 Z
0:8 2Z
2
dZ ¼ 2:8 (g)
The required reactor volume is
V
R
new
¼ v
0
t
cr
t ¼ (2000 L=min)(0:2278 min)(2:8) ¼ 1275 L (h)
An additional 275 L should be added to the reactor.
Example 7.3 The elementary, gas-phase reaction
A þ B ! C
252 PLUG-FLOW REACTOR

is carried out in an isothermal plug-flow reactor operated at 2 atm and 1708C. At
this temperature, the reaction rate constant is k ¼ 90 L/(mol min), and the vapor
pressure of the product, C, is 0.3 atm. The reactor is fed with two gas streams: the
first one consists of 80% A, 10% B, 10% inert (I), and is at 2.5 atm and 1508C;
the second consists of 80% B, 20% I, and is at 3 atm and 1808C. The first stream
is fed at a rate of 100 mol/min and the second at a rate of 120 mol/min.
Determine:
a. The conversion of reactant A when C begins to condense.
b. The reactor volume where C starts to condense.
c. The reactor volume needed for 85% conversion of A.
Solution In the first section of the reactor, all the species are gaseous, and the
reaction is
A(g) þ B(g) ! C(g)
and its stoichiometric coefficients are
s
A
¼1 s
B
¼1 s
C
¼ 1 D ¼1
First, we have to select a reference stream. Since the two streams are mixed at the
reactor inlet, we select a fictitious reference stream that consists of the molar flow
rates of the two streams, and is at 2 atm and 1708C (the reactor operating con-
ditions). Hence,
F
A
0
¼ 0:8F
1
¼ 80 mol=min
F
B
0
¼ 0:1F
1
þ 0: 8F
2
¼ 106 mol=min
F
I
0
¼ 0:1F
1
þ 0: 2F
2
¼ 34 mol=min
(F
tot
)
0
¼ F
A
0
þ F
B
0
þ F
I
0
¼ 220 mol=min
The composition of the reference stream is y
A
0
¼ 0:364, y
B
0
¼ 0:482, and
y
I
0
¼ 0:154. Assuming ideal-gas behavior, the concentration of the reference
stream is
C
0
¼
P
0
RT
0
¼
2 atm
(0:08206 L atm=mol K)(443 K)
¼ 0:055 mol=L
Using Eq. 7.1.14, the volumetric flow rate of the reference stream is
v
0
¼
(F
tot
)
0
C
0
¼ 4000 L=min
7.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 253
a. Species C starts to condense when its partial pressure in the reactor
is 0.3 atm. At a given point in the reactor, the partial pressure of product
Cis
P
C
¼ y
C
P ¼
F
C
F
tot
P
Using Eqs. 2.7.8 and 2.7.10,
P
C
¼
y
C
0
þ s
C
Z
1 þ D Z
P ¼
Z
1 Z
(2 atm) ¼ 0:3 atm (a)
Solving (a), Z ¼ 0.130, and using Eq. 2.6.5, the conversion is
f
A
¼
s
A
y
A
0
Z ¼ 0:358 (b)
b. To determine the reactor volume for Z ¼ 0.130, we use Eq. 7.2.2,
dZ
dt
¼ r
t
cr
C
0
(c)
Since the reaction is elementary, r ¼ kC
A
C
B
, and, using Eq. 7.2.6,
r ¼ kC
0
2
( y
A
0
Z)( y
B
0
Z)
(1 þ D Z)
2
(d)
Using Eq. 3.5.4, for a second-order reaction, the characteristic reaction
time is
t
cr
¼
1
kC
0
¼ 0:202 min (e)
Substituting (d) and (e) into (c), the design equation reduces to
dZ
dt
¼
(0:364 Z)(0:482 Z)
(1 Z)
2
(f)
254 PLUG-FLOW REACTOR

To determine the dimensionless space time for Z ¼ 0.130, we solve (f) by
separation of variables,
t ¼
ð
0:130
0
(1 Z)
2
(0:364 Z)(0:482 Z)
dZ ¼ 0:9306 (g)
Using Eq. 7.1.3, the volume of the reactor for Z ¼ 0.130 is
V
R
¼ tv
0
t
cr
¼ (0:9306)(4000 L=min)(0:202 min) ¼ 751:9L
c. Any product C generated after dimensionless extent of 0.130 is reached
cannot be in the vapor phase, as shown in Figure E7.3.1. Hence, the follow-
ing reaction takes place downstream in the reactor:
A(g) þ B(g) ! C(liq)
For this reaction, D
gas
¼ 22. For Z . 0.130, a portion of product C is in the
gas phase and a portion in the liquid phase (with negligible volume). The
total molar flow rate of the gas-phase is now (for Z . 0.130)
(F
tot
)
gas
¼ (F
tot
)
0
[1 þ (1)0:130 þ (2)(Z 0:130)]
¼ (F
tot
)
0
(1:13 2Z) (h)
Using Eq. 7.2.6, for Z . 0.130, the concentrations of the two reactants are
C
A
¼ C
0
0:364 Z
1:13 2Z
C
B
¼ C
0
0:482 Z
1:13 2Z
(i)
Substituting (i) and (d) into (c), for Z . 0.130, the design equation is
dZ
dt
¼
(0:364 Z)(0:482 Z)
(1:13 2Z)
2
for Z . 0:13 ( j)
Figure E7.3.1 Zones of product C.
7.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 255
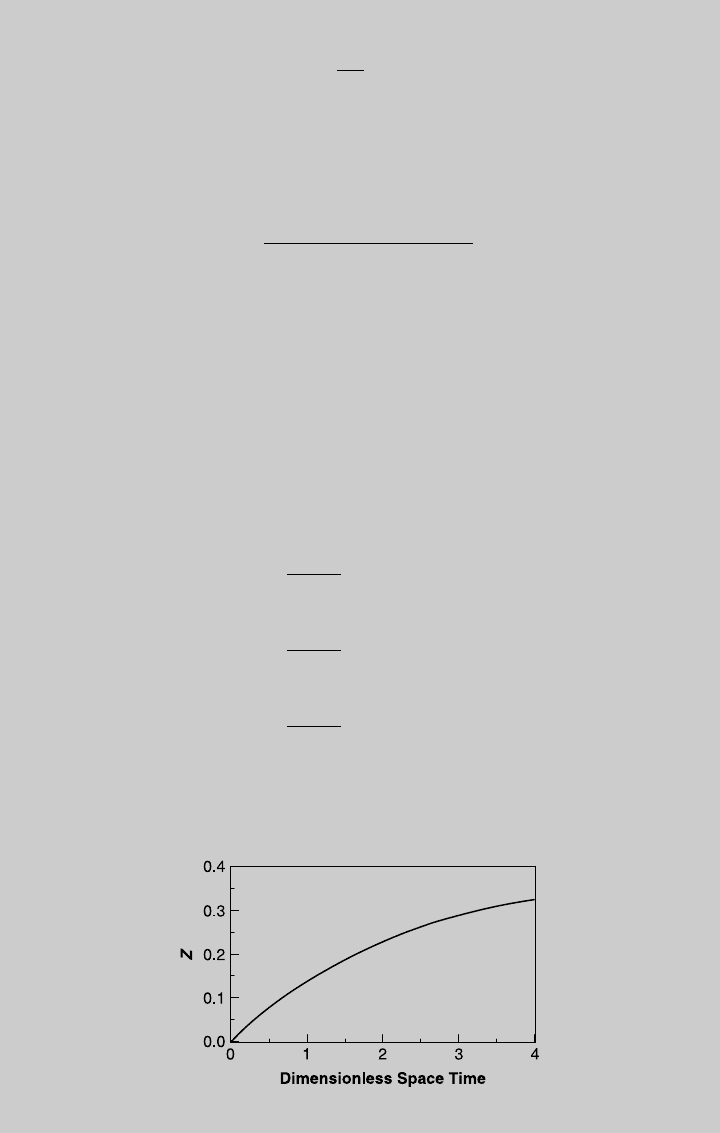
Using Eq. 2.6.5, for f
A
¼ 0.85,
Z ¼
y
A
0
s
A
f
A
¼ 0:3094
To determine the dimensionless space time of the reactor section
where product C is formed as a liquid, we integrate ( j) between 0.130 and
0.3094:
t ¼
ð
0:3094
0:130
(1:13 2Z)
2
(0:364 Z)(0:482 Z)
dZ ¼ 2:52 (k)
Using Eq. 7.1.3, the volume of the reactor section where condensed C is
formed is
V
R
¼ tv
0
t
cr
¼ 2036 L
The total volume of the reactor is V
R
¼ 751.9 þ 2036 ¼ 2788 L.
d. The reactor operation is described by two design equations: (f) for
0 Z 0.13 and (k) for Z . 0.13. Figure E7.3.2 shows the reaction operat-
ing curve. Using Eq. 2.7.8, the species curves are
F
A
(t)
(F
tot
)
0
¼ y
A
0
Z(t)
F
B
(t)
(F
tot
)
0
¼ y
B
0
Z(t)
F
C
(t)
(F
tot
)
0
¼ Z(t)
Figure E7.3.3 shows the species curves.
Figure E7.3.2 Reaction operating curve.
256 PLUG-FLOW REACTOR

Example 7.4 Design a packed-bed reactor for the gas-phase, heterogeneous
catalytic cracking reaction
A ! B þC
The reactor should produce 20 metric ton per day of product B at 75% conver-
sion of A. A process stream at 3878C and 2 atm, consisting of 90% A and 10%
inert (I), is available in the plant. Based on the kinetic data below:
a. Determine the feed flow rate to the reactor.
b. Derive the design equation and plot the reaction and species curves.
c. Determine the volume of the reactor.
d. Determine the mass of the catalyst in the bed.
Data: Molecular mass of product B is 28 g/mol
Bulk density of the catalyst bed is 1.30 kg/L
The mass-based reaction rate expression is
r
w
¼
k
w
C
A
1 þ KC
A
mol (g catalyst)
1
s
1
where k
W
¼ 0.192 cm
3
/g-cat s, and K ¼ 60 L/mol.
Solution This example sho ws the use of the design equation when the ra te
expression is given on the basis of mass, and it is not a po wer function of the species
concentrations. The s toichiometric coefficients of the chemical reaction ar e
s
A
¼1 s
B
¼ 1 s
C
¼ 1 s
I
¼ 0 D ¼ 1
Figure E7.3.3 Species operating curves.
7.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 257

W e select the feed str eam as the reference s tream; hence, y
A
0
¼ 0:9, y
A
I
¼ 0:1, and
y
B
0
¼ y
C
0
¼ 0. The reference concentration is
C
0
¼
P
0
RT
0
¼
2atm
(0:08206 L atm/mol K)(660 K)
¼ 0:0369 mol=L
a. To determine the feed flow rate, we use Eq. 2.7.8 to express the production
rate of product B in terms of (F
tot
)
0
. Selecting the inlet stream as the reference
stream,
F
B
out
¼ (F
tot
)
0
( y
B
0
þ s
B
Z
out
) (a)
Using Eq. 2.6.5,
Z
out
¼
y
A
0
s
A
f
A
out
¼ 0:675
and the molar flow rate of product B at the reactor outlet is
F
B
out
¼
_
m
B
MW
B
¼ 8:267 mol=s
Hence, using (a), the molar flow rate of the reference stream is
(F
tot
)
0
¼
F
B
out
s
B
Z
out
¼ 12:25 mol=s
The volumetric flow rate of the reference stream is
v
0
¼
(F
tot
)
0
C
0
¼ 332:0L=s (b)
b. Using Eq. 3.2.3, the volume-based rate expression is
r ¼ r
bed
k
w
C
A
1 þ KC
A
¼
kC
A
1 þ KC
A
(c)
where k ¼ r
bed
k
w
¼ 0.250 s
21
is the volume-based reaction rate constant. We
use Eq. 7.2.6 to express the species concentrations:
C
A
¼ C
0
0:9 Z
1 þ Z
(d)
258 PLUG-FLOW REACTOR

Substituting (c) and (d) in Eq. 7.2.2, the design equation becomes
dZ
dt
¼ kC
0
0:9 Z
1 þ Z
1 þ KC
0
0:9 Z
1 þ Z
0
B
@
1
C
A
t
cr
C
0
(e)
We define the characteristic reaction time by
t
cr
¼
1
k
¼ 4s (f)
and the design equation reduces to
dZ
dt
¼
0:9 Z
1 þ Z þ KC
0
(0:9 Z)
(g)
Substituting the numerical values of K and C
0
, (g) becomes
dZ
dt
¼
0:9 Z
2:99 1:21Z
(h)
We solve (h) numerically subject to the initial condition that at t ¼ 0, Z ¼ 0.
The reaction curve is shown in Figure E7.4.1. Using Eq. 2.7.8, the species
curves are
F
A
(t)
(F
tot
)
0
¼ y
A
0
Z(t)
F
B
(t)
(F
tot
)
0
¼
F
C
(t)
(F
tot
)
0
¼ Z(t)
Figure E7.4.2 shows the species curves.
Figure E7.4.1 Reaction operating curve.
7.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 259
