Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.

c. To determine the reactor volume, we use either the reaction operating curve
or the integral form of the design equation:
t ¼
ð
0:675
0
2:99 1:21Z
0:9 Z
dZ ¼ 3:458 (i)
Using Eq. 7.1.3, the volume of the reactor is
V
R
¼ tv
0
t
cr
¼ 4584 L ( j)
d. The mass of the catalyst bed is
M
bed
¼ r
bed
V
R
¼ 5959 kg
We conclude the discussion with two comments. First, note that the design
equation provides us with the reactor volume needed to obtain a given extent (or
conversion). However, it does not indicate whether we should use a long reactor
with a small diameter or a short reactor with a large diameter (provided, of
course, that the plug-flow assumption is valid). The reactor diameter is determined
by other considerations such as the heat-transfer area needed to provide (or remove)
heat to the reactor, and the pressure drop (pumping cost). The effect of heat-transfer
on the performance of plug-flow reactors is discussed in Section 7.4. The effect of
pressure drop on the performance of gaseous plug-flow reactors is discussed in
Section 7.5. Second, in the examples above, we designed the reactor for a given
specified extent. However, the question of what is the most suitable reaction
extent for the operation has not been discussed. The cost of the reactants, the
value of the products, the cost of the equipment, and the operating expenses
(including separation costs) affect the optimal level of the extent. These points
are covered in Chapter 10.
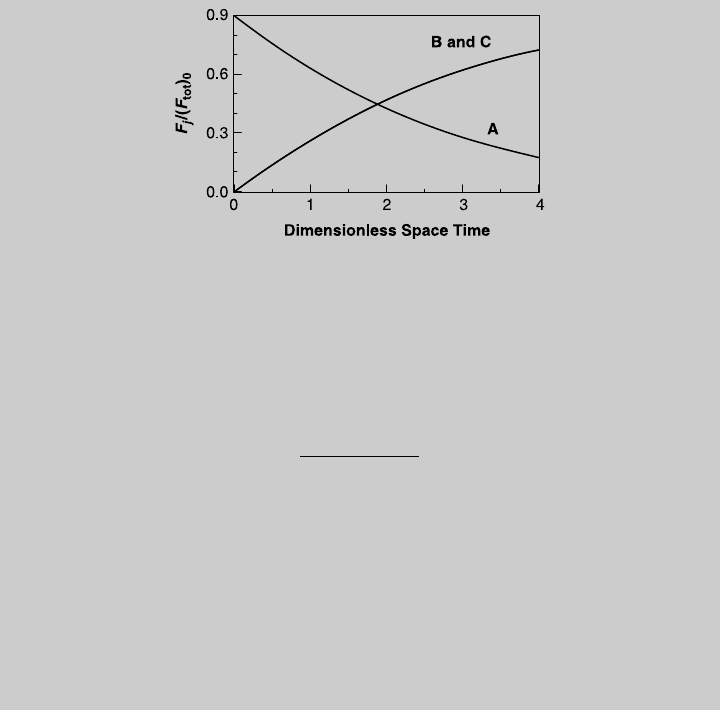
Figure E7.4.2 Species operating curves.
260 PLUG-FLOW REACTOR
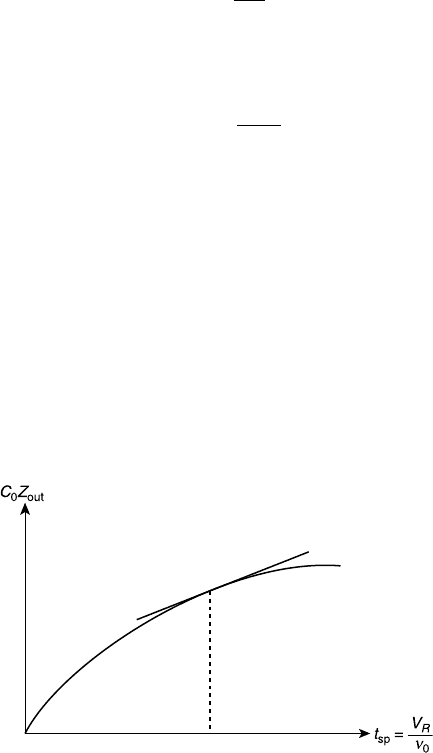
7.2.2 Determination of Reaction Rate Expression
In the preceding section, we described how to apply the design equation when the
reaction rate expression is given. Now, we will discuss how to determine the rate
expression from data obtained in plug-flow operations. The method is based on
measuring the exit composition of the reactor at different space times, and then,
by differentiating the data, we obtain the reaction rate at the exit conditions.
Different space times are obtained by either withdrawing samples at different
points along the reactor (different reactor volumes) or by varying the feed flow
rate. The approach is similar to the differential method applied to batch reactors.
To derive a relation between the reaction rate and the extent, we rearrange Eq.
7.2.2 and use Eq. 7.1.3 to obtain
r ¼ C
0
dZ
dt
sp
(7:2:10)
where t
sp
¼ V
R
/v
0
is the space time defined by Eq. 4.4.6. For the reactor outlet,
r
out
¼ C
0
dZ
out
dt
sp
(7:2:11)
Hence, by plotting C
0
Z
out
versus t
sp
and taking the derivatives, we can determine
r
out
as shown schematically in Figure 7.4. Note that r
out
occurs at the outlet
concentration.
The main difficulty in using Eq. 7.2.11 is that the extent is not a measurable
quantity. Therefore, we have to derive a relationship between Z
out
and an appropri-
ate measured quantity. We do so by using the design equation and relevant stoichio-
metric relations. In most applications, we measure the concentration of a species at
the reactor outlet and calculate the extent by either Eq. 7.2.5 for liquid-phase reac-
tions or Eq. 7.2.6 for gas-phase reactions. We can then determine the orders of the
individual species for power rate expressions.
Figure 7.4 Determination of reaction rate from PFR operating data.
7.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 261

Example 7.5 A stream of gaseous reactant A at 3 atm and 308C
(C
A
0
¼ 120 mmol=L) is fed into a 500-L plug-flow reactor where it decomposes
according to the reaction:
A ! 2B þ C
The concentration of reactant A is measured at the outlet of the reactor for differ-
ent feed flow rates. Based on the data below, determine:
a. The order of the reaction
b. The rate constant at 308C
Data:
v
0
(L=min) 5:010:012:516:725:050:0 125 250 1
C
A
out
(mmol=L) 7:113:516:220:026:840:662:679:5 120
Solution The stoichiometric coefficients are:
s
A
¼1 s
B
¼ 2 s
C
¼ 1 D ¼ 2
We select the inlet stream as the reference stream; hence,
y
A
0
¼ 1, y
B
0
¼ y
C
0
¼ 0, and C
0
¼ C
A
0
¼ 120 mol=L. Using Eq. 7.2.6, we cal-
culate Z
out
for each outlet concentration:
Z
out
¼
C
0
y
A
0
C
A
out
D C
A
out
s
A
C
0
(a)
The reactor space time is
t
sp
¼
V
R
v
0
(b)
For each run, we calculate Z
out
using (a), and t
sp
using (b), and then calculate
C
0
Z
out
:
a. To determine the reaction rate at the reactor outlet for each run (using Eq.
7.2.11), we differentiate the data. Since the data points are not equally
v
0
(L/min) 5.0 10.0 12.5 16.7 25.0 50.0 125 250 1
C
A
out
(mmol=L) 7.1 13.5 16.2 20 26.8 40.6 62.6 79.5 120
Z
out
0.841 0.724 0.681 0.622 0.537 0.395 0.234 0.145 0
t
sp
(min) 100 50 40 30 20 10 4 2 0
C
0
Z
out
(mmol/L) 101 86.9 81.7 74.6 64.4 47.4 28.1 17.4 0
262 PLUG-FLOW REACTOR
spaced, we calculate the derivative at the midpoints of each of two adjacent
points. The calculated values are given in the table below:
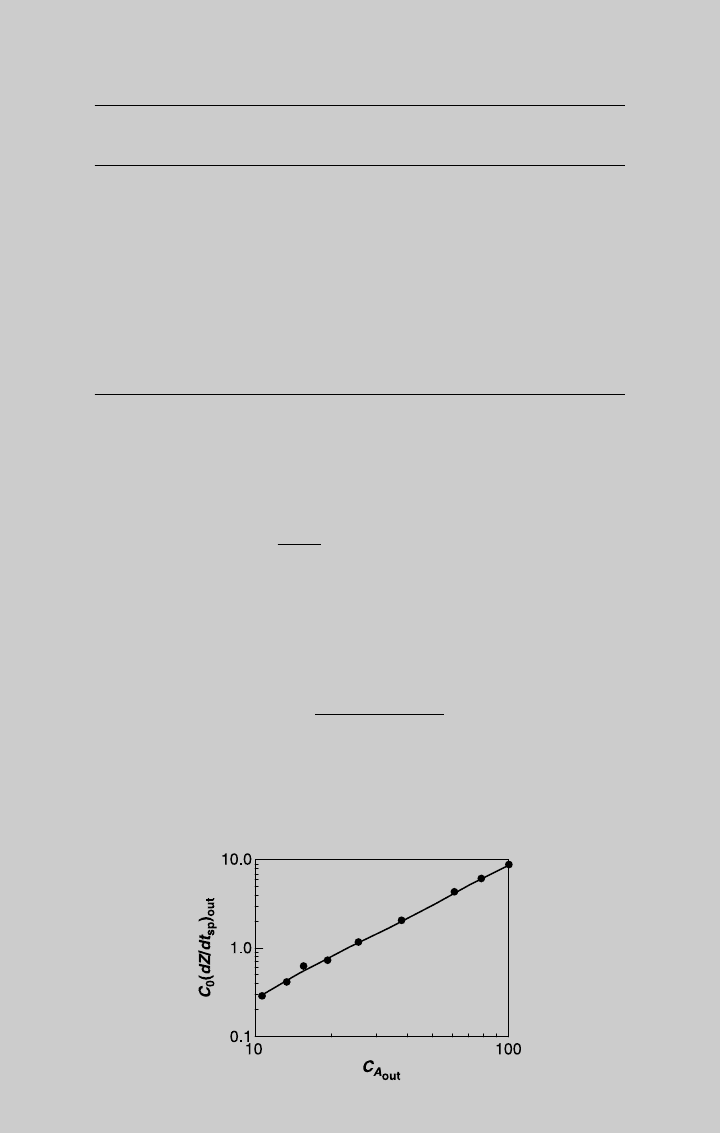
Assuming the rate expression is of the form r ¼ kC
a
A
, substituting
Eq. 7.2.11, and taking the log,
ln C
0
dZ
out
dt
sp
¼ ln (k) þ a ln (C
A
out
) (c)
By plotting ln(C
0
dZ
out
/dt
sp
) versus ln(C
A
out
), we should get a straight line
whose slope is a. The plot is shown in Figure E7.5.1. The slope is
Slope ¼
ln 10 ln 0:35
ln 100 ln 10
¼ 1:48 (d)
b. Now that the order of the reaction is known ( a ¼ 1.5), we use the integral
form of the design equation (Eq. 7.2.4) to determine the value of k.For
t
sp
(min)
C
0
Z
out
(mmol/L)
C
A
out
(mmol=L)
DðC
0
Z
out
Þ=D t
sp
ðmmol=L minÞ
(C
A
out
)
ave
(mmol=L)
0 0.0 120
2 17.4 79.5 8.70 99.7
4 28.1 62.6 5.35 71.0
10 47.4 40.6 3.21 51.6
20 64.4 26.8 1.70 33.7
30 74.7 20.2 1.03 23.5
40 81.7 16.2 0.70 18.2
50 86.9 13.5 0.52 14.9
100 101.0 7.1 0.28 10.3
Figure E7.5.1 Determination of reaction order.
7.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 263

a ¼ 1.5, Eq. 7.2.2 reduces to
dZ
dt
¼
1 Z
1 þ 2Z
1:5
(e)
Using Eq. 3.5.4, the characteristic reaction time is
t
cr
¼
1
kC
0:5
A
0
(f)
Separating the variables and integrating (e),
t ;
t
sp
t
cr
¼
ð
Z
out
0
(1 þ 2Z)
1:5
(1 Z)
1:5
dZ ¼ G(Z
out
) (g)
The right-hand side of (g) is a function of Z
out
, G(Z
out
). Hence, by plotting
G(Z
out
) versus the space time, t
sp
, we obtain a line whose slope is 1/t
cr
.
The table below provides the calculated data, and the plot is shown in
Figure E7.5.2.
The slope is
Slope ¼
1
t
cr
¼ kC
0:5
A
0
¼ 0:1 min
1
(h)
and the rate constant at 308Cisk ¼ 9.13 10
23
(L/mmol)
0.5
min
21
.
Figure E7.5.2 Determination of reaction rate constant.
t
sp
(min) 100 50 40 30 20 10 4 2 0
Z
out
0.841 0.724 0.681 0.622 0.537 0.395 0.234 0.145 0
G(Z
out
) 10.01 5.00 4.00 3.00 2.00 1.00 0.40 0.20 0
264 PLUG-FLOW REACTOR
7.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS
When more than one chemical reaction takes place in a plug-flow reactor, we
have to address several issues before we start the design. First, we have to deter-
mine how many independent reactions there are among the given reactions and
then select a set of independent reactions. Next, we have to identify all the reac-
tions that actually take place in the reactor (including dependent reactions) and
express their rates. As discussed in Chapter 4, we have to write the design
equation for each independent chemical reaction. To solve the design equations
(obtain relationships between Z
m
’s and t), we have to express the rates of the
individual chemical reactions, r
m
’s and r
k
’s, in terms of the Z
m
’s and t. The
procedure for designing plug-flow reactors with multiple chemical reactions
goes as follows:
1. Identify all the chemical reactions that take place in the reactor and define
the stoichiometric coefficients of each species in each reaction.
2. Determine the number of independent chemical reactions.
3. Select a set of independent reactions from among the reactions whose rate
expressions are given.
4. For each dependent reaction, determine its a
km
multipliers with each of the
independent reactions using Eq. 2.4.9.
5. Select a reference stream [determine (F
tot
)
0
, C
0
, v
0
] and its species compo-
sitions, y
j
0
’s.
6. Write Eq. 7.1.1 for each independent chemical reaction.
7. Select a leading (or desirable) chemical reaction and determine the
expression and the value of its characteristic reaction time, t
cr
.
8. Express the reaction rates in terms of the extents of the independent
reactions, Z
m
’s.
9. Specify the inlet conditions.
10. Solve the design equations for Z
m
’s as functions of the dimensionless space
time, t, and obtain the reaction operating curves.
11. Determine the species operating curves using Eq. 2.7.8.
12. Determine the reactor volume using Eq. 7.1.3.
Below, we describe the design formulation of isothermal plug-flow reactors with
multiple reactions for various types of chemical reactions (reversible, series, paral-
lel, etc.). In most cases, we solve the design equations numerically by applying a
numerical technique such as the Runge-Kutta method or using commercial math-
ematical software such as HiQ, Mathcad, Maple, and Mathematica. In some
simple cases, we can obtain analytical solutions. Note that, for isothermal oper-
ations, du ¼ 0, and we do not have to solve the energy balance equation simul-
taneously with the design equations.
7.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 265

Example 7.6 Product B is produced in an isothermal tubular reactor where the
following gas-phase, first-order chemical reactions take place:
Reaction 1: A ! 2B
Reaction 2: B ! C þ D
A gaseous stream of reactant A (C
A
0
¼ 0:04 mol=L) is fed into a 200 L reactor at
a rate of 100 L/min. At the reactor operating temperature, k
1
¼ 2 min
21
and
k
2
¼ 1 min
21
. The pressure drop along the reactor is negligible.
a. Derive the design equations and plot the reaction and species curves.
b. Derive and plot the reaction, yield, and selectivity curves.
c. Determine the conversion of reactant A and the production rate of products B,
C, and D for the given reactor.
d. Determine the optimal reactor volume to maximize the production rate of
product B.
e. Determine the conversion of A, the production rate of products B and C, and
the yield of product B in the optimal reactor.
Solution Since each reaction has a species that does not participate in the
other, the two reactions are independent, and there is no dependent reaction.
The stoichiometric coefficients are
s
A
1
¼1 s
B
1
¼ 2 s
C
1
¼ 0 s
D
1
¼ 0 D
1
¼ 1
s
A
2
¼ 0 s
B
2
¼1 s
C
2
¼ 1 s
D
2
¼ 1 D
2
¼ 1
We write Eq. 7.1.1 for each of the independent reactions (m ¼ 1, 2),
dZ
1
dt
¼ r
1
t
cr
C
0
(a)
dZ
2
dt
¼ r
2
t
cr
C
0
(b)
We select the inlet stream as the reference stream; hence, Z
1
in
¼ Z
2
in
¼ 0. Since
only reactant A is fed into the reactor, C
0
¼ C
A
0
¼ 4 mol=L, y
A
0
¼ 1, and
y
B
0
¼ y
C
0
¼ y
D
0
¼ 0. Also,
(F
tot
)
0
¼ v
0
C
0
¼ 4 mol=min
a. For gas-phase reactions, isothermal operation (u ¼ 1), and negligible
pressure drop, we use Eq. 7.1.15 to express the species concentrations, and
266 PLUG-FLOW REACTOR
the reaction rates are
r
1
¼ k
1
C
0
1 Z
1
1 þ Z
1
þ Z
2
(c)
r
2
¼ k
2
C
0
2Z
1
Z
2
1 þ Z
1
þ Z
2
(d)
We define the characteristic reaction time on the basis of Reaction 1; hence,
t
cr
¼
1
k
1
¼ 0:5 min (e)
Substituting (c), (d), and (e) into (a) and (b), the design equations become
dZ
1
dt
¼
1 Z
1
1 þ Z
1
þ Z
2
(f)
dZ
2
dt
¼
k
2
k
1
2Z
1
Z
2
1 þ Z
1
þ Z
2
(g)
We solve (f) and (g) numerically subject to the initial condition that at t ¼ 0,
and Z
1
¼ Z
2
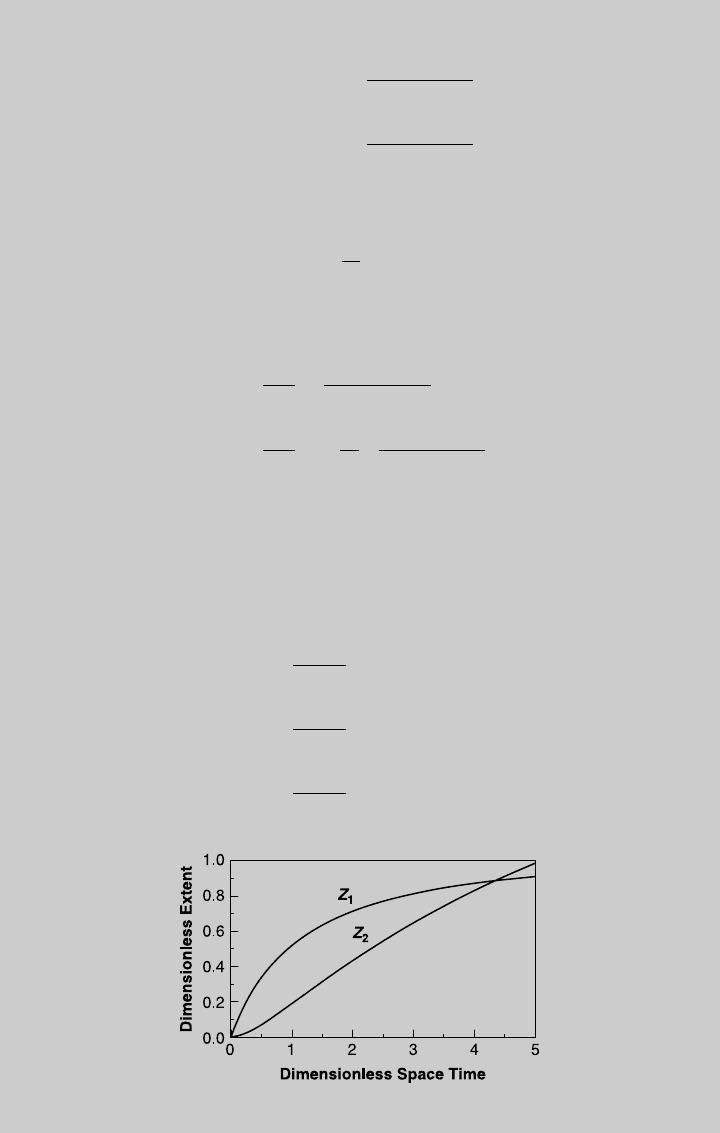
¼ 0. The reaction curves are shown in the Figure E7.6.1. Once
we have Z
1
and Z
2
as a function of the dimensionless space time, we use
Eq. 2.7.8 to determine the species curves:
F
A
(F
tot
)
0
¼ 1 Z
1
(h)
F
B
(F
tot
)
0
¼ 2Z
1
Z
2
(i)
F
C
(F
tot
)
0
¼ Z
2
(j)
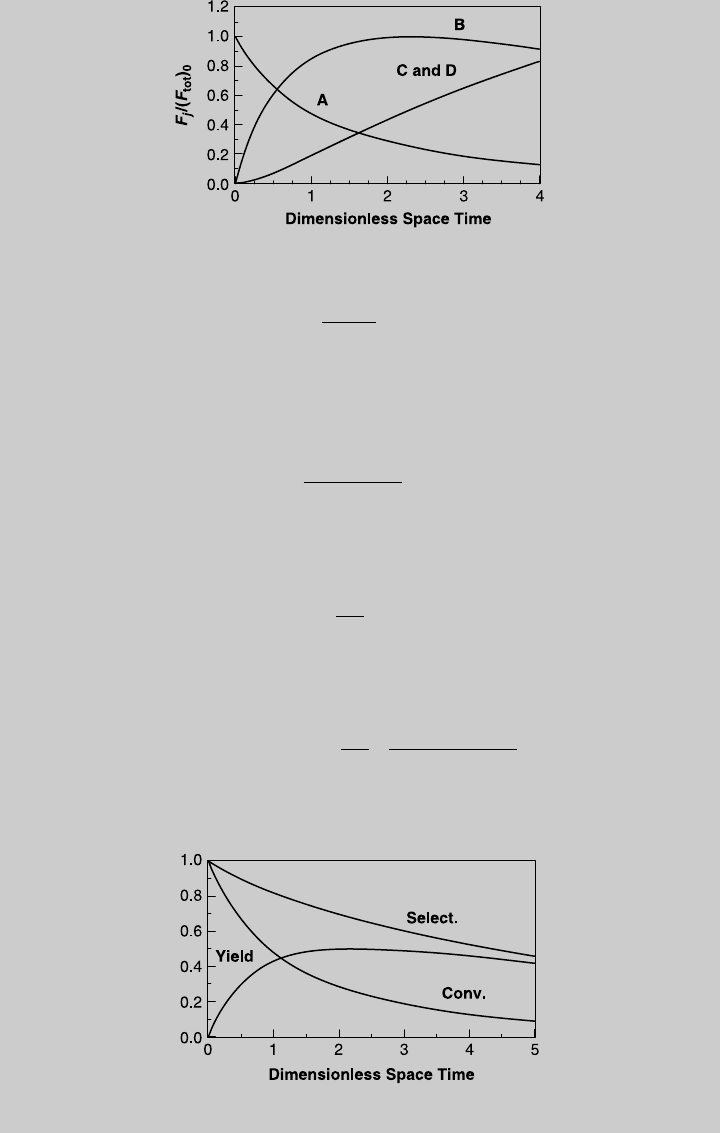
Figure E7.6.1 Reaction operating curves.
7.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 267
F
D
(F
tot
)
0
¼ Z
2
(k)
Figure E7.6.2 shows the species curves.
b. Using Eq. 2.6.2,
f
A
(t) ¼
F
A
in
F
A
out
F
A
in
¼ 1 Z
1
(t) (l)
The desirable reaction is Reaction 1. Using Eq. 2.6.15, the yield of product B is
h
B
(t) ¼
1
2
[2Z
1
(t) Z
2
(t)] (m)
Using Eq. 2.6.19, the selectivity of product B is
s
B
(t) ¼
1
2
2Z
1
(t) Z
2
(t)
Z
1
(t)
(n)
Figure E7.6.3 shows the conversion, yield, and selectivity curves.
Figure E7.6.2 Species operating curves.
Figure E7.6.3 Conversion, yield, and selectivity curves.
268 PLUG-FLOW REACTOR

c. For the given reactor and feed flow rate, using Eq. 7.1.3 and (i), the dimen-
sionless space time is
t ¼
V
R
v
0
k
1
¼ 4
From the reaction curves (or tabulated calculated data), for t ¼ 4, Z
1
¼ 0.874
and Z
2
¼ 0.832. Using (c) through (f), the species molar flow rates at
the reactor outlet are F
A
¼ 0.504 mol/min, F
B
¼ 3.66 mol/min, F
C
¼
F
D
¼ 3.33 mol/min. Using (l), f
A
¼ 0.874, using (m), the yield of product
B is 0.458, and using (n) the selectivity is 0.524.
d. To determine the reactor volume that provides the highest production rate of
product B, we use the species curves (or tabulated calculated data) and find
that the highest F
B
is reached at t ¼ 2.30. Using Eq. 7.1.3 and (e), the opti-
mal reactor volume is
V
R
¼
tv
0
k
1
¼ 115 L
From the reaction operating curves, at t ¼ 2.30, the solutions of (f) and (g)
are Z
1
¼ 0.751 and Z
2
¼ 0.501. Using (h) through (k), the species molar flow
rates at the optimal reactor outlet are: F
A
¼ 0.996 mol/min, F
B
¼ 4.00 mol/
min, F
C
¼ F
D
¼ 2.00 mol/min. Using (l), the conversion of reactant A is
0.751. Using (m), the yield of product B is 0.500. Using (n), the selectivity
of product B is 0.667. The table below provides a comparison between the
performance of the given reactor and the optimal reactor.
We see that the volume of the optimal reactor is slightly more than half that
of the given reactor. While the feed rate is the same, the conversion of
reactant A is slightly lower, the production rate of product B is about 10%
higher, and the production of the by-products (products C and D) is reduced
by 40%.
Given Reactor Optimal Reactor
Reactor volume (L) 200 115
Volumetric feed rate (L/min) 100 100
Conversion of reactant A 0.874 0.751
Yield of product B 0.458 0.500
Selectivity of product B 0.524 0.667
Production rate of product B (mol/min) 3.66 3.99
Production rate of product C (mol/min) 3.33 2.00
7.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 269
